Abstract
Objective
To assess the diagnostic delay (DD) and physician-related DD (pDD) in patients with axial spondyloarthritis (SpA) and the potential benefits of a multidisciplinary clinic (MDC) approach.
Methods
A retrospective study was undertaken among patients with axial SpA, which aimed to analyse DD, pDD and their risk factors. The influence of pDD on disease outcomes was examined. The pDDs among consecutive SpA patients in an MDC cohort were compared with propensity score matched historical controls (1:1).
Results
A total of 208 patients with axial SpA formed the historical control group and 49 patients with axial SpA formed the MDC cohort after introduction of the MDC. The median DD and pDD in the historical controls were 25.5 and 10.0 months, respectively. A cut-off of pDD > 4 months was associated with more active disease and functional impairment. An initial visit to a non-rheumatologist was the most significant risk factor for pDD. Following MDC introduction, the median pDD decreased from 13 months to 1 month after adjustments were made for confounders such as sex, education level, history of smoking, human leukocyte antigen-B27 status and SpA/ankylosing spondylitis classification criteria.
Conclusion
The MDC was a promising approach that resulted in a reduced pDD among patients with axial SpA.
Introduction
Ankylosing spondylitis (AS) is a chronic disabling inflammatory disorder primarily affecting enthesis and axial skeleton joints.1–3 Due to the insidious onset of the symptoms such as low back pain, along with the slow radiographic progression of the disease (according to the modified New York criteria 4 ), prolonged diagnostic delay (DD) is a well-recognized feature of AS management.5–9 The DDs in AS, or the time interval between the onset of the symptoms and the correct diagnosis being made, were reported to range from 5 to 10 years.1,3,5 To identify the disease at an early stage, the Assessment of Spondyloarthritis International Society (ASAS) proposed new criteria for axial spondyloarthritis (SpA). 10 Axial SpA includes not only classic AS, but also nonradiographic disease, for which sensitive imaging such as magnetic resonance imaging has improved the ability to achieve an early diagnosis.5,7 However, DD still remains an issue in axial SpA, with a reported time gap of 2–8 years.11–13 It has been proposed that early diagnosis is probably related to better outcomes for axial SpA.6,14 Patients with late diagnosis (> 5 years) have significantly higher structural damage and more severely limited spinal mobility. 6 In addition, early diagnosis is crucial in order to increase the chance of achieving treatment targets. 14
Physicians play an important role in the early identification of disease, but in clinical practice in mainland China, due to a lack of a fully developed screening and triage system for rheumatic diseases by general practitioners, patients ‘randomly’ choose different medical specialists without professional guidance. Thus, physician-related DD (pDD), which is defined as the time interval between the initial visit to a physician and the time of axial SpA diagnosis, may contribute significantly to the overall DD. In this regard, the present study aimed to assess the DD and pDD of patients with axial SpA in a clinic in Shanghai, China. The risk factors and potential solutions to reduce DD or pDD were explored.
Patients and methods
Study design and patients
This retrospective study enrolled patients with axial SpA who visited the Department of Rheumatology, Renji Hospital, School of Medicine, Shanghai Jiaotong University, Shanghai, China between April 2014 and February 2016. The primary aim of the study was to investigate the DD and pDD of patients with axial SpA in this clinic. All patients fulfilled the axial SpA classification criteria of the ASAS, 10 and/or the modified New York criteria for AS. 4 Peripheral SpA and patients with psoriasis or inflammatory bowel disease were excluded. The DD and pDD in all axial SpA/AS patients were analysed and the relevant risk factors were identified. These patients were considered the historical controls.
The secondary aim of the study was to evaluate whether a multidisciplinary clinic (MDC) could shorten the pDD. An MDC was created in March 2016 in Renji Hospital, School of Medicine, Shanghai Jiaotong University, which composed of a rheumatologist (T.L.) and an orthopaedic surgeon (W.X.), who were responsible for the final diagnostic decision-making. In addition, a nurse specialist (C.Y.) and a medical assistant (J.H.), both of whom had received an arthritis nurse-training programme from the Prince Wales Hospital (Hong Kong), also played essential roles in the MDC. For this part of the study, consecutive newly diagnosed patients with axial SpA were enrolled between March 2016 and February 2017 by the MDC clinic using the same inclusion/exclusion criteria as described above. The pDDs and DDs in the MDC cohort were compared with the propensity score matched historical controls (1:1).
The study protocol was approved by the Institutional Review Board of Renji Hospital (no. 2013084k). The written informed consent was acquired from each patient and patient anonymity was ensured.
Data collection
Data on age at disease onset, time between onset of symptoms and initial visit to a physician, time of axial SpA diagnosis, education level, history of uveitis, history of smoking, family history, human leukocyte antigen (HLA)-B27 and medication use (such as tumour necrosis factor inhibitors [TNFi]) were collected from the medical charts. DD and pDD were calculated. The Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), 15 Bath Ankylosing Spondylitis Functional Index (BASFI) 16 and Bath Ankylosing Spondylitis Metrology Index (BASMI) 17 scores were collected.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 22.0. (IBM Corp., Armonk, NY, USA). Continuous data are presented as mean ± SD or median (interquartile range [IQR]), as appropriate based on the results of the Kolmogorov–Smirnov test. According to risk factors for DD and pDD, propensity score matching (1:1) was performed to minimize selection bias between the MDC cohort and the historical controls. Comparisons between the two groups were performed with the non-parametric Mann–Whitney U-test. Analysis of covariance (ANCOVA) was used to illustrate the influence of pDD on disease activity, physical function and axial flexibility. Two-sided P-values < 0.05 were considered statistically significant.
Results
The first part of this retrospective study enrolled 208 patients with axial SpA to provide historical control data regarding DD and pDD. Their demographic and clinical characteristics are summarized in Table 1. The mean ± SD age of this patient group was 35.5 ± 12.8 years and 149 (71.6%) were male. According to the available radiographic data at the time of diagnosis, 128 of 179 (71.5%) fulfilled the AS criteria. HLA-B27 positivity was demonstrated in 158 of 185 (85.4%) patients. The overall median (IQR) DD was 25.5 (4.0, 74.8) months and the overall mean ± SD DD was 57.9 ± 81.0 months. The median (IQR) pDD was 10.0 (0.5, 59.3) months. The median (IQR) disease duration after diagnosis to the time of retrospective chart review was 9.5 (0.0, 51.8) and the mean ± SD was 40.8 ± 65.1 months. Of note, during this period, 108 of 208 (51.9%) patients had prior exposure to at least one TNFi treatment.
Demographic and clinical characteristics of patients with axial spondyloarthritis (SpA) (n = 208) who participated in a study of the risk factors associated with diagnostic delay.
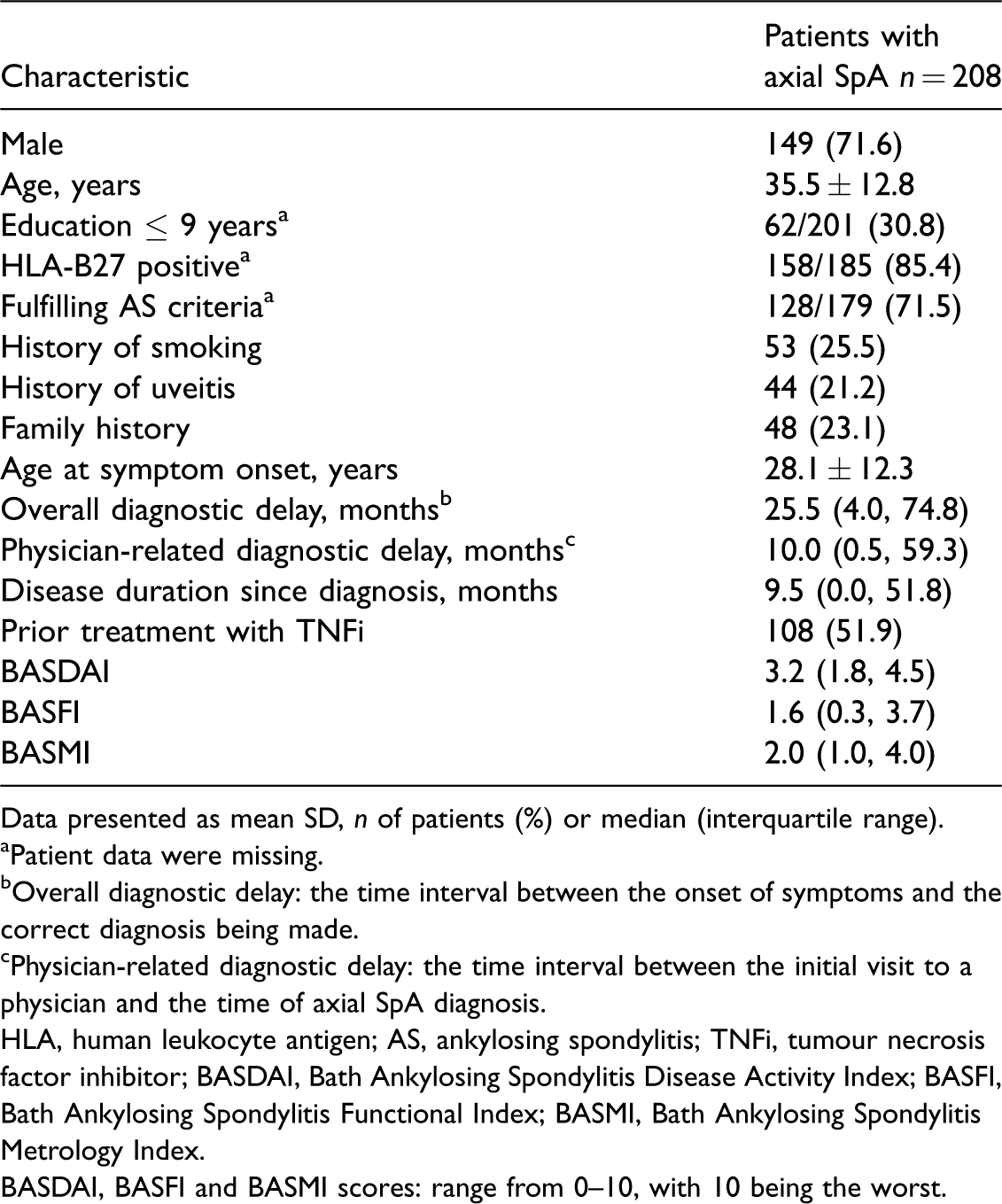
Data presented as mean SD, n of patients (%) or median (interquartile range).
aPatient data were missing.
bOverall diagnostic delay: the time interval between the onset of symptoms and the correct diagnosis being made.
cPhysician-related diagnostic delay: the time interval between the initial visit to a physician and the time of axial SpA diagnosis.
HLA, human leukocyte antigen; AS, ankylosing spondylitis; TNFi, tumour necrosis factor inhibitor; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index.
BASDAI, BASFI and BASMI scores: range from 0–10, with 10 being the worst.
Patients with a cut-off of pDD > 4 months showed significantly higher BASDAI and BASMI scores than those with pDD ≤ 4 months (P = 0.025 and P = 0.004, respectively; Table 2). These differences of BASDAI and BASMI scores between the two groups were similar (P = 0.025 and P = 0.003, respectively) after adjustment for the time since diagnosis and exposure to TNFi using ANCOVA.
Comparison of Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), Bath Ankylosing Spondylitis Functional Index (BASFI) and Bath Ankylosing Spondylitis Metrology Index (BASMI) between patients with axial spondyloarthritis (n = 208) stratified according to the physician-related diagnostic delay.

Data presented as median (interquartile range).
aNon-parametric Mann–Whitney U-test; bAdjusted for the time since diagnosis and exposure to tumour necrosis factor inhibitor using analysis of covariance; NS, not statistically significant (P ≥ 0.05).
Male patients and patients with HLA-B27, history of smoking, patients with AS (compared with non-radiographic axial SpA [nr-aSpA]) and those who had an initial visit to a non-rheumatologist had a significantly longer overall DD (Table 3) (P < 0.05 for all comparisons). Moreover, pDD was significantly longer in patients who initially visited to a non-rheumatologist and in patients with a lower education level (<9 years). With regard to physician speciality, 125 of 208 (60.1%) patients had their first consultation with an orthopaedist, but only 42 of 208 (20.2%) patients received their diagnosis of axial SpA from an orthopaedist (Figure 1). In comparison, only 21 of 208 (10.1%) patients had their first consultation with a rheumatologist and 143 of 208 (68.8%) patients received their diagnosis of axial SpA from a rheumatologist.

The physician’s speciality at the first consultation compared with the speciality of the physician who made the diagnosis of axial spondyloarthritis (SpA) in patients (n = 208) who participated in a study of the risk factors associated with diagnostic delay.
Risk factors for diagnostic delays in patients with axial spondyloarthritis (SpA) (n = 208) who participated in a study of the risk factors associated with diagnostic delay.
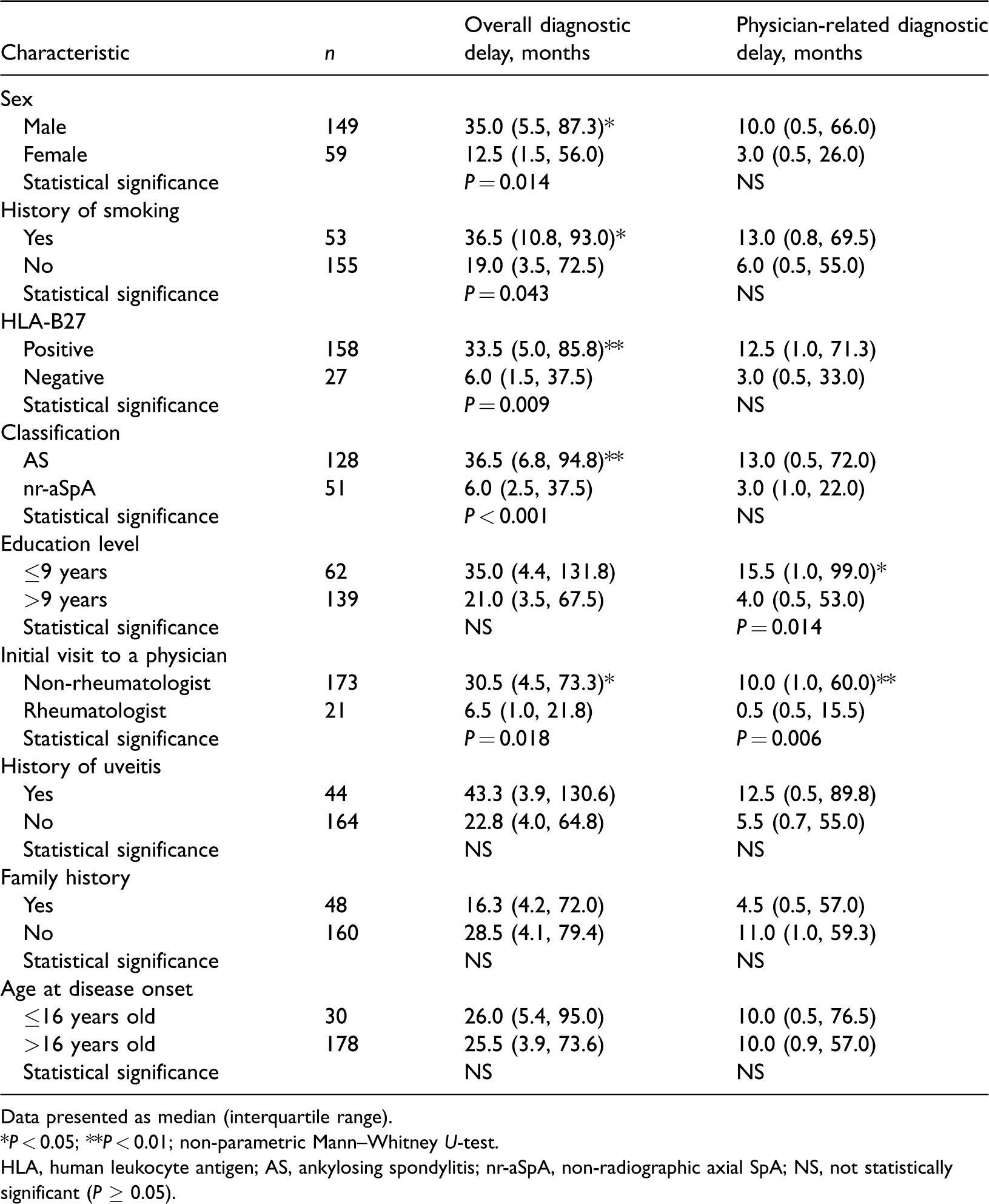
Data presented as median (interquartile range).
*P < 0.05; **P < 0.01; non-parametric Mann–Whitney U-test.
HLA, human leukocyte antigen; AS, ankylosing spondylitis; nr-aSpA, non-radiographic axial SpA; NS, not statistically significant (P ≥ 0.05).
The difference of pDD was compared between the MDC cohort (n = 49) and the historical controls. Propensity scores (1:1) were used to match all the relevant confounders, e.g. sex, history of smoking, HLA-B27, AS or nr-aSpA diagnosis and education level (Table 4). The median pDD was significantly shorter in the MDC cohort compared with the historical controls (1.0 versus 13.0 months, respectively; P = 0.026). The median overall DD was also significantly shorter in the MDC cohort compared with the historical controls (12.0 versus 46.0 months, respectively; P = 0.049) (Table 5).
Comparison of the characteristics of the patients with axial spondyloarthritis (SpA) in the multidisciplinary clinic (MDC) cohort and the historical controls using propensity score matching (1:1).

Data presented as n of patients (%).
aPatient data were missing.
SMD, standardized mean difference: an SMD < 0.2 can be considered as a sign of balance.
Comparison of the diagnostic delays (DDs) of the patients with axial spondyloarthritis (SpA) in the multidisciplinary clinic (MDC) cohort and the historical controls using propensity score matching (1:1).

Data presented as median (interquartile range).
Discussion
The issue of DD in axial SpA/AS, and pDD in particular, to some extent reflect the efficiency of the health system dealing with this disease. According to this current retrospective study, the median (IQR) overall DD was 25.5 (4.0–74.8) months and the median (IQR) pDD was 10.0 (0.5–59.3) months in the historical controls. These current findings were comparable with a recent cross-sectional study undertaken in France, which reported a median DD of 2.0 years; 13 but significantly longer than another study from Spain, which reported a median DD of 4.2 months. 18 The direct comparison of DDs among studies is difficult not only because of the disparities in the models of health care delivery, but also because of the subjective nature of the patients’ reporting of their symptom onset. The estimates of overall DD are likely to be less reliable than the measurement of pDD, which relies on documented dates in the patient’s medical records. Furthermore, these current data demonstrated that a pDD with a cut-off > 4 months was associated with more active disease and more functional impairment, even after adjusting for treatment exposure to TNFi biological agents. This underscores the significance of prolonged pDD in terms of its impact on disease outcome.
To further investigate the possible factors that contribute to pDD, the current study observed that an initial visit to a non-rheumatologist was the most significant risk factor. Only 21 of 208 (10.1%) patients had their first consultation with a rheumatologist and 125 of 208 (60.1%) patients initially saw an orthopaedist. In contrast, 143 of 208 (68.8%) patients received their diagnosis of axial SpA from a rheumatologist and only 42 of 208 (20.2%) patients received their diagnosis of axial SpA from an orthopaedist. These findings appear to be unique to our clinical centre and should only be cautiously extrapolated to a wider region. For example, in a study of 102 patients with axial SpA in Korea, the specialities of the physicians who were involved before the diagnosis of SpA being made by a rheumatologist included orthopaedists (86%), neurosurgeons (28%), general physicians (5%), physiatrists (5%) and others (22%). 11 A study in Japan showed that 69% of the patients with AS visited two or more doctors prior to AS diagnosis and orthopaedic surgeons were the most commonly visited (62%). 19 In Indian patients with AS, most incorrect initial diagnoses were made by orthopaedists (75.9%), followed by general physicians (50%) and rheumatologists (12%). 20 Thus, setting up an MDC combining a rheumatologist and an orthopaedist working together would appear to be a reasonable way to reduce the pDD in axial SpA.
The MDC is a well-recognized working model designed to deliver patient-centred care by centralizing relevant practitioners at a single physical site. Evidence has shown that the MDC model is associated with improved efficiency and completeness of care for many chronic diseases.21–23 Indeed, the current data demonstrated that the pDD was significantly shorter in patients with axial SpA who were managed by the MDC compared with the propensity score matched historical controls (1 versus 13 months, P = 0.026), after adjustments for possible confounders such as sex, education level, history of smoking, HLA-B27 status and SpA/AS criteria.
In conclusion, the formation of an MDC appears to be a promising approach to partially solving the problem of pDD in patients with axial SpA, although follow-up studies to see whether patients managed by the MDC ultimately have a better long-term outcome will be crucial to further validate this approach.
Footnotes
Acknowledgements
We would like to thank Professor LS Tam from the Prince of Wales Hospital, Hong Kong, for providing the nurse training programme; and Dr Malcolm Handel from Janssen-Cilag Pty Ltd for his valuable comments on the manuscript.
Author contributions
All authors took part in drafting the article or revising it critically for important intellectual content and approved the final version to be published. Weiwei Xin, Ting Li and Shuang Ye contributed to the conception and design of the study. Jiajie Li, Yuan Xu, Yuling Chen, Chunhua Ye, Jiaxian Huang and Liping Qian contributed to data collection. Jiajie Li, Yuan Xu, Yuling Chen and Shuang Ye performed the analysis and interpretation of data. Jiajie Li, Yuan Xu and Yuling Chen contributed equally to this article.
Declaration of conflicting interest
Dr Shuang Ye reports receiving honoraria from Pfizer, Roche, Johnson & Johnson and Abbive. All authors declare that there were no other potential conflicts of interest relevant to this article.
Funding
A Research Project Grant (no. 11411951700; to S.Y.) was provided by the Foundation of Science and Technology Commission of Shanghai Municipality. Support was also provided by Janssen Asia Pacific.
