Abstract
Objective
Previous studies have shown that patients with obstructive sleep apnea syndrome (OSAS) may have various vestibular abnormalities, and these mainly present as subclinical lesions. In this study, we attempted to combine ocular vestibular-evoked myogenic potentials (VEMPs) and cervical (VEMPs) to investigate vestibular dysfunction in patients with OSAS.
Methods
Thirty-eight patients with severe OSAS and 42 healthy controls were enrolled and divided into two groups according to results of polysomnography. All parameters of VEMP graphs were analyzed.
Results
The response rate of ocular VEMPs was significantly lower in patients with severe OSAS (85.5%) than in controls (96.4%). The mean n1 latency of patients with severe OSAS (10.6 ± 1.0 months) was significantly longer than that of controls (10.2 ± 1.1 months). With regard to cervical VEMP, p1-n1 amplitudes were significantly different between the groups. Other VEMP parameters were not significantly different between the groups.
Conclusions
VEMPs can potentially be used to evaluate vestibular system lesions at the early stage of OSAS, including not only the vestibular nerve, but also the otolith organs involved.
Introduction
Obstructive sleep apnea syndrome (OSAS), which is characterized by chronic intermittent hypoxia during sleep, adversely affects the function of the nervous system. Consequently, various symptoms of an impaired vestibular system emerge. A previous study showed that daytime neuromuscular coordination and equilibrium are associated with repeated decreases in oxygen saturation. 1 Another study showed that up to 20% of patients with OSAS complaining of dizziness and vertigo have sleep disturbance due to idiopathic or unknown causes. 2 The uncomfortable feeling of dizziness and vertigo severely disturbs these patients’ daily life.
The mechanism underlying neurological dysfunction in patients with OSAS remains poorly understood because of technical limitations. However, some recent studies have suggested that the brainstem tends to be involved in neurological dysfunction and that symptoms of vestibular dysfunction may emerge afterwards.3,4 The mechanism underlying impairment in the vestibular system, especially in the brainstem, contributes to OSAS. Some researchers have attempted to develop techniques, such as vestibular-evoked myogenic potentials (VEMPs), to evaluate vestibular function. VEMPs are important as a type of potential diagnostic tool for detecting detrimental effects of the nervous system.5–7
VEMPs are recorded from the muscles in response to vestibular stimulation. If potentials are recorded from the sternocleidomastoid (SCM) muscles, this is termed cervical VEMPs (cVEMPs), while if they are recorded from the extraocular muscles, this is termed ocular VEMPs (oVEMPs). These two methods, which test different pathways, have provided great clinical value in identifying if lesions of the central nervous system are restricted to descending (cVEMP) or ascending (oVEMP) tracts in the brainstem. 6 cVEMP responses are more significantly correlated with non-motor features, including fatigue, cognition, perception, and memory, while oVEMP responses show less correlation with motor and non-motor features. 8 Therefore, combining the use of oVEMP and cVEMP to detect and locate impairment resulting from hypoxia in the vestibular pathways for patients with OSAS as early as possible might be useful.
In recent years, some studies have confirmed that OSAS contributes to vestibular disability. Two of these studies adopted caloric testing, brainstem auditory-evoked potentials, and head impulse testing to evaluate semicircular canal function of patients with OSAS.9,10 In line with this evaluation, cVEMP was used to evaluate brainstem involvement in OSAS. 11 Although these tests have clinical value, brainstem auditory-evoked potentials are not as sensitive as VEMPs in detecting lesions of the brainstem. 12 Furthermore, no studies have reported the combined use of oVEMP with cVEMP to evaluate clinically silent lesions in patients suffering from OSAS. The caloric test is the gold standard for testing vestibular disorder, but it is not an appropriate tool for differentiating central from peripheral vestibular disorders. Notably, assessing VEMPs is less invasive, less expensive, and more comfortable than other techniques. Therefore, the integrity of ascendant and descendant vestibular pathways can be tested by using cVEMPs and oVEMPs together.
This study aimed to examine the clinical roles of cVEMPs and oVEMPs. This could be a promising technique for diagnostic and localization use for dysfunctional vestibular collic reflexes resulting from severe OSAS.
Participants and methods
Participants
This prospective clinical study included patients with OSAS who were diagnosed in our department and sex-matched healthy controls. Data on the subjects were collected from July 2016 to June 2018. All participants aged 20 to 50 years old who were informed about the purpose of the study were enrolled for a polysomnography (PSG) test.
The subjects had an otoscopic examination, and a pure tone audiometry (PTA) test (recording frequencies of 250, 500, 1000, 2000, 4000, and 8000 Hz). The exclusion criteria were as follows: 1) previous diagnosis of neurological issues; 2) any middle or inner ear deficit; 3) hearing threshold >30 dB HL; 4) diabetes and/or uncontrolled hypertension; 5) a history of cervical disease and/or cervical surgery; and 6) body mass index >32 kg/m2. This study was approved by the Ethical Committee of the Second Hospital Affiliated to Xi’an Jiaotong University. All subjects signed an informed consent form.
PSG
An overnight PSG test (≥7 hours) was performed on a polysomnographic system (Grael; Compumedics Inc., Melbourne, Australia) for all qualified participants who had not been permitted to take sedatives 1 week before the test in the Sleep Research Center of The Second Affiliated Hospital of Xi’an Jiaotong University School of Medicine. Recruited subjects were categorized according to apnea–hypopnea index (AHI) criteria set by the American Academy of Sleep Medicine Criteria in 1999. The lowest arterial oxygen saturation was also recorded, and the subjects were graded according to the Chinese OSAS criteria. 13 The participants were considered to have severe OSAS if the AHI was >30 and oxygen saturation was <65%, while they were considered to be healthy controls if the AHI was <5.
VEMP recordings
After PSG, cVEMP and oVEMP tests were performed by a specialist who was blinded to AHI values and grouping in a sound-proofed examination room. Methods of recording and equipment were designed according to previous studies.14,15 Short tone-bursts of 500 Hz were used as the acoustic stimuli in the test with 1 ms of rise/fall time and 2 ms of plateau time. The electromyographic signal from the stimulated side was amplified by using an ICS Chartr EP analyzer (GN Otometrics, Taastrup, Denmark) and then bandpass-filtered (10–1000 Hz). Cumulative 50 stimuli were performed twice to confirm the reproducibility of results. The initial intensity was set at 100 dB nHL, which was reduced by 5 dB nHL until the VEMP response was abolished. This process was then repeated. The intensity of the last characteristic wave form of VEMP was defined as the response threshold. VEMPs, which were evoked by conventional air conducted (AC) stimuli, were performed on both sides for each participant.
cVEMPs
During the cVEMP test, the participants stayed in the supine position and were instructed to push their heads forward as far as possible while looking at their toes to activate the SCM muscles. Electromyographic activity was monitored and measured to ensure muscular potential >50 µV and then a cVEMP test was carried out. After the electromyographic signal was filtered, tone bursts were presented through headphones. The evoked response was recorded from the upper part of contracting SCM muscles and referred to the response from the tendon of the same SCM muscle. Simultaneously, the ground electrode was placed on the glabella (impedance of the electrodes was not >5 kΩ).
oVEMPs
Participants who took an oVEMP test stayed in the supine position and were stimulated by the same short tone-bursts as cVEMP. The active electrode was situated on the orbital margin below the eye and the reference electrode was placed approximately 15 mm below the active electrode. The stimulus parameter was same as that used in cVEMPs. When acoustic stimuli were delivered to the ear, the subjects were instructed to gaze at the median point above the head directly to activate ocular muscles. The visual angle was approximately 30 degrees.
Data analysis
We evaluated each ear’s VEMP responses separately. Therefore, 76 ears in the OSAS group and 84 ears in the control group were tested. The wave forms of positive (p1) and negative (n1) at a stimulation of 100 dBHL were analyzed. The main outcome measures were the latency (p1 and n1 latency), peak-to-peak amplitude (p1-n1 amplitude), interval (p1-n1 interval), and response rate. The Fisher’s exact or chi-square test was used to compare the response rate between the groups. The numerical results were analyzed by the two-sided paired Student’s t test using statistical software (Statistical Package for the Social Sciences, version 18.0 for Windows, SPSS Inc., Chicago, IL, USA). A P value <0.05 was considered statistically significant.
Results
Clinical characteristics
In the present study, we analyzed 42 controls and 38 patients with severe OSAS, with a total of 160 ears. The mean (standard deviation) age of the OSAS group was 39.5 ± 8.3 years and that in the control group was 36.5 ± 9.6 years. There was no significant difference in sex between the two groups. Clinical characteristics of the participants are shown in Table 1.
Baseline characteristics of the study groups.

Data are expressed as number or mean (standard deviation). OSAS: obstructive sleep apnea syndrome; BMI: body mass index; AHI: apnea–hypopnea index; PTA: pure tone audiometry; NS: non-significant (P > 0.05).
cVEMP abnormalities
The cVEMP response rate was not significantly different between the OSAS group (75%) and control group (86.9%) (P = 0.054) (Figures 1, 2). Nineteen 19 ears in the severe OSAS group and 11 ears in the control group had no cVEMP responses. The mean value of p1-n1 amplitude in the OSAS group (108.5 ± 47.3 µV) was significantly lower than that in the control group (135.7 ± 1.30 µV) (Table 2). There were no significant differences in other cVEMP parameters between the groups.

VEMP response rates in patients with severe OSAS and healthy controls. *P < 0.05. VEMP: vestibular-evoked myogenic potential; OSAS: obstructive sleep apnea syndrome; cVEMPs: cervical vestibular-evoked myogenic potentials; oVEMPS: ocular vestibular-evoked myogenic potentials.
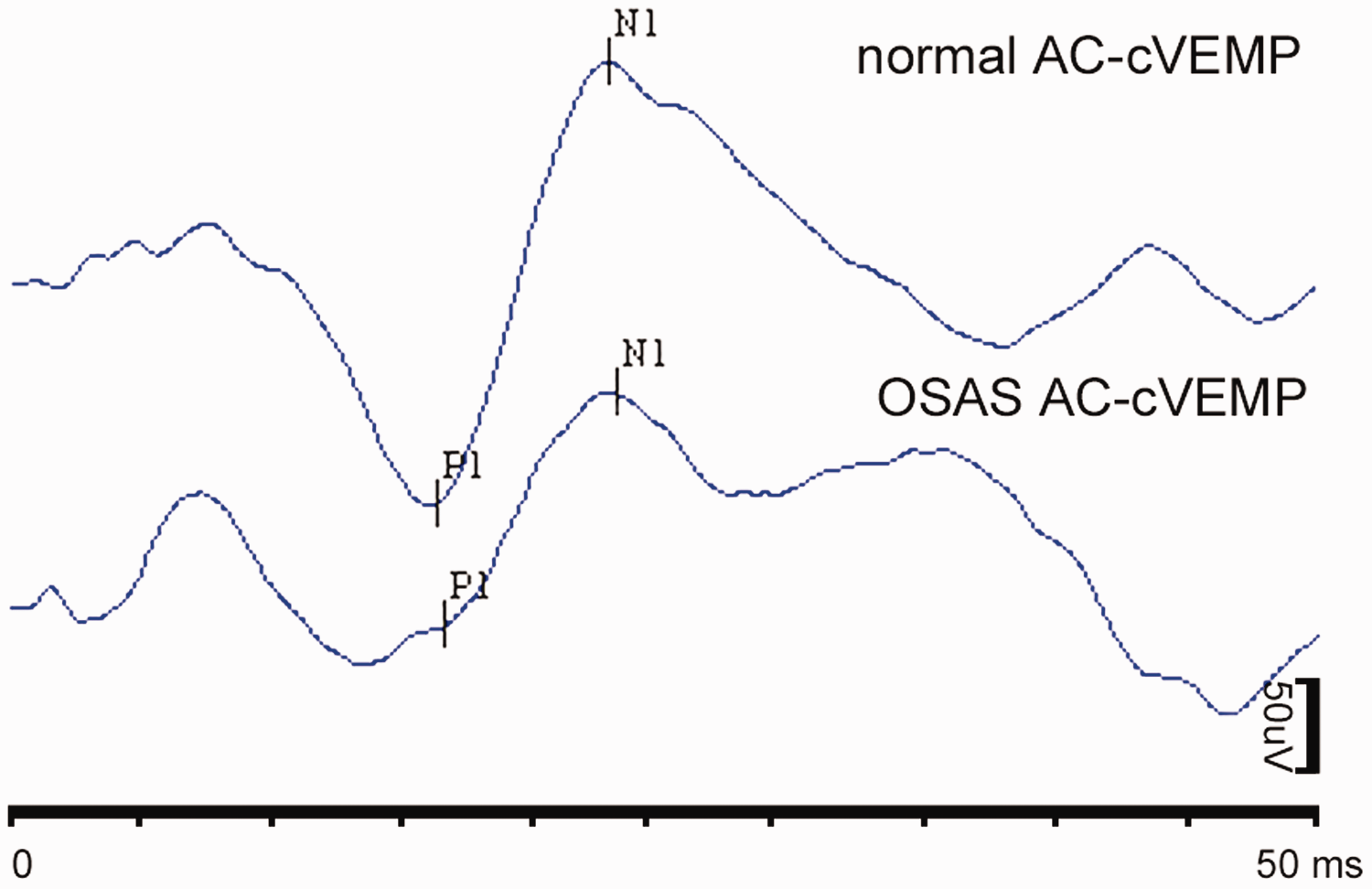
Sample cVEMP tracings from a patient with OSAS and a control. AC: air conducted; cVEMP: cervical vestibular-evoked myogenic potential; OSAS: obstructive sleep apnea syndrome.
Comparison of VEMPs in patients with OSAS and controls.
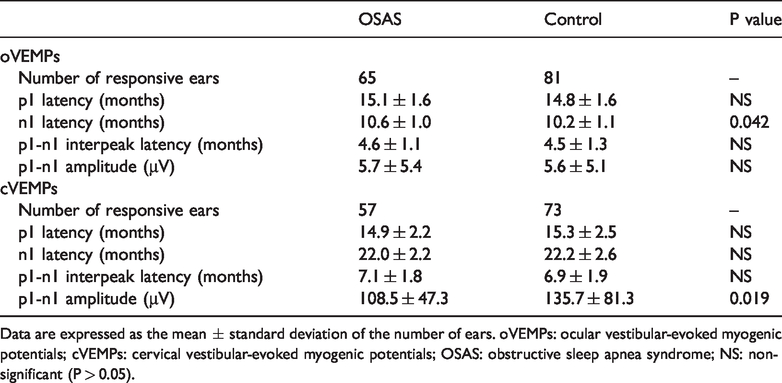
Data are expressed as the mean ± standard deviation of the number of ears. oVEMPs: ocular vestibular-evoked myogenic potentials; cVEMPs: cervical vestibular-evoked myogenic potentials; OSAS: obstructive sleep apnea syndrome; NS: non-significant (P > 0.05).
oVEMP abnormalities
The oVEMP response rate in the OSAS group (85.5%) was significantly lower than that in the control group (96.4%) (Figures 1, 3). The mean value of p1 latency in the OSAS group was not significantly different to that in the control group. However, the mean value of n1 latency was significantly higher in the OSAS group than in the control group (P = 0.042). Although values of p1-n1 amplitude appeared to be greater in the OSAS group than in the control group, this was not significant. Additionally, values of p1-n1 interpeak latency were not significantly different between the groups (Table 2).

Sample oVEMP tracings from a patient with OSAS and a control. AC: air conducted; cVEMP: cervical vestibular-evoked myogenic potential; OSAS: obstructive sleep apnea syndrome.
Discussion
In the present study, 85.5% of patients with severe OSAS showed oVEMP responses and 75% showed cVEMP responses, which were lower than those in controls. These data indicate that VEMP testing is a sensitive diagnostic tool for examining aberrant conduction of the vestibular system in patients with OSAS.
oVEMP and cVEMP have long been widely used to investigate peripheral vestibular disorders by ear-nose-throat specialists. Kim et al. 16 found that cVEMPs were absent in patients with acute vestibular neuritis. Another study showed that reduced or absent, rather than delayed, cVEMP and oVEMP responses were observed in abnormalities of benign paroxysmal positional vertigo disease (even after successful repositioning in benign paroxysmal positional vertigo). Patients with vestibular migraine and Meniere’s disease showed reduced cVEMP amplitudes compared with controls, whereas no difference was found in latency between the groups. 17 Although the results of these studies are not exactly the same, evidence has shown that disorders involving the peripheral vestibular system are more likely to have attenuated or absent VEMP potentials.16,17
In this study, we also found significantly lower amplitudes of cVEMP responses in patients with OSAS compared with controls. This abnormality was also found in a previous study. 11 Some clinical researchers have found that reduced vestibular sensitivity is associated with absent VEMPs, and that a high amplitude of VEMPs occurs when the sensitivity of the vestibular system increases.7,18 This conclusion supports our observation that an impaired vestibular nerve resulted in a smaller wave amplitude and lower cVEMP response rate in patients with OSAS than in controls.
In our study, with the exception of the response rate, latency and latency intervals of oVEMP were significantly affected in patients with OSAS. The findings in this study are consistent with previous studies that showed prolonged latency, which may be associated with demyelination of axons (this reduces conduction velocity).19,20 Macambira et al. 21 reported that the latency component of oVEMP was delayed in older adults and it may be associated with a reduction in the number of neurons. Severe demyelination of axons due to OSAS may contribute to absent oVEMP responses because of conduction block.
Notably, in our study, there was no significant difference in the amplitudes of oVEMP responses between the two groups. A minor difference in wave latency and latency intervals of cVEMP responses was also detected between these two groups. Prolongation of oVEMP latency with a normal latency of cVEMP might be an early sign of upper brainstem involvement in severe OSAS. The findings in the present study are consistent with previous observations of patients with central nervous dysfunction, in whom the wave amplitude of the cVEMP response showed a significant decrease, but latency only showed slight changes.7,11 Another study showed that the amplitude of the oVEMP response was normal, but latency was shortened in patients with migraines, which is consistent with the findings in our study. 22 Some investigators consider that VEMP amplitudes can be used to measure otolith function independently. 23 Based on the evidence shown above, we consider that severe OSAS can impair not only the central vestibular system, but also vestibular end organs. This possibility indicates that dissociation between the results of cVEMP and oVEMP is important for localizing subclinical lesions. Furthermore, impairment is likely to occur at the early stage of OSAS.
Our results are consistent with a previous study, which reported that less cVEMP response was obtained in patients with OSAS. 11 However, this previous study was an incomprehensive valuation in which the researchers did not use oVEMP to investigate injury to the vestibular system. A previous study that was designed to investigate the relevancy of cVEMP and Parkinson’s disease reported that cVEMP amplitudes of patients were significantly decreased. 8 Ivankovic et al. 12 found that the combination of cVEMP and oVEMP was comparable with magnetic resonance imaging, but superior to a clinical examination or auditory-evoked potentials in detecting lesions of the brainstem.
Excitation occurs at saccules, and then typically ascends via the descending medial vestibulospinal tract in the descending brainstem, which is considered as the main cause of cVEMP. oVEMP represents a crossed excitatory vestibulo-ocular reflex following activation of the utricular nerve that mainly passes through the medial longitudinal fasciculus from the upper brainstem to the midbrain. 6 Consequently, combined measurement of oVEMPs and cVEMPs should be a complementary approach for investigating clinically silent lesions in patients with OSAS. Accumulating evidence shows that cVEMPs combined with oVEMPs can detect a significantly higher percentage of abnormalities in the central vestibular pathway, despite some research groups reporting inconsistent results.6,22
An intriguing finding in our study was that the rate of oVEMP wave acquisition (96.4%) was higher than that of cVEMP (86.9%) in the control group. This finding is in contrast to a previous study that investigated the relationship between VEMPs and OSAS and showed that the rate of obtaining oVEMP waves was lower than that of cVEMP. 11 Patients in previous studies underwent an auditory examination before VEMP testing, which was not based on the same standardization because of many factors, such as the electrode used, the inclusion criteria of the PTA test, and patient cooperation. This suggests that future research on VEMP tests needs to be performed in a standardized manner.
Conclusions
The present study shows that abnormalities of the vestibular nerve and otolith organ in patients with severe OSAS can be electrophysiologically observed at the early stage. Combined use of cVEMPs and oVEMPs may help localize subclinical lesions. Therefore, recording VEMPs is a potential tool for evaluating alterations of the vestibular system in patients with OSAS. However, pathological changes of the vestibular nervous system in these patients need to be examined in future investigations.
Footnotes
Acknowledgement
The authors thank Yan-fei Chen for his helpful support in VEMP tests.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
