Abstract
Objective
To evaluate the effect of Zhigancao decoction on reversal of right atrial myocardial fibrosis after rapid atrial pacing (RAP)-induced atrial fibrillation (AF).
Methods
New Zealand white rabbits were randomly divided into four groups: sham operation group (Group A: implanted electrodes, no RAP), pacing group (Group B: RAP-induced AF), Zhigancao soup water decoction Yin group (Group C: RAP-induced AF followed by Zhigancao soup Yin prescription twice a day for 30 days), and Zhigancao soup group (Group D: RAP-induced AF followed by Zhigancao water decoction twice a day for 30 days). The atrial myocardium was then examined for myocardial fibrosis by Masson staining, and protein expression of matrix metalloproteinase-9 (MMP-9) was immunohistochemically assessed. The right atrial appendage tissue field action potential duration (fAPD) was measured by microelectrode arrays.
Results
RAP successfully induced AF. Myocardial fibrosis was more severe in Groups B and C and less severe in Group D. Protein expression of MMP-9 was strongly positive in Groups B and C and weakly positive in Group D. The fAPD was significantly decreased in Groups B and C, but the decrease in Group D was not significant.
Conclusion
Zhigancao decoction can reverse AF-induced myocardial fibrosis in rabbits and shorten the fAPD.
Keywords
Introduction
Atrial fibrillation (AF) is one of the most common arrhythmias. The occurrence rate of AF in the general population is about 0.4% to 1.0%,1,2 and this arrhythmia is associated with a high probability of disability and death. The combination of atrial electric and structural remodeling causes the occurrence and maintenance of AF. As an important representation of atrial structural remodeling during AF, atrial fibrosis can isolate the myocardial cells. Myocardial interstitial fibrosis can cause delayed conduction of intra-atrial electrical activity and abnormal pathway conduction, resulting in increased anisotropy of intra-atrial electrical activity conduction and facilitating the formation of re-entrance. Therefore, atrial fibrosis is considered to play a crucial role in the occurrence and maintenance of AF.
The present study focused on the mechanism of Zhigancao soup in preventing and treating myocardial fibrosis in an effort to provide a theoretical basis and scientific foundation for clinical treatment and promote the use of traditional Chinese medicine in the management of myocardial fibrosis.
Materials and methods
General materials
Thirty-two adult New Zealand rabbits (22 male, 10 female; body weight, 2.5–3.0 kg) were provided by the Animal Experiment Center of Xinjiang Medical University. The quality of the animals was of the first class standard (License No. SCXK(Xin)2003-001). The rabbits were randomly divided into four groups of eight animals each: the sham operation group (Group A), in which pacing electrodes were placed without performing any rapid atrial pacing (RAP); the pacing group (Group B), in which short-term RAP was performed in the right atrium for 12 h to induce AF; the Zhigancao soup water decoction Yin group (Group C), in which short-term RAP was performed in the right atrium for 12 h to induce AF and a Zhigancao soup Yin prescription was then perfused into the stomach twice a day for 30 days; and the Zhigancao soup group (Group D), in which short-term RAP was performed in the right atrium for 12 h to induce AF and Zhigancao water decoction was perfused into the stomach twice a day for 30 days.
All studies were performed in accordance with the guidelines specified by the Animal Experimentation and Ethics Committee of Xinjiang University. The present study conforms to the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health (Publication No. 85-23, revised 1996).
Main drugs
Preparation of Zhigancao soup water decoction
The following ingredients were mixed and immersed in 1200 mL of cold water (according to 6 mL of water per gram of crude drug) for 30 minutes: Zhigancao 20 g, ginger 15 g, Rehmannia 80 g, cassia twig 15 g, donkey-hide gelatin (melted by heating) 15 g, lilyturf root 10 g, edestan 10 g, and 9 dates. The mixture was boiled and then stewed with a mild fire for 40 minutes. The liquid was then filtered while hot to drop completely naturally. Next, 800 mL of water (according to 4 mL of water for per gram of crude medicine) was added for secondary stewing. After the mixture was boiled, it was stewed with a mild fire for 40 minutes. The liquid was then filtered while hot to drop completely naturally. Next, the second decocting raw material was added to 800 mL of water and concentrated in a water bath at 95ºC to 200 mL (concentration of 1 g/mL). The rabbits were orally administered the decoction by stomach perfusion using the “General Method” scheme of Zhigancao soup. The amount perfused was based on the body weight (1 mL of Zhigancao soup water decoction per 100 g of body weight), and the dosage including the crude herbs was 20 g/kg per day; this is six times the regular dose for adults. The decoction was administered twice a day for 30 days.
Preparation of Yin Zhigancao soup
Yin Zhigancao soup comprised ginger 15 g, Rehmannia 80 g, cassia twig 15 g, donkey-hide gelatin (melted by heating) 15 g, lilyturf root 10 g, edestan 10 g, and 9 Chinese dates. The preparation method was the same as that of the Zhigancao soup water decoction.
Preparation of RAP model
For anesthetic induction, 3% pentobarbital sodium (30 mg/kg) was injected into the veins along the edges of the rabbits’ ears. The rabbits were fixed on a platform with an endotracheal tube inserted into the trachea and ventilator-aided breathing. The right side of the neck was incised to separate the right jugular vein, which was then ligated. All positioning was performed under the direction of B-mode ultrasonography. An electrical physiological apparatus (LEAD-2007; Sichuan Jinjiang Electronics, Sichuan, China) was applied to conduct RAP for 24 h with a pacing frequency of 600 times/min, pulse width of 0.5 ms, and strength of 2 V using a continuous single stimulation model. Rabbits in the sham operation group underwent placement of the pacing electrode without RAP.
Masson staining
Masson staining was performed as follows. Regular dewaxing was followed by application of a Masson compound staining solution for 5 minutes, slight washing with 0.2% acetic acid solution, application of 5% phosphatotungstic acid, immersed washing with 0.2% acetic acid solution twice, washing with acetic acid water twice, dewaxing with absolute ethyl alcohol, transparency treatment with dimethylbenzene, and sealing treatment with neutral gum. The results of Masson staining showed either green or blue collagen fibrils, either gray-black or gray-blue cell nuclei, and red myocardial fibers and red blood cell cytoplasm. Photographs were taken under the microscope.
Immunohistochemical detection of matrix metalloproteinase 9 (MMP-9)
The right auricular tissue was embedded in ceresin wax after being fixed. The coronal surface was cut into pieces with a height of 4 µm. Next, the specimen was dewaxed, and high-pressure heat-induced antigen retrieval was performed. The streptavidin–alkaline phosphatase immunohistochemical staining method was applied for MMP-9 staining. Negative comparison was made with phosphate-buffered saline to substitute for the primary antibody. MMP-9 positivity was indicated by yellow staining of the ground substance. The degree of staining was classified as follows: negative, colorless; positive, slightly brown-yellow; and strongly positive, brown. The positive cells had a clear cellular structure, good positioning and qualitative determination of positive particles, and significantly higher coloring than that of the background. The primary antibody was rabbit anti-rat MMP-9 multiple antibody (Wuhan Boster Biological Engineering Co., Ltd., Wuhan, China). The streptavidin–alkaline phosphatase immunohistochemical detection kit was obtained from Beijing ZSGB Biotechnology Co., Ltd. (Beijing, China).
Recording of right auricular field potential with microelectrode arrays
The specimen was fixed in the microelectrode array recording dish, the temperature of which was controlled at 37°C using a carbon dioxide temperature control apparatus. Tyrode’s salt solution was perfused into the atrial tissue at a constant speed (3 mL/minute) with mixed gas comprising 95% oxygen + 5% carbon dioxide. The stimulus generator sent out pulses to drive the stimulation at twice the threshold with a wave width of 2 ms. After the specimen had remained steady for 30 minutes, the MEA device began to record the field action potential duration (fAPD) of the right auricular tissue (Figure 1). The software MC_Rack (Multi Channel Systems, Reutlingen, Germany) was used to analyze the fAPD.
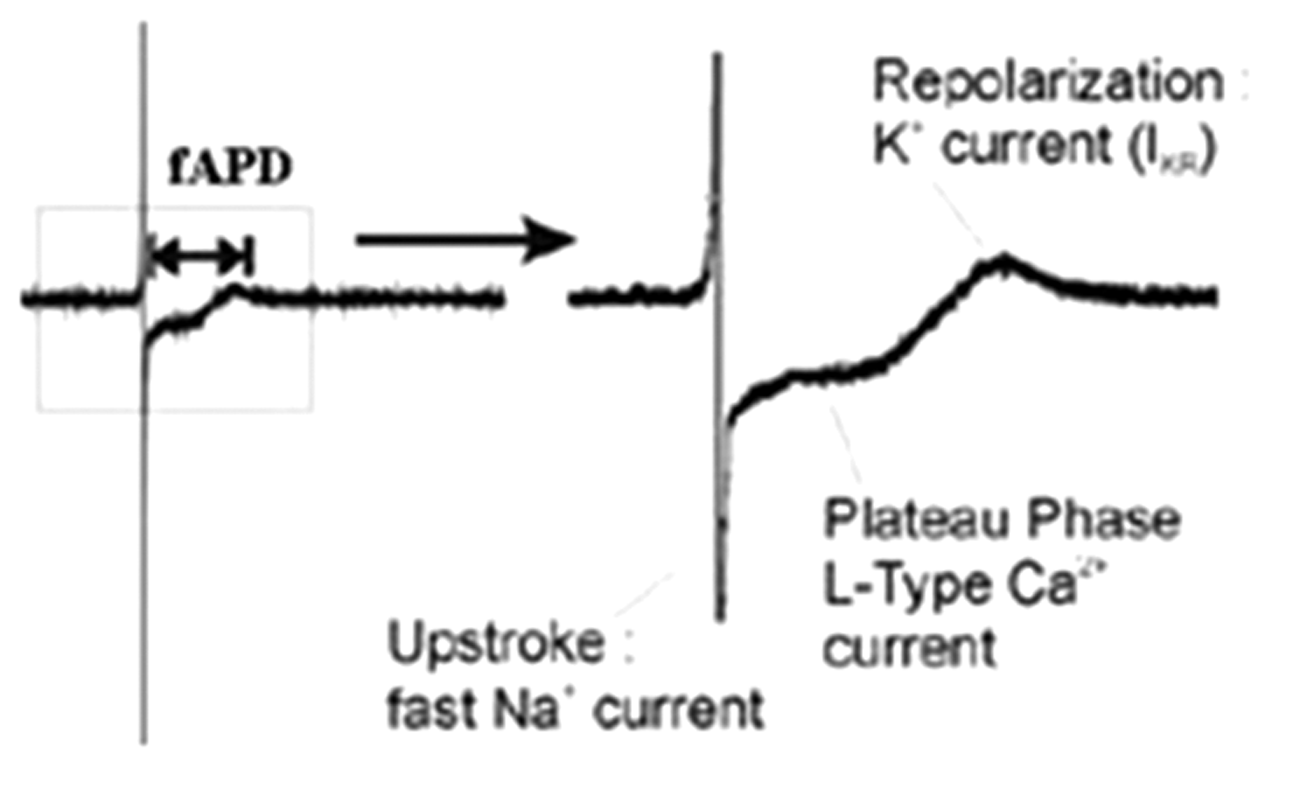
Schematic diagram of field action potential duration and ion current figure with microelectrode array.
Statistical analysis
The software SPSS for Windows, Version 16.0 (SPSS Inc., Chicago, IL, USA) was used to conduct the statistical analysis. Measurement data are presented as mean ± standard deviation. Comparisons among several groups were performed by one-way analysis of variance, and comparisons between two groups were performed by the least significant difference method. Differences were considered to be statistically significant at a P value of <0.05.
Results
Changes in intracardiac electrophysiologic mapping and surface electrophysiological parameters after RAP of the right atrium
At 12 h after RAP of the right atrium, disorderly electrical activities appeared on the surface electrophysiological map of the atrium. Specifically, the p-wave disappeared, and instead, an f-wave appeared with uneven separation and forms and a frequency of 450 to 600 times/minute. The R-R interval was unequal and lasted for >10 s. These findings indicated that the AF models were successfully established in the rabbits (Figures 2 and 3).
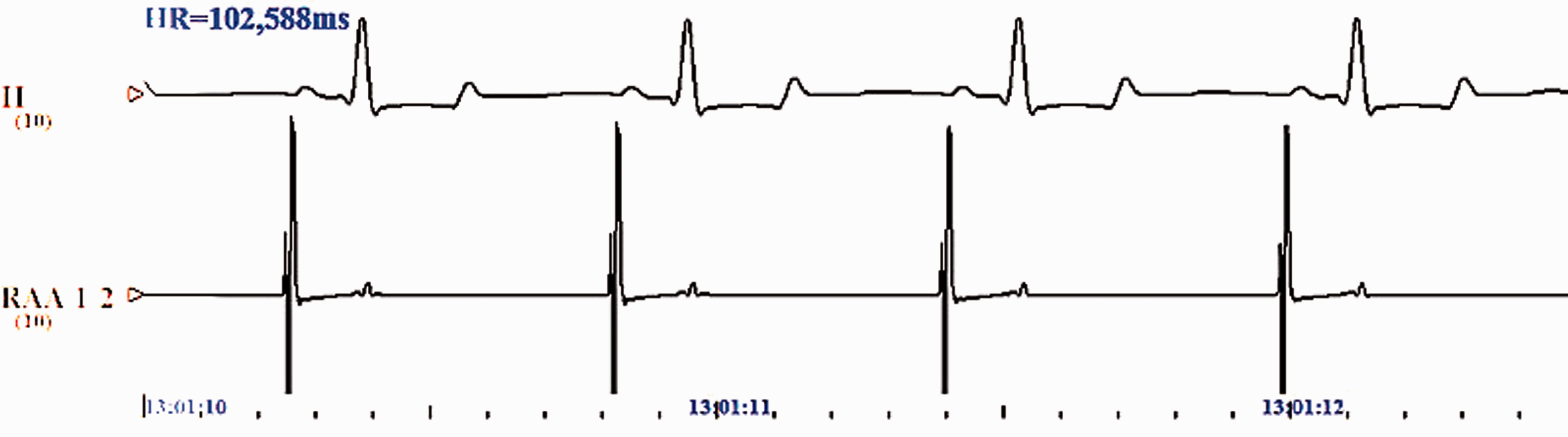
Endocardial electrophysiology and electrocardiogram of the right atrium of normal rabbits. Lead II of the electrocardiogram of rabbits in the sham-operated group shows regular P waves and QRS waves. The a-wave (atrial wave) and v-wave (ventricular wave) are found in the high right atrium.

Endocardial electrophysiology and electrocardiogram of the right atrium of rabbits that have undergone rapid atrial pacing. Lead II of the electrocardiogram of rabbits in the rapid atrial pacing group shows P waves with varying intervals. Rapid pacing of the high right atrium occurs with an S1S1 interval of 100 ms and voltage of 4 V.
Masson staining
In Group A, a small quantity of collagen fibrils were stained blue with Masson stain, the myocardial fibrils were stained red, and the collagen fibrils formed a connective network. In Groups B and C, a large quantity of collagen fibrils were stained blue-green. The network was distributed among the red myocardial fibrils. The distribution of collagen fibrils was disorderly, and myocardial bundles were winded and separated by many collagen fibrils (Figures 4(b) and 5(c)). In the Group D, the myocardial fibrils were stained red and the collagen fibrils formed a connective network (Figure 4(d)).

Masson staining of right atrial appendage tissue in the four groups of rabbits (×400). (a) Group A. (b) Group B. (c) Group C. (d) Group D.
Protein expression of MMP-9
In Group A, protein expression of MMP-9 was indicated by pale yellow or brown (weak positive) staining and showed a patchy distribution in the myocardium (Figure 5(a)). In Groups B and C, protein expression of MMP-9 was indicated by deep brown (strong positive) staining in the myocardial cytoplasm (Figure 5(b) and (c)). In Group D, protein expression of MMP-9 was indicated by brown (weak positive) staining (Figure 5(d)).

Protein expression of right atrial appendage tissue in four groups of rabbits (×400). (a) Group A. (b) Group B. (c) Group C. (d) Group D.
Comparison of fAPD
The fAPD in Group B (162.48 ± 0.88 ms) and Group C (163.04 ± 1.18 ms) was shorter than that in Group A (174.30 ± 1.36 ms), and the difference was statistically significant (P < 0.05). In Group D, the mild decrease in fAPD (172.67 ± 0.87 ms) was not statistically significant. Compared with Group A, the negative peak in Groups B and C increased remarkably and the plateau period extended with back elevation. Group D exhibited no increase in the negative peak or extension of the plateau period with back elevation (Figure 6).
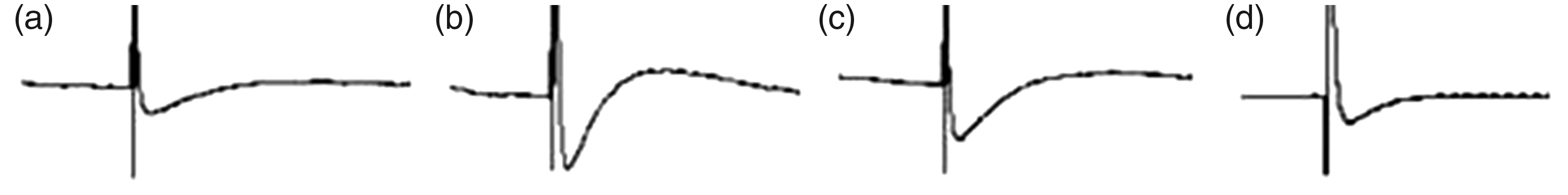
Graphic changes in the field action potential duration in the four groups of rabbits. (a) Group A. (b) Group B. (c) Group C. (d) Group D.
Discussion
At present, research on the mechanism of AF in China is mainly centered on abnormal electrophysiology. Most cases of AF are paroxysmal in the early stage; with time, the AF becomes continuous and chronic. Atrial structural remodeling is the most prominent change in patients with AF and may prohibit the ability to reverse the AF. However, no effective measures to stop the transformation of AF from paroxysmal to chronic have yet been established. The 2006 guidelines of the American Heart Association/American College of Cardiology/European Society of Cardiology (AHA/ACC/ESC) 3 clearly state that treatment with an angiotensin-converting enzyme and angiotensin receptor antagonist can reduce the pressure within the atria and tension of the atrial walls, thus helping to prevent atrial remodeling and associated arrhythmias. Myocardial fibrosis is mainly characterized by increasing deposition of collagen in the interstitial substances, an imbalance in the original proportions of collagens, and a disorderly arrangement of collagen fibers.4,5 The 2006 guidelines of the AHA/ACC/ESC reflect the important roles of substrate changes, including atrial fibrosis, on one side of the heart. The present study builds on the concept of RAP by inducing AF in rabbits and then administering Zhigancao soup to treat AF-induced myocardial fibrosis. This study showed that the degree of myocardial fibrosis in the right auricula of rabbits in the RAP group worsened, the degree of myocardial fibrosis in the Zhigancao Yin group remained unchanged, and the degree of myocardial fibrosis in the Zhigancao soup improved.
Studies outside of China have shown that MMPs may cause myocardial fibrosis and that the level of MMPs is an important index reflecting the severity of myocardial fibrosis, which in turn is closely associated with the occurrence of multiple types of cardiovascular disease.6,7 As the MMP-9 level gradually increases, myocardial interstitial fibrosis also gradually worsens, and this phenomenon may be an important factor in the evolution of AF. Structural remodeling plays a key role in the development of AF. Nakano et al. 8 evaluated both the expression and activity of MMP-9 in the atrial tissues of patients with AF. They found that the MMP-9 activity was positively correlated with the left atrial diameter, suggesting that MMP-9 is associated with the remodeling of atrial structures and expansion of atrial tissue in the course of AF. Hoit et al. 9 induced atrial dysfunction by RAP in dogs and found that compared with the normal control group, the number of myocardial calcium pumps in the left atrium did not change but that the activity of MMP-9 increased by 50%. In the present study, we used the protein expression of MMP-9 as an index with which to measure myocardial fibrosis. The results showed strong positive protein expression of MMP-9 in the right auricula of rabbits in the RAP group and Zhigancao soup Yin group but weak positive expression in the Zhigancao soup group. These findings suggest that MMP-9 participates in the evolution of AF-induced structural remodeling, which uses myocardial fibrosis as the physiological matrix. After intervention with the Zhigancao soup, the MMP-9 expression markedly decreased, the degree of fibrosis lessened, and the AF-induced structural remodeling reversed.
Another study outside of China showed that in an animal model of chronic atrial fibrosis, the worsening of atrial fibrosis was associated with an increase in induced AF. 10 When the atrial fibrosis worsened, the structure between myocardial cells and electrical activities changed accordingly, resulting in increased divergence of the refractory period of the atria or abnormal electrical activity of local myocardial cells, slowing conduction and promoting the occurrence and maintenance of AF. Because of the close coupling of cells/tissues and electrodes at detection, MEA can sensitively detect the influence of drugs on the electrophysiological signals. Moreover, the sensor has stability and can analyze the effect of drugs according to the changes in electrophysiological functions. Some studies have suggested that atrial fibrosis can also cause changes in the capacitance of myocardial cellular membranes and the density of ion channels, leading to remodeling of the ion channels and facilitating electrical remodeling of the atrium.11,12 The present study applied MEA technology to study electrical activity disorders and ion current changes produced by AF-induced myocardial fibrosis in rabbits after treatment with Zhigancao soup. The results showed that in the pacing group, the fAPD of the right auricula decreased and electrical remodeling took place. Secondary to the clear increase in negative waves, the plateau period decreased with back elevation, suggesting that both sodium and calcium participate in the occurrence of electrical remodeling and that Zhigancao reduces the occurrence of myocardial fibrosis and thereby influences the development and maintenance of electrical remodeling. In experimental studies, the preventive effect of traditional Chinese medicine against myocardial fibrosis is continuing to develop. For example, research is being performed on active substances of cardiovascular vessels and calcium ions, making it possible to further elucidate the mechanism of how traditional Chinese medicine resists myocardial fibrosis at a deeper level. In the past, research on the preventive effects of Zhigancao against arrhythmia mainly centered on the influence of this drug on automatic rhythmicity, excitableness, the refractory period, triggered activities, and other factors. The present study further reveals the influence of Zhigancao soup on reversing AF-induced structural remodeling in rabbits from the viewpoint of resisting myocardial fibrosis.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Footnotes
Funding
This work was supported by The National Natural Science Fund (grant 81560065).
