Abstract
Objective
Intravenous thrombolysis with recombinant tissue plasminogen activator (rt-PA) is considered the most effective treatment method for AIS; however, it is associated with a risk of hemorrhage. We analyzed the risk factors for digestive and urologic hemorrhage during rt-PA therapy.
Methods
We retrospectively analyzed patients with AIS who underwent intravenous thrombolysis with rt-PA during a 5-year period in a Chinese stroke center. Data on the demographics, medical history, laboratory test results, and clinical outcomes were collected.
Results
338 patients with AIS were eligible and included. Logistic regression multivariate analysis showed that gastric catheter was significantly correlated with digestive hemorrhage, while age and urinary catheter were significantly correlated with urologic hemorrhage. Most hemorrhagic events were associated with catheterization after 1 to 24 hours of rt-PA therapy.
Conclusions
In summary, gastric and urinary catheters were correlated with digestive and urologic hemorrhage in patients with AIS undergoing rt-PA therapy. Well-designed controlled studies with large samples are required to confirm our findings.
Keywords
Introduction
Acute ischemic stroke (AIS) accounts for about 80% of all types of stroke. 1 The estimated annual incidence of stroke is 0.25% and increases with aging. 2 The clinical syndrome of AIS involves a sudden onset of focal or global disturbance of central nervous system function caused by an interruption in the cerebral circulation. Therefore, the main therapeutic goals for AIS are improvement of the circulation, neuroprotection, and prevention of complications. In addition to endovascular therapy, intravenous thrombolysis with recombinant tissue plasminogen activator (rt-PA) has been considered the most effective method against AIS, 3 showing significant improvement in the survival rate and quality of life of patients with AIS.
However, intravenous thrombolysis with rt-PA is associated with a risk of hemorrhage. Patients with serious hemorrhage usually have a poor clinical prognosis with a mortality rate of up to 90%. 4 Most studies to date have focused on cerebral hemorrhage as a complication of intravenous thrombolysis. For instance, one study showed that the presence of leukoaraiosis might increase the risk of intracerebral hemorrhage after intravenous thrombolysis in patients with AIS. 5 However, the events associated with peripheral hemorrhage have been rarely discussed despite the fact that these events also affect the survival of patients with AIS who undergo intravenous thrombolysis with rt-PA. 6 In the present study, we analyzed the risk factors for digestive and urologic hemorrhage with the aim of enabling more patients with AIS to gain benefits from rt-PA therapy.
Materials and methods
Study design
We retrospectively analyzed patients with AIS who underwent intravenous thrombolysis with rt-PA during a 5-year period (April 2011–April 2016) in the most well-known stroke center in China (Xuanwu Hospital, Capital Medical University, Beijing). Data on the patients’ demographics, medical history, laboratory test results, and clinical outcomes were collected. For intravenous thrombolysis, rt-PA (Boehringer Ingelheim Pharma GmbH, Ingelheim, Germany) was administered at dose of 0.9 mg/kg.
Patient selection
Eligible patients were required to meet the following criteria: age of ≥18 years, onset within 4.5 hours, ≥1-hour duration of clinical signs related to stroke, no findings of intracranial hemorrhage or early large-area cerebral infarction on brain computed tomography, and provision of informed consent. The exclusion criteria were absence of intact records during rt-PA therapy, performance of arterial thrombolysis after failure of intravenous thrombolysis, and contraindications for rt-PA therapy.
Data collection
We used an electrocardiogram monitor (IntelliVue MP70; Philips Healthcare, USA) to collect the patients’ vital signs. The registered clinical data included sex, age, vital signs, height, weight, body mass index, thrombolysis time window, drug administration, and history of hypertension, diabetes, hyperlipidemia, smoking, drinking, coronary heart disease, atrial fibrillation, stroke, transient ischemic attack, peptic ulcer, surgery, and trauma/falls. Twenty-five laboratory parameters were measured from routine blood, biochemical, and coagulation tests, and brain computed tomography scans were performed. All patients were evaluated using the National Institutes of Health Stroke Scale (NIHSS) and activities of daily living (ADL) scale.
Study outcomes
Peripheral hemorrhage such as digestive and urologic hemorrhage occurring within 36 hours of intravenous thrombolysis were the main outcomes in the present study. The diagnosis of digestive hemorrhage was dependent on gastric juice and feces test results, while the presence of positive red blood cells on routine urinalysis was necessary for a diagnosis of urologic hemorrhage.
Statistical analysis
We used EpiData 3.1 (EpiData, Odense, Denmark) to set up the database with double-personnel data entry and SPSS v.18.0 (SPSS Inc., Chicago, IL, USA) to analyze the data. For all tests, differences were considered statistically significant at a p value of <0.05.
Ethical considerations
This study was approved by the ethics committee of Capital Medical University. Prior to entering the study, all patients were informed of the study procedures.
Results
In total, 338 patients with AIS were eligible and included in the study. Of these patients, 18 (5.3%) developed digestive hemorrhage and 24 (7.1%) developed urologic hemorrhage within 36 hours of intravenous thrombolysis.
Comparison of clinical data between patients with and without digestive hemorrhage
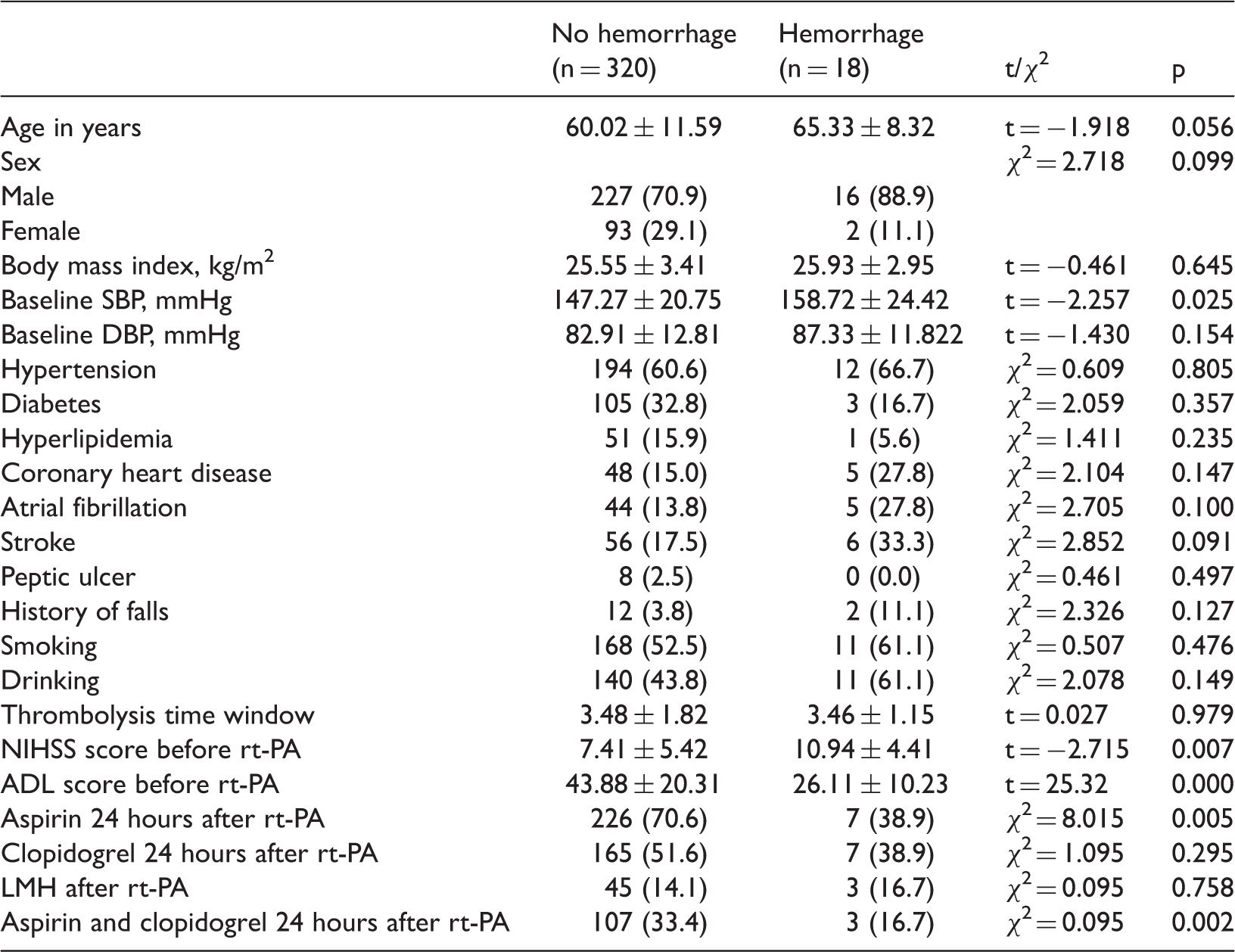
Data are presented as n (%) or mean ± standard deviation unless otherwise indicated.
ADL: activities of daily living; DBP: diastolic blood pressure; LMH: low-molecular-weight heparin; NIHSS: National Institutes of Health Stroke Scale; rt-PA: recombinant tissue plasminogen activator; SBP: systolic blood pressure.
Comparison of laboratory parameters between patients with and without digestive hemorrhage

Data are presented as mean ± standard deviation unless otherwise indicated.
INR: international normalized ratio.
Comparison of gastric catheter between patients with and without digestive hemorrhage

Data are presented as n (%) unless otherwise indicated.
GC: gastric catheter.
Logistic regression analysis of risk factors for digestive hemorrhage

Likelihood ratio test = 98.298, Cox and Snell R2 = 0.118, Nagelkerke R2 = 0.346.
ADL: activities of daily living; BUN: blood urea nitrogen; CI: confidence interval; NIHSS: National Institutes of Health Stroke Scale; OR: odds ratio; RBC: red blood cell count; rt-PA: recombinant tissue plasminogen activator; SE: standard error.
Comparison of clinical data between patients with and without urologic hemorrhage
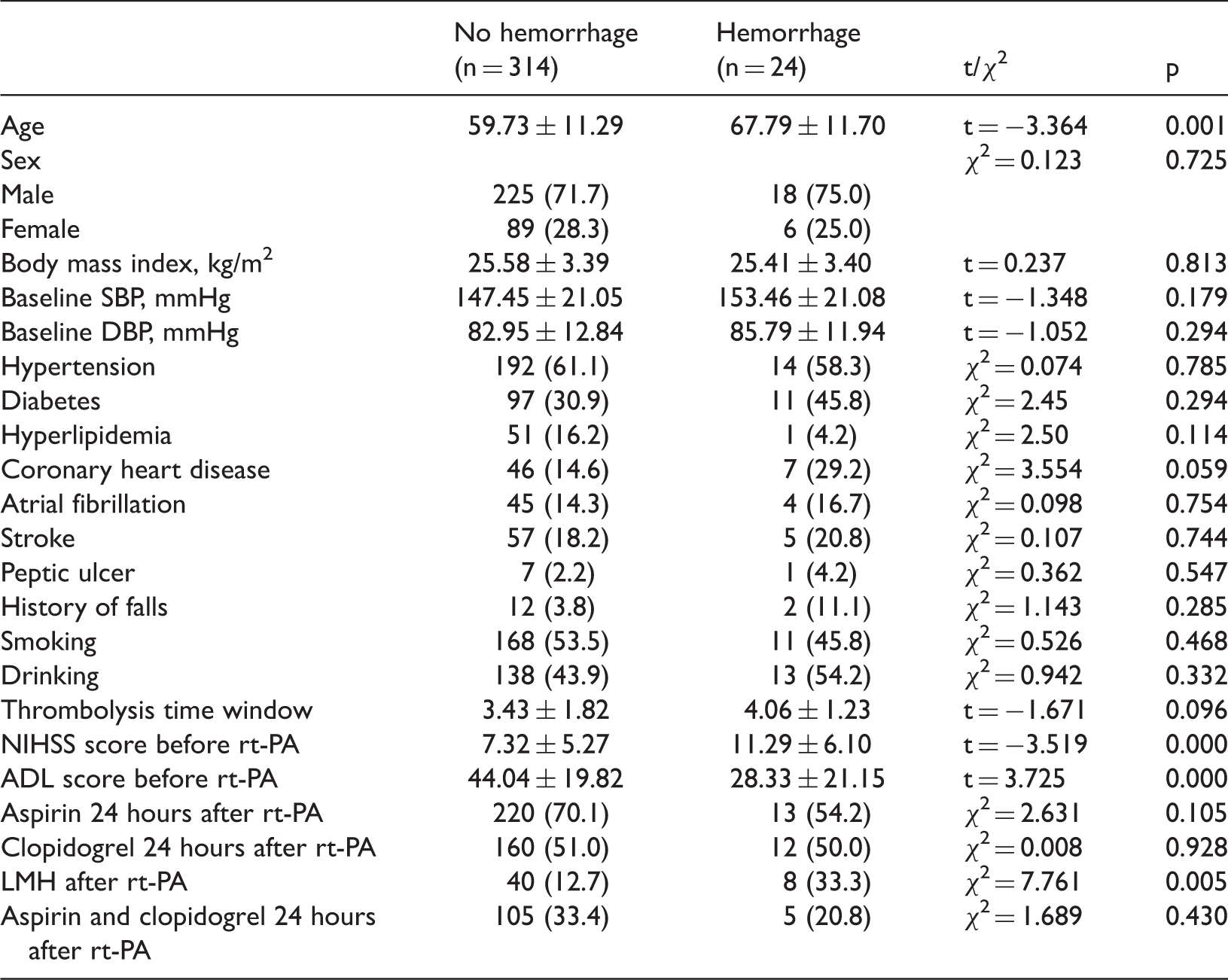
Data are presented as n (%) or mean ± standard deviation unless otherwise indicated.
ADL: activities of daily living; DBP: diastolic blood pressure; LMH: low-molecular-weight heparin; NIHSS: National Institutes of Health Stroke Scale; rt-PA: recombinant tissue plasminogen activator; SBP: systolic blood pressure.
Comparison of laboratory parameters between patients with and without urologic hemorrhage

Data are presented as mean ± standard deviation unless otherwise indicated.
INR: international normalized ratio.
Comparison of urinary catheter between patients with and without urologic hemorrhage

Data are presented as n (%).
UC: urinary catheter.
Logistic regression analysis of risk factors for urologic hemorrhage

Likelihood ratio test = 132.044, Cox and Snell R2 = 0.115, Nagelkerke R2 = 0.286.
ADL: activities of daily living; BUN: blood urea nitrogen; CI: confidence interval; LMH: low-molecular-weight heparin, NIHSS: National Institutes of Health Stroke Scale; OR: odds ratio; rt-PA: recombinant tissue plasminogen activator; PTA: prothrombin time activity; SE: standard error.
Analysis of the timepoint of catheterization and peripheral hemorrhage.

Data are presented as n (%).
rt-PA: recombinant tissue plasminogen activator; CTR: catheterization.
With respect to the relationship between the time of catheterization and the development of peripheral hemorrhage, most adverse events were related to catheterization after 1 to 24 hours of rt-PA therapy. The proportion was 75.0% (9 of 12 events) among patients with digestive hemorrhage and 58.3% (7 of 12 events) among patients with urologic hemorrhage.
Discussion
At present, rt-PA is the most widely used therapy for intravenous thrombolysis worldwide. 7 Most studies have mainly focused on the development of cerebral hemorrhagic events caused by rt-PA 8 ; relatively little is known about the risk factors for peripheral hemorrhage. One study focused on 1044 patients with AIS who underwent intravenous thrombolysis with rt-PA and found that hypertension at baseline was closely correlated with hemorrhagic events. 9 Blood pressure control (systolic blood pressure < 185 mmHg, diastolic blood pressure < 185 mmHg) has been confirmed to be beneficial in preventing hemorrhagic events after rt-PA therapy. 10 The NIHSS score is another important index for predicting hemorrhagic events after rt-PA therapy. An NIHSS score of >25 is generally thought to be a contraindication for rt-PA therapy. 11 Our analyses revealed the following risk factors: the systolic blood pressure at baseline, the NIHSS and ADL scores before rt-PA therapy, and administration of a single aspirin dose or aspirin combined with clopidogrel within 24 hours after rt-PA therapy. Various drugs such as cortisol, aspirin, and nonsteroidal anti-inflammatory drugs are known to potentially result in stomach ulcers and bleeding. 12
We identified the following risk factors for urologic hemorrhage: age, the NIHSS and ADL scores before rt-PA therapy, use of low-molecular-weight heparin after rt-PA therapy, the triglyceride concentration, and the prothrombin time activity. Low-molecular-weight heparin is generally believed to have satisfactory safety and a low risk of bleeding. 13 However, more attention should be given to patients undergoing rt-PA therapy, especially when they have other risk factors such as high age. No significant differences were found in sex, although this can likely be attributed to the small sample size. Male patients are usually more vulnerable to hemorrhagic events. 14 Potential selection bias could be another reason. For instance, fewer older patients underwent rt-PA therapy because of the higher risk with age. The male:female ratio was 2.6:1.0, which meant male-dominant sample. Solutions to these problems might include expansion of the sample size, inclusion of multiple populations, and proper randomization. The multivariate logistic regression analysis showed that catheterization was an independent risk factor for patients with AIS undergoing rt-PA therapy. Impairments during catheterization might give rise to hemorrhagic events.
In summary, gastric and urinary catheterization were respectively correlated with digestive and urologic hemorrhage in patients with AIS undergoing rt-PA therapy. Well-designed controlled studies with large samples are required to confirm our findings. Classification of bleeding as minor, moderate, and severe according to the Global Use of Strategies To Open coronary arteries (GUSTO) criteria should also be considered. 15
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was supported Beijing Municipal Administration of Hospitals Youth Program (QML20150805), as well as the Biological Medicine and Life Sciences Innovation Project (Z151100003915088), Beijing Science and Technology Commission.
