Abstract
Objective
The optimal surgical approach for Siewert type II adenocarcinoma of the esophagogastric junction (AEG) is controversial. In this study, we evaluated the outcomes of total gastrectomy for Siewert type II/III AEG via the left thoracic surgical approach that is used at our center.
Methods
We identified 41 patients with advanced AEG in our retrospective database and analyzed their 3-year survival rate, upper surgical margin, postoperative complications, and index of estimated benefit from lymph node dissection.
Results
The 3-year overall survival rate of the whole group was 63%, but no difference was observed between Siewert type II and III AEGs. Esophageal exposure and lymphadenectomy were sufficient. Eight patients developed postoperative complications, but none of the patients developed anastomotic leakage. Dissection of lymph node station Nos. 19 and 110 may be necessary for patients with Siewert type II AEG. Multivariate analysis revealed that the cT category was the only independent risk factor.
Conclusions
Total gastrectomy via an approach from the abdominal cavity into the thoracic cavity may be an optimal surgical technique for advanced Siewert type II AEG.
Introduction
During the past several decades, the incidence of adenocarcinoma of the esophagogastric junction (AEG) has distinctly increased in Western countries. 1 Additionally, gastroesophageal reflux disease, increased body weight, premalignant Barrett’s esophagus, and alcohol consumption may result in an increased risk of gastric cancer in Western countries; these factors may also be the key factors that contribute to AEG. No research to date has shown similar results in Asian countries, such as China and Japan; however, AEG is a very common malignancy in Asian countries, and most AEGs are Siewert type II/III.2,3
AEG is a malignancy of the esophagogastric junction (EGJ). In 1998, the German scholars Siewert and Stein 4 classified AEG into types I, II, and III. The center of the malignancy is located from 1 to 5 cm above the EGJ in Siewert type I, from 2 cm below to 1 cm above the EGJ in Siewert type II, and from 2 to 5 cm below the EGJ in Siewert type III. 4 This classification has become a common clinical tool to guide therapeutic decision-making. 5 Esophagectomy is performed using a transthoracic approach for the treatment of Siewert type I AEG because these tumors have a high frequency of thoracic lymph node involvement, while total gastrectomy is performed using an abdominal approach for the treatment of Siewert type III AEG. 6 Neoadjuvant chemoradiotherapy or perioperative chemotherapy significantly improves the outcome of advanced Siewert type II/III AEG over surgery alone.7,8 However, the optimal surgical approach for Siewert type II AEG is controversial.9–11 Three approaches are used for these tumors: a right-sided transthoracic approach (Ivor Lewis procedure), an abdominal approach, and a left-sided thoracoabdominal approach. 12
These three approaches can achieve similar outcomes, but they involve different types of esophageal and gastric resections and reconstructions as well as variations in the patient’s quality of life after surgery. Some studies have shown that the abdominal approach with transhiatal esophagectomy results in fewer pulmonary complications and shorter hospital stays, but it is not sufficient for exposing the upper esophagus or for performing thoracic lymph node dissection. 13 The transthoracic approach is useful for exposing the upper esophagus, but it is not sufficient for performing lymph node dissection, especially for the abdominal lymph nodes. 14 In contrast, the thoracoabdominal approach allows extensive dissection of the thoracic and abdominal lymph nodes but leads to more postoperative complications and a reduced quality of life. 15 Thus, it is necessary to identify a new surgical approach that allows extensive lymph node dissection and has a low occurrence of complications.
This study was performed to introduce a left thoracic surgical approach for treatment of Siewert type II/III AEG at our center. To the best of our knowledge, the proposed approach may be the optimal surgical approach because it allows exposure of the upper esophagus and extensive dissection of the lymph nodes, and it is associated with a low occurrence of complications.
Material and methods
Patients
We retrospectively analyzed the data of consecutive patients with AEG who underwent curative surgical resection from June 2013 to August 2015 at the Gastrointestinal Surgery Department of the First Affiliated Hospital of Zhejiang Chinese Medical University and Zhejiang Cancer Hospital, China. AEG was confirmed by gastroscopy in all patients. The inclusion criterion was the presence of either advanced Siewert type II AEG or highly advanced Siewert type III AEG. None of the patients had supraclavicular lymph node metastasis or upper mediastinal node metastasis, as confirmed by preoperative computed tomography and B-ultrasound. Some patients underwent preoperative chemotherapy, and some underwent laparoscopic exploration.
The study was approved by the ethics committee of the First Affiliated Hospital of Zhejiang Chinese Medical University and Zhejiang Cancer Hospital. The study conformed to the tenets of the Declaration of Helsinki. All patients provided written informed consent before taking part in the study.
Surgical operation
Selection of incision: A midline incision was performed in the upper abdomen, and abdominal exploration was performed to determine the tumor location, tumor classification, extent of esophageal infiltration, and presence of lymph node metastasis. After verification of the indications for surgery, the incision was extended up to the xiphoid process and the xiphoid was removed. The surgeon decided whether to perform an incision around the navel according to the patient’s body type. An incision protector was placed, and both sides of the costal arch were retracted toward the outside by hooks to create a sufficient surgical field.
Abdominal resection and lymph node dissection: The surgical procedures were similar to those of radical total gastrectomy with routine D2 lymphadenectomy. The splenic hilar lymph nodes (station No. 10) were resected with in situ preservation of the spleen. Two patients’ tumors involved the splenogastric ligament, and these patients underwent resection combined with splenectomy; three patients’ tumors involved the pancreas, and these patients underwent splenectomy and distal pancreatectomy.
Thoracic resection and lymph node dissection: The left triangular ligament of the liver was dissected, and the left coronal ligament was separated to the left edge of the inferior vena cava. The left lateral lobe of the liver was then folded. The esophageal hiatus was opened, and a transverse incision was made approximately 10 cm into the chest on the left diaphragm at about the 3-o’clock position (Figure 1). If the tumor directly involved the diaphragm, parts of the diaphragm were resected to ensure complete tumor removal. If blood vessels of the left diaphragm were present in the pre-cut line, the vessels were ligated. Next, a large S pull-hook was applied to open the diaphragm, the left pulmonary ligament was loosened, parts of the diaphragm muscle were resected, and the left pleura was incised to attain sufficient mediastinal exposure (Figure 2). Lymph node dissection was then performed. The replexed peritoneum of the esophagus was opened at the crura of the diaphragm, and the inferior phrenic lymph node and phrenic esophageal hiatus lymph node (station Nos. 19 and 20) were dissected. The esophagus was separated cephalically along the pericardium with a retractor and along the side of the pericardium. The lower lymph nodes of the mediastinum and its surrounding adipose tissue were then dissected while pulling the pericardium along the right pleura and thoracic aorta. The upper vagus nerve periesophageal plexus was dissected to the greatest extent possible. The periesophageal lymph nodes, epiphrenic lymph nodes, and posterior mediastinal lymph nodes were also dissected (station Nos. 110, 111, and 112). According to the tumor size, the esophagus could be exposed at its upper portion up to 12 cm. Purse-string forceps were used to place sutures 3 to 5 cm away from the upper margin of the tumor, and part of the esophagus was dissected. A No. 25 stapler base was then inserted.

The esophageal hiatus was opened, and a transverse incision was made approximately 10 cm into the chest on the left diaphragm at approximately the 3-o’clock position. The organs in the operative field are as follows: A: liver, B: diaphragm, C: esophagus, D: stomach, E: abdominal aorta, F: lung, G: pancreas, H: spleen.
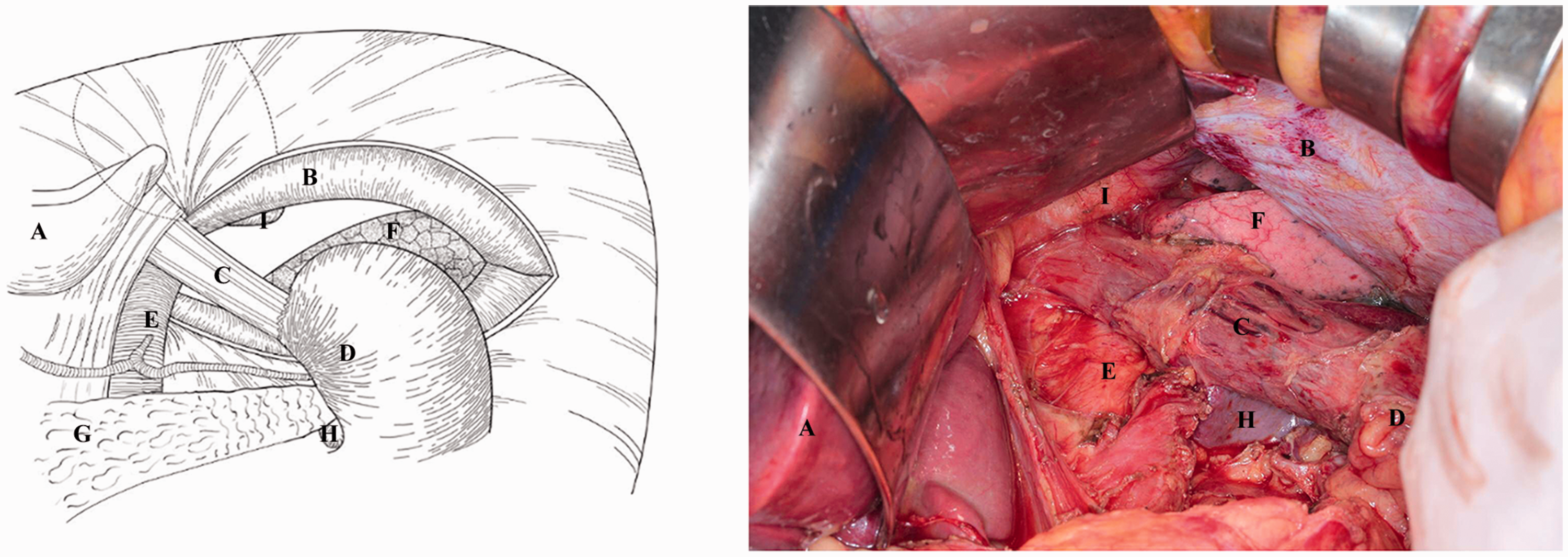
A large S pull-hook was used to open the diaphragm, the left pulmonary ligament was loosened, part of the diaphragm muscle was resected, the left pleura was incised, and a sufficient amount of the mediastinum was exposed. The organs in the operative field are as follows: A: liver, B: diaphragm, C: esophagus, D: stomach, E: abdominal aorta, F: lung, G: pancreas, H: spleen, I: heart.
Gastrointestinal reconstruction: Roux-en-Y esophagojejunostomy was performed, and esophagus–jejunum anastomosis was performed behind the transverse colon to reduce anastomotic tension in some cases. A jejunum substitute with long mesenteric and vascular arcades was selected. One to two blood vessels of the mesenteric artery were then dissected, and the intestine was dissected at this location. If a satisfactory blood supply to the intestine and sufficient dissociation of the mesenteric artery was verified, a circular stapler was used for the esophagojejunostomy approximately 15 cm from the ligament of Treitz. Finally, a jejunum–jejunum Roux-en-Y anastomosis was performed approximately 40 cm from the esophagogastric anastomosis, and the mesenteric fissures were closed.
Reconstruction of the esophageal hiatus: A closed thoracic drainage tube was inserted through the seventh or eighth intercostal space of the left chest, and the diaphragm was closed with interrupted 7-0 silk sutures. The surgeon ensured that the esophageal hiatus was neither too loose nor too tight after reconstruction (a width of two fingers was appropriate) and that the anastomosis was in the mediastinum.
Postoperative evaluation and follow-up
Postoperative chemotherapy was performed according to each patient’s individual pathological diagnosis. If an infiltrating margin was found, adjuvant irradiation was necessary. The anastomotic stomas were observed by gastrointestinal imaging at 5 to 9 days postoperatively, and abdominal computed tomography was performed 1 month after the operation. Re-examination was then performed every 3 months. The patients were examined regularly every 3 months during the first year and every 6 months thereafter.
Statistical analysis
Statistical analysis was performed with IBM SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, NY, USA). Survival curves were estimated using the Kaplan–Meier method, and multivariate and univariate analyses were performed using a Cox stepwise proportional hazard model. A p value of <0.05 was considered statistically significant.
Results
This study included 41 consecutive patients (26 male, 15 female) with a mean body mass index of 21.1 ± 2.7 kg/m2 and mean age of 58 years (range, 37–71 years). Among the 41 patients, 32 had advanced Siewert type II AEG and 9 had highly advanced Siewert type III AEG. Nine patients underwent preoperative chemotherapy, and 21 patients underwent laparoscopic exploration. The characteristics of the study patients are shown in Table 1.
Characteristics of the study patients.

Data are presented as n or mean (range). AEG, adenocarcinoma of the esophagogastric junction.
All 41 patients underwent total gastrectomy via the new approach from the abdominal cavity into the thoracic cavity. Among these 41 patients, 3 underwent combined resection of the spleen and pancreatic body and tail, 2 underwent combined resection of the spleen, and 5 underwent combined resection of the partial diaphragm. The mean length of esophageal exposure was 9.7 cm (range, 7–13 cm), the mean number of total lymph nodes excised was 46 (range, 29–68), and the mean number of mediastinal lymph nodes excised was 6 (range, 3–11). Thirty-two patients were confirmed to have differentiated adenocarcinoma, and nine patients were confirmed to have undifferentiated adenocarcinoma. Twenty-seven patients underwent R0 resection, and two patients underwent R1 resection. The surgical results are shown in Table 2.
Surgical results.

Data are presented as n or mean ± standard deviation (range).
Eight patients had postoperative complications: nine grade I complications (not requiring special treatment), one grade II complication (requiring special treatment such as a blood transfusion), two grade IIIa complications (requiring surgical, endoscopic, or radiological intervention without anesthesia), and no grade IIIb or higher complications (some patients had more than one complication). The postoperative complications are shown in Table 3.
Postoperative complications.

*Some patients had more than one complication.
The evaluation of each lymph node station is shown in Table 5. The lymph node metastasis rate, 3-year overall survival rate, and index of estimated benefit from lymph node dissection (IEBLD) of the patients with lymph node metastasis were 14.63%, 83.33%, and 12.2 for station No. 19; 5.00%, 50.00%, and 2.5 for station No. 20; 12.20%, 80.00%, and 9.8 for station No. 110; 7.32%, 66.67%, and 4.8 for station No. 111; and 12.50%, 60.00%, and 7.5 for station No. 112, respectively.
Frequency of lymph node metastasis and 3-year survival in patients with Siewert type II/III adenocarcinoma of the esophagogastric junction based on lymph node station.
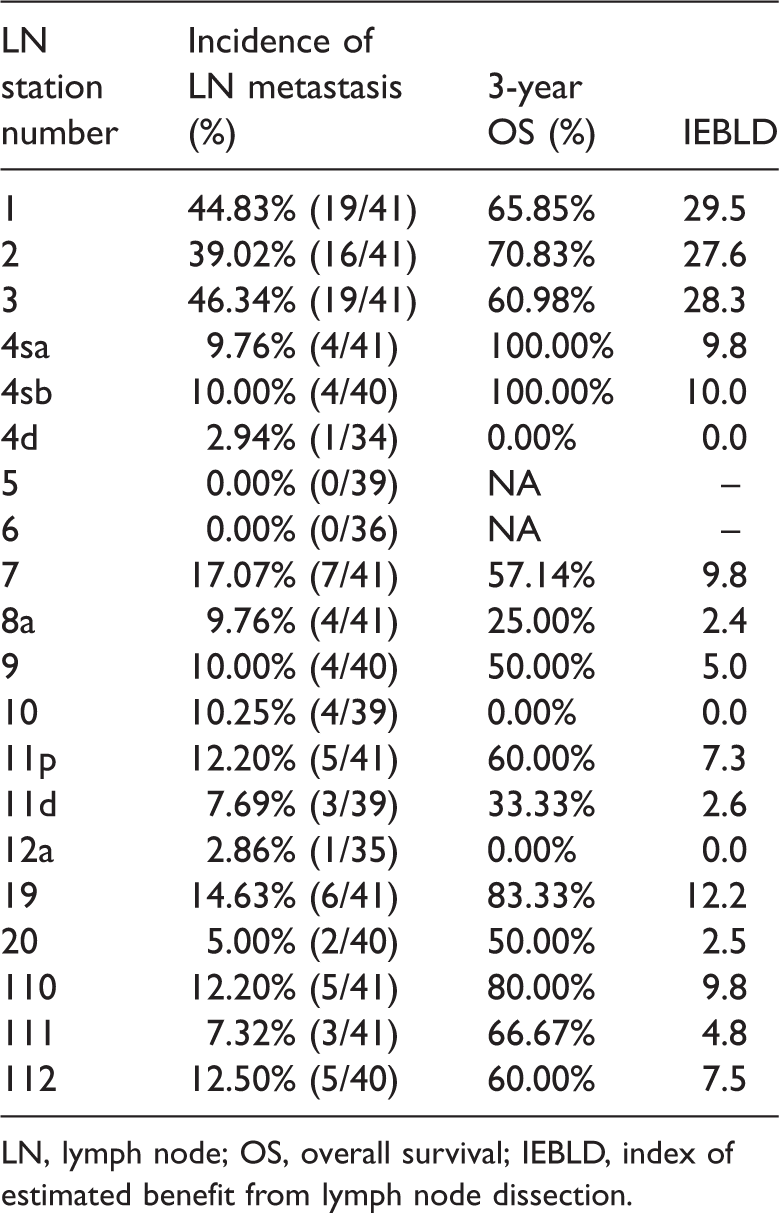
LN, lymph node; OS, overall survival; IEBLD, index of estimated benefit from lymph node dissection.
The 3-year survival rate of the entire group was 63% (Figure 3). Four patients died of liver metastasis, one of lung metastases, one of anastomotic recurrence, three of peritoneal metastasis, and four of psychological disorders. The significant prognostic factors for survival according to the univariate analysis were the cT category, cN category, infiltrating margin, presence of a vessel carcinoma embolus, and performance of pancreatic dissection. Multivariate analysis using the Cox proportional hazard model confirmed that the cTNM category was the only independent risk factor. No difference was found between the 3-year survival rates of patients with type II and III AEG (66% versus 56%, respectively) (Figure 4). The prognostic factors in the univariate and multivariate analyses are shown in Table 4.

Three-year overall survival rates of all 41 patients with adenocarcinoma of the esophagogastric junction.

Three-year overall survival according to the Siewert classification in patients with adenocarcinoma of the esophagogastric junction.
Prognostic factors in univariate and multivariate analyses.

*Statistically significant (p < 0.05)
HR hazard ratio; CI, confidence interval.
Discussion
The incidence of AEG has increased in recent years, and controversy exists regarding which surgical approach should be used to treat patients with AEG, especially those with Siewert type II AEG. In the past, esophagogastrectomy via the left transthoracic approach was a commonly used surgical technique for the treatment of Siewert type II AEG because of its benefits for thoracic lymph node dissection. Lymphatic drainage in AEGs includes drainage of the paracardial lymph nodes (station Nos. 1 and 2), greater curvature lymph nodes (station Nos. 4sa, 4sb, and 4d), lesser curvature lymph nodes (station No. 3), and lower mediastinal lymph nodes (station Nos. 110, 111, and 112).16–19 Parry et al. 20 observed that 11% of patients with Siewert type II AEG had upper mediastinal nodal involvement and that 18% of patients had middle mediastinal nodal involvement. A study by Blank et al. 13 showed that a less aggressive surgical approach may result in a high risk of postoperative recurrence and that thoracoabdominal esophagectomy may be a better treatment for advanced type II AEG. A meta-analysis by Aurello et al. 16 showed that the transthoracic approach can provide better 5-year overall survival and 5-year disease-free survival rates. However, Yamashita et al. 21 confirmed that the IEBLD of the mediastinal lymph nodes was marginal but that the IEBLD of the periesophageal nodes (station No. 110) was exceptional. 21 Furthermore, some studies have shown that the incidence of pulmonary complications in the transthoracic approach can be reduced.11,22 In contrast, a phase III trial conducted by Omloo et al. 23 showed that extended transthoracic resection was a more hazardous surgery and that it was only appropriate for patients with Siewert type I AEG. In Japan, a randomized controlled trial by Sasako et al. 9 compared the left thoracoabdominal approach with the transhiatal approach for patients who had AEG with esophageal invasion of <3 cm. Their results confirmed the lack of a significant survival benefit between these two approaches for Siewert type II/III AEG, but the left thoracoabdominal approach group had a higher in-hospital mortality rate and more complications. 9 The transhiatal approach can reduce the risk of postoperative pulmonary complications compared with thoracotomy.14,24,25 Therefore, transhiatal D2 lymphadenectomy has become a common surgical approach for Siewert type II/III AEG.
However, the transhiatal approach is not optimal. The disadvantages of this approach are as follows. First, it cannot guarantee a safer upper surgical margin, which is a significant prognostic factor. Second, the lower mediastinal lymph node dissection is not sufficient. Lower mediastinal lymph node dissection is difficult due to the narrow space of the lower mediastinal anatomy, thus increasing the risk of complications such as bradycardia and cardiac arrest. Third, the safety of Roux-en-Y esophagojejunostomy cannot be ensured, 26 and it may result in anastomotic stricture or anastomotic leakage.
This study was performed to evaluate a left thoracic surgical approach for Siewert type II/III AEG at our center. The advantages of the left thoracic surgical approach are as follows. First, this approach allows for sufficient low mediastinal lymphadenectomy. Extended esophageal resection and mediastinal lymph node dissection are essential for treating AEG, especially for esophagus-predominant EGJ tumors.20,27 However, Yamashita et al. 21 showed that the rate of low mediastinal node dissection, including station Nos. 110, 111, and 112, was <20%. In the present study, we excised a large number of total lymph nodes (46 ± 14), including the low mediastinal lymph nodes (6 ± 2), and those at station Nos. 110, 111, and 112 were routinely dissected. The low rate of lymph node metastasis at station Nos. 20 and 111 led to the relatively low IEBLD in these lymph nodes. Additionally, the rate of lymph node metastasis at station Nos. 19, 110, and 112 was >10%. A considerable 3-year overall survival rate and high metastasis rate were observed in patients with Siewert type II AEG with lymph node metastasis at station Nos. 19 and 110. These factors led to the high IEBLD in these patients. Interestingly, patients with No. 110 lymph node metastasis also commonly had No. 19 lymph node metastasis (4 of 5 patients). Notably, these patients were confirmed to have Siewert type II AEG. This may explain one mechanism of gastric cancer lymph node metastasis in which the No. 110 lymph node station is downstream of the No. 19 lymph node station. Therefore, we propose that the lymph nodes at station Nos. 19 and 110 may be better treated as regional lymph nodes in Siewert type II AEG. However, the sample size in the present study was small; a more extensive controlled study is needed to verify the benefit of lower mediastinal lymphadenectomy in patients with Siewert type II AEG. The second advantage of the left thoracic surgical approach is that a longer esophageal length is exposed. The standard safety margin has not yet been confirmed. Mine et al. 28 showed that a safety margin of 5 to 8 cm is necessary, but Barbour et al. 29 suggested that a safety margin of >3.8 cm is sufficient. According to the National Comprehensive Cancer Network Guideline, an adequate margin of ≥4 cm is necessary to prevent recurrence. In the present study, the upper surgical margin (9.7 ± 1.9 cm) was exposed; this exposure is essential to not only improve the negative surgical margin rate (R0: 39/41) but also safely restructure the Roux-en-Y esophagojejunostomy with no anastomosis. These are all benefits of the large operative field obtained after incising the left diaphragm. Complete tumor resection with free resection margins and adequate lymphadenectomy are the only curative treatments for patients with cancer. The third advantage is that the occurrence of postoperative complications can be reduced when using this new surgical approach. Postoperative complications caused by radical gastrectomy are common. Among the 41 patients in our study, only 8 patients developed complications [nine grade I complications (not requiring special treatment), 1 grade II complication (requiring special treatment such as a blood transfusion), 1 grade IIIa complication (requiring surgical, endoscopic, or radiological intervention without anesthesia), and no grade IIIb or higher complications]. The rate of pulmonary complications caused by the treatment in our study was lower than that caused by thoracoabdominal esophagectomy in a study by Blank et al. 13 Ott et al. 30 showed that approximately 50% of deaths after surgery were induced by pulmonary complications. Furthermore, some studies have focused on anastomotic leakage because of its high occurrence rate.31,32 Additionally, depending on the anastomosis sites and techniques used, anastomotic leakage can be reduced. Patients also benefit from the shorter operation time (168 ± 28 minutes) and shorter postoperative treatments. It is important to diagnose and treat postoperative complications to avoid additional complications.
In the present study, the 3-year overall survival rate for patients with Siewert type II/III AEG was 63% after total gastrectomy using the new abdominal approach. Huang et al. 33 confirmed that after laparoscopic-assisted total gastrectomy for AEG or open total gastrectomy, the 3-year overall survival rates of patients for AEG were not significantly different (72.0% versus 61.5%, respectively). As mentioned in some studies, total gastrectomy using the abdominal approach is used to treat advanced Siewert type III AEG.18,34 Although no difference was found in the 3-year survival rates between Siewert type II and III AEGs in the present study (66% versus 56%, respectively), this new surgical approach may be recommended for Siewert type II/III AEGs. Additionally, adequate mediastinal lymphadenectomy was shown in this study.
Many studies of the risk factors for AEG have focused on the TNM stage, depth of tumor invasion, and complications.35,36 In the present study, the prognostic factors identified by the univariate analysis were complete tumor resection (p = 0.004), pancreatectomy (p = 0.017), cT tumor category (p = 0.001), cN tumor category (p = 0.001), and vessel carcinoma embolus (p = 0.020); however, the cT category was the only independent risk factor (p = 0.008). This finding is consistent with several previous studies. Zhang et al. 37 confirmed that the TNM stage was significantly associated with AEG (p = 0.002). Many studies have shown that the T stage and N stage are independent factors for overall survival.38,39
On the basis of the above results, R0 resection, adequate lower mediastinal lymphadenectomy, safe Roux-en-Y esophagojejunostomy, and low postoperative complications can be achieved using this new approach for total gastrectomy. Dissection of lymph node station Nos. 19 and 110 may be necessary for treatment of AEG, especially Siewert type II AEG. However, the present study has limitations, including the single postoperative assessment and retrospective design. A more extensive control study is needed to verify the benefit of this new surgical approach for the treatment of Siewert type II/III AEG. Additionally, examination of a larger number of patients with Siewert type II/III AEG may be necessary.
Conclusions
Total gastrectomy via an approach from the abdominal cavity to the thoracic cavity may be the optimal surgical technique for advanced Siewert type II/III AEG, especially Siewert type II AEG.
Footnotes
Authors’ contributions
Can Hu and Hao-te Zhu contributed equally to this work. Study conception or design: Xiang-dong Cheng and Zhi-yuan Xu. Drafting of the manuscript: Can Hu, Hao-te Zhu, Jian-fa Yu, Yi-an Du, and Li-jing Wang. Data acquisition: Can Hu, Hao-te Zhu, Zhi-yuan Xu, and Ling Huang. Data analysis: Can Hu, Hao-Te Zhu, and Peng-fei Yu. Supervision or mentorship: Xiang-dong Cheng and Zhi-yuan Xu. All of the authors contributed important intellectual content to the overall work. Xiang-dong Cheng takes responsibility for the honesty and accuracy of the present study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by grants from the Natural Science Foundation of Zhejiang Province (Grant Nos. LY16H280011 and LY18H290006), the Key Research Program of Traditional Chinese Medical Science and Technology Plan of Zhejiang Province (Grant No. 2016ZZ012), and the Medical Science and Technology Project of Zhejiang Province (Grant Nos. WKJ-ZJ-1728 and 2016KYB220).
