Abstract
We herein report two cases of paradoxical carbon dioxide (CO2) embolism during laparoscopic nephrectomy and hepatic left lateral lobectomy without evidence of a right-to-left shunt or obvious rupture of blood vessels. Transesophageal echocardiography detected paradoxical CO2 embolism before the end-tidal CO2 partial pressure (PETCO2) dropped from baseline. The pneumoperitoneum was reduced or stopped immediately after detection of the embolism. One patient developed a postoperative epileptiform seizure. In the other patient, many gas bubbles were drawn out from the central venous line. We speculate that rapid introduction of pneumoperitoneum pushed a large amount of CO2 into the abdominal blood vessels, exceeding the gas exchange capacity of the lung and causing CO2 bubble formation in the left-side cardiac system. These two cases indicate that intraoperative transesophageal echocardiography can reduce the influence of CO2 embolism during laparoscopic tumor surgery by early diagnosis of the embolism and provide helpful information to establish a list of differential diagnoses of postoperative complications.
Keywords
Introduction
Laparoscopic surgery is performed worldwide. Research has shown that in laparoscopic nephrectomy, gas bubbles may enter the right atrium (RA) from the inferior vena cava (IVC) during renal vein dissection as shown by transesophageal echocardiography (TEE). 1 In a report of laparoscopic cholecystectomy, carbon dioxide (CO2) embolism was observed by TEE in 69% of patients. 2 Moreover, in a study of laparoscopic hepatectomy, intraoperative TEE consistently identified gas bubbles in the RA, right ventricle (RV), and right ventricular outflow tract. 3 The surgical site, pneumoperitoneal pressure, respiratory parameters, and presence of abdominal adhesion were found to affect the occurrence of gas embolism. Furthermore, paradoxical CO2 embolism in the systemic circulation has been reported during laparoscopic liver resection, 4 laparoscopic cholecystectomy, 5 gynecological laparoscopy, 6 and laparoscopic nephrectomy. 7 We herein present two cases of paradoxical CO2 embolism during laparoscopic radical nephrectomy for renal cell carcinoma (Case 1) and laparoscopic hepatic left lateral lobectomy, hepatic radiofrequency ablation, and resection of a sigmoid tumor (Case 2). TEE detected CO2 bubbles before the end-tidal CO2 partial pressure (PETCO2) dropped. Neither patient showed evidence of an intracardiac right-to-left shunt.
Case report
Case 1. The first patient was a 55-year-old woman with a height of 146 cm and weight of 42 kg. She was diagnosed with a right renal mass of about 63 × 58 × 62 mm in the lower pole of the right kidney. Laparoscopic right nephrectomy under general anesthesia was planned. Her preoperative electrocardiogram, chest X-ray, and laboratory parameters were within the normal range.
In the operating room, the patient’s blood pressure was 170/100 mmHg, heart rate was 80 beats/minute, and pulse oxygen saturation (SpO2) was 98%. The left internal jugular vein and left radial artery were accessed for monitoring. Standard induction was performed. The ventilation mode was volume-controlled ventilation with a tidal volume of 8 mL/kg, respiratory rate of 12 breaths/minute, and inspired oxygen fraction of 50% with no positive end-expiratory pressure. The patient was then placed in the left lateral recumbent position. Her PETCO2 before the introduction of CO2 pneumoperitoneum was 37 mmHg, and her SpO2 was 100%.
TEE (Vivid E95; GE Healthcare, Chicago, IL, USA) examination revealed no abnormality. Trocars were placed in the abdominal cavity at three sites (the navel, the subcostal margin at the midline of the clavicle, and the intersection of the anterior axillary line and level of the navel). The CO2 pneumoperitoneum was established at a pressure of 15 mmHg. Ten minutes after the introduction of CO2 pneumoperitoneum, while the surgeons were dissecting the perirenal fat without significant bleeding, TEE showed a sudden increase in small gas bubbles in the RA and small bubbles in the RV. The patient was immediately placed in the Trendelenburg position. About 8 minutes later, TEE showed a large amount of gas bubbles in the left atrium, left ventricle, ascending aorta, and pulmonary artery (Figure 1). At the same time, the PETCO2 began to fall to 20 mmHg from 37 mmHg, and this decrease lasted for 20 minutes. There were no changes in the patient’s pulse, blood pressure, SpO2, or electrocardiogram parameters.

Intraoperative transesophageal echocardiography (TEE) showed gas bubbles in the heart. (a) In the midesophageal aortic valve short-axis view, TEE showed a large amount of gas bubbles in the right atrium, right ventricle, left atrium, left ventricle, and aortic valve. (b) In the transgastric basal short-axis view, intraoperative TEE showed a large amount of gas bubbles in the left ventricle. (c) In the midesophageal ascending aortic short-axis view, TEE showed many gas bubbles in the ascending aorta and pulmonary artery.
We quickly increased the fraction of inspired oxygen to 100%. Arterial blood gas analysis revealed a pH of 7.27, arterial oxygen pressure of 161 mmHg, arterial CO2 pressure of 65 mmHg, and lactic acid concentration of 2.4 mmol/L. After communication with the surgeon, the pneumoperitoneum pressure was reduced to 10 mmHg. Meanwhile, gas bubbles could still be seen in the whole heart, the hepatic vein, and the IVC (Figure 2). There was no regional wall motion abnormality, and the RV was normal. Color ultrasonography showed no defect in the atrial septum or ventricular septum and no patent foramen ovale (PFO).

Intraoperative transesophageal echocardiography showed a large number of gas bubbles in the hepatic vein and a small number of bubbles in the inferior vena cava.
Nine hours after the operation, the patient exhibited slurred speech and developed an epileptiform seizure in the ward. After the seizure, the muscle strength of both limbs was weakened. The blood myoglobin and lactic acid concentrations were increased. However, a computed tomography scan showed no obvious infarction or hemorrhage. Postoperative transthoracic echocardiography was performed by an independent ultrasound physician, who confirmed that there was no defect in the atrial septum or ventricular septum and no PFO.
Case 2. The second patient was a 47-year-old man with hepatic metastasis of colonic carcinoma who presented for laparoscopic hepatic left lateral lobectomy, hepatic radiofrequency ablation, and resection of a sigmoid tumor.
TEE was used intraoperatively to assess the presence of gas emboli in the mid-esophagus right ventricular inflow–outflow view. Forty minutes after the beginning of the surgery, a large amount of gas bubbles was found to have entered the RA and RV from the IVC (Figure 3) when the surgeon was dissecting the hepatic veins. Two minutes later, the PETCO2 began to fall from 36 to 32 mmHg. Three minutes after observation of the gas bubbles in the RA, gas bubbles were also present in the left heart and ascending aorta. The PETCO2 then fell to 17 mmHg, tachycardia occurred (74–108 beats/minute), and the SpO2 decreased (from 99% to 91%); a slight increase in the central venous pressure (from 5 to 12 mmHg) and blood pressure followed. The pneumoperitoneal pressure was 12 mmHg. The pneumoperitoneum site was urgently closed, and the patient was immediately placed in the left lateral recumbent position with the head down. Gas bubbles were aspirated from the right internal jugular vein central line using a 50-mL syringe (Figure 4). The patient recovered and was extubated after surgery in the operation room. He complained of numbness in the ring finger and little finger of the right hand the night of the operation, but he recovered normal feeling in the fingers the next morning.

Intraoperative transesophageal echocardiography showed that gas bubbles had entered the right atrium and right ventricle from the inferior vena cava.
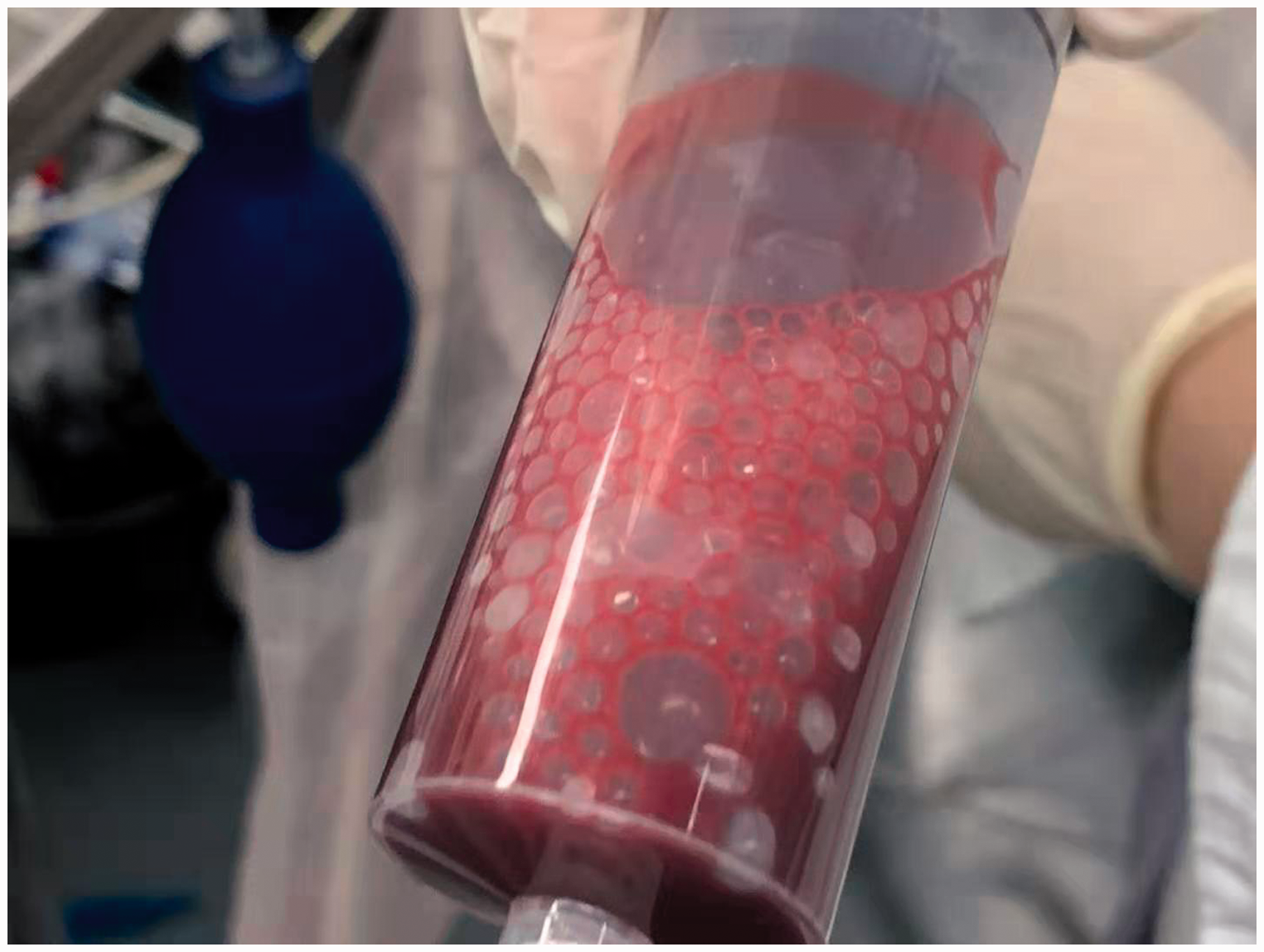
Gas bubbles were drawn out from the right internal jugular vein central line using a 50-mL injection syringe.
Discussion
Risk factors for gas embolism during laparoscopic surgery include high intra-abdominal pressure, 8 , 9 a history of previous pelvic and abdominal surgery, 10 and intraoperative venous injury. 1 , 2 , 4 , 5 , 11 Both of our patients had cancer and no history of surgery, and TEE showed a large amount of small bubbles in the RA without obvious abdominal blood vessel rupture or bleeding. Such bubbles may be absorbed from uninjured veins or ruptured venules in the abdominal wall, omentum, and mesentery under high pneumoperitoneal pressure, subsequently entering the RA through the IVC. The vasculature within tumors is structurally abnormal; it is not organized in the conventional hierarchical arrangement and is more permeable than the normal vasculature. 12 , 13 These features contribute to leaky, tortuous, and dilated blood vessels. Therefore, we speculate that tumor resection and surgeries involving a complex vascular plexus, such as hepatectomy, cholecystectomy, and nephrectomy, may also be risk factors for gas embolism during laparoscopic surgery.
A few reports have described gas embolism during laparoscopic surgeries, 3 , 4 , 11 ,14–16 but none have directly described bubbles in the hepatic vessels during non-hepatic laparoscopic surgeries as observed by TEE. In this case report, TEE showed many small bubbles in the hepatic vein in Case 1, which may indicate that the bubbles observed in the heart came from the hepatic vessels.
Cardiac structural abnormalities such as PFO, ventricular septal defect, and patent ductus arteriosus may lead to paradoxical gas embolization. However, several reports have described paradoxical CO2 embolism during laparoscopic surgery without evidence of an intracardiac right-to-left shunt. 4 , 6 , 7 In a reported case of unilateral limb hemiplegia after gynecological laparoscopy, 6 intraoperative TEE showed many bubbles in the left atrium and ventricle, but pulmonary angiography and transthoracic echocardiography with contrast agent failed to show any evidence of a right-to-left shunt. In another case report of cerebral infarction after laparoscopic hepatectomy, 4 intraoperative TEE showed numerous gas bubbles in the left atrium and ventricle after intraoperative right hepatic vein injury. However, echocardiography, electrocardiography, and 99mTc-macroaggregated albumin lung perfusion scintigraphy also showed no evidence of a right-to-left shunt. Three possible explanations proposed by the authors were bubbles entering the systemic circulation through instantaneous opening of the PFO, pulmonary vasodilation and arteriovenous communication from cirrhosis leading to an intrapulmonary shunt and contradictory gas embolism, and venous bubbles overflowing and entering the systemic circulation through the lung. 4
In our patients, intraoperative TEE showed no evidence of an atrial or ventricular septal defect or PFO, which was verified by another independent ultrasound physician postoperatively. Intraoperative TEE also showed a large number of small bubbles appearing in the left atrium near the orifice of the pulmonary vein. We speculate that the amount of CO2 from the right heart may have exceeded the gas exchange capacity of the lung and entered the left atrium through the pulmonary vein.
There are two reported mechanisms of paradoxical CO2 embolism leading to nervous system injury. One is that reduced cardiac output indirectly causes cerebral ischemia and hypoxia. 17 and the other is that CO2 directly embolizes cerebral blood vessels and causes ischemia and hypoxia of brain cells.4–7 In one of the above-mentioned reports, after large amounts of CO2 emboli were found in the cerebral vessels, magnetic resonance imaging and other examinations showed cerebral infarction. 4 Additionally, bubbles were found in the cerebral vessels during autopsy in a case of fatal CO2 embolism. 18 One report indicated that imaging examinations might show negative results after less severe CO2 embolism. 7 In that same report, CO2 might have been absorbed or a cerebral microvascular embolism might have been responsible for the symptoms of nervous system injury, 7 which is consistent with Case 1 of the present report.
The clinical manifestations of gas embolism are various. The most common manifestations of gas embolism include a sudden drop in the PETCO2, a decrease in the SpO2, deterioration of the hemodynamic status, electrocardiogram changes, or even cardiac arrest. 19 , 20 However, not all gas embolism produces obvious symptoms. 2 , 21 One study indicated that gas embolism commonly occurred during laparoscopic cholecystectomy, but few cases of cardiorespiratory instability were found. 22 Delayed wakefulness, 23 postoperative chest tightness, chest pain, or neurological abnormalities may be the initial symptoms. 4 Delayed cardiac arrest and persistent cortical blindness after the surgery have also been reported. 24
It is important to quickly identify gas embolism events. TEE can detect a small amount of bubbles in the heart cavity and blood vessels during laparoscopic surgery without other clinical signs of gas embolism. Moreover, we found that the emergence of gas bubbles in the heart as observed by TEE occurred earlier than the changes in vital signs. TEE can also monitor the outcome of gas embolism in real time. The severity of venous air embolism and paradoxical air embolism can be quantitatively graded from 0 to 3 by the microbubble score. 25 Secretain et al. 26 created a software algorithm (“Detection of Emboli using Transesophageal Echocardiography for Counting, Total volume, and Size estimation”, or DETECTS™) to identify and measure potential emboli present during cardiac surgery using two-dimensional ultrasound. The microbubble score and DETECTS™ may be useful in studies of CO2 embolism in future. McConnell’s sign has been used to diagnose pulmonary embolism for more than 20 years. However, we found no evidence of McConnell’s sign in either patient.
Once gas embolism is diagnosed, CO2 insufflation should be stopped immediately. When SpO2 begins to drop, the fraction of inspired oxygen needs to be increased. The patient may benefit from placement in the Trendelenburg position or the left lateral decubitus with head-down position, which allows the gas to enter the apex of the RV and prevents its entry into the pulmonary artery. 14 TEE may help to monitor bubble changes during adjustments in the body position. Hyperbaric oxygen therapy is reportedly effective for paradoxical gas embolization.15,27,28 The patient in Case 2 of the present report had no adverse outcomes; this might have been related to our timely management of the gas embolism, including urgent stopping of the pneumoperitoneum, adjustment of the patient’s position, and aspiration of the gas bubbles from the central line.
In view of the scarce reports about complications caused by air embolism, prevention is very important. Patients may benefit from careful trocar placement by surgeons and a low CO2 inflation pressure and speed. Reducing the pneumoperitoneum pressure to 6 to 10 mmHg during laparoscopic surgery may be useful to prevent CO2 embolism. 29 An animal study showed that under low airway pressure, the risk of pulmonary gas embolism increases when the pneumoperitoneum pressure is higher than the central venous pressure. 30
In conclusion, we have reported two cases of the detection of paradoxical CO2 embolism by TEE, which occurred earlier than the changes in the vital signs during laparoscopic surgeries without obvious rupture of blood vessels or evidence of a right-to-left shunt. TEE is helpful for quick identification of gas embolism and prevention of serious complications.
Footnotes
Authors’ contributions
WTH, JZ, and BP were involved in the care of the patient and writing of the paper. BYW and ZRS performed the chart review and literature review and collaborated on the paper writing. CHM was involved in the care of the patient and collaborated on the paper writing. JPH revised and reviewed the paper. JZ revised the manuscript and performed the final review of the paper. All authors read and approved the final manuscript.
Availability of data and materials
Data are available only upon request because of privacy or other restrictions. The data that support the findings of this study are available on request from the corresponding author Jing Zhong. The data are not publicly available because they contain information that could compromise patient privacy/consent.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of Fudan University Shanghai Cancer Center. The patient provided written informed consent to participate.
Funding
This work was sponsored by the National Natural Science Foundation of China (Nos. 81971868, 81873948, 81601712) and the Shanghai Pujiang Program (17PJD008). Neither of the funding bodies had any influence on the collection of data or writing of the manuscript.
