Abstract
Background:
Tumor necrosis factor inhibitors are the most widely used biological disease-modifying antirheumatic drugs for radiographic axial spondyloarthritis. After achieving remission with tumor necrosis factor inhibitor treatment, experts recommend tapering tumor necrosis factor inhibitor. However, biomarkers for successful tumor necrosis factor inhibitor tapering in radiographic axial spondyloarthritis have not been identified.
Objectives:
To identify biomarkers associated with successful tumor necrosis factor inhibitor tapering in patients with radiographic axial spondyloarthritis.
Design:
We prospectively collected blood samples from radiographic axial spondyloarthritis patients at single tertiary hospital.
Methods:
Patients with radiographic axial spondyloarthritis who achieved remission (axial spondyloarthritis disease activity score < 1.3) after treatment with tumor necrosis factor inhibitor were enrolled. Baseline demographics, medication history, and laboratory data were collected when the tumor necrosis factor inhibitor dose was tapered. The percentage of helper T cell subtypes (Th1/Th2/Th17/Th22) in peripheral blood, and serum levels of tumor necrosis factor-α, interleukin-12, IL-17A, IL-22, IL-23, interferon (IFN)-γ, soluble CD14, and zonulin, were measured. Patients were assigned to tumor necrosis factor inhibitor tapering success (axial spondyloarthritis disease activity score < 2.1) or failure (axial spondyloarthritis disease activity score ⩾ 2.1) groups according to disease activity (assessed at 12 weeks posttumor necrosis factor inhibitor tapering).
Results:
Twenty radiographic axial spondyloarthritis patients were enrolled (median age, 31.0 years; 65% males). Most (80%) were positive for human leukocyte antigen-B27. The change of axial spondyloarthritis disease activity score in the tumor necrosis factor inhibitor-tapering failure group was 1.36, while that in the tumor necrosis factor inhibitor-tapering success group was 0.07. The percentage of Th1 and Th17 cells was significantly lower, and that of Th2 cells higher, in the tumor necrosis factor inhibitor-tapering success group. In addition, serum levels of IL-12, IL-17A, IL-22, IFN-γ, tumor necrosis factor-α, zonulin, and soluble CD14 were significantly lower in the tumor necrosis factor inhibitor-tapering success group.
Conclusion:
Patients with radiographic axial spondyloarthritis who achieve successful tumor necrosis factor inhibitor tapering had lower percentages of Th1 and Th17 cells, a higher percentage of Th2 cells, and lower serum levels of IL-12, IL-17A, IL-22, IFN-γ, tumor necrosis factor-α, zonulin, and soluble CD14 at the time of tumor necrosis factor inhibitor tapering. These findings may help to identify patients with radiographic axial spondyloarthritis for whom tumor necrosis factor inhibitor tapering is appropriate.
Introduction
Radiographic axial spondyloarthritis (r-axSpA), previously called ankylosing spondylitis (AS), is a systemic inflammatory arthritis, the predominant manifestations of which are inflammation and pain in axial joints.1,2 The spectrum of SpA includes r-axSpA, nonradiographic-axSpA, psoriatic arthritis, inflammatory bowel disease-related SpA, reactive arthritis, and SpA, which can accompany several extra-articular symptoms such as uveitis, psoriasis, and inflammatory bowel disease.3,4 Treatment goals for axSpA include reducing pain and stiffness, improving quality of life, controlling extra-articular manifestations, preventing critical comorbidities such as cardiovascular disease, and reducing spinal structural progression (i.e., syndesmophyte development).5 –7 The first-line treatment of choice is nonsteroidal anti-inflammatory drugs; biologic disease-modifying antirheumatic drugs (bDMARDs), and Janus kinase (JAK) inhibitors are recommended for patients who do not respond to first-line therapy.5 –7 Although many studies have been conducted to find biomarkers of r-axSpA and develop therapeutic agents by targeting key molecules, 8 only blocking agents for tumor necrosis factor (TNF)-α and interleukin (IL)-17 have demonstrated significant therapeutic effects in patients with r-axSpA and are currently in use. TNF-α plays a critical role in the pathogenesis of axSpA 9 ; indeed, several TNF inhibitors (TNFis) are used to currently treat patients with r-axSpA.5 –7
With respect to axSpA, TNFis are the most widely used bDMARDs in clinical practice. A meta-analysis shows that the monoclonal antibody (Ab) form of TNFis is the most effective for controlling disease activity in patients with axSpA. 10 TNFis also slow spinal structural progression and improve quality of life in patients with axSpA.11,12 Although TNFis are a well-known effective treatment for axSpA, they are associated with several potential adverse events and are inconvenient to administer. TNFi also increase the risk of tuberculosis and other infections.13,14 The risk of nasopharyngitis, headache, and injection-site reactions is also higher 15 ; indeed, pain and skin reactions caused by TNFi injections can reduce drug adherence significantly. 16 The current treatment guidelines for axSpA recommend dose quotient (DQ) reduction (tapering) of TNFi in patients who achieve sustained remission; however, the strength of this recommendation is weak.5,6 One meta-analysis shows that TNFi tapering increases the incidence of disease flares in patients with spondyloarthritis (SpA). 17 Several studies reveal that human leukocyte antigen-B27 status, disease activity, comorbidities, duration of remission, and DQ of TNFi affect disease flares after TNFi tapering; however, these studies evaluated only demographic and clinical information, and were retrospective in nature.18,19 Identifying patients for whom TNFi tapering is appropriate is important, and further research to identify predictors for successful tapering is needed.
Here, we enrolled patients with r-axSpA who achieved remission after TNFi treatment and then investigated several biomarkers, including cytokine levels and percentages of helper T (Th) cells, to identify biomarkers associated with successful TNFi tapering.
Methods
Patients
Patients with r-axSpA who satisfied the following criteria were enrolled from a single tertiary hospital (Konkuk University of Medical Center): (1) fulfilled the 1984 modified New York criteria for AS 20 ; (2) achieved remission by TNFi treatment according to the Axial Spondyloarthritis Disease Activity Score (ASDAS < 1.3) for at least 6 months 21 ; (3) were over 18 years of age; and (4) consented to reduce the DQ of TNFi. Patients with other forms of arthritis, autoimmune diseases, or cancers were excluded. The patients were enrolled during December 2022 to May 2024. The study was conducted as a prospective cohort study, and the study duration was 12 weeks. The DQ of each TNFi was reduced consistently to 66.6% by increasing the interval of injection (i.e., adalimumab subcutaneous (SC), 40 mg injection every 3 weeks; golimumab SC, 50 mg injection every 6 weeks; golimumab IV, 2 mg/kg intravenous injection every 12 weeks; infliximab SC, 120 mg injection every 3 weeks). Baseline characteristics, including demographic, laboratory, and clinical information, as well as peripheral blood samples, were collected on the day of TNFi tapering. Disease activity at baseline was measured using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) 22 and the ASDAS-erythrocyte sedimentation rate (ESR)/C-reactive protein (CRP), 21 and then again 12 weeks after TNFi tapering. The functional impairment index (Bath Ankylosing Spondylitis Functional Index (BASFI)) 23 was also measured. At 12 weeks post-TNFi tapering, patients with an ASDAS < 2.1 were assigned to the TNFi tapering success group, and those with an ASDAS ⩾ 2.1 were assigned to the TNFi tapering failure group. 21 The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Prior to enrolment, all participants were made aware of the purpose of the study and the involved, and full understanding was ensured. All participants provided written informed consent. The experimental protocol was approved by the Institutional Review Board of Konkuk University Medical Center (KUMC 2022-03-050).
Peripheral blood sampling of patients with r-axSpA
Peripheral blood samples were taken, and peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll–Paque density gradient centrifugation (GE Healthcare Biosciences, Uppsala, Sweden). Serum was collected in an Eppendorf tube and stored at −20°C prior to testing by enzyme-linked immunosorbent assay (ELISA).
Flow cytometry
CD4+ T cells were isolated from PBMCs using magnetic beads (StemCell Technologies, Vancouver, BC, USA) and then stained with monoclonal antibodies. Surface staining was performed using FITC-conjugated anti-CD196 (CCR6, BioLegend, San Diego, CA, USA), PE-conjugated anti-CD194 (CCR4, BioLegend), PE-Cy7-conjugated anti-CCR10 (BD Biosciences, San Diego, CA, USA), and APC-conjugated anti-CD183 (CXCR3, BioLegend). Cells were analyzed using a FACSCalibur flow cytometer (BD Pharmingen, Franklin Lakes, NJ, USA). The following markers were used to identify different Th cell subsets: Th1: CXCR3+CCR4−CCR10−CCR6−; Th2: CXCR3−CCR4+CCR10−CCR6−; Th17: CXCR3−CCR4+CCR10−CCR6+; and Th22: CXCR3−CCR4+CCR10+CCR6+. 24
Enzyme-linked immunosorbent assay
ELISA was used to measure the serum levels of IL-12, IL-17A, IL-22, IL-23, interferon (IFN)-γ, TNF-α, soluble CD14 (sCD14, all from R&D Systems, Minneapolis, MN, USA) and zonulin (Cusabio, Houston, TX, USA). Cytokine levels ranged from 10–2000 pg/mL. A standard curve was constructed by plotting the optical density against the log concentration of each recombinant cytokine, and the curve was used to determine cytokine concentrations in the test samples at 450 nm using a microplate reader (SpectraMax ABS plus, Molecular Devices, CA, USA) with SoftMax pro ver. 7.1 software (Molecular Devices, San Jose, CA, USA).
Statistical analysis
Continuous data are presented as the mean ± standard deviation or as the median and interquartile range (IQR). Noncontinuous variables are presented as numbers and percentages. Student’s t-test or the Mann–Whitney test was used to assess continuous variables after checking for normality, and the Chi-square or Fisher’s exact test was used to assess noncontinuous variables. Receiver operator characteristic (ROC) curves, along with the area under the curve (AUC), sensitivity, and specificity were evaluated to assess the diagnostic accuracy of each biomarker associated with successful TNFi tapering. The percentage of each Th subtype and serum cytokine levels were assigned to one of four categories based on the median value and the 25% and 75% IQR when applied to the ROC curve. Since the sample size was small and no previous studies have investigated biomarkers for TNFi tapering in r-axSpA, we determined the cut-off values based on our dataset. Given the limited sample size, setting a continuous cut-off value was not feasible; therefore, we employed the median and IQR for classification. In category 1, the 1st quartile was lower than the 25th percentile; in category 2, the 2nd quartile was higher than the 25th percentile but lower than the median value; in category 3, the 3rd quartile was higher than the median value but lower than the 75th percentile; and in category 4, the 4th quartile was higher than the 75th percentile. The cut-off values were selected considering both sensitivity and specificity. Statistical significance was set at p < 0.05. All statistical analyses were performed using Prism 9.0 (GraphPad Software Inc., San Diego, CA, USA).
Results
Comparison of baseline characteristics between the TNFi-tapering success and failure groups
A total of 20 patients with r-axSpA were enrolled; of these 12 were assigned to the TNFi success group. The median age of the 20 enrolled patients was 31.0 years, and 13 (65.0%) were male. The baseline age, sex, BMI, disease duration, and duration of TNFi treatment prior to enrolment were similar between the TNFi-tapering success and failure groups. Adalimumab was the most frequently used TNFi (14/20, 70.0%); however, fewer patients in the TNFi-tapering failure group than in the success group received adalimumab (3/8 (37.5%) vs 11/12 (91.7%), respectively; p = 0.009). The baseline disease activity (BASDAI, ASDAS-ESR, ASDAS) and functional impairment (BASFI) indices were comparable between the two groups. There was no difference in laboratory findings and axSpA manifestations (i.e., peripheral arthritis, uveitis, psoriasis, dactylitis, and inflammatory bowel disease) between the groups (Table 1). Net changes in the ASDAS, ASDAS-ESR, BASDAI, and BASFI scores between baseline and 12 weeks after TNFi tapering were significantly higher in the TNFi-tapering failure group (Table 2).
Baseline characteristics of patients with r-axSpA.
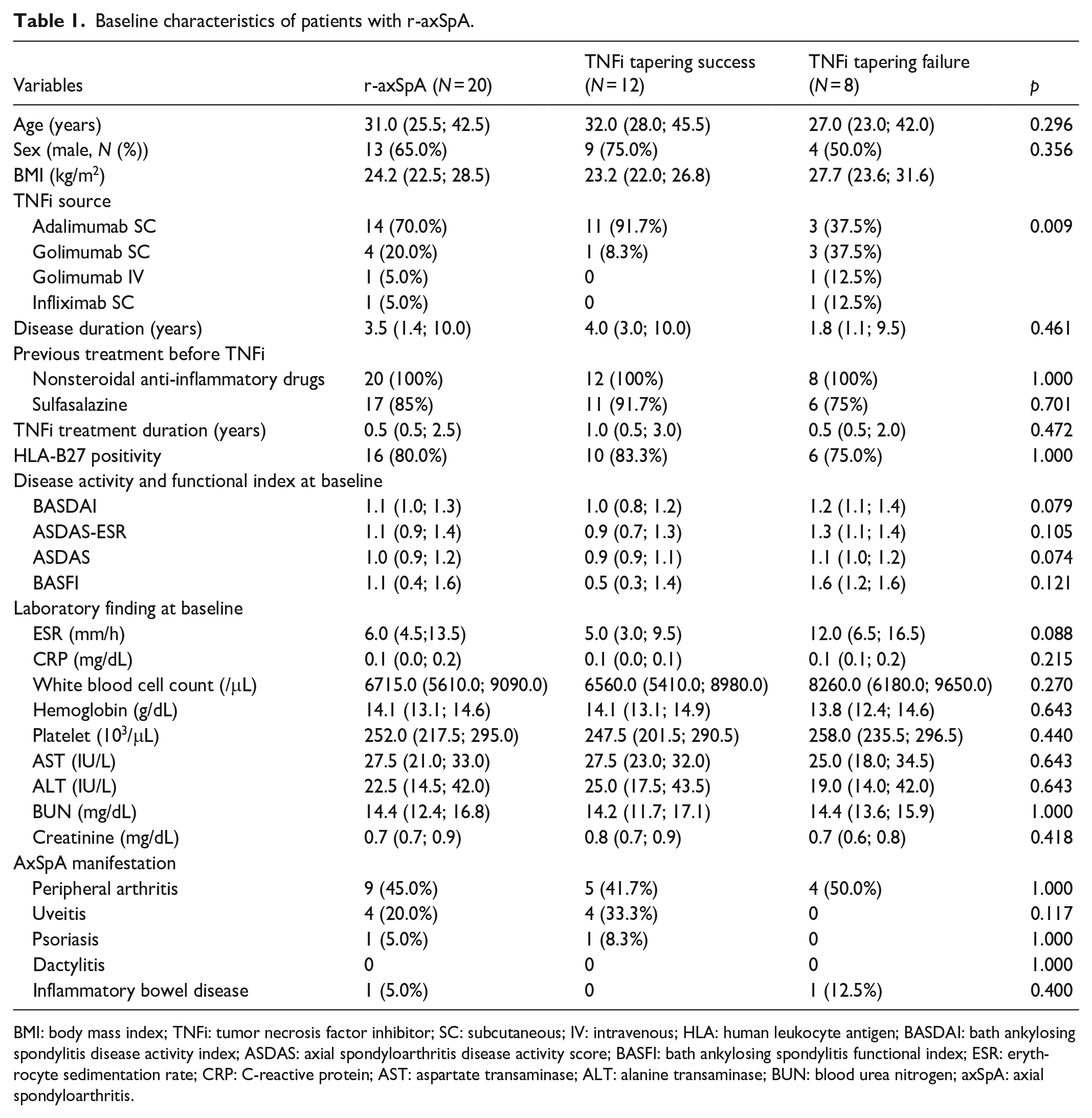
BMI: body mass index; TNFi: tumor necrosis factor inhibitor; SC: subcutaneous; IV: intravenous; HLA: human leukocyte antigen; BASDAI: bath ankylosing spondylitis disease activity index; ASDAS: axial spondyloarthritis disease activity score; BASFI: bath ankylosing spondylitis functional index; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; AST: aspartate transaminase; ALT: alanine transaminase; BUN: blood urea nitrogen; axSpA: axial spondyloarthritis.
Comparison of net change of disease activity score, functional index, and acute phase reactants between TNFi tapering success and failure groups.

BASDAI: bath ankylosing spondylitis disease activity index; ASDAS: axial spondyloarthritis disease activity score; BASFI: bath ankylosing spondylitis functional index; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein.
Values are presented as median with interquartile range.
Percentage of different Th cell subtypes and serum cytokine levels in the TNFi-tapering success and failure groups
Peripheral blood was collected at the time of TNFi tapering, and PBMCs were isolated. CD4+ T cells were isolated from the PBMCs and the percentage of each Th cell subtype within the CD4+ population was measured. CD4+ T cells comprised 22.52% of PBMCs (mean total PBMC count, 3.852 × 107 cells/mL; mean CD4+ T cell count, 0.867 × 107 cells/mL). The percentage of CXCR3+CCR4-CCR10−CCR6− (Th1) and CXCR3−CCR4+CCR10−CCR6+ (Th17) cells was significantly lower in the TNFi-tapering success group than in the failure group (median 16.8% vs 56.8%, respectively, p < 0.001 for Th1; median 20.1% vs 25.8%, respectively, p < 0.001 for Th17). The percentage of CXCR3−CCR4+CCR10−CCR6− (Th2) cells in the TNFi-tapering success group was higher (median 48.9%; IQR 45.1%–56.0%) than that in the failure group (median 27.9%; IQR 23.1%–28.6%). The percentage of CXCR3−CCR4+CCR10+CCR6+ (Th22) cells was comparable between the two groups (Figure 1 and Supplemental Table 1).

Percentage of helper T (CD4+) cells in the TNFi-tapering success and failure groups. CD4+ T cells were extracted from PBMCs and expression of cell surface markers (CXCR3, CCR4, CCR10, and CCR6) was analyzed by flow cytometry. The percentages of CXCR3+CCR4−CCR10−CCR6- (type 1 helper T cell (Th1)), CXCR3-CCR4+CCR10−CCR6− (Th2), CXCR3−CCR4+CCR10−CCR6+ (Th17), and CXCR3−CCR4+CCR10+CCR6+ (Th22) were measured.
There were significant differences between the groups with respect to serum cytokine levels. IL-17A, IL-12, IL-22, TNF-α, and IFN-γ levels in the TNFi-tapering success group were lower than those in the failure group. There was no significant difference in serum IL-23 levels between the two groups. Levels of the intestinal inflammatory marker (sCD14) and the intestinal permeability marker (zonulin) were significantly lower in the TNFi-tapering success group than in the failure group (Figure 2). The flow cytometry and ELISA data are summarized in Supplemental Table 1.

Serum levels of cytokines and markers related to gut dysbiosis/inflammation. Serum samples were obtained from the enrolled r-axSpA patients and the levels of interleukin (IL)-12, IL-17A, IL-22, IL-23, interferon-γ, TNF-α, zonulin, and soluble CD14 were measured by ELISA.
Accuracy of Th cell percentages and serum cytokine levels for successful TNFi tapering
The AUC for the ROC was evaluated based on the quartile for each Th cell subtype (Figure 3(a)) and on the quartile for each serum cytokine (Figure 3(b)). The percentage of Th1 (cut-off value, lower than 16.4%) and Th2 (cut-off value, upper than 52.5%) cells showed the greatest diagnostic accuracy (AUC = 0.969, 95% CI: 0.904–1.000 for both), whereas IL-17A (cut-off value, lower than 0.141 ng/mL), IFN-γ (cut-off value, lower than 0.447 ng/mL), and sCD14 (cut-off value, lower than 1.854 ng/mL) levels were the most accurate markers for successful TNFi tapering (AUC = 0.969, 95% CI: 0.904–1.000 for IL-17A, IFN-γ, and sCD14, Figure 3(a) and (b)).
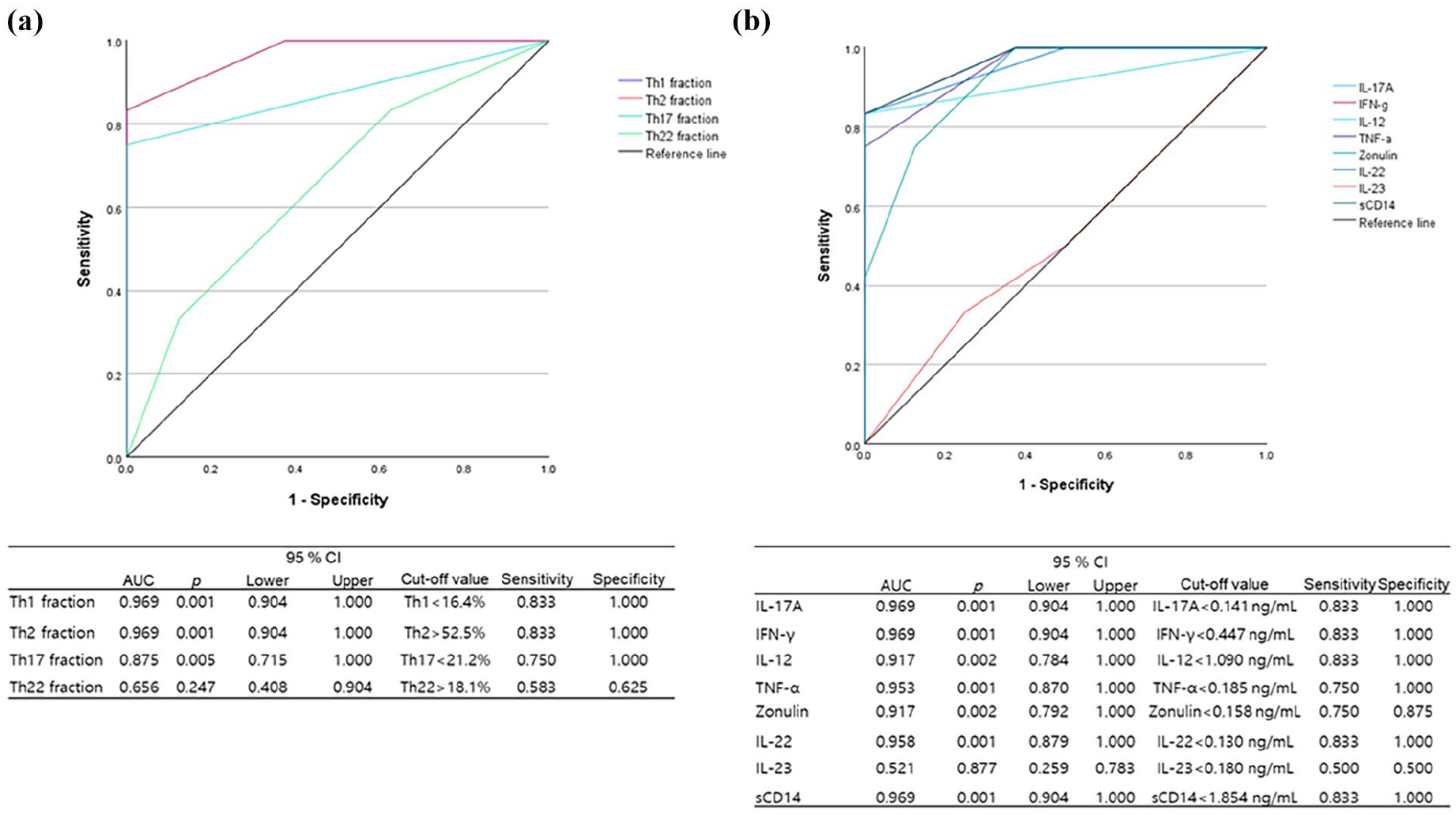
ROC curve analysis, and the sensitivity/specificity of potential biomarkers for successful TNFi tapering. ROC curve and AUC; the cut-off value, along with sensitivity/specificity for successful tapering of TNFi, were calculated with respect to (a) the percentage of different helper T cell subsets, and (b) serum cytokine levels.
Discussion
This study compared the percentages of Th cell subtypes, as well as cytokine levels, in the peripheral blood of r-axSpA patients who succeeded in tapering TNFi with those who did not, and identified and biomarkers that were associated with successful TNFi tapering. Patients with lower percentages of Th1 and Th17 cells, and a higher percentage of Th2 cells, at the time of TNFi tapering were more likely to succeed. In addition, low serum levels of IL-17A, IFN-γ, IL-12, TNF-α, IL-22, zonulin, and sCD14 were associated with successful TNFi tapering. These findings may help to identify r-axSpA patients most suited to TNFi tapering after they have achieved sustained remission with TNFi therapy.
Various pathological immune cells play a role in the pathogenesis of axSpA. 2 Innate cells such as type 3 innate lymphoid cells (ILC3), 25 mucosa-associated invariant T (MAIT) cells, 26 invariant natural killer cells, 27 γδ T cells, 28 mast cells, 29 neutrophils, 30 and adaptive immune cells (e.g., Th17, 31 Th22, 32 and enthesial-resident RAR-related orphan receptor-γt+CD3+CD4-CD8- T cells 33 ), also play pathological roles in r-axSpA. Although many types of immune cell play pathological roles in axSpA, most are not easy to obtain because of their location (i.e., they are gut-, joint-, or enthesis-resident). In addition, ILC3, MAIT, and γδ T cells comprise <1% of the total PBMC population,25,34 which limits analysis of these cells as clinically useful biomarkers in axSpA. Analysis of the Th cell fraction in peripheral blood is easier, making it a more suitable source of potentially clinically useful biomarkers; indeed, Th cells comprise approximately 25%–50% of the total PBMC population. 35 Patients with r-axSpA have high levels of circulating Th17. 31 Nonresponders to TNFi have a high percentage of Th17 cells, whereas TNFi responders show a significant reduction in circulating Th17 cells. 36 The Th22 population in the peripheral blood of r-axSpA patients is higher than that in healthy individuals, and these cells are a major of source of IL-22.31,32 IL-22 plays a critical role in de novo formation of abnormal bone in the spine of r-axSpA mice model, 33 and it also increases osteoclastogenesis of patients with r-axSpA in vitro. 25 The percentage of Th1 cells in the peripheral blood of patients with r-axSpA is higher, whereas the percentage of Th2 cells in lower, than that in in healthy controls. 37 Treatment with TNFi leads to a reduction in Th1 cytokine (IFN-γ and TNF-α) levels, and an increase in Th2 cytokine levels (IL-4 and IL-10), compared with before treatment. 38 In this study, we found that suppression of Th1 and Th17 cells, and an increase in the percentage of Th2 cells, was associated with successful TNFi tapering; however, there was no significant difference in the percentage of Th22 cells between the two groups. These findings suggest that clinicians should check that r-axSpA patients have low enough Th1 and Th17 levels and high enough Th2 levels before considering TNFi DQ reduction.
The IL-23/IL-17 axis has been suggested as a key pathological cytokine pathway in axSpA, and other cytokines such as TNF-α and IL-22 are known to play important roles in axSpA pathogenesis. 39 TNFi and IL-17A inhibitors are recommended therapies for r-axSpA.5–7 Patients with r-axSpA also have high serum levels of IL-23, IL-12, and IFN-γ.38,40,41 Here, we found that all of the aforementioned cytokines, except IL-23, were lower in the TNFi-tapering success group than in the failure group. Ustekinumab (an inhibitor of IL-12/IL-23 p40) failed in clinical trials involving patients with axSpA. 42 Some hypothetical explanations for the failure of IL-23 inhibitors have been suggested, 43 and no further clinical trials of IL-23 inhibitors are being conducted in patients with axSpA. In this study, we found that suppression of both Th1 (IFN-γ) and Th17 cytokines (IL-17A) was associated with successful TNFi tapering. A previous study of patients with rheumatoid arthritis (RA) showed that patients in whom drug reduction was successful had lower serum IFN-γ and IL-17A levels at the time of drug tapering than those who had a disease flare after drug reduction. 44 The main difference between the present study and previous study is that the disease is fundamentally different (RA vs r-axSpA); also, the previous study was not limited to TNFi (it included all conventional synthetic and bDMARDs). 44 However, the interesting point is that concomitant suppression of Th1- and Th17-related cytokines may associate with successful drug reduction, regardless of the type of inflammatory arthritis or medication used.
Gut dysbiosis, increased gut permeability, and inflammation are well-established pathological processes in patients with axSpA. 45 Zonulin increases gut permeability, which is linked to gut dysbiosis. 46 sCD14 is a marker of inflammation-induced gut dysbiosis.47,48 Previous studies report that both zonulin and sCD14 are increased in patients with axSpA, reflecting the degree of gut dysbiosis and inflammation.46,49 Here, we found that serum levels of zonulin and sCD14 were significantly lower in the TNFi-tapering success group than in the failure group, suggesting that the recovery from gut inflammation/permeability may be associated with successful drug reduction.
AxSpA is a heterogeneous disease; therefore, clinical manifestations, prognosis, and treatment responses differ between patients. In such cases, a personalized (precision) medicine approach is required to treat the disease and predict prognosis. 50 Several studies aimed to establish precision medicine approaches to axSpA, including identification of markers that predict a good response to TNFi 51 and multiomics analyses. 52 Patients with r-axSpA who had baseline enthesitis, psoriasis, and low CRP levels had lower probability of achieving partial remission. 53 Female sex, concomitant steroid use, persistently high inflammatory markers, high BASFI and BASDAI indices were negative predictors for TNFi response and continuation rate in patients with r-axSpA.54,55 Biologic tapering is recommended for patients who achieve sustained remission; however, the supporting evidence is weak.5,6 In addition, a recent meta-analysis revealed that TNFi tapering increases the risk of flares in patients with SpA. 17 Tapering TNFi showed benefits in term of patient compliance, drug exposure risk, and economic burden.56,57 Two retrospective cohort studies identified some clinical predictors of successful TNFi tapering in patients with r-axSpA18,19; however, these studies only evaluated clinical and demographic parameters. This study is, to the best of our knowledge, the first prospective study to identify potentially meaningful biomarkers that are associated with successful tapering of TNFi in patients with r-axSpA.
This study has several limitations. First, the sample size is small, and sample size calculation was omitted because no related previous study existed. Second, more adalimumab users were included in the TNFi success group, and the influence of each TNFi on tapering may differ. Third, the follow-up duration after tapering TNFi (12 weeks) is relatively short. Further studies should enroll more patients and use extended follow-up times. Fourth, the TNFi DQ reduction was set at 66.6%; therefore, the results may not be the same if a greater reduction in TNFi dose was attempted. Fifth, we only evaluated TNFi in this study; the results may be different for IL-17 and JAK inhibitors. Finally, the study was limited to patients with r-axSpA; patients with other subtypes of SpA were not included. Further studies, including other forms of bDMARDs, JAK inhibitors, and other subtypes of SpA, should be conducted.
Conclusion
In conclusion, a reduction on the percentage of Th1 and Th17 cells, r-axSpA-related cytokines (IL-17A, IFN-γ, TNF-α, IL-12, and IL-22), and gut dysbiosis-related molecules (i.e., zonulin and sCD14) in peripheral blood are associated with successful tapering of TNFi in patients with r-axSpA. In addition, a higher percentage of Th2 cells was associated with successful tapering of TNFi. Measuring these helper T cell subsets, in addition to specific serum cytokines and molecules related to gut dysbiosis/inflammation may help identify patients with r-axSpA who are suitable for TNFi tapering.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251330812 – Supplemental material for Biomarkers for successful tapering of a tumor necrosis factor inhibitor in patients with radiographic axial spondyloarthritis: A pilot study
Supplemental material, sj-docx-1-smo-10.1177_20503121251330812 for Biomarkers for successful tapering of a tumor necrosis factor inhibitor in patients with radiographic axial spondyloarthritis: A pilot study by Hong Ki Min and Ji-Yeon Lee in SAGE Open Medicine
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
