Abstract
Objective
To evaluate otolithic functions in patients with residual dizziness after successful canalith repositioning procedures (CRPs) for unilateral posterior canal benign paroxysmal positional vertigo (BPPV), and to investigate possible risk factors.
Methods
This case–control observational study included healthy controls and patients with residual dizziness after improvement following CRP for BPPV. All participants were subjected to full history taking, otoscopy, audiological basic evaluation, Dix-Hallpike test to search for posterior canal BPPV, residual dizziness screening, and vestibular evoked myogenic potential (VEMP) testing. Between-group differences were assessed and possible factors associated with residual dizziness were identified by univariate analysis.
Results
A total of 50 patients with residual dizziness (mean age, 56.53 ± 7.46 years [29 female: 21 male]) and 50 healthy controls (mean age, 58.13 ± 7.57 years [20 female: 30 male]) were included. A significant difference in VEMP latencies was found between the patient and control group (delayed in the patient group), with no significant between-group difference in amplitude in both ears. Aging, female sex, long duration of BPPV, number of CRPs, cervical VEMP and ocular VEMP abnormalities, and winter onset, were significantly associated with the risk of residual dizziness.
Conclusions
Residual dizziness is a frequent sequel of BPPV that may relate to otolithic dysfunction. VEMP changes were revealed in the form of delayed latencies.
Keywords
Introduction
Benign paroxysmal positional vertigo (BPPV) is the most common known cause of peripheral vertigo. 1 The prevalence of BPPV increases with ageing, with a peak onset in the fifth to sixth decades, and women are reported to be more affected than men. 2 The posterior semicircular canal is mostly involved, followed by the lateral canal. 3
Different canalith repositioning procedures (CRPs) are used to manage BPPV, with most patients usually experiencing marked improvement. However, residual dizziness is reported to affect about 38–61% of patients with BPPV, even after successful CRP.4,5 These residual manifestations include a non-specific sense of unsteadiness, continuous or intermittent lightheadedness, disorientation, or even drowsiness. 6 Several possible explanations have been postulated, including: the presence of debris in the canal; 7 otolithic dysfunctions; 8 coexistence of other vestibular insults; and/or autonomic dysfunctions. 9
Otolith function can be assessed with both ocular (o) and cervical (c) vestibular evoked myogenic potentials (VEMPs). While cVEMPs are known to reflect the function of ipsilateral sacculo-collic inhibitory pathways, oVEMPs reflect functions of the crossed utriculo-ocular reflex pathway. 10
The purpose of the present study was to evaluate otolithic functions in patients with residual dizziness after successful repositioning manoeuvres to treat unilateral posterior canal BPPV, and to search for other possible risk factors.
Patients and methods
Study type and setting
This case–control observational study was conducted at the Audio-Vestibular Unit, Alzahraa Hospital, Al-Azhar University, Cairo, Egypt, between 1 November 2021 and 1 May 2023. The reporting of this study conforms to STROBE guidelines. 11
Sample size
Sample size was calculated using G*power software, version 3.1.9.2 (www.psycho.uni-duesseldorf.de/abteilungen/aap/gpower3/). To reduce the risk of a type II error and increase the statistical power, and based on independent samples t-test to compare the two studied groups, a minimum sample size of 26 subjects in each group was calculated, considering a significance level of 0.05 (two-tailed), an effect size (d) of 0.8, and a power (1 − β) of 0.80, and α was set a priori at 0.05.
Study population
Participants were categorized into two groups: a control group and a study group. The control group comprised healthy adults, aged 40–70 years, with normal middle ear functions, no complaints regarding balance, and no history of chronic diseases, who were selected by simple random sampling from patients attending different outpatient clinics at Alzahraa hospital, Al-Azhar University, Cairo, Egypt for follow-up examinations. The study group comprised patients aged 40–70 years who were sequentially enrolled after fulfilling the following inclusion criteria: history of BPPV in the form of recurrent, short, and violent attacks of true vertigo and nystagmus induced by head movements; 12 and disappearance of the attacks of positional vertigo and nystagmus after successful canalith repositioning manoeuvres and persistence of unsteadiness or lightheadedness without receiving medications. 9 Subjects known to have history of chronic ear diseases, chronic systemic diseases, neurological diseases, or secondary BPPV were excluded from the study.
Procedures
All participants underwent full audiological and medical history taking, otoscopy and basic audiological evaluation to ensure normal peripheral hearing sensitivity and normal middle ear pressure.
Posterior canal BPPV was assessed by Dix-Hallpike test before and 30 min after the CRP. If the positional vertigo and nystagmus were still present, the CRP was repeated. After 1 week, patients were reassessed by Dix-Hallpike test to ensure that they had been successfully treated. However, if the positional vertigo and/or the nystagmus were persistent, the CRP was again repeated. In addition, to ensure resolution of positional vertigo and persistence of residual dizziness, patients were asked 10 Arabic questions that were suitable for the lifestyle of an Egyptian patient, adapted from the Dizziness Handicap Inventory (question Nos. 1, 3, 5, 9, 11, 13, 16, 18, 21 and 25). 13
Both study groups underwent VEMP testing using an Eclipse evoked potentials system (Interacoustics, Middelfart, Denmark), as follows:
First, the skin was prepared to verify that impedance was appropriate (<5 kΩ). Participants were instructed to sit upright and turn their heads opposite to the stimulated ear in cVEMP testing, while maintaining an upward gaze of 30°. Regarding cVEMPs, the active electrode was located on the upper third of the ipsilateral sternocleidomastoid, while negative and ground electrodes were located on the sternum and forehead, respectively. 14 The active electrode for oVEMPs was placed on the chin, the reference electrode was placed at the orbital midline below the eye, and the ground electrode was located on the forehead. 15
Tone burst stimuli, at an intensity of 97 dBnHL and frequency of 500 Hz, were delivered through insert receivers (Eclipse evoked potentials system) as 200 sweeps in total and 5/s rating, with a duration of 50 ms.14,16 Two consecutive traces were obtained, and amplitudes were rectified. The following formula was used to obtain the asymmetric ratio: 100 ([AR–AL]/[AR+AL]) where AR is the amplitude on right side and AL is the amplitude on left side. 17
Ethical considerations
This research was conducted according to the Declaration of Helsinki and was approved by the Institutional Review Board of the Faculty of Medicine for Girls, Al-Azhar University, Cairo, Egypt (Approval No. 1069; 20/10/2021). All participants provided written informed consent and rights for participation or withdrawal were preserved. All participant details were de-identified and participant confidentiality and security of data and materials were ensured through all stages of the study.
Statistical analyses
Data were analysed using IBM SPSS Statistics for Windows, version 23.0 (IBM Corp., Armonk, NY, USA). Quantitative data are presented as mean ± SD or range for data with parametric distribution, or as median with interquartile range for data with nonparametric distribution. Qualitative variables are presented as n (%) prevalence. Qualitative data were analysed using Fisher’s exact test, while independent samples t-test and Mann–Whitney U-test were used to assess the differences in means between quantitative variables. Factors related to residual dizziness were identified by univariate analysis with odds ratio (OR) and 95% confidence interval (CI). A P-value <0.05 was considered statistically significant.
Results
A total of 100 participants who fulfilled the study criteria were included, comprising 50 healthy control participants and 50 patients with residual dizziness after successful CRP for BPPV. Their mean age was 58.13 ± 7.57 and 56.53 ± 7.46 years, respectively, and the control group comprised 20 female and 30 male participants, while 29 (58%) of the patient group were female and 21 (42%) were male, with no statistically significant between-group differences (P > 0.05; Table 1). Upon Dix-Hallpike testing, four patients were found to have cupulothiasis.
Comparison of age and sex between control and patient groups.

Data presented as mean ± SD or n (%) prevalence.
NS, no statistically significant between-group difference (P > 0.05; independent samples t-test for continuous variables and Fisher’s exact test for categorical variables).
Comparison of cVEMP measurements showed statistically significant between-group differences in wave absolute latencies (positivity at 13 ms and negativity at 23 ms [P13 & N23 latency]–delayed in the patient group in both ears; all P < 0.05); and no significant between-group differences in P13N23 amplitude in either ear (P > 0.05; Table 2).
Comparison of cervical vestibular-evoked myogenic potential (cVEMP) measurements between control and patient groups.

Data presented as mean ± SD or median (interquartile range).
P13 N23, biphasic wave with an initial positive peak at 12–13 ms latency and a subsequent negative peak at 22–23 ms latency.
NS, no statistically significant between-group difference (P > 0.05; independent samples t-test for parametric continuous variables or Mann–Whitney U-test for nonparametric continuous variables).
Comparison of oVEMP measurements showed statistically significant between-group differences in wave absolute latencies (latency of biphasic surface potential with peaks at approximately 10 and 15 ms, beginning with negativity [N1 & P1 latency]–delayed in the patient group; all P < 0.001) and no significant between-group differences in (N1P1) amplitude in either ear (P > 0.05; Table 3).
Comparison of ocular vestibular-evoked myogenic potential (oVEMP) measurements between control and patient groups.

Data presented as mean ± SD or median (interquartile range).
N1 P1, a biphasic surface potential with peaks at approximately 10 and 15 ms, beginning with a negativity.
NS, no statistically significant between-group difference (P > 0.05; independent samples t-test for parametric continuous variables or Mann–Whitney U-test for nonparametric continuous variables).
Univariate analysis of the study group of risk factors for residual dizziness showed that aging, being female, long duration of BPPV, number of CRPs, oVEMP and cVEMP abnormalities, and winter onset were significantly associated with the incidence of residual dizziness (Table 4).
Univariate analysis of factors associated with residual dizziness.
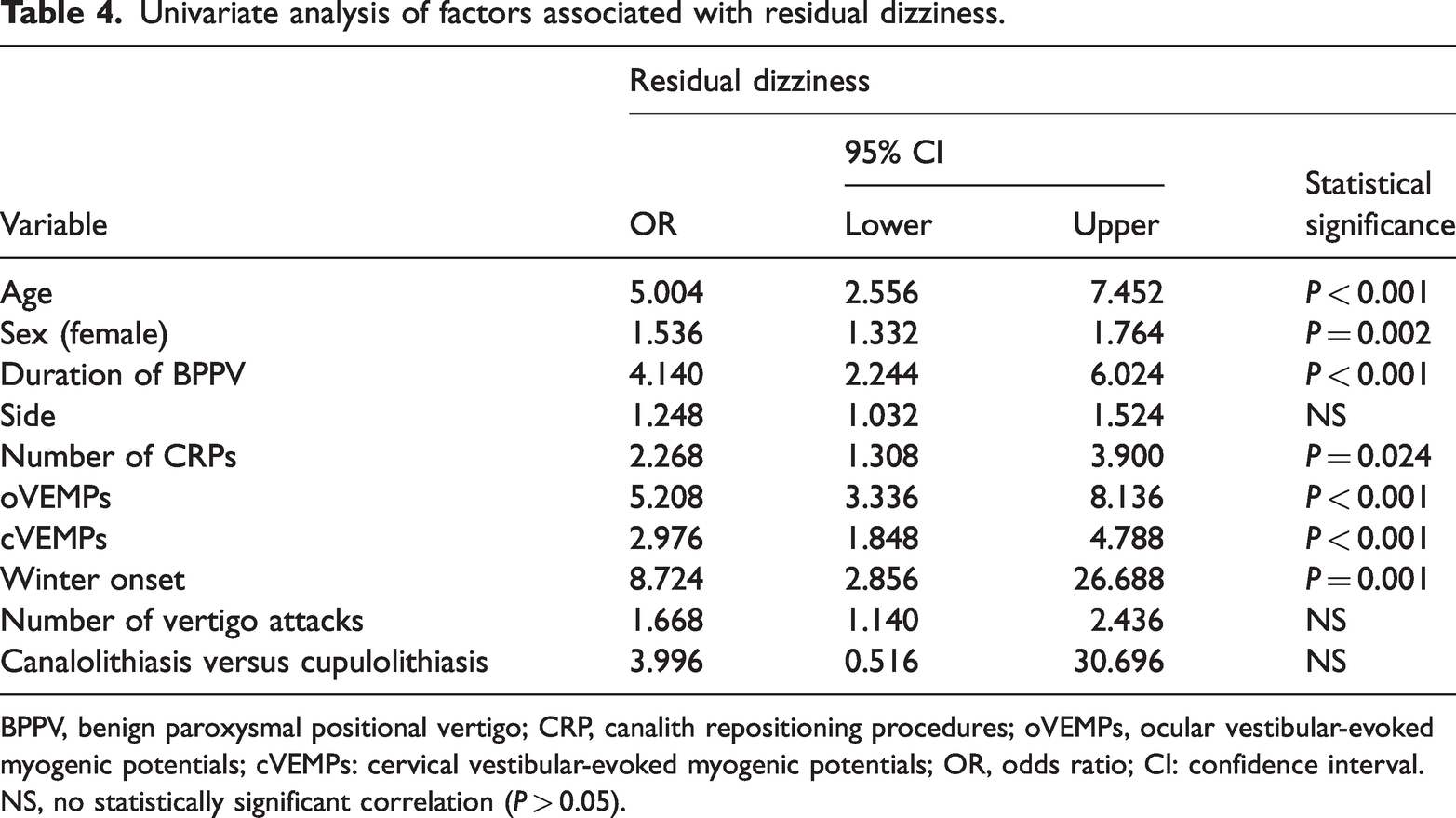
BPPV, benign paroxysmal positional vertigo; CRP, canalith repositioning procedures; oVEMPs, ocular vestibular-evoked myogenic potentials; cVEMPs: cervical vestibular-evoked myogenic potentials; OR, odds ratio; CI: confidence interval.
NS, no statistically significant correlation (P > 0.05).
Discussion
Residual dizziness is reported to be a common consequence of BPPV. 8 This dizziness may adversely affect a patient’s daily life due to the associated psychological and social issues that may decrease daily living activities and increase the risk of falling in the elderly.18,19 Consequently, dizziness evaluation and treatment can be crucial to prevent falls in the elderly. 20
The present results revealed that residual dizziness was significantly associated with being female, and concurred with the finding of a systematic review and meta-analysis of risk factors for residual dizziness in patients with BPPV after successful repositioning. 21 This may be explained by the high prevalence of osteoporosis and osteoarthritis among females, 22 and the protective benefits of testosterone hormones. Also, women usually experience mood changes due to hormonal flux, 23 and mood disorders have been reported to increase the risk of BPPV and subsequent residual dizziness. However, after menopause, the effects of sex hormones decrease, and gender differences in residual dizziness decrease. 24
In contrast to the current study, Zang et al. 25 showed that residual dizziness was not linked to gender. The disagreement with the present results may be explained by differences in sample sizing and environmental factors.
In the present study, a statistically significant difference was found between groups regarding cVEMP (P13&N23) latencies, which were delayed in the patient group, while no significant difference was found regarding (P13N23) amplitude. These findings agreed with the results of Oya et al., 26 who reported infrequent abnormalities of the cVEMPs (a delay in latency of P13&N23 and a decrease in P13N23 amplitude), and the results of Ke et al., 21 who revealed that abnormal cVEMPs may be identified as a risk factor for residual dizziness.
Regarding oVEMP test results, a statistically significant between-group difference was found regarding (N1&P1) latency, which was delayed in the patient group, but no statistically significant difference was found in (N1P1) amplitude. These findings concurred with those of a previous meta-analysis reporting that abnormal oVEMP measurements could be identified as a risk factor for residual dizziness after being improved, 21 and a previous study that showed an association between residual dizziness and abnormal oVEMP measurements. 27
A degeneration process has been postulated to affect the macula of the saccule and utricle. 28 Also, on postmortem temporal bone examination, cellular loss has been reported in both of the saccular macula and the inferior vestibular nerve in patients with BPPV. 29 This gave a theoretical basis for using cVEMPs to predict the occurrence of residual dizziness. 30 Also, microscopic scanning has shown that surface pitting, hollowing out with fragmentation and dislocations of saccular otoconia start earlier than utricular degenerations. 31 In contrast, another study has shown that there was no significant association between abnormal cVEMPs and residual dizziness, and proposed that residual dizziness is due to utricular dysfunction. 32
In the present study, four patients had cupolithiasis. The pathophysiology of the vast majority of posterior canal BPPV is thought to be due to canalithiasis and not cupolithiasis. 33 Cupolithiasis can be treated by CRP, however, since the debris is adhering to the cupula and not free floating, some basic principles of physics are needed to dislodge this debris from the cupula. 34
The present results revealed an association between residual dizziness and the number of repositioning manoeuvres. However, this finding contrasts with previous studies reporting no correlation. 8
The results of the present study may be limited by several factors. First, the case–control observational design may be used to establish a correlation between exposures and outcomes, but cannot establish causation. Second, no randomization was performed. Third, recruitment of patients was limited to a single centre, which may restrict the applicability of the findings. Fourth, no postural, balance, or biochemical tests were performed to provide further insights into the occurrence of residual dizziness. Fifth, patients were recruited only from those attending the audio-vestibular unit. Therefore, findings should be viewed with caution as other patients may seek care in the neurology department. Sixth, the study did not consider all potential factors that might be associated with the risk of developing residual dizziness in patients with BPPV. Finally, confounding variables that appeared significant in the univariate analysis were not eliminated through multivariate analysis. All of these factors affect the generalizability of the study results and prospective multiple-centre follow-up studies are required to thoroughly examine the incidence and predictors of residual dizziness after CRP and long-term outcomes.
Conclusions
Residual dizziness is a frequent sequel of BPPV that may relate to otolithic dysfunction. VEMP changes were revealed in the form of delayed latencies. Aging, female sex, long duration of BPPV, number of CRPs, oVEMP and cVEMP abnormalities, and winter onset were associated with the risk of residual dizziness.
Footnotes
Author contribution
All authors contributed equally to this work design, data collection, analysis, and interpretation, drafting and revision of the article, and final approval of the version for publishing.
Data availability statement
The datasets generated during this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
