Abstract
Objective
To investigate the association between serum angiopoietin-like 4 (ANGPTL4) levels and recurrence of atrial fibrillation (AF) after catheter ablation.
Methods
This retrospective study recruited patients with AF undergoing catheter ablation and they were divided into two groups (new-onset AF group and recurrent AF group). Demographic, clinical, and laboratory parameters were collected.
Results
A total of 192 patients with AF were included, including 69 patients with recurrence of AF. Serum ANGPTL4 levels were lower in patients with recurrent AF than in those with new-onset AF. Serum ANGPTL4 levels were positively correlated with superoxide dismutase and peroxisome proliferator-activated receptor γ, and negatively correlated with the CHA2DS2-VASC score, left atrial diameter, and levels of brain natriuretic peptide, malondialdehyde, high-sensitivity C-reactive protein, and interleukin-6. The receiver operating characteristic curve showed that the best cut-off for recurrent AF was serum ANGPTL4 levels < 19.735 ng/mL, with a sensitivity and specificity of 63.9% and 74.5%, respectively. Serum ANGPTL4 levels were significantly associated with recurrence and new onset of AF (odds ratio, 2.241; 95% confidence interval, 1.081–4.648).
Conclusions
Serum ANGPTL4 levels are lower in patients with recurrent AF than in those with new-onset AF, and are associated with cardiac hypertrophy, oxidative stress, and inflammation.
Keywords
Introduction
Atrial fibrillation (AF) is a common persistent and recurrent arrhythmia in clinical practice with an increased risk of stroke and mortality. AF originates in the abnormal atrial matrix and can be triggered and accelerated by multiple disorders, such as heart failure, coronary heart disease, aging, and hypertension.1,2 The prevalence of AF is high in various medical conditions, such as hypertension, chronic heart failure, atherosclerosis, cardiac hypertrophy, and valvular heart disease. 3 Catheter ablation (CA) has been used for treating patients with AF for two decades and has high efficacy compared with antiarrhythmic drugs. However, CA cannot eradicate arrhythmia and recurrence of AF, which are common problems after application of CA. 4 Many clinical scoring systems can be applied in predicting recurrence of AF after catheter ablation, but they have limited efficacy. 5 Therefore, new biomarkers of AF are urgently required for their predictive effect on recurrence of AF.
Angiopoietin-like 4 (ANGPTL4) is a stromal cell protein, which is involved in development of many inflammation-related diseases. ANGPTL4 inhibits lipoprotein lipase activity, thus reducing hydrolysis of plasma triglycerides to free fatty acids. ANGPTL4 expression is regulated by fatty acids. Omega-3 polyunsaturated fatty acids (ω-3 PUFAs) strongly enhance ANGPTL4 expression in rat liver cancer cells compared with saturated fatty acids. 6 Free fatty acids also induce ANGPTL4 expression in cardiomyocytes and show protective function through inhibiting lipoprotein lipase activity.7,8 Furthermore, ANGPTL4 inhibits phenylephrine-induced cardiac hypertrophy, 9 which is an important contributor to arrhythmia. 10
In this study, we assessed changes in serum ANGPTL4 levels in patients with AF, and evaluated their association with recurrence of arrhythmia after CA.
Patients and methods
Study population
This retrospective study was carried out in patients with AF from the Department of Cardiology at Zhoupu Hospital between July 2016 and December 2018. This study procedure was examined and approved by the Ethics Committee of Zhoupu Hospital (approval number: WZ-009). Written informed consent was obtained from all patients or their relatives. Patients with AF were diagnosed by an electrocardiogram (ECG)/Holter recording and echocardiography, and all patients had CA performed on admission to hospital. Healthy controls were people who had a physical examination with normal sinus rhythm and no prior history of AF, and they were matched for age and sex. Exclusion criteria were as follows: patients who received anticoagulation treatment within 1 month (e.g., warfarin and dabigatran); patients with valvular heart disease, venous thrombosis, pulmonary embolism, hepatic and renal insufficiency, or cancer; and patients who had surgery and trauma within 1 month. All patients were followed up after CA for at least 3 months, and all patients with AF were treated with anticoagulants during the follow-up. When relapse of arrhythmia occurred within 3 months, the patients were defined as having recurrent AF, and other patients were defined as having new-onset AF. 4
Radiofrequency CA
A routine blood test, blood biochemistry, coagulation function, myocardial enzyme spectrum, thyroid function, an electrocardiogram, and transthoracic echocardiography were evaluated before the operation. Preoperative transesophageal echocardiography was performed to exclude atrial thrombus. Patients were anesthetized with 1% lidocaine, and the right femoral vein was punctured using the Seldiger technique to establish a pathway. After successful puncture, the LASSO electrode (Biosense Webster, Diamond Bar, CA, USA) and COOLFLEX cryoablation electrode (Abbott, Abbott Park, IL, USA) were inserted into the left atrium from the right atrium through atrioventricular septum puncture. The opening position of the pulmonary veins was determined under the guidance of left atrial modeling, ablation energy and temperature were set to 25 to 35 Watts and 43°C, and cold saline (30°C–35°C) was infused at 17 mL/minute. Fentanyl was used for analgesia and midazolam was used for sedation. Pulmonary vein isolation was performed for CA, with linear ablation of the left and right atria using the superior and inferior pulmonary veins. The end point of CA was bidirectional electrical resistance of the pulmonary vein and left atrium. If AF did not stop during the operation, coronary sinus ablation and fragmentation potential ablation were performed. If AF was still present, synchronous direct current (100–150 J) was applied. At the end of the operation, the sheath tube was pulled out, the bleeding was stopped by compression, and an elastic bandage was applied.
Follow-up
All patients who underwent radiofrequency CA were followed up after discharge. The patients were followed up at 1, 2, 3, 4, 8, and 12 weeks after the operation, and were followed up regularly at 3 and 6 months. All patients at follow-up were asked whether there were any symptoms indicating recurrence of AF. A 12-lead electrocardiogram, dynamic electrocardiogram, and echocardiography were routinely performed, and other relevant examinations were carried out on the basis of the clinical condition of the patients. Patients without palpitation symptoms were routinely examined by ECG or Holter depending on the follow-up date. Patients with palpitation were examined by ECG or Holter immediately. Recurrence of AF was defined as AF, atrial flutter, or atrial tachycardia lasting longer than 30 s after 3 months of radiofrequency ablation.
Clinical data and laboratory tests
Peripheral blood was collected in the morning from each fasting patient on admission to hospital, and samples were sent to a central laboratory as quickly as possible. We recorded the following clinical variables for each patient: age, sex, smoking, hypertension, diabetes mellitus; heart failure, and coronary artery disease. The congestive heart failure, hypertension, age 75 years or older, diabetes mellitus, previous stroke or transient ischemic attack, vascular disease, age 65 to 74 years, female (CHA2DS2-VASC) score was applied to assess the risk of stroke (congestive heart failure: score of 1; hypertension: score of 1; age of 65–74 years: score of 1; age >75 years: score of 2; diabetes: score of 1; stroke/transient ischemic attack: score of 2; vascular disease: score of 1; female: score of 1). Serum brain natriuretic peptide (BNP), high-sensitivity C-reactive protein (hs-CRP), interleukin (IL)-6, and peroxisome proliferator-activated receptor γ (PPARγ) levels were measured by enzyme-linked immunosorbent assay (ELISA). Serum malondialdehyde (MDA) and superoxide dismutase (SOD) levels were measured by colorimetry.
ELISA detection of serum ANGPTL4 levels
Serum ANGPTL4 levels in controls and in patients with AF were measured by an ELISA kit (cat. no. DY3485; R&D Systems, Minneapolis, MN, USA), and absorbance at 450 nm was measured with a microplate reader (ELx800; Bio-Rad Laboratories, Inc., Hercules, CA, USA). ANGPTL4 levels were calculated using a standard curve, which was established by different levels of recombinant human ANGPTL4, and expressed as ng/mL for each case.
Statistical analysis
Data were analyzed by IBM SPSS version 19.0 (IBM Corp., Armonk, NY, USA) statistical software. Continuous variables are expressed as mean ± standard deviation for a normal distribution or expressed as median (interquartile range) for a non-normal distribution, and were analyzed using the t test or Wilcoxon–Mann–Whitney test. Categorical variables are expressed as frequency (percentage) and were analyzed using the chi-square test. The correlations between ANGPTL4 levels and continuous variables were analyzed using Spearman’s rank correlation test. The receiver operating characteristic curve was constructed to determine the cut-off point of serum ANGPTL4 levels between new-onset AF and recurrent AF. Multivariate logistic regression analysis was performed to determine the effect of low serum ANGPTL4 levels (<19.735 ng/mL) on recurrence of AF, and results are expressed as the odds ratio and the 95% confidence interval. P < 0.05 was considered as a significant difference.
Results
Baseline characteristics
A total of 192 patients with AF who underwent CA ablation were included in this study (mean age: 66.64 ± 11.11 years; 54.2% were men). The follow‐up period was at least 3 months and recurrence of AF occurred in 69 (35.9%) patients. Clinical, echocardiographic, and biochemical data of all patients with AF are shown in Table 1. Patients with recurrence of AF had a higher mean CHA2DS2-VASc score, larger left atrial diameter (LAD), higher serum BNP, MDA, hs-CRP, and IL-6 levels, and lower serum SOD and PPARγ levels compared with those with new-onset AF (all P < 0.05).
Clinical characteristics of patients with newly-diagnosed and recurrent AF.

Values are mean ± standard deviation or n (%).
AF, atrial fibrillation; BMI, body mass index; CHA2DS2-VASc, congestive heart failure, hypertension, age 75 years or older, diabetes mellitus, previous stroke or transient ischemic attack, vascular disease, age 65–74 years, female; T2DM, type 2 diabetes mellitus; HF, heart failure; CAD, coronary artery disease; LVEF, left ventricular ejection fraction; LAD, left atrial diameter; BNP, B‐type natriuretic peptide; TG, triglycerides; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; MDA, malondialdehyde; SOD, superoxide dismutase; hs-CRP, high-sensitivity C-reactive protein; IL-6, interleukin-6; PPARγ, peroxisome proliferator-activated receptor γ; ANGPTL4, angiopoietin-like 4.
Serum ANGPTL4 levels in patients with recurrent AF
Serum ANGPTL4 levels in patients with AF (19.59 ± 3.57 ng/mL) were significantly lower than those in controls (26.92 ± 3.66 ng/mL, P < 0.001) (Figure 1a). Among patients with AF, serum ANGPTL4 levels were significantly lower in those with recurrent AF than in those with new-onset AF (P < 0.001) (Table 1, Figure 1b).

Serum ANGPTL4 levels in patients with AF and controls. (a) Serum ANGPTL4 levels were lower in patients with AF (n = 192) than in healthy controls (n = 170). (b) Serum ANGPTL4 levels were lower in patients with recurrent AF (n = 69) than in those with new-onset AF (n = 123). ***P < 0.001
Correlations of serum ANGPTL4 with severity of AF
We performed Spearman’s rank correlation test to determine the associations of ANGPTL4 with clinical and biochemical variables in patients with AF. Serum ANGPTL4 levels were significantly negatively correlated with the CHA2DS2-VASC score, LAD, and BNP, MDA, hs-CRP and IL-6 levels, and positively correlated with serum SOD and PPARγ levels (all P < 0.05, Table 2).
Correlations of serum angiopoietin-like 4 levels with clinical characteristics and biomarker levels in patients with AF.
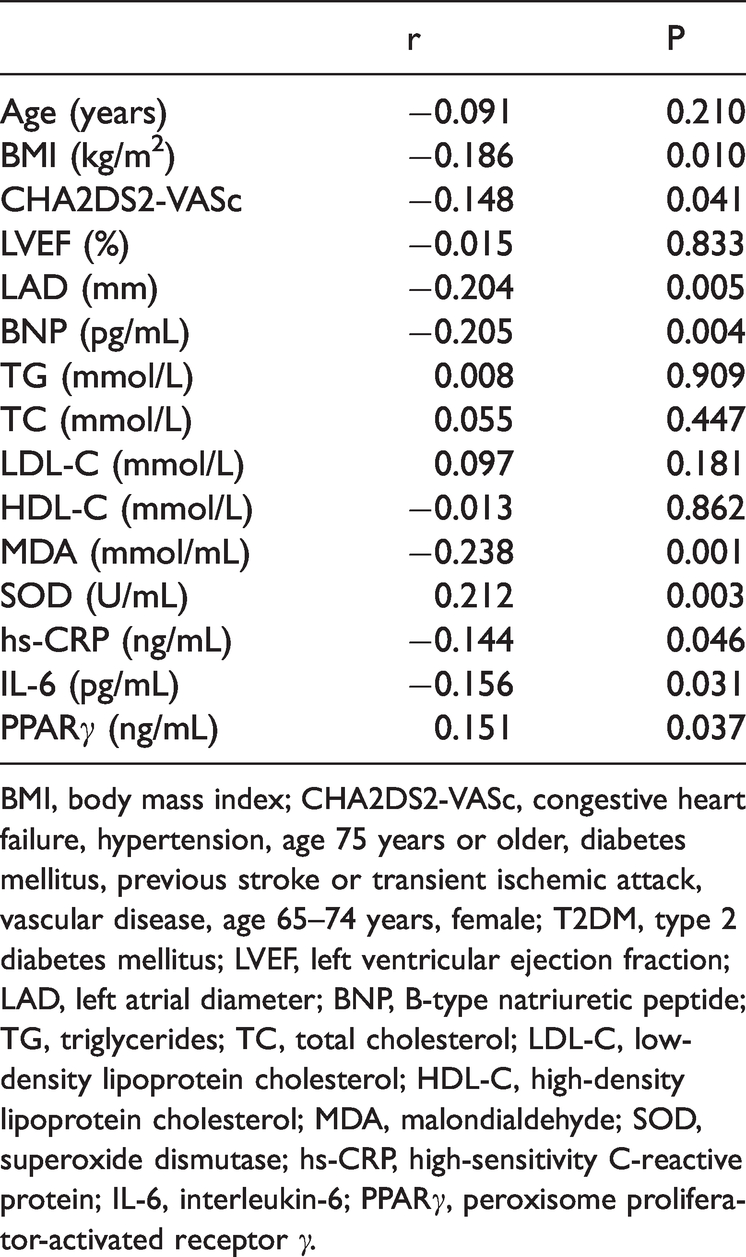
BMI, body mass index; CHA2DS2-VASc, congestive heart failure, hypertension, age 75 years or older, diabetes mellitus, previous stroke or transient ischemic attack, vascular disease, age 65–74 years, female; T2DM, type 2 diabetes mellitus; LVEF, left ventricular ejection fraction; LAD, left atrial diameter; BNP, B‐type natriuretic peptide; TG, triglycerides; TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; MDA, malondialdehyde; SOD, superoxide dismutase; hs-CRP, high-sensitivity C-reactive protein; IL-6, interleukin-6; PPARγ, peroxisome proliferator-activated receptor γ.
Logistic multivariate regression for new-onset and recurrent AF.

LAD, left atrial diameter; BNP, B‐type natriuretic peptide; MDA, malondialdehyde; IL-6, interleukin-6; PPARγ, peroxisome proliferator-activated receptor γ; ANGPTL4, angiopoietin-like 4.
Serum ANGPTL4 predicts recurrence of AF within 3 months
To differentiate between new-onset and recurrent AF, the receiver operating characteristic curve showed that the cut-off point of serum ANGPTL4 levels was 19.735 ng/mL (sensitivity = 63.9%, specificity = 74.5%; area under the curve = 0.708, P < 0.001) (Figure 2). Multivariate logistic regression analysis showed that various variables, including serum ANGPTL4 (odds ratio, 2.241; 95% confidence interval, 1.081–4.648; P = 0.03) and BNP (odds ratio, 1.046; 95% confidence interval, 1.010–1.084; P = 0.012) levels were independent contributors to recurrence of AF and new-onset AF. Additionally, lower serum ANGPTL4 levels (<19.735 ng/mL) had a significantly higher risk of recurrence (P < 0.001).

Receiver operating characteristic curve for calculating the area under the curve and the optimal cut-off point for serum ANGPTL4 levels for distinguishing between new-onset and recurrent AF. The optimal cut-off point was 19.735 ng/mL. The AUC was 0.708.
Discussion
This study investigated serum ANGPTL4 levels in patients with AF. We found the following main findings. Patients with AF had significantly lower serum ANGPTL4 levels compared with age- and sex-matched healthy controls. Additionally, serum ANGPTL4 levels were significantly lower in patients with recurrent AF compared with those with new-onset AF. Serum ANGPTL4 levels were negatively correlated with the CHA2DS2-VASC score, LAD, and levels of BNP, MDA, hs-CRP and IL-6, and positively correlated with serum SOD and PPARγ levels in patients with AF. Patients with AF who had lower serum ANGPTL4 levels (<19.735 ng/mL) had a higher risk of recurrence of AF. Therefore, ANGPTL4 may be a good serum biomarker for patients with AF high a risk of recurrence.
The study showed a correlation between serum ANGPTL4 levels and the risk of recurrence of AF. ANGPTL4 is a lipoprotein lipase inhibitor, which reduces hydrolysis of plasma triglycerides to free fatty acids. Enzyme activity of ANGPTL4 is also regulated by negative feedback of fatty acids, thus maintaining a steady state.
11
This indicates that ANGPTL4 expression is increased by fatty acids, especially by polyunsaturated fatty acids in liver cancer cells and in cardiomyocytes.6,7 Patients with AF show disorder of fatty acid metabolism and a change in atrial fatty acid transport, which is thought to be related to an increased risk of AF attack after a heart operation.
12
Additionally, patients with AF show increased saturated fatty acids and decreased polyunsaturated fatty acids in plasma, suggesting that free fatty acids may be involved in development and progression of AF.
13
A series of
We found an association of ANGPTL4 with cardiac hypertrophy, as shown by negative correlations of serum ANGPTL4 levels with LAD and BNP levels, which are three important indicators of cardiac hypertrophy. Serum BNP levels are increased in patients with AF and increased atrial pressure, 19 and they are regarded as an independent predictor of recurrence of AF. 20 Our study supports the above-mentioned results because we found markedly higher serum BNP levels in recurrent AF compared with new-onset AF. Multivariate analysis also showed that serum BNP levels were an independent predictor for recurrence of AF. This finding supports the hypothesis that hypertrophy involves progression and recurrence of AF.21,22 A previous study showed that ANGPTL4 had an inhibitory effect on cardiomyocyte hypertrophy. 9 Whether ANGPTL4 modulates cardiomyocyte hypertrophy in AF is unclear and requires further investigation.
Our study showed that serum PPARγ levels were lower in recurrent AF compared with new-onset AF, and they were positively correlated with ANGPTL4. Transcription and expression of ANGPTL4 are directly regulated by peroxisome proliferator-activated receptors (PPARs), such as PPARα, PPARβ/δ, and PPARγ. 23 PPARα and PPARγ are related to AF. Elderly patients with AF show an inflammatory status and atrial remodeling, and lower serum PPARγ protein levels. 24 In a rat model, the PPARγ activator pioglitazone prevents age-related AF susceptibility by increasing antioxidant capacity and reducing apoptosis. 25 PPARγ activators also inhibit cardiac fibrosis caused by diabetes, hypertension, and AF. 26 Atrial PPARα tissue expression is reduced in patients with AF and is associated with atrial metabolic remodeling. 27 ANGPTL4 also inhibits cardiac hypertrophy by enhancing PPARα expression. 9 Our study indicates that PPARγ is an independent predictor for recurrence of AF. Whether PPARα and PPARγ modulate ANGPTL4 and related mechanisms in AF are unknown.
Limitations of this study are as follows. First, the source of serum ANGPTL4 in patients with AF is still unclear. Whether ANGPTL4 is derived from cardiomyocytes requires further study, and this should be verified in excised myocardial tissue of patients with AF and valvular heart disease. Second, in this study, serum ANGPTL4 levels were measured only at the beginning of admission, and detailed time series of serum ANGPTL4 levels after admission and recurrence were lacking. Third, an animal model is required to determine the possible mechanisms of ANGPTL4 in the pathophysiology of AF. Fourth, ANGPTL4 expression can be affected by many factors, such as PPARγ. Therefore, whether serum ANGPTL4 can be upregulated by PPARγ activators should be investigated in clinical studies and animal models.
In conclusion, serum ANGPTL4 levels are decreased in patients with recurrent AF, and are associated with cardiac hypertrophy, oxidative stress, inflammation, and serum PPARγ levels. Serum ANGPTL4 levels are an independent predictor for recurrence of AF, and might act as a biomarker for predicting recurrence of arrhythmia post‐CA.
Footnotes
Availability of data and materials
The raw data used and analyzed for the current study are available from the corresponding author on reasonable request.
Author contributions
Zhongping Ning conceived the idea of the study, designed and supervised the study, and wrote the manuscript. Xinming Li designed the study and wrote the manuscript. Xi Zhu collected clinical data and performed statistical analysis. Jun Luo and Yingbiao Wu performed the experiments. All authors have read and approved the manuscript.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by (1) the Clinical Plateau Discipline of Pudong New Area Health Committee (Grant No: PWYgy2018-03); (2) the Key Specialty Construction Project of Pudong New District Health Committee (Grant No: PWZzk2017-19); and (3) the General Project of Science and Technology Committee of Pudong New Area (Grant No: PKJ2019-Y40).
