Abstract
Multiple brown tumors represent a rare variant of osteitis fibrosa cystica. Brown tumors are associated with primary, secondary, or tertiary hyperparathyroidism. Brown tumors have been reported in patients with chronic kidney disease resulting in mineral and bone disorders. Chronic kidney disease resulting in mineral and bone disorder is a result of increased osteoclast activity and excessive production of parathormone due to parathyroid gland hyperactivity. Brown tumors are frequently overlooked in patients with end-stage renal disease since calcimimetics and vitamin D analogs were introduced as pharmacological therapy for secondary hyperparathyroidism. We present a case of a 79 year-old pre-dialysis woman, with multiple brown tumors secondary to a parathyroid adenoma despite being treated with cinacalcet for secondary hyperparathyroidism. In addition, we review the corresponding literature.
Keywords
Introduction
Secondary hyperparathyroidism (SHPT) is a common complication of chronic kidney disease (CKD). Renal osteodystrophy is defined by bone morphology alterations related to CKD. 1 Osteitis fibrosa cystica (OFC), its skeletal expression, is characterized by increased bone turnover. Brown tumors (BTs) are a rare variant of OFC and are associated with late presentation of primary (PHPT), secondary (SHPT), or tertiary hyperparathyroidism (THPT). Since calcimimetics and vitamin D analogs were introduced as pharmacological therapy for SHPT, BTs are not an expected complication and easily overlooked. There are many similarities in the radiological and histological features of BTs and giant cell tumors (GCTs), but these lesions rarely coexist. 2 The treatment of these tumors mainly consists of partial or total resection of the parathyroid glands which induces spontaneous tumor regression.
The purpose of this case report is to describe the unusual presentation of multiple BTs that were initially misdiagnosed as GCT, in a patient with end-stage renal disease (ESRD) and severe SHPT, until the histological confirmation of a PHPT due to parathyroid adenoma.
Case presentation
We present the case of a 79-year-old woman with a history of hypertension, ischemic heart disease and type-2 diabetes mellitus with microvascular (CKD and retinopathy) and macrovascular complications. In June 2018, the serum and urinary chemistry revealed parathormone levels (iPTH) of 600 pg/mL (normal range: 15–65 pg/mL), normocalcemia, hyperphosphatemia, low levels of calcitriol, low-normal calciuria, and declining CKD to advance stages (CKD5 not in dialysis) consistent with SHPT. She was treated with calcifediol, paricalcitol, phosphate binders, and cinacalcet (off-label compassionate use indication). During the next 18 months, despite an increase in her cinacalcet dose, a progressive increase in iPTH levels (up to 1266 pg/mL) without the development of hypocalcemia or hyperphosphatemia were observed (Table 1). Considering that her SHPT was caused by a hyperplasia parathyroid, a parathyroid scintigraphy with 99mTc-sestamibi was performed (Figure 1(a) and (b)). This showed an increased focal uptake in the inferior right parathyroid gland. Given the severity of the HPT, surgical treatment was recommended, but the procedure had to be postponed due to the COVID-19 pandemic.
Evolution of treatment and analytic parameters.

GFR: glomerular filtration rate; CKD-EPI: chronic kidney disease epidemiology collaboration; CrCl: creatinine clearance in 24 h; PTHi: parathyroid hormone intact; SCa: serum calcium; SCa-corrected SA: corrected serum calcium for SA; SA: serum albumin; SPhos: serum phosphorus; Mg, serum magnesium; calcitriol: 1,25-(OH)2 vitamin D3; SNa: serum sodium; SK: serum potassium; SCl: serum chlorine; ALP: alkaline phosphatase; NA: not applicable.
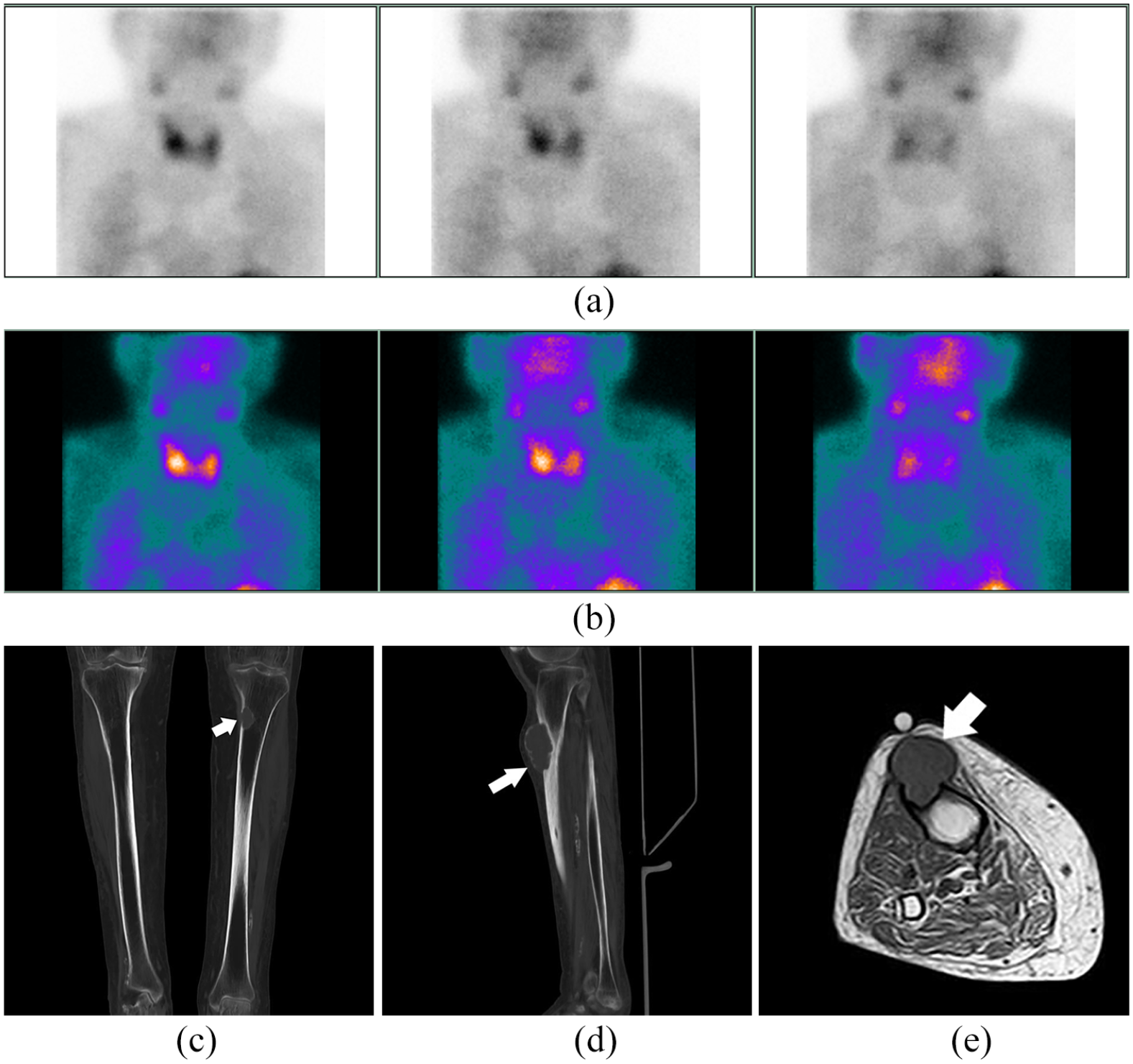
Scintigraphy study of planar parathyroid in anterior cervical projection in two phases (with acquisition of an early phase image at 20 “and two late-phase images at 60” and “120”) after the administration of 740 MBq of 99mTc-sestamibi in which an overactive focus is observed superimposed on the lower pole of the inferior right parathyroid gland with a slightly slowed lavage compatible with hyperfunctioning parathyroid tissue (a, b). CT coronal view showing proximal tibial metaphysis, the white arrow points to a radiolucent mass (brown tumor) that breaks the cortical of the left tibia (c). The sagittal CT view shows the Brown tumor as an exophytic mass invading soft tissue of the right tibia (d). MRI shows the brown tumor as a lesion with low T1; the white arrow points to a mildly hyperintense patchy area, suggesting intralesional hemorrhage (e).
Six months later, the patient presented to our internal medicine outpatient clinic with a 2-month history of severe musculoskeletal pain in both lower limbs and constitutional symptoms. On physical examination, there was a palpable mass in the proximal right tibia with reduced range of movements of knee flexion and extension. The rest of the physical examination was normal. The X-rays, computed tomography (CT), and magnetic resonance imaging (MRI) scans revealed multiple eccentric osteolytic and expansible trabeculated lesions in both tibias and patellae (Figure 1(c)—(e)).
Given the initial diagnostic impression of bone metastasis, we focused our tests to rule out malignancies. A whole-body scan with 20 mCi 99mTc-sestamibi was performed to look for multifocal disease; which showed multiple deposits at the level of the cranial diploe, patella, and diaphysis of both tibiae and costal arches. Additional investigations were carried out including mammography and gastrointestinal endoscopy with biopsies. All these tests were negative for malignancy. Laboratory studies revealed elevated iPTH level of 3051 pg/mL (Table 1). Tumor markers and serum protein electrophoresis were normal. An ultrasonography-guided biopsy of the right proximal tibia mass was performed. Histopathological tissue examination revealed the presence of diffusely shaped giant cells (Figure 2(a)—(d)). The patient underwent radio-guided subtotal parathyroidectomy with intraoperative SPECT/CT imaging. iPTH was measured post-operatively, and the levels decreased to 363.9 pg/mL. A histopathological examination of the mass confirmed the diagnosis of parathyroid adenoma. Based on the evolution clinical and histological confirmation, the final diagnosis was BT with PHPT due to solitary parathyroid adenoma in the setting of a patient with CKD. One-month post-surgery, her iPTH levels were 277.7 pg/mL.

CD68 immunostaining was performed (20×), using this marking for giant cells, the tendency to form dense lobes or groups separated by areas with lower density is appreciated from a panoramic point of view (a). In greater detail, oval cells and occasional more elongated cells are observed between the multinucleated cells in the center of the image (H-E., 400×) (b). In p63 immunostaining, nuclear staining is seen in elongated oval cells (400×) (c). In the CD68 immunostaining showed marking of the osteoclastic cells of the accompanying macrophages (400×) (d).
The patient had a satisfactory clinical outcome with better bone pain control and reduction in lower limb masses, which improved her right knee range of movement. Despite the satisfactory post-operative outcome, chronic maintenance hemodialysis was initiated due to uremic syndrome and low glomerular filtration rate of <8 mL/min (Table 1).
Discussion
Here, we report a case of multiple BTs as a presumed consequence of advanced CKD-MBD due to poorly controlled SHPT, however, was found to be a result of PHT due to parathyroid adenoma after parathyroidectomy. Lajolo et al., 3 presented four new cases of oral cavity BTs and a review of 163 existing cases in the literature. The results of the systematic analysis of the report the cases highlight that BTs seem to mainly affect PHPT and SHPT, and they rarely occur in patients with THPT (44.3%, 51.5%, and 4.2%, respectively).
BTs are the expression of severe OFC with an increased incidence in female and elderly patients, especially those over 50 years. These tumors can occur in any bone; usually in large bones, such as the ribs, clavicles, pelvis, and jaw, however are rare in cranial and facial bones.3,4 In our case, the location of the lesions in the metaphysis of long bones, especially in distal femur and proximal tibia is more typical of GCT. 5 Differential diagnosis includes bone metastases, amyloid cysts, chondroma, aneurysmal bone cyst, osteosarcoma, GCT, or myeloplax tumor.5–7 Biochemical markers, parathyroid ultrasonography, parathyroid scintigraphy (99mTc-MIBI), and bone scintigraphy (99mTc-MDP) are often necessary to reach the correct diagnosis. 8 In patients with negative results on conventional tests, 18F-Fluorocholine PET/CT has shown its usefulness. 9
Treatment of BTs depends on the severity of the bone lesions. The mainstay of treatment is normalization of iPTH levels with pharmacological therapy, aiming for a regression or resolution of the tumor. 2 The Kidney Disease Improving Global Outcomes (KDIGO) Guidelines on CKD-MBD 10 suggest the combination of calcimimetics with calcitriol or vitamin D analogs in patients with CKD5D requiring PTH-lowering therapy. However, its use in CKD patients not requiring renal replacement therapy is not recommended due to the possible development of hypocalcemia and hyperphosphatemia. However, some studies have shown beneficial effects of cinacalcet in patients with CKD not on dialysis. They reported a measured reduction in iPTH levels and similar rates of side effects as those observed in dialysis patients.11,12 Considering the severity of our patient’s disease and high surgical risk, we decided to start treatment with calcimimetics with a close monitoring of MBD profiles.
Since the start of medical therapy, the patient’s iPTH levels were never normalized. Paradoxically, they rocketed to astonishing values when she presented to us. These abnormal levels can be explained by the presence of PHPT due a parathyroid adenoma resistant to the proposed medical therapies, although refractory SPHT has been described in postmenopausal women. 13 OFC is a direct consequence of parathyroid adenoma in 80%–85% of cases; however, only 2% of parathyroid adenomas will present with this condition. 14 In our case, the absence of hypercalciuria, hypophosphatemia, and nephrocalcinosis led us to consider initially that OFC was secondary to SPHT until the histological confirmation of a PHPT due to solitary parathyroid adenoma.
Finally, parathyroidectomy in PHPT is the gold standard treatment. However, in high-risk surgical candidates, cinacalcet is an option, especially for those presenting with hypercalcemia. 14 Likewise, in SPHT due to CKD, parathyroidectomy is indicated in patients with hyperparathyroidism resistant to medical therapy. 15 The indications for surgical management of severe SHPT according to the Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines are persistently elevated iPTH values >800 pg/mL (>6 months) associated with hypercalcemia and/or hyperphosphatemia and symptoms/complications attributable to SHPT. 16
Conclusion
This case report emphasizes the need of considering alternative causes for presumed SPTH, and the role for a multidisciplinary follow-up in the management of BT. We hope our experience may lead to a better understanding and management of this rare condition in the context of CKD.
Footnotes
Acknowledgements
The authors are grateful to Dr Oscar A. Sanchez Valenzuela for documenting the images of the partial parathyroidectomy surgical procedure.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The research was conducted ethically in accordance with the World Medical Association Declaration of Helsinki.
Informed consent
The patient has given their written informed consent to collect anonymous and for publish their case (including publication of images).
Data availability
The data used to support the findings of this study are available from the corresponding author on request (Contact JDF,
