Abstract
Objective
Cerebral infarction has a poor prognosis and causes a serious burden on families and society. Recombinant tissue plasminogen activator (rt-PA) and urokinase (UK) are commonly used thrombolytic agents in the clinic. However, direct and powerful clinical trial evidence to determine the therapeutic effect of rt-PA and UK on intravenous thrombolysis is lacking.
Methods
In this study, 180 patients with acute cerebral infarction were treated with rt-PA or UK. The National Institutes of Health Stroke Scale (NIHSS) scores, Barthel index, bleeding complications, and biomarkers were evaluated.
Results
No significant differences in NIHSS or Barthel scores were found between the groups. However, UK increased the risk of intracranial haemorrhage compared with rt-PA. rt-PA had increased activity in reducing serum levels of MMP-9 than UK.
Conclusion
Intravenous thrombolysis with rt-PA and UK in the time window of acute cerebral infarction can achieve similar therapeutic effects, but rt-PA can further reduce the risk of cerebral haemorrhage and is relatively safer than UK.
Keywords
Introduction
Cerebral infarction is the most common stroke-type clinical disease. Clogged blood vessels result in severe cerebral infarction, which lead to high disability and mortality rates and seriously threaten human health. 1 Timely and effective treatment of cerebral infarction during an acute attack is closely related to prognosis. The treatment for acute cerebral infarction focuses on restoring the blood supply to the ischemic brain tissue, restoring the cerebral vessels of the occlusion, and promoting the recovery of the neurological function. Currently, early thrombolysis guided by a time window is the effective and clinically recognised treatment.2,3
In 1995, the National Institute of Neurological Disease and Stroke confirmed for the first time that intravenous application of recombinant tissue plasminogen activator (rt-PA) could treat ultra-early ischemic stroke through evidence-based medical studies. As such, rt-PA intravenous thrombolysis therapy for acute cerebral infarction has become the clinical standard and the most effective treatment method that is popular worldwide. 4 Urokinase (UK) is an endogenous plasmin-activating agent that can be extracted from human plasma and urine. UK is a thrombolytic agent commonly used in the clinic, especially in China.5,6 UK degrades the chemical bond between arginine and conjugates in the molecule by binding to plasminogen in clots, producing fibrinolytic enzymes, which play a role in thrombolysis.7,8
However, direct and powerful clinical trial evidence to determine the therapeutic effect of rt-PA or UK for intravenous thrombolysis is lacking. Although the majority of experts and guidelines tend to use alteplase based on medical evidence, the most suitable intravenous thrombolytic drugs for treating acute cerebral infarction remain unclear.9–11 There are few studies on the efficacy of intravenous thrombolysis for treating acute cerebral infarction.
Results
In this study, 180 patients with acute cerebral infarction were treated with rt-PA or UK between January 2017 and January 2019 to evaluate the therapeutic efficacy of intravenous thrombolysis with rt-PA or UK in patients with acute cerebral infarction. Patients with known inflammation, infectious tumours, and unavailable blood samples were excluded from this study. A total of 180 patients (106 men and 74 women; mean age of 59.9 years ± 6.4 years [44–74]) who were hospitalised within 12 hours of the onset of ischemic stroke were retrospectively examined. The following patients were included: 90 patients for the UK group (54 men and 36 women; mean age of 57.2 years ± 5.6 years) and 90 patients for the rt-PA group (60 men and 30 women; mean age of 58.5 years ± 5.9 years).
The difference in the National Institutes of Health Stroke Scale (NIHSS) scores before treatment between the two groups was non-significant. After treatment, the NIHSS scores of patients in the rt-PA and UK groups decreased. The difference in the NIHSS scores after treatment (after 24 hours and 7 days) was also non-significant between the two groups (Table 1). The Barthel index was used to evaluate the quality of the daily life of patients. The difference in the Barthel index before treatment was non-significant between the two groups. After treatment, the Barthel index of patients in the rt-PA and UK groups increased. The difference in the Barthel index after treatment (after 24 hours and 7 days) between the two groups was non-significant (Table 2). The percentage of bleeding complications was also determined, and the results indicated that the difference between the two groups was non-significant (Table 3). Furthermore, serial changes in biomarkers were detected. A significant difference was observed in hs-CRP and IL-6 24 hours and 7 days after onset and in MMP-9 7 days after onset (Table 4). However, repeated-measures ANOVA did not demonstrate any significant difference between the two groups regarding hs-CRP and IL-6. For MMP-9, a significant interaction of the groups within the three timepoints was observed with repeated-measures ANOVA. The concentration in the rt-PA group was significantly lower than in the UK group (F = 20.157, P = 0.004), as shown in Figure 1.
NIHSS scores of patients.

NIHSS, National Institutes of Health Stroke Scale; rt-PA, recombinant tissue plasminogen activator; UK, urokinase.
Barthel index of patients.

rt-PA, recombinant tissue plasminogen activator; UK, urokinase.
Bleeding complications in the patients (%).

rt-PA, recombinant tissue plasminogen activator; UK, urokinase.
Changes in biomarkers.

rt-PA, recombinant tissue plasminogen activator; UK, urokinase.
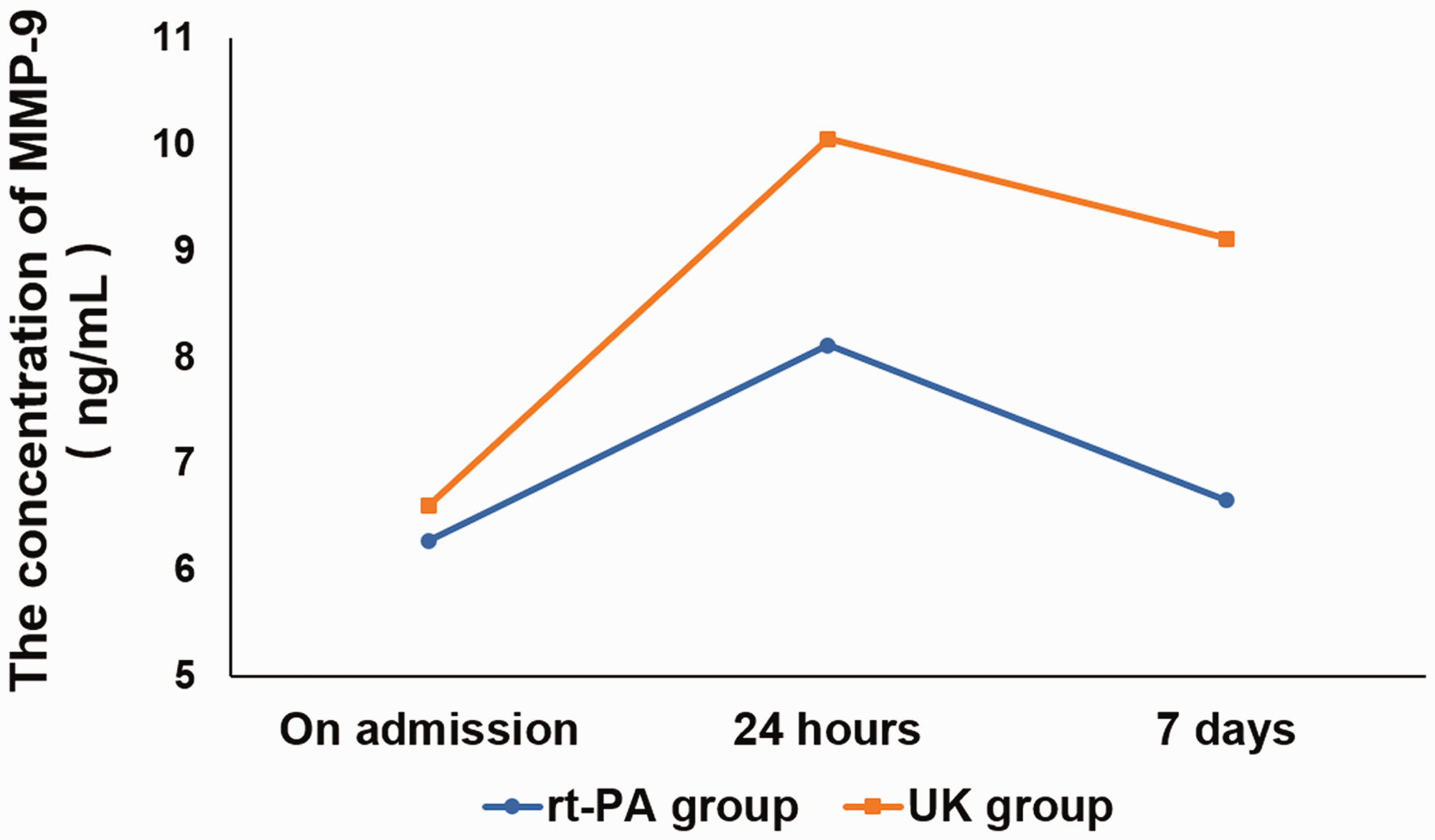
Timeline of MMP-9 concentrations in the two groups. MMP-9 levels were measured by ELISA before treatment and 24 hours and 7 days following treatment in patients of the rt-PA (blue) and UK (red) groups.
Discussion
Acute cerebral infarction is the most common cerebrovascular disease, accounting for approximately 70% of strokes. Within 10 minutes of an ischemic attack, irreversible necrosis occurs at the centre of the local ischemic area, and ischemic penumbra gradually forms around the necrotic area. The occurrence of ischemic penumbra is the theoretical basis for treating acute cerebral infarction.12,13 With early recovery of blood perfusion in the infarction area, this part of the ischaemia-injured brain cells can be repaired, indicating that the condition and prognosis of patients with cerebral infarction can be improved. Therefore, the application of thrombolytic drugs is crucial. Early thrombolytic therapy for acute cerebral infarction has also become a reliable and safe method.
rt-PA is a commonly used thrombolytic drug worldwide, but the effects of UK on intravenous thrombolysis in patients with acute cerebrovascular disease is controversial.14–17 Only China and Japan use UK in clinical practice. UK is not widely recognised internationally because of the increased risk of bleeding. Our results showed that the difference in NIHSS and Barthel scores between the rt-PA and UK groups at 24 hours and 7 days after thrombolysis under the same time window was non-significant. However, UK increased the risk of intracranial haemorrhage compared with rt-PA. Serum concentrations of hs-CRP, IL-6, MMP-2, and MMP-9 were serially measured at admission. hs-CRP and IL-6 are common inflammatory cytokines that play an important role in the occurrence and development of acute cerebral infarction when overexpressed.18,19 MMP‑2 and MMP‑9 can degrade collagen fibres, elastic fibres, and other extracellular matrix components, resulting in a weakened fibrous cap function and an unstable carotid plaque, which increases the risk of acute cerebral infarction. 20 Serum levels of MMP-9 were reduced by rt-PA compared with UK, indicating that rt-PA has a lower risk of haemorrhagic transformation and is safer for clinical use than UK.
However, UK remains widely used in China. Although UK is not an ideal therapeutic drug, it is in line with the national conditions of many developing countries. UK has a low cost and is easily accepted by the majority of the population. Many community hospitals may not have alteplase, and if they are no longer advised to use UK, then medicine will be unavailable. When both drugs are available, the reasonable and effective A drugs are suggested for use in clinical practice; however, UK is also an optional thrombolytic drug in the appropriate time window when only it is available.
Our results show that intravenous thrombolysis with rt-PA and UK in the time window of acute cerebral infarction can achieve similar therapeutic effects, but rt-PA can reduce the risk of cerebral haemorrhage and is relatively safer than UK. In this study, NIHSS scores, Barthel index, bleeding complications, and biomarkers were used to evaluate the efficacy of rt-PA and UK in patients with acute cerebral infarction. However, ultra-high-field time-of-flight (TOF) magnetic resonance angiography (MRA) has become a more valuable method to visualise small perforating arteries and should be used in the future as a diagnostic tool in clinical practice. 21 High-field TOF-MRA may be helpful to enhance our understanding of the anatomical patterns in vivo, ischemic processes, diagnosing cerebral vessel malformations, and planning neurosurgical procedures. 22 In future studies, ultra-high-field TOF-MRA will be used as a more effective and accurate diagnostic method to determine improved treatments.
Patients and methods
This study was approved by the Human Ethics Committees of Chengde Medical College. Informed consent was obtained from all patients or their relatives prior to inclusion in the study.
Patients with the following conditions were excluded: (1) slight neurologic deficits at admission (a NIHSS score of <4), (2) serious concurrent diseases, (3) stroke caused by arterial dissection or uncommon diseases, (4) severe inflammatory or autoimmune diseases, and (5) those taking steroids or nonsteroidal anti-inflammatory drugs. All patients were treated with rt-PA (0.6–0.9 mg/kg, intravenous drip, Boehringer Ingelheim, Germany) or UK (1 × 106–1.5 × 106 IU/kg, intravenous drip, Nanjing Nanda Pharmaceutical Co., Nanjing, China) within 4 hours ± 1.5 hours of acute ischemic stroke.
Blood samples of all patients were obtained within 60 minutes of arrival to the hospital. Serum was separated and stored at −80°C until analysis. hs-CRP, IL-6, MMP-2, and MMP-9 levels were measured with enzyme-linked immunosorbent assay (ELISA) kits (CardioPhaseTM High-Sensitivity C-reactive Protein System, Siemens, Germany; Biotrak High-Sensitivity Human IL-6 ELISA System, GE Healthcare, Piscataway, NJ, USA; human MMP-2 and MMP9 kits, GE Healthcare).
IBM SPSS Statistics for Windows, version 24.0 (IBM Corp., Armonk, NY, USA) was used for data analysis. Data are displayed as percentage values and absolute numbers. Patient characteristics are presented as the mean and standard deviation. Unpaired Student’s t tests were used to compare continuous variables, and chi-square tests were used for nominal parameters. P ≤ 0.05 was considered statistically significant. Two-way repeated-measures analysis of variance (ANOVA) was used to compare changes over time for the concentrations of biomarkers between groups. Lack of correlation was evaluated using the Mauchly sphericity test. If this assumption was not satisfied, the Greenhouse–Geisser correction was used at a significance level of P ≤ 0.05.
