Abstract
Objective
This study was performed to investigate the risk factors for severe hypocalcemia (SH) after total parathyroidectomy without autotransplantation (TPTX) in patients with secondary hyperparathyroidism.
Methods
We retrospectively analyzed the records of 136 patients with secondary hyperparathyroidism treated by TPTX. The patients were categorized as having SH or non-SH based on their postoperative blood calcium concentration. The risk factors for SH were identified by comparing the clinical characteristics between the two groups and by performing multiple logistic regression analyses.
Results
After surgery, 46.0% of the patients had hypocalcemia and 31.7% had SH. Evidence of renal osteodystrophy on lumbar radiographs and the serum concentrations of intact parathyroid hormone (iPTH), calcium, and alkaline phosphatase (ALP) were different between the two groups. The SH group had higher preoperative iPTH and ALP concentrations than the non-SH group. In addition, more patients with SH showed renal osteodystrophy. Logistic regression analyses indicated that preoperative hypocalcemia and a high ALP concentration were independent predictors of SH.
Conclusion
Preoperative hypocalcemia and a high ALP concentration were identified as risk factors for SH following TPTX.
Keywords
Introduction
Secondary hyperparathyroidism (SHPT) is a common complication of end-stage renal disease. It is characterized by progressive parathyroid gland hyperplasia and increased synthesis and secretion of parathyroid hormone (PTH).1,2 SHPT promotes bone disease with disordered calcium transport, vascular calcium deposition, and cardiovascular disease, which are life-threatening conditions that negatively impact quality of life.3–6 Many patients with refractory SHPT depend on long-term dialysis and require surgical intervention. 7 Parathyroidectomy (PTX) is required for 20% of patients with SHPT after 3 to 10 years of hemodialysis and for 40% of patients after 20 years of hemodialysis.8,9 The standard surgical treatments for SHPT are subtotal PTX and total PTX with autotransplantation. PTX with autotransplantation is performed to reduce the risk of hypoparathyroidism after total PTX and is typically recommended for younger patients. 10 However, this technique leaves behind a portion of active parathyroid tissue, which may promote recurrence of SHPT.
Total PTX without autotransplantation (TPTX) is an effective treatment for SHPT, but it is associated with postoperative hypocalcemia, which limits its clinical applicability.11,12 The incidence of postoperative hypocalcemia ranges from 72% to 97% 13 and severe hypocalcemia (SH) can cause convulsions, arrhythmia, and even sudden death. Thus, detection of risk factors for hypocalcemia combined with calcium supplementation may prevent hypocalcemia and avoid life-threatening consequences. The specific risk factors for low blood calcium are unclear. To elucidate the risk factors for postoperative SH, we retrospectively analyzed the records of patients with SHPT treated by TPTX at the China-Japan Friendship Hospital from October 2016 to May 2018.
Materials and methods
Study design
We retrospectively evaluated the medical records of patients with SHPT who underwent TPTX at the China-Japan Friendship Hospital from October 2016 to May 2018. The data included in the analysis were the patients’ age, sex, height, body weight, body mass index, dialysis duration, hemodialysis duration, symptoms, X-ray evidence of renal osteodystrophy, and preoperative concentrations of blood calcium, blood phosphorus, alkaline phosphatase (ALP), calcium-phosphorus product, and intact PTH (iPTH). Written informed consent was obtained from all patients. The Medical Research Ethics Committee of the China-Japan Friendship Hospital approved this study (reference number: No. 2016-011).
Surgical indications
The surgical indications for end-stage renal disease with SHPT were persistent elevation of the iPTH concentration above 800 pg/mL, uncontrolled hypercalcemia accompanied by hyperphosphatemia or refractory hyperparathyroidism with clinical symptoms, resistance to active vitamin D, Doppler ultrasound images indicating more than one enlarged (>1 cm) parathyroid gland, and rich blood flow. 14
With the patient under general anesthesia, an experienced surgeon examined the posterior aspect of the thyroid gland. Complete surgical resection was performed for all identified parathyroid glands. If parathyroid glands were not detected, the thymus was examined and the suspected parathyroid gland was removed.
Specimen collection and patient classification
Preoperative blood samples were collected 1 day prior to surgery, and postoperative samples were collected within 48 hours of surgery. The patients were classified into either the SH or non-SH group based on their postoperative blood calcium concentrations. Postoperative SH was defined as a blood calcium concentration of <1.875 mmol/L (7.5 mg/L) accompanied by symptoms of hypocalcemia.2–7 In clinical practice, calcium concentrations below this threshold necessitate calcium supplementation. In the present study, a calcium concentration of <1.8 mmol/L with symptoms of hypocalcemia was an indication for intravenous calcium supplementation.
Statistical analyses
Statistical analyses were performed using SPSS version 17 (SPSS Inc., Chicago, IL, USA). Group differences were detected using single-factor analysis of variance and the chi-square test. Data from single-factor analyses with a p value of <0.1 were included in the multivariate logistic regression analyses. The significance level was 0.05 for all tests.
Results
We reviewed the medical records of 136 patients and identified 126 patients who had undergone total PTX (postoperative PTH concentration lower than normal [88.0 pg/L] and identification of at least four parathyroid glands in the postoperative pathologic examination). The patients’ features, clinical manifestations, preoperative laboratory test results, and imaging results are listed in Table 1. The non-SH group comprised 24 men (60%) and 16 women (40%). The SH group comprised 37 men (43%) and 49 women (57%).
Preoperative indicators of patients in the SH and non-SH groups.
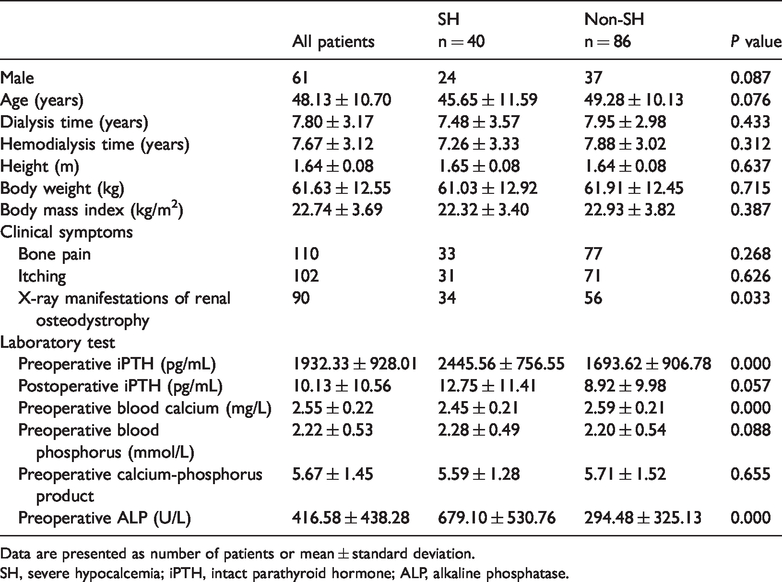
Data are presented as number of patients or mean ± standard deviation.
SH, severe hypocalcemia; iPTH, intact parathyroid hormone; ALP, alkaline phosphatase.
Among all patients who had undergone TPTX, 46.0% had a calcium concentration of <2 mmol/L, indicative of hypocalcemia, and 31.7% were classified as having SH. Patients with SH and non-SH significantly differed with respect to X-ray evidence of renal bone disease (p = 0.033) and the concentrations of iPTH (p = 0.000), calcium (p = 0.000), and ALP (p = 0.000). The SH group had higher preoperative iPTH and ALP concentrations and lower preoperative calcium concentrations. Significantly more patients had X-ray evidence of renal osteodystrophy in the SH than non-SH group.
Factors associated with p values of <0.1 were included in the logistic regression analyses (Table 2). A low preoperative calcium concentration (p = 0.002) and high ALP concentration (p = 0.023) were independent predictors of SH after surgery.
Logistic regression analysis results of indicators between SH and non-SH groups.

P value refers to the significance between SH and non-SH groups.
SH, severe hypocalcemia; OR, odds ratio; iPTH, intact parathyroid hormone; ALP, alkaline phosphatase.
Discussion
The incidence of hypocalcemia in patients with SHPT after PTX ranges from 20% to 85%.15–18 SH, or “hungry bone syndrome,” was first described by Albright and Reifenstein 19 in 1948 and is common in patients with hyperparathyroidism. This state is triggered by a sudden postoperative decrease in PTH, which stimulates rapid migration of blood calcium to the bone tissue and a significant increase in bone remineralization.20–22 However, the diagnostic criteria for SH after TPTX are not clear. In the present study, postoperative SH was defined as a blood calcium concentration of <1.875 mmol/L (7.5 mg/L) in the 48 hours following surgery accompanied by symptoms of hypocalcemia. Studies performed in an attempt to predict postoperative hypocalcemia after hyperparathyroidism surgery have produced inconsistent and unclear findings.14,17,23,24
The influence of age on the postoperative calcium concentration is uncertain. A direct relationship between age and postoperative hypocalcemia has been reported in patients with hyperparathyroidism,21,25 possibly due to vitamin D deficiency and insufficient nutritional intake.21,26,27 However, other studies have identified an indirect relationship between age and the risk of postoperative hypocalcemia.15,17,20,27 Bone remodeling is a complex process that includes both reabsorption and formation. 28 Bone formation increases significantly after TPTX. Because younger individuals have higher osteogenic ability, they are more likely to develop SH. In the present study, the SH group included more younger patients than the non-SH group (SH group, 45.65 ± 11.59 years vs. non-SH group, 49.28 ± 10.13 years), but the difference was not statistically significant.
Males are more likely to develop postoperative hypocalcemia. 17 Low rates of reabsorption in the male cortical bone leads to lower cortical bone loss than in females. As a result, males who undergo TPTX show greater bone formation and a greater likelihood of hypocalcemia. 17 In the present study, the SH group included more men than women (60.0% in the SH group vs. 43.0% in the non-SH group), but the difference was not statistically significant. The sample size may have been insufficient to detect sex-related differences in the incidence of hypocalcemia. Therefore, future studies should address this relationship with larger sample sizes.
Single-factor analyses have identified the preoperative PTH concentration as an independent risk factor for SH. Excess PTH may stimulate bone formation and resorption;17,29 however, one study showed that PTH increases bone resorption but reduces formation. 30 The rapid decline in circulating PTH following TPTX triggers reduced bone resorption and increased bone formation, consistent with the features of hypocalcemia. In the present study, the preoperative iPTH concentration did not predict SH. The inclusion of multiple variables might have precluded the detection of a significant relationship, or the preoperative blood iPTH concentration might not be an independent risk factor for SH development after TPTX. The latter explanation is supported by observations that a uremia status is related to PTH resistance in bone cells31–33 and the inconsistent relationship between the serum iPTH concentration and the degree of bone remodeling. In addition, iPTH quantification includes smaller PTH molecules (besides iPTH), some of which have opposite effects on bone metabolism.31–33
Both the single-factor and multiple-factor logistic regression analyses identified preoperative hypocalcemia as an independent risk factor for postoperative hypocalcemia. This finding is in accordance with previous studies, particularly results from rapid movement of blood calcium into bone and extensive bone remineralization.11,13,14,29
The concentrations of different types of ALP (liver and bone isoenzymes) are elevated in patients who have bone diseases with high osteogenic activity. In patients with hyperparathyroidism, both osteogenic activity and bone resorption are increased.21,34 Previous studies have demonstrated that osteoclast activity is significantly diminished after hyperparathyroidism surgery, whereas osteoblast activity remains stable.35,36 Thus, disrupted bone metabolism may contribute to the development of hypocalcemia after surgery. Our single-factor and multiple-factor analyses identified the preoperative ALP concentration as a risk factor for SH after surgery, which is consistent with previous findings.21,37
We found that 34 patients with SH and 56 patients with non-SH had X-ray results indicating renal osteodystrophy. Chronic kidney disease or renal injury impairs bone synthesis and metabolism, which reduces bone cell function and formation. In patients with end-stage renal disease, increased iPTH concentrations impair bone remodeling, leading to increased bone resorption and reduced bone formation. Therefore, for patients with renal osteodystrophy, reduced postoperative iPTH and calcium concentrations contribute to SH development. 14
Collectively, our results indicate that a low calcium concentration and high ALP concentration are risk factors for SH after TPTX. Thus, high-risk patients should receive calcium supplementation as an intervention to reduce the likelihood of postoperative SH. The use of high-concentration calcium supplements can inhibit residual PTH activity; however, these effects are temporary with intravenous calcium supplementation, and patients should gradually return to oral calcium supplementation within weeks to months after surgery.
