Abstract
An accessory cavitated uterine mass (ACUM) is a very rare obstructive genital malformation characterized by pelvic pain and severe dysmenorrhea. It is easily mistaken for other obstructive genital malformations in women, such as cystic uterine adenomyosis or cystic degeneration of uterine fibroids. This case report describes a 30-year-old patient with a huge uterine cornual mass. Successful resection was performed by surgical excision, and the lesion was diagnosed as an ACUM. Given the rarity of a giant ACUM, this report also includes a brief review of the relevant literature.
Keywords
Introduction
An accessory cavitated uterine mass (ACUM) is often located in the anterior lateral wall of the uterus below the attachment of the round ligament. 1 This cystic lesion is lined with functional endometrial tissue and is commonly seen in young childless women. It presents with progressive worsening of dysmenorrhea and is easily misdiagnosed as cystic uterine adenomyosis. In 2010, Acién et al. 2 discussed theories regarding the development of ACUM based on previous similar cases; they subsequently summarized the clinical characteristics of such patients and coined the term ACUM. This mass was historically considered a special type of cystic adenomyosis, but its pathogenesis remains unclear. The disease can usually be treated with drugs and surgery. Drug treatments, including nonsteroidal anti-inflammatory drugs and oral contraceptives, are often temporarily effective, but symptoms tend to relapse after discontinuing medication. By contrast, surgical treatment can effectively relieve dysmenorrhea symptoms and improve reproductive health outcomes. Because of the extremely rare occurrence of ACUM in clinical practice, its clinical diagnostic rate is low and treatment is often delayed. The present case report describes the diagnosis and treatment of a huge ACUM in a young woman and is being shared with the aim of providing insight into the diagnosis and treatment of ACUM.
Case report
A 30-year-old woman underwent a physical examination at Chongqing Jinyi Hospital on 8 February 2023. Ultrasound examination revealed a 2.8- × 2.2-cm cystic echogenic mass in the right ovary, considered physiological, and an 8.6- × 5.0-cm cystic echogenic mass of uncertain nature in the right adnexal region. The patient was recommended to seek further diagnosis at a higher-level hospital, but she did not seek further treatment. On 27 February 2023, the patient experienced intermittent tolerable lower abdominal pain after intercourse. The pain worsened after eating cold food and improved with rest.
On 28 February, the patient presented to Chongqing Hospital of Traditional Chinese Medicine. She was married with no children (G0P0). Her menstrual history was regular with a duration of 4 days and a cycle of 28 days. Her menstrual flow was slightly reduced, and she had significant dysmenorrhea requiring painkillers. She denied a history of surgery or trauma. Her last menstrual period had occurred on 16 February 2023, with reduced flow, dark red color, small clots, and significant dysmenorrhea. The patient had experienced menarche at 12 years of age, with only mild initial menstrual cramps. However, after turning 16 years of age, her dysmenorrhea progressively intensified, reaching a pain score of 5 to 7. The discomfort was most severe on the first day of her menstrual cycle and persisted throughout. Although she occasionally resorted to pain medication, the overall effect on her quality of life remained minimal.
Gynecological examination revealed an enlarged uterus with normal activity, medium consistency, and no tenderness; there also was no tenderness in the bilateral adnexal regions. A complete blood count, liver function tests, and the erythrocyte sedimentation rate were within normal limits, as were the concentrations of alpha fetoprotein, carcinoembryonic antigen, and human chorionic gonadotropin (<1.3 ng/mL, <0.50 ng/mL, and <1.20 mIU/mL, respectively). However, the concentration of the tumor marker CA125 was slightly elevated (65.0 U/mL), and that of CA19-9 was significantly elevated (127.12 U/mL). Endovaginal ultrasound examination revealed a right adnexal anechoic mass measuring approximately 6.4 × 4.9 × 7.4 cm. It had clear boundaries, regular edges, and poor internal echoes with dense weak echoes. Additionally, there was an area of slightly high echo on the inner wall, measuring approximately 1.5 × 0.6 cm, surrounded by thick muscle with low echoes. These findings were not clearly separated from the right side of the uterus and were not connected to the uterine cavity or the right ovary. Color flow Doppler imaging (CFDI) showed scattered punctate blood flow signals around the anechoic mass, and no blood flow signals were seen within the area of slightly high echo (Figure 1(a)–(c)). Pelvic magnetic resonance imaging (MRI) showed a mass-like shadow with abnormal signal intensity in the upper posterior part of the uterine fundus, with a maximum size of 8.3 × 5.7 cm in the largest cross-section. T1-weighted imaging showed high signal intensity, and T2-weighted imaging showed high signal intensity with internal patches of slightly low signal intensity. The lesion wall was thick, approximately 0.8 cm, with iso intensity in both T1- and T2-weighted imaging. The lesion wall was significantly enhanced on contrast imaging, and the boundary between the lesion and the uterine fundus was unclear (Figure 1(d), (e)). Based on the above-mentioned examination results, we preliminarily considered the lesion to be cystic adenomyosis, possibly with an associated uterine malformation. Because of the large size of the mass and obvious symptoms, we planned to perform laparoscopic exploration and resection. The reporting of this study conforms to the CARE guidelines.3,4
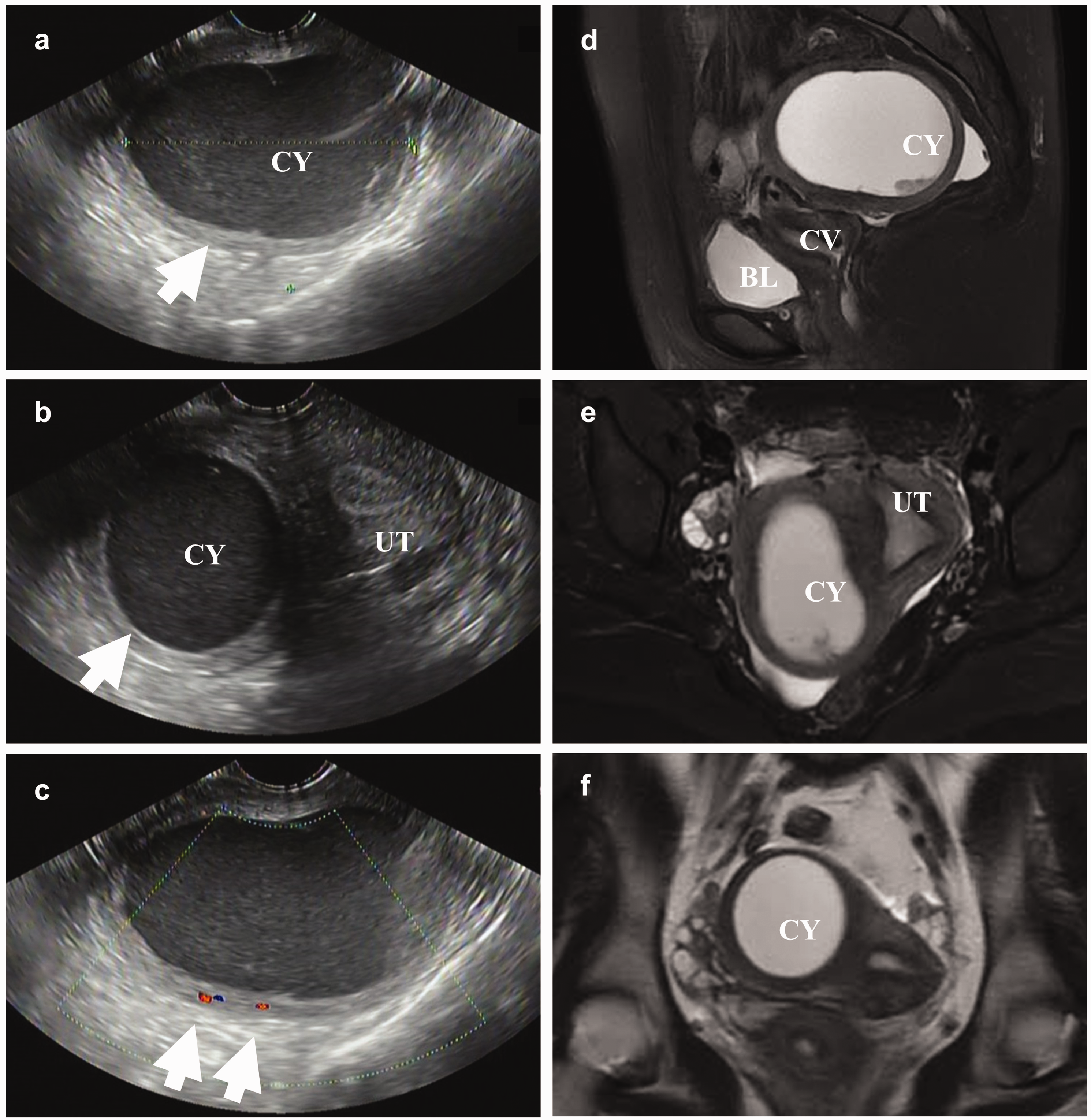
Representative pelvic ultrasound and magnetic resonance imaging scans of the 30-year-old woman in this case. (a) An echoless area with clear borders, regular morphology, and poor internal acoustic transparency was seen in the right adnexal region, exhibiting a “ground-glass” appearance. (b) The echoless area was poorly demarcated from the right wall of the uterus and had a boundary with the endometrium. (c) Color flow Doppler imaging showed dot-like blood flow signals around the echoless area, but no blood flow signals were seen inside. (d) Sagittal fat-suppression T2-weighted imaging showed that the posterior uterus had a well-defined, regularly shaped lesion with high signal intensity located on the right side of the fundus. (e) Transverse fat-suppression T2-weighted imaging showed a lesion located on the right side of the fundus, surrounded by muscular layers and not connected to the endometrium and (f) coronal T2-weighted imaging showed a lesion located on the right side of the fundus, surrounded by muscular layers and not connected to the endometrium. UT, uterus; BL, bladder; CV, cervix; CY, cyst.
Laparoscopic exploration and treatment were performed on 6 March 2023. During the procedure, the uterus was found to be posteriorly positioned with an irregular morphology, and a 9-cm-diameter tuberous protuberance was detected on the anterior wall of the right uterine fundus and the right uterine cornu (Figure 2(a), (b)). The posterior wall of the tumor exhibited partial lamellar adhesions to the right tubal mesentery and round ligament (Figure 2(c)). After careful separation of these adhesions, the right fallopian tube and ovary were exposed. The bilateral ovaries and fallopian tubes were normal in appearance. The posterior wall of the uterus had formed lamellar membranous adhesions to the anterior wall of the rectum and the peritoneum of the right pelvic floor. These adhesions were separated through a combination of blunt and sharp dissection techniques. An ultrasonic knife was used to make an incision in the right anterior uterine wall and right uterine cornu, where the tumor was conspicuously protruding. The mass was located within the intermuscular wall and cystic in nature, and its boundary was clear. Therefore, the tumor removal procedure was complex but proceeded smoothly. The base of the tumor was attached to the starting point of the round ligament of the right uterine horn. When the base of the tumor was manipulated, the tumor ruptured spontaneously and released approximately 300 mL of chocolate-like fluid (Figure 2(d)). The inner wall of the cyst was smooth, with no apparent endometrial-like glandular tissue or papillary projections. The cyst wall was composed of uterine muscle of uneven thickness. The cystic tumor was eventually removed intact, and exploration of the surgical trauma site revealed no communication with the uterine cavity (Figure 2(e)). Following tumor removal, the residual cavity was irrigated with saline, and any active bleeding points were electrocoagulated to achieve hemostasis. A 1/0 micro-bridging suture was used to close the base of the tumor and the tumor fossa. Additionally, a continuous 0/1 barbed suture was used to close the myometrium and the serosal membrane layer, ensuring complete closure of the tumor cavity and restoration of normal uterine morphology.

Intraoperative findings. (a)–(c) During the surgery, a mass of approximately 9 cm in diameter was visible in the anterior wall of the right uterine fundus and the right uterine horn. (d) The tumor spontaneously ruptured during the operation, releasing approximately 300 mL of chocolate-like fluid and (e) the clinical condition after surgical resection. The arrow indicates the surgical wound left after removing the lesion. M, mass; UT, uterus; OV, ovary; OD, oviduct; RL, round ligament.
The surgical specimen was sent for pathological examination. The cyst wall tissue weighed 80 g and was tough and unevenly thick. The cyst wall morphology was regular, the inner wall was smooth, and chocolate-like liquid was visible inside the cyst. No endometrial tissue was found on the inner wall. Pathological examination showed that the cyst wall was covered with columnar epithelium comprising neatly arranged epithelial cells with no atypia. A few oval or short spindle-shaped endometrial stromal cells were visible around the focal cyst wall. The glandular epithelium surrounding the cyst wall was composed of smooth muscle tissue, with well-differentiated smooth muscle cells and no prominent atypia (Figure 3). The final diagnosis of ACUM was derived from a combination of the clinical features, anatomical site, and pathologic features. The patient recovered well after surgery. Her dysmenorrhea symptoms were significantly relieved without any medication for menstrual suppression or pain management throughout the 6-month postoperative follow-up.

Histopathological examination findings. (a), (b) The wall of the capsule was lined with a large number of smooth muscle cells (black solid arrows), and the inner wall was locally lined with endometrial glands and mesenchyme (red hollow arrows). (a) Hematoxylin–eosin, ×40 and (b) Hematoxylin–eosin, ×100. SMT, smooth muscle tissue.
Discussion
ACUM is a lesion initially reported and defined by Acién et al. 2 It is a solitary cystic lesion located in the anterior lateral wall of the uterus below the attachment of the round ligament. 5 ACUM is an important cause of pelvic pain during menstruation in young women. The origin of ACUM is currently unclear. One hypothesis suggests that duplication or persistence of the Müllerian duct tissue may be associated with the development of round ligament dysfunction. 6 In such cases, the abnormal Müllerian ducts undergo normal fusion and absorption with the symmetrical Müllerian ducts on the opposite side. However, the endometrium is not absorbed, resulting in the formation of free abnormal chambers and leading to a new Müllerian duct malformation, namely ACUM. The current diagnostic criteria for ACUM are mainly a normal uterine cavity and endometrial cavity, normal ovaries and fallopian tubes, surgically resected and pathologically confirmed cavity-like masses, endometrial tissue containing glandular and stromal tissue in the uterine adnexal cavity, chocolate-like fluid in the adnexal cavity, and no adenomyosis (however, the possibility of small foci existing in the myometrium adjacent to the mass cannot be excluded).
The incidence of ACUM remains unknown. To date, more than 30 articles have reported more than 80 cases of ACUM (including diagnostic terms for juvenile cystic adenomyoma and cystic adenomyoma), with an average lesion size of approximately 3.35 cm. 7 Only one article reported a 10-cm lesion in a 37-year-old woman. 8 In the present study, we have reported an extremely rare case of a giant ACUM with a total lesion length of 9 cm, which is significantly larger than those reported in the literature. This large size may have been related to the patient’s longer clinical history (13-year history of dysmenorrhea). Most cases of ACUM occur in patients aged <30 years, especially in adolescent patients. ACUM can also occur in women aged >30 years, but it less frequently found in perimenopausal and postmenopausal women. Takeuchi et al. 9 reported an average age of 25 years among the nine patients in their study. Naftalin et al. 10 reported that 20 patients were diagnosed with ACUM at a median age of 29.2 years (interquartile range: 25.0–35.8 years). The patient in this case was 30 years old, which is consistent with previous reports. Our patient’s clinical symptoms included severe dysmenorrhea and chronic pelvic pain, and medication did not effectively relieve the pain. The pain often occurred on the same side as the lesion, which may have been due to the periodic bleeding and continuous accumulation of pressure within the enclosed space of the lesion cyst. Previous experience suggests that careful examination is required because of the generally small size of ACUM lesions. The present case suggests that even for larger lesions in patients with consistent age and clinical symptoms, clinicians should be alert to the possibility of ACUM.
Imaging is the key technique for early and precise diagnosis of ACUM.10,11 The most commonly used imaging modalities for ACUM include ultrasound and MRI. Transvaginal ultrasound is easy to perform and highly accurate, making it the imaging modality of choice for ACUM. Ultrasound can clearly show the lesion’s location, shape, and general composition. CFDI technology can be used to visualize the blood flow of the lesion, which helps suggest the nature of the lesion. MRI has the advantages of good soft tissue contrast and multi-sequence scanning imaging, allowing for a more reliable assessment of the lesion’s composition. It is currently the method of choice for comprehensive examination of female genital malformations. MRI can clearly show the location of the lesion and its relationship with the uterine cavity, which is helpful for diagnosing ACUM and choosing appropriate surgical procedures. Peyron et al. 12 reported a series of MRI features in patients with ACUM and proposed common MRI signs, including a non-communicating functional accessory horn in a normal uterus as well as round masses with a central hemorrhagic cavity and a surrounding regular ring. MRI is helpful for timely diagnosis and appropriate laparoscopic resection. In addition, hysterosalpingography can show abnormalities in the uterine cavity and bilateral fallopian tubes, which is helpful for the differential diagnosis of ACUM and other reproductive tract abnormalities. 13 However, hysterosalpingography cannot be performed in adolescents without a history of sexual activity.
Medication and surgery are the common management modalities for ACUM. 14 Commonly used medications include nonsteroidal anti-inflammatory drugs, oral contraceptives, and gonadotropin-releasing hormone agonists; however, most medications have poor efficacy, and symptoms are prone to recurrence after drug withdrawal. 15 Common methods of surgical treatment of ACUM include removal of the uterine appendage mass (laparoscopic or open surgery), hysterectomy (laparoscopic or open surgery), and ethanol sclerotherapy.16,17 Ethanol sclerotherapy can significantly alleviate the pain in most patients, but it is highly operator-dependent and may result in recurrence. Laparoscopic mass resection with uterine preservation not only rapidly alleviates pain but also prevents postoperative recurrence and does not affect the patient’s fertility, making it the preferred method for treating ACUM. After undergoing laparoscopic uterine-preserving surgery, our patient’s symptoms significantly improved, and she developed no symptoms of recurrence during the 6-month follow-up period. Elective hysterectomy or unilateral salpingectomy with mass resection is also a safe and effective treatment option for women with ACUM who have additional pathology. Because Müllerian duct anomalies sometimes coexist with urinary tract anomalies, hysterectomy is associated with a risk of urinary tract injury. Therefore, before planning the surgery, the surgeon should carefully choose the treatment plan based on the patient’s condition and treatment needs.
Finally, with the advancements in imaging technology, ACUM has been reported more frequently during the past two decades, although it is still an extremely rare lesion. ACUM is a clearly defined category of uterine malformation, but its diagnostic rate is extremely low. Gynecologists and radiologists should be aware of this lesion’s characteristic clinical features and imaging signs, especially for small lesions that require careful detection and differential diagnosis. It is important to select an appropriate individualized treatment plan based on the patient’s clinical condition. The prognosis of this disease remains unknown, and clinicians must perform careful long-term follow-up of patients with ACUM. In reporting this extremely rare case of a giant ACUM, we aim to offer clinicians and imaging physicians a new perspective that ACUM should be considered and investigated as a differential diagnosis in atypical cases of chronic pelvic pain in women with giant pelvic lesions. However, we lacked long-term follow-up data in this case, necessitating accumulation of further cases to obtain a more comprehensive understanding of the treatment prognosis.
In conclusion, we have herein reported a rare case of a giant ACUM. Such lesions are highly susceptible to preoperative misdiagnosis and require improved clinical and imaging expertise for a full understanding of their features. When drug treatment is ineffective, appropriate surgical plans should be promptly adopted for resection and long-term follow-up. Compared with the more common small ACUM (<4 cm), understanding and recognizing large ACUM remains crucial, and its long-term efficacy should be monitored through follow-up.
Footnotes
Author contributions
Fenglian Deng, Lu Zhang, Yanzhuo Huang, Qiuling Chen, and Xinqi Xiao: collection of clinical and radiological materials and writing of the manuscript. Lijuan Wang: collection of the pathological data. Ke Liu: guiding of the writing of the manuscript. All authors contributed to the article, and all read and approved the final version of the manuscript.
Data availability statement
The datasets generated during this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Ethics statement
The patient provided consent to undergo all treatments. She also provided written informed consent for publication of this report. This study was approved by the Ethics Committee of Chongqing Traditional Chinese Medicine Hospital (2023-ky-41).
Funding
This work was supported by the Chongqing Municipal Research Institutions Performance Incentive Guidance Special Program (No. cstc2021jxjl130030).
