Abstract
Objective
To investigate if hydrogen-rich saline (HRS), which has been shown to have antioxidant and anti-inflammatory properties, could mitigate cardiac remodelling and reduce the incidence of atrial fibrillation (AF) in the rat model of cardiac hypertrophy.
Methods
Pressure overload was induced in rats by abdominal aortic constriction (AAC). The animals were separated into four groups: sham; AAC group; AAC plus low dose HRS (LHRS); AAC plus high dose HRS (HHRS). The sham and AAC groups received normal saline intraperitoneally and the LHRS and HHRS groups received 3 or 6 ml/kg HRS daily for six weeks, respectively. In vitro research was also performed using cardiotrophin-1 (CT-1)-induced hypertrophy of cultured neonatal rat cardiomyocytes.
Results
Cardiac hypertrophy was successfully induced by AAC and low and high dose HRS mitigated the pressure overload as shown by lower heart and atrial weights in these treatment groups. AF incidence and duration of the HRS groups were also significantly lower in the HRS groups compared with the AAC group. Atrial fibrosis was also reduced in the HRS groups and the JAK-STAT signalling pathway was down-regulated. In vitro experiments showed that hydrogen-rich medium mitigated the CT-1-induced cardiomyocyte hypertrophy with a similar effect as the JAK specific antagonists AG490.
Conclusions
HRS was found to mitigate cardiac hypertrophy induced by pressure overload in rats and reduce atrial fibrosis and AF which was possibly achieved via inhibition of the JAK-STAT signalling pathway.
Keywords
Introduction
Affecting approximately 26 million population worldwide, heart failure (HF) is one of the most common life-threatening chronic cardiovascular diseases and is increasing in prevalence. 1 Atrial fibrillation (AF) is the most common persistent arrhythmia and reduces cardiac function and increases the chance of stroke. 2 The conditions of HF and AF are closely inter-related. Patients with HF have a significantly high risk of AF, while patients with AF always have a low cardiac function. 3 In addition, both conditions have many risk factors in common, including, hypertension, ischemic heart disease, diabetes, obesity, arteriosclerosis and age. 4 Although increased fibrosis and altered subcellular Ca2+ homeostasis are involved in the pathophysiology of AF, the exact underlying molecular changes in HF have not been delineated. 4
Several signalling pathways have been implicated in the development of cardiac hypertrophy. 5 Of these, the Janus kinase/signal transducer and activation of transcription (JAK/STAT) signalling pathway has been shown to play a crucial role in cardiac remodelling and cell inflammatory response.5,6 In particular, the JAK/STAT3 signalling pathway has been shown to be involved in the production of interleukin (IL)-6 in neutrophils and activated macrophages. 7 Cardiotrophin-1 (CT-1) is a cardiac hypertrophy-inducing factor and a member of the IL-6 family of cytokines. 5, Studies suggests that CT-1 achieves its effect via activation of JAK/STAT pathway. 5, However, limited information is available on the association of JAK/STAT, CT-1 and pressure- overload-induced atrial remodelling.
Molecular hydrogen has been shown to have antioxidant and anti-inflammatory properties and several studies have shown that it has beneficial effects in animal models and human disease including ischemia-reperfusion injury, 8 ischemic stroke, 9 and oxidative-stress-induced radiation injury. 10 Indeed, hydrogen-rich saline (HRS) has been shown in animal models to have beneficial effects in doxorubicin-induced HF. 11 In addition, one study found that HRS could mitigate pressure-overload-induced hypertrophy and promote cardiac function in rats. 5 To our knowledge, there has been no previous investigation on the impact of HRS on the incidence of HF associated AF. Therefore, in this present study we used animal in vivo and in vitro methods (i.e., the rat model of cardiac hypertrophy induced by pressure overload due to abdominal aortic constriction [AAC] and the CT-1-induced hypertrophy of cultured rat cardiomyocytes) to investigate if HRS could mitigate cardiac remodelling and reduce the incidence of AF. In addition, we investigated the involvement of the JAK/STAT signalling pathway.
Materials and methods
Rat model of cardiac hypertrophy
Adult male Sprague-Dawley rats (200–220 g) were housed in laboratory cages in a controlled environment and kept under a 12/12 h dark/light cycle with free access to food and water. The animals were acclimated for seven days before any experimental procedures took place. HRS was prepared by saturating saline with hydrogen (0.6 mmol/l).
In total, 81 rats were used in the in vivo study and randomly separated into four groups:
sham group, n = 23 AAC (abdominal aortic constriction) group, n = 19 LHRS (AAC + low dose HRS), n = 19 HHRS (AAC + high dose HRS), n = 20.
With the exception of the sham group, all rats underwent AAC surgery, as previously described. 9 In brief, rats were anaesthetized, placed in a supine position and a 2-cm incision was made along the midline of the abdomen. A length of 4–0 silk suture (8cm) was passed underneath the abdominal aorta between the origins of the right and left renal arteries. A knot was made around the aorta and a 22G needle; the needle was then removed to achieve a 0.7-mm diameter constriction. Sham-operated rats received the same surgical procedure without constriction. The sham and AAC groups received normal saline intraperitoneally and the LHRS and HHRS groups received 3 or 6 ml/kg HRS daily for six weeks, respectively. At 6 weeks the rats were sacrificed, with quick decollation to obtain heart samples. Body weight was recorded and hearts, atria and left ventricles were weighed separately.
All animal procedures were performed according to the NIH and ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines for the Care and Use of Laboratory Animals. The study was approved by the Experimental Animal Administration Committee of Nanchang University and Biomedical Ethics Committee of Nanchang University.
Echocardiography
At 6 weeks before euthanasia, echocardiography was performed using MyLab 30CV ultrasound system (Biosound Esaote, Inc., Indianapolis, IN) on anesthetized (1.5% isoflurane) rats using a 10-MHz linear array transducer. Echocardiographic parameters including heart rate (beats/min), left ventricular anterior wall thickness at end-diastole (LVAWd), left ventricular posterior wall thickness at end-diastole (LVPWd), fractional shortening, and left ventricular ejection fraction (LVEF) were measured. Both parasternal long- and short-axis views were adopted to measure the left ventricle at a frame rate of 120 Hz.
High frequency electrical stimulation
Also at 6 weeks, electrophysiological characteristics of the animals’ hearts were determined using intracardiac programmed electrical stimulation. 12 Rats were anesthetized and a surface electrocardiogram (ECG) and intracardiac electrogram were recorded simultaneously for each animal. P wave duration and intra-atrial conduction time were measured. An electrode catheter (1.1 F, Science) was inserted into the right atrium through the right jugular vein. A burst of electrical stimuli for 30 seconds was applied to test the inducibility of atrial arrhythmias. Thereafter, three more burst tests were conducted. AF was judged to have occurred on P wave disappearance and QRS wave irregularity lasting more than 2 seconds. LabChart Software 7.0 was used to record and analyse ECG data.
Western blotting
After the heart samples had been collected from the rats, key protein expressions of JAK-STAT pathways in atrial myocardia were examined using western blotting analysis as previously described. 13 The following antibodies were used: primary anti-IL-6 (1:500, ab9324, Abcam); anti-JAK (1:1000, ab32101, Abcam); anti-STAT3 (1:1000. ab76315, Abcam); anti-β-actin (1:5000, ab179467, Abcam). The Gel Imaging System and Image J software were used to image and analyse Western blotting bands.
Masson's trichome staining
Masson's trichrome staining of heart tissues was used to evaluate the extent of atrial fibrosis. As previously described,
14
following euthanasia, the hearts were removed rinsed in PBS, fixed in 4% paraformaldehyde, and embedded in paraffin. The paraffin‐embedded tissues were then cut into 5‐µm sections and stained with Masson's trichrome staining kit (#HT15, Sigma Aldrich) according to the manufacturer's instructions. Atrial fibrosis was assessed using Image J software and collagen volume fraction (CVF) was calculated according to the following formula:
Cardiotrophin-1-induced hypertrophy of cultured rat cardiomyocytes in vitro
Cardiomyocytes were obtained from 20 neonatal rats (1-3 days old), CT-1 was obtained from R&D Systems, Minneapolis, MN and AG490, a specific JAK inhibitor, was obtained from Gibco, Fort Worth, TX. As described previously, 15 the cardiomyocytes were incubated in Dulbecco's Modified Eagle Medium (DMEM, Gibco, Fort Worth, TX) for 48 h. Thereafter, the medium was replaced with serum-free DMEM with 10 µg/ml transferrin, 10 µg/ml insulin, 1.5 µM Vitamin B12 and 100 µM Vitamin C and the cell culture continued for an additional 24 h. Afterwards, the cells were incubated for 72 hours in one of the following: diluent (control); CT-1 0.1 nM; CT-1 0.1 nM +HRS; CT-1 0.1 nM +AG490 0.1 mM.
Scion Image Software (HJ2000) was used to measure the surface area of the cardiomyocytes in a total of 100 cells from 4 fields. For the measurement of cell size, each field in phase-contrast pictures of the cultured cardiomyocytes was randomly selected and photographed and 25 individual cell surface areas were measured by planimetry. The cardiomyocytes were washed with D-Hanks solution, and scraped off the well. After preparing cell lysates, the protein content of cardiomyocytes was measured by Lowry’s modified assay.
Statistical analyses
Data were analysed using Graphpad Prism 7.0 software and a P-value < 0.05 was considered to indicate statistical significance. Normally distributed continuous variables were expressed as mean ± standard deviation (SD) and differences between groups were compared using one-way analysis of variance (ANOVA) and post hoc Tukey's test. Non-normally distributed variables were expressed as median and interquartile range and differences between groups were compared using the Kruskal-Wallis test and post hoc Dunn's multiple comparisons test. In addition, the AF incidence was compared using χ2 test.
Results
Heart weights
Compared with the sham group, the ratio of heart, atria and left ventricle weights to body weights in the AAC group were statistically significantly greater; these results confirmed extensive cardiac hypertrophy (Figure 1a, 1b, 1c, and 1d). The administration of HRS, whether low or high dose, mitigated the hypertrophy. The reduction was more evident for the high dose HRS group than for the low dose group. However, the ratio of atria to heart weight (AW/HW) showed a reverse trend (Figure 1e). The AW/HW for the AAC group was lower than that of sham group (P < 0.0001) and the AW/HW of the high dose HRS group was higher than that of both the AAC and low dose HRS group (P < 0.01).

Cardiac hypertrophy in the rat abdominal aortic constriction (AAC) model in vivo.
Echocardiography
With the exception of LVEF, statistically significant differences were found among the four groups in heart rate, LVAWd, LVPWd and fractional shortening (Table 1). As expected, by contrast with the sham group, heart rate, LVAWd, and LVPWd were statistically significantly increased in the AAC group. Although values for these parameters were lower in the low dose HRS group compared with the AAC group, they only achieved statistical significance in the high dose HRS group. By contrast with the sham group, fractional shortening was statistically significantly lower in the AAC but only administration of high dose HRS mitigated this effect.
Echocardiographic results from the different treatment groups.

AAC, abdominal aortic constriction; HRS, hydrogen-rich saline; LVAWd, LV anterior wall thickness at end-diastole; LVPWd, LV posterior wall thickness at end-diastole. LVEF = Left ventricular ejection fraction.
**P < 0.05, ***P < 0.005, ****P < 0.001, *****P < 0.0001 vs Sham group,
#P < 0.05, ##P < 0.005, ###P < 0.001 vs AAC group,
+P < 0.05, ++P < 0.005, +++P < 0.001 vs Sham group vs LHRS group.
AF inducibility and duration
Intracardiac programmed electrical stimulus was used to examine the AF inducibility and duration. Representative ECG traces of burst pacing and following heart rhythm are shown for the four groups in Figure 2a. Incidences of AF were, 8.7% (2/23), 52.6% (10/19), 15.8% (3/19) and 15.0% (3/20) for sham, AAC, LHRS and HHRS groups, respectively (Figure 2b). While the incidence of AF was statistically significantly increased from controls in the AAC group, the administration of low and high dose HRS alleviated the effect.
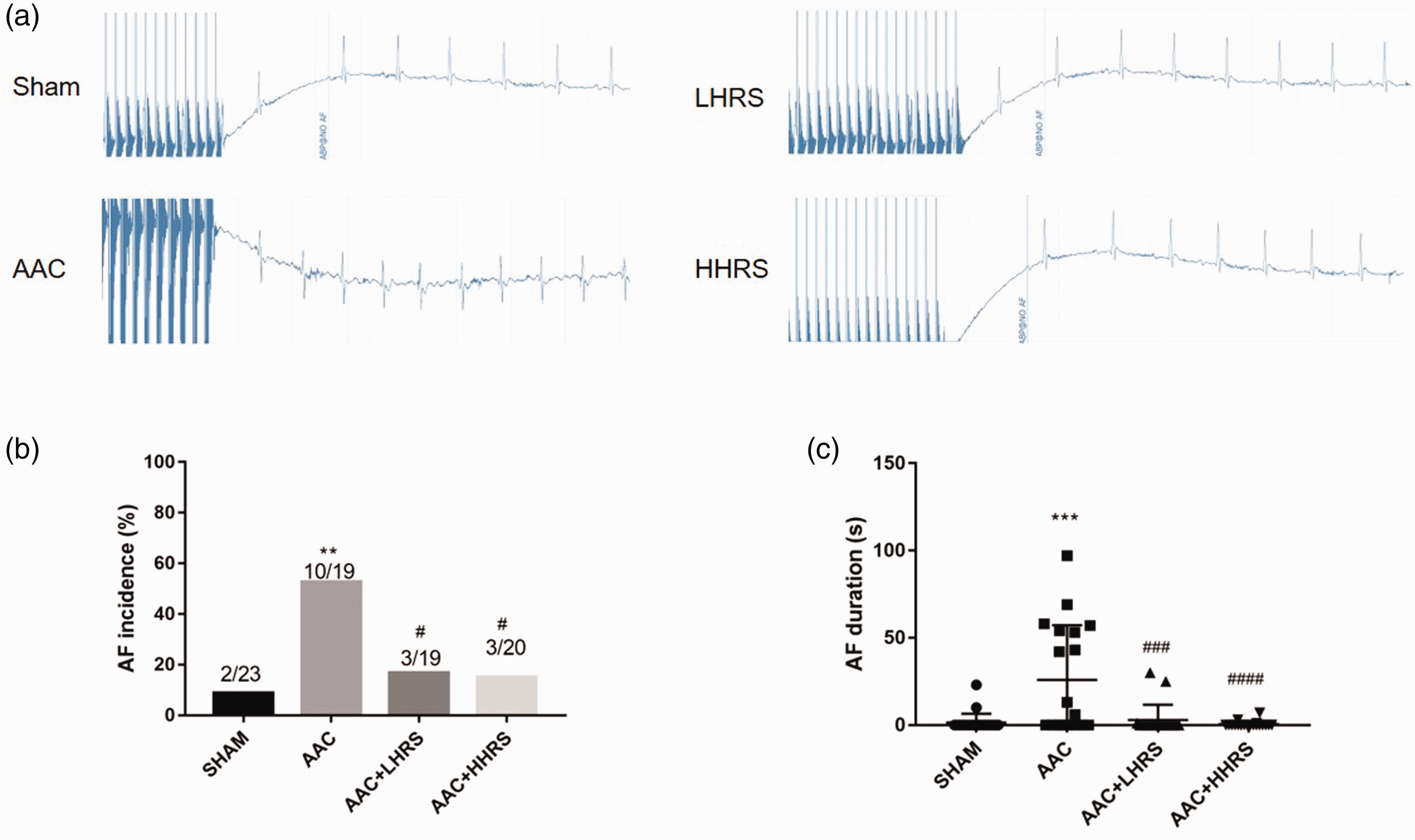
Intracardiac programmed electrical stimulation results. (a) Representative burst pacing electrocardiograph (ECG) traces from the four groups. (b) Incidence (%) of atrial fibrillation (AF) in the four groups. (c) Duration (s) of atrial fibrillation (AF) in the four groups.
The durations of AF were 0 (0, 0) s, 6 (0, 54) s, 0 (0, 0) s, 0 (0, 0) s for sham, AAC, LHRS and HHRS groups, respectively (Figure 2c). While the duration of AF was statistically significantly increased from controls in the AAC group, the administration of low and high dose HRS alleviated the effect.
Atrial fibrosis
Masson trichome staining was used to evaluate atrial remodelling. The AAC group showed more extensive atrial fibrosis than the other groups and the low and high dose HRS groups had less fibrosis than the AAC group (Figure 3a). Using Image J software to calculate the collagen volume fraction (CVF), the AAC group had statistically significantly higher values compared with the sham group (P < 0.0001) and the addition of low and high dose HRS significantly alleviated the effects of AAC (Figure 3b). While the values for LHRS remained significantly higher than the values for the sham group, the values for HHRS were no different from those of the sham group. However, there was a statistically significant difference (P < 0.01) between the low and high dose HRS groups.
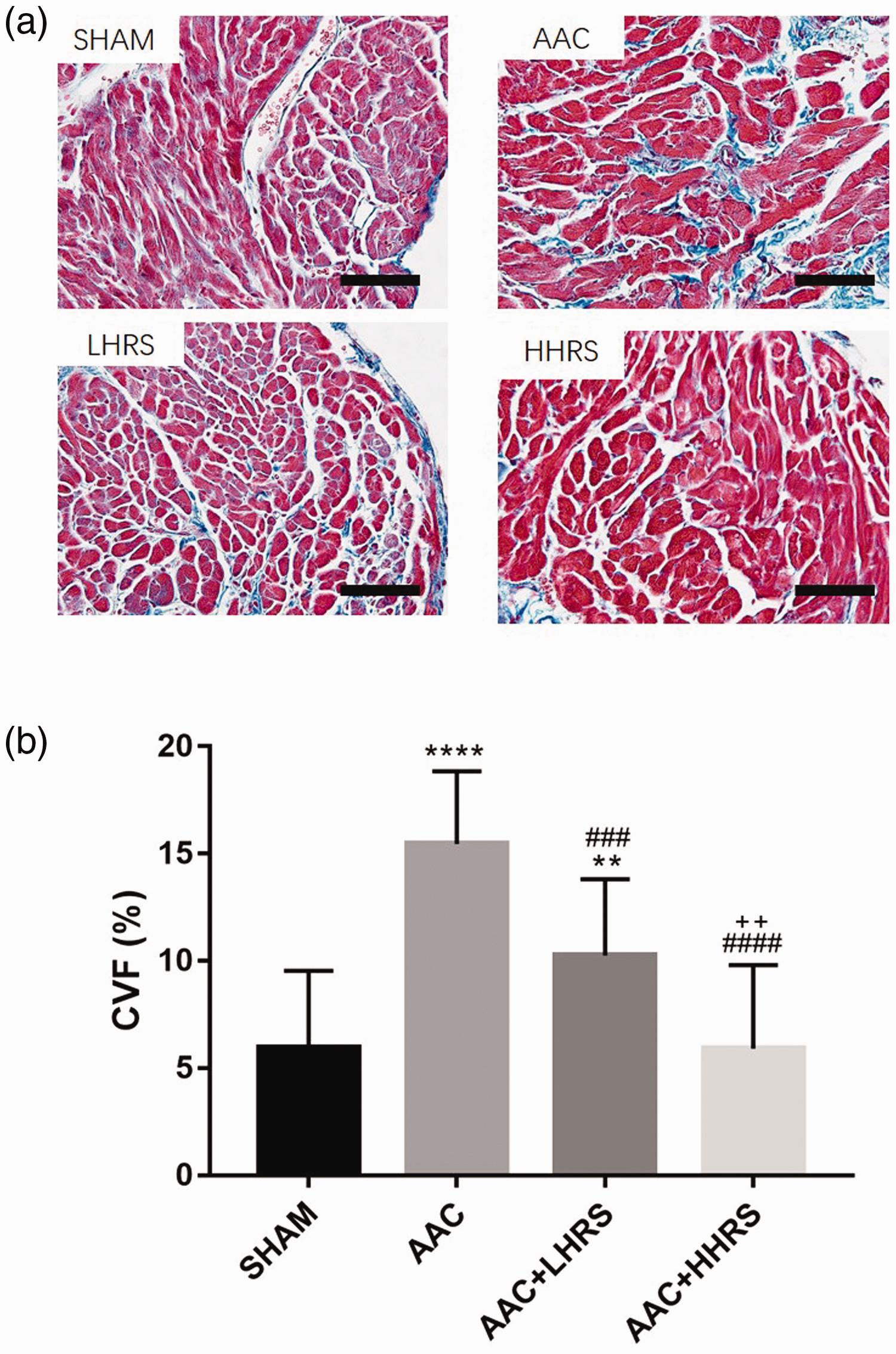
Atrial structural remodelling in vivo.
IL-6 expression and JAK/STAT3 signalling pathway
A representative Western Blot analysis of proteins in cardiomyocytes showing bands of IL-6/JAK/STAT3/β-actin is shown in Figure 4a. Western blot analysis indicated that that IL-6 expression ratio in the AAC group was statistically significantly increased from the sham group and the administration of low and high dose HRS alleviated this trend (Figure 4b). In addition, while the JAK/STAT3 signalling pathway was substantially upregulated in the AAC group compared with the sham group, it was mitigated by the addition of low and high dose HRS (Figures 4c and d).
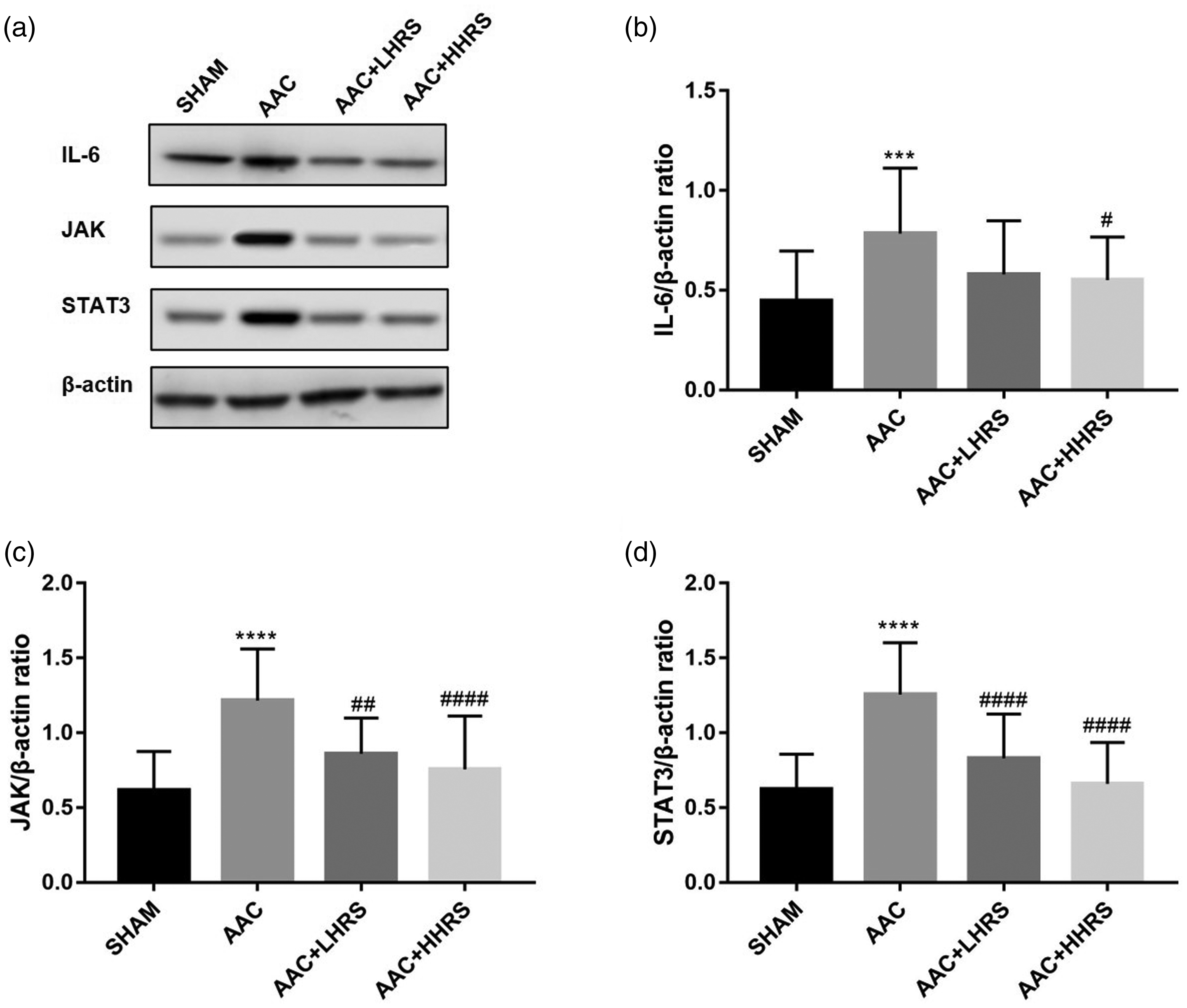
Interleukin (IL)-6/JAK/STAT3 signalling pathway expression in vivo.
Cardiac hypertrophy in vitro
The addition of HRS inhibited CT-1-induced hypertrophy of cultured cardiomyocytes in vitro. The cell surface area and protein content of the cardiac cells treated with CT-1 and the CT-1+ AG490 were statistically significantly greater than controls and those treated with CT-1+HRS (Figures 5a and b). The JAK/STAT3 signalling pathway was activated in the CT-1 group (Figures 5c). Administration of HRS significantly down-regulated the expression of IL-6 and JAK/STAT3 signalling pathway expression, which was similar to the effects of the JAK specific antagonists AG490 (Figures 5d-f).

Effects of hydrogen-rich medium on Cardiotrophin-1 (CT-1) -induced hypertrophy of cultured cardiomyocyte in vitro.
Discussion
Hypertensive heart disease is one of the key contributing factors involved in the cardiac remodelling that is associated with HF. In this study, we successfully induced cardiac pressure overload in the AAC rat model, a commonly used method that mimics cardiac hypertrophy and HF. 16 We found that in the rat model, HRS mitigated the cardiac hypertrophy and HF induced by the pressure overload and also reduced atrial fibrosis and AF which was possibly achieved via the JAK-STAT signalling pathway.
Molecular hydrogen, a potential organ protective agent, has been demonstrated to be effective in ischemia/reperfusion in several organs.18,19 Studies have shown that molecular hydrogen is effective in reducing oxidative stress and cell injury.8–10 Indeed, one study showed that infusion with HRS exerted cardiovascular protective effects against isoproterenol-induced myocardial infarction in part via interactions that evoked antioxidant and anti-inflammatory activities. 20 Another study that focused on the impact of hydrogen on myocardial fibrosis induced by diabetes, suggested that the protective effect of hydrogen against diabetes-induced myocardial fibrosis might be associated with the attenuation of autophagy via the upregulation of the PI3K/AKT1 signalling pathway. 21 In the present study, we used low and high doses of HRS and although both doses alleviated the cardiac hypertrophy, atrial fibrosis, increased duration of AF induced by AAC, the high dose achieved better results than the lower dose. However, there was no difference between doses in AF incidence, which may be attributed to the small sample size.
Cardiac remodelling is a common critical mechanism of both HF and AF. We measured heart weight, left ventricle weight and atrial weight of all treated groups and found that all parts of the heart were enlarged following the AAC procedure indicating significant cardiac remodelling had occurred. Echocardiography demonstrated a significant increase in the thickness of the ventricular wall and a decrease in LVEF, indicating hypertrophy and HF. However, the relative ratio of the atria to the whole heart was lower for the AAC group than the sham group which may have been due to the pressure overload chain which starts from the aorta goes to the left ventricle, then the left atrium, and lastly to the right heart. Consistent with our results, another study that did not measure atrial weight, but measured heart weight (HW) and body weight (BW) found that the ratio of HW/ BW and LVW/BW were in order of AAC > low dose HRS > high dose HRS > sham groups. 5
Cardiac fibrosis plays a crucial role in the pathogenesis of HF and AF. We found that the AAC group had severe atrial fibrosis and as a result of atrial remodelling, demonstrated a higher incidence and longer duration of AF compared with the sham group. Similar to other findings, 5 we also found that the expression of the JAK-STAT signalling pathway was up-regulated in the AAC group. Following the administration of HRS, the atrial fibrosis was mitigated with a dose-dependent response, and the JAK/STAT3 signalling pathway was also inhibited. The AF incidence and duration of both the HRS groups were also significantly lower than those of the AAC group.
To further validate our results, we conducted cell experiments in vitro. Similar to our in vivo findings, the hydrogen rich medium reduced cell surface area and protein content of the cardiomyocytes that had been induced by CT-1 and confirmed that hydrogen was effective in reversing cardiac remodelling. We also found that the JAK/STAT3 signalling pathway was down-regulated by H2 rich medium in vitro, which had a similar effect to the JAK specific antagonists AG490. These results suggest that JAK/STAT3 signalling pathway is a critical factor involved in the inhibitory effects of HRS.
Our study had several limitations. For example, despite the positive effect of HRS on overload pressure-induced AF and atrial fibrosis, other mechanisms such as inflammation and apoptosis were not investigated. Therefore, further research is needed to explore the precise mechanisms involved in cardiac hypertrophy. In addition, only the AAC rat model was used in this study and other HF models were not investigated. More studies are required in the future to confirm our findings.
In summary, HRS was found to mitigate cardiac hypertrophy induced by pressure overload in rats and reduce atrial fibrosis and AF which was possibly achieved via inhibition of the JAK-STAT signalling pathway.

