Abstract
Objective
This study was performed to evaluate two classic procedures guided by anatomic markers for harvesting the anterolateral thigh (ALT) flap: one began with an incision on the lateral side to identify perforators emerging from the muscle to the superficial tissue and to track the perforators upward to the upper stem vessel, and the other began with an incision on the medial side to identify the vessel branch from the stem artery and to track it downward to the flap perforators.
Methods
Twenty-eight consecutive patients with tissue defects repaired with ALT flaps were investigated; 13 and 15 patients underwent the lateral and medial incision technique, respectively. The surgeon’s subjective view regarding procedural difficulty and the operative times were statistically analyzed.
Results
All flaps were harvested successfully. A two-paddle flap from one thigh in the medial group failed due to necrosis; all others survived completely. Subjectively, harvesting of flaps starting with a lateral incision was somewhat difficult, and the operative time was significantly longer using the lateral technique.
Conclusions
Classic procedures to harvest the anterolateral thigh flap are still practicable, and starting with a medial incision is more efficient than starting with a lateral incision.
Introduction
The anterolateral thigh (ALT) flap is widely considered a workhorse flap for repair of tissue defects, and its advantages include its long pedicle and low donor site morbidity. Moreover, the supine position during harvesting of this flap facilitates anesthetic management. The main limitation of the ALT flap is the anatomic variation of the perforators.1–5 Dissecting the perforators is the most important procedure when harvesting this flap. Recent studies on flap planning have mostly focused on predicting the vascular information with Doppler ultrasonography, computed tomography angiography, magnetic resonance angiography, and some dye techniques, although each technique has its own shortcomings and merits.6,7 Surgeons are striving to simplify and universalize the operative procedures in flap surgery.8,9 The classic harvesting technique for the ALT flap is based on anatomic markers and involves an open surgical procedure in which perforators are identified; this technique still has vitality in clinical practice. Two harvesting procedures may be used: that beginning from a lateral incision and that beginning from a medial incision. We hypothesized that beginning from a medial incision would result in a more rapid, economical, and efficient ALT flap harvesting procedure in a selected population.
Patients and methods
Our institutional review board approved this study. Informed consent was not required because the two approaches used in the study are commonly performed in our institution. Consecutive patients underwent tissue defect repair with ALT flaps using the lateral harvesting technique from October 2011 to September 2014 and using the medial harvesting technique from October 2014 to December 2016. The flaps harvested with the lateral technique ranged in size from 9 × 7 to 20 × 15 cm2. A representative case is shown in Figure 1. A line was depicted between the anterior superior iliac spine (ASIS) and superior lateral border of the patella, and the midpoint of this line was marked. The flap was designed with its center at this point. The lateral border was incised, and the flap with or without the deep muscular fascia was retracted medially. All available perforators to the flap were carefully protected and dissected to find the upper trunk. The flaps harvested with the medial technique ranged in size from 8 × 6 to 21 × 10 cm2. This procedure involved a few modifications of the technique described by Adler et al., 8 and a representative case is shown in Figure 2. A line was depicted between the ASIS and the midpoint of the superior border of the patella, using the anterolateral muscular septum as the axis of the ALT flap. The middle one-third of this line was incised, and the deep muscular fascia of the rectus femoris muscle directly under the incision was severed. The first lateral branch from the descending branch of the lateral circumflex femoral artery to the vastus lateralis muscle was easily identified while the rectus femoris muscle was retracted medially. We estimated the approximate perforator sites according to the course of the branches; we sometimes transected and deroofed a small amount of the overlying muscle to observe the course of these branches. Careful dissection was performed between the deep muscular fascia and the underlying vastus lateralis muscle to identify the perforators emerging from the muscle to the superficial tissues. Generally, dense connective tissue indicated the presence of a vessel bundle (perforator), and no difficulty was encountered in separating the deep fascia and its encapsulated muscle. A point on the skin over the perforator was marked, and we centered and mapped the flap based on this point. If the width of the recipient defect was more than 6 cm, we searched for two adequate perforators; if such vessels were found, we harvested two long, narrow flaps nourished with these two perforators. The two branches were identified before they entered the vastus lateralis muscle. The flaps were realigned so that they were suitable for the defect reconstruction. The donor site was directly closed if its width was less than 6 or 7 cm. Otherwise, a skin graft was used to cover the donor site. All procedures were performed by the same surgical team.

Representative case of harvesting of the anterolateral thigh flap beginning with a lateral incision (Patient 10). (a) Mapping of the flap. (b) Identification of the perforator emerging from the deep fascia. (c) Deroofing of the encapsulating muscle and tracing to the upper stem vessel. (d) Harvesting of the flap with the corresponding length vascular pedicle.
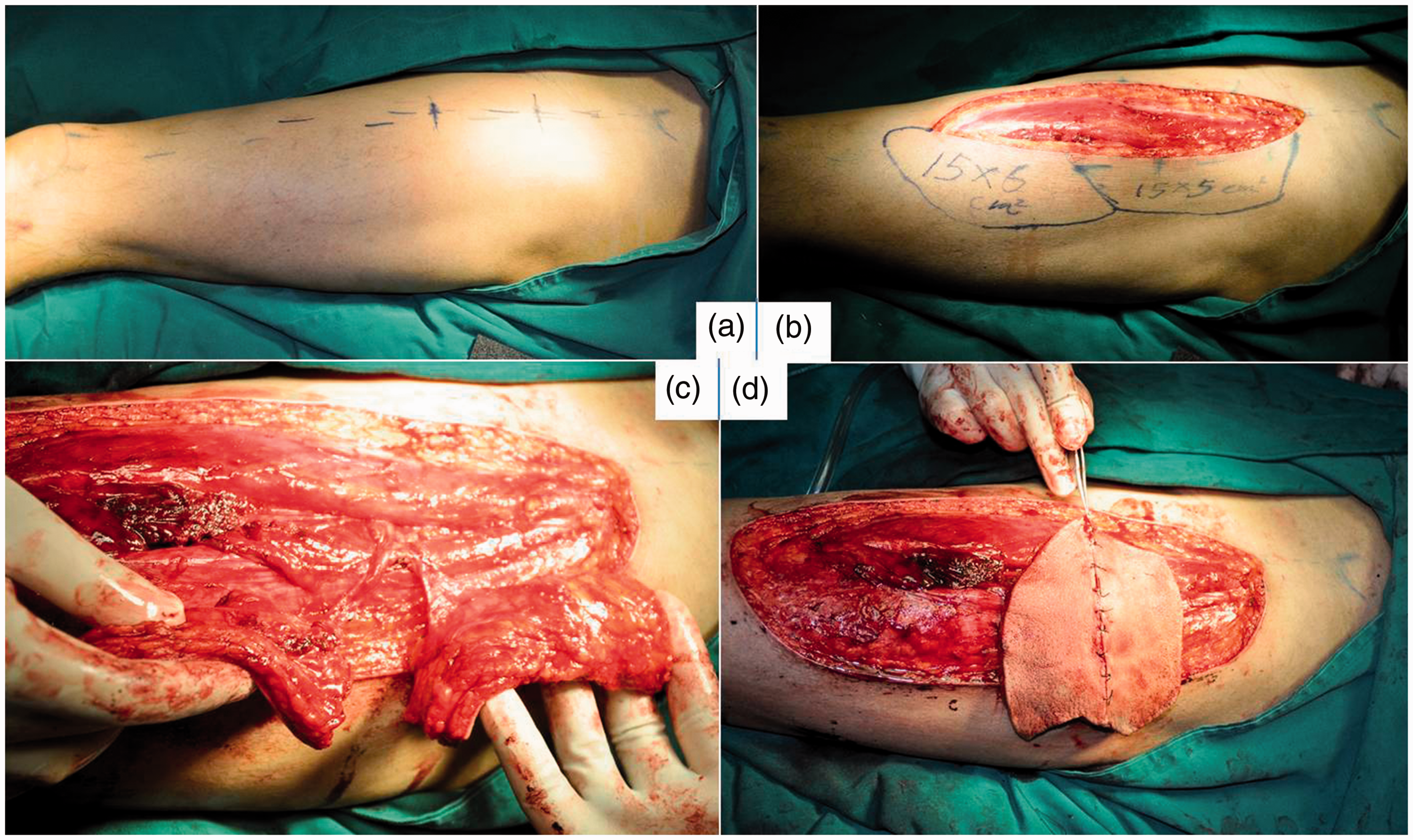
Representative case of harvesting of the anterolateral thigh flap beginning with a medial incision (Patient 18). (a) Mapping of the incision. (b) Identification of perforators branching from the descending branch of the lateral circumflex femoral artery and emerging from the muscle and use of these perforators to map the flap. (c) Deroofing of the encapsulating muscle and harvesting of two separate perforator flaps with a common vascular pedicle. (d) Repositioning of the flaps to form a flap available for the recipient site.
The operative time, subjective difficulty level of the surgery, perforator location, and other patient-related data were collected and evaluated (Tables 1 and 2). We analyzed the operative time and subjective data with a t-test and the cross-table exact Fisher method of the chi square test, respectively, using SPSS 22.0 software (IBM Corp., Armonk, NY, USA).
Flaps harvested by starting with a lateral incision.
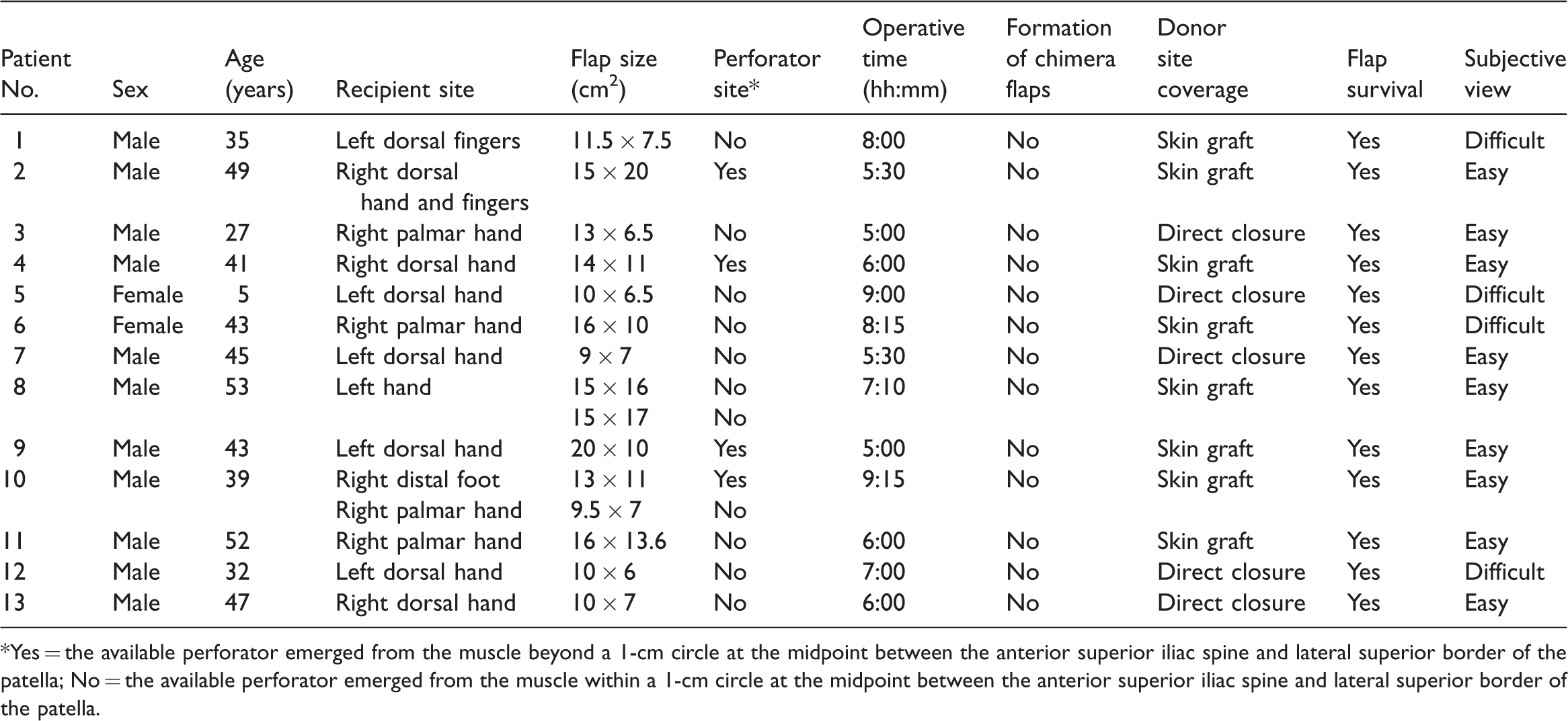
*Yes = the available perforator emerged from the muscle beyond a 1-cm circle at the midpoint between the anterior superior iliac spine and lateral superior border of the patella; No = the available perforator emerged from the muscle within a 1-cm circle at the midpoint between the anterior superior iliac spine and lateral superior border of the patella.
Flaps harvested by starting with a medial incision.

*Yes = the available perforator emerged from the muscle beyond a 1-cm circle at the midpoint between the anterior superior iliac spine and lateral superior border of the patella; No = the available perforator emerged from the muscle within a 1-cm circle at the midpoint between the anterior superior iliac spine and lateral superior border of the patella.
Results
Thirteen and 15 patients underwent tissue defect repair using the lateral and medial ALT harvesting technique, respectively. The patients’ demographic information is shown in Tables 1 and 2. In total, 30 flaps were harvested (in Patients 8 and 10, flaps were harvested from both thighs for a total of 4 flaps between these patients). In Patients 17, 18, 23, and 27, a flap with two perforators was harvested from only one thigh; each of these was counted as one donor site/flap. Twenty-eight flaps were harvested successfully, and all but one (a two-paddle flap from one thigh) survived completely. This flap failed due to gradual necrosis within 2 days postoperatively. We considered the cause to be infection, and exploration revealed purulent discharge beneath the flap. In four patients, we successfully used two available perforators to form a two-paddle flap. Eighteen perforator flaps among the total 30 flaps emerged from the muscle beyond a circle of 3 cm in diameter. Double-sided ALT flaps were harvested in two patients (degloving injury of the whole hand in Patient 9 and tissue defects of the anterior arm and dorsal hand in Patient 10). When harvesting the ALT flap in Patient 5, we initially found a perforator with a visibly adequate diameter; during dissection; however, we found that it did not course in an upward and medial direction and that it contained thick muscle covering. We avoided transection of this thick muscle because of the risk of dysfunction of the vastus lateralis muscle. Instead, we continued to dissect medially and found another perforator. Fortunately, its course was directed upward and medially toward the trunk, and its muscle coverage was thin. We ligated the previous perforator and harvested this subsequently identified perforator to nourish the flap.
The operative time in the lateral technique group was about 60 minutes longer than that in the medial technique group, with a significant difference (p < 0.05). However, the surgeon’s subjective view regarding the difficulty of harvesting the ALT flap was not significantly different between the two procedures. We identified the stem vessel and perforator and dissected the muscle to free the vascular pedicle much more confidently in the medial than lateral technique group. In the lateral technique group, we identified only the perforator and performed tentative dissection to determine whether it would course back to the expected stem vessel.
Discussion
Since the ALT flap was first described by Song et al. 10 in 1984, it has become popular and is now considered a workhorse flap in the field of tissue reconstruction with benefits including versatility, a long pedicle, and low donor site morbidity.3–5,11 The supine position facilitates anesthetic management, flap harvesting, and most tissue defect repair techniques. However, a main drawback is the variance in the vascular anatomy. Preoperatively, the ALT flap is usually plotted based on the perforator location. Hand-held Doppler examination, color duplex sonography, digital subtraction angiography, computed tomography angiography, magnetic resonance angiography, and some specific fluorescent staining angiography techniques are currently used to analyze the anatomic characteristics of the perforator, such as its course, size, number of vessels, and adjacent connections. Some of these techniques have been limited to experimental studies, some are expensive or time-consuming, and some have insufficient sensitivity and specificity.6,7 Additionally, some contrast agents can jeopardize the host’s tissues, preventing these agents from being routinely used. Because of the above-mentioned shortcomings and the fact that some clinics are not equipped with the required instruments and technology, the preoperative flap design in the clinical setting mainly depends on general anatomic characteristics and the surgeon’s experience. Many surgeons have no devices with which to preoperatively locate the perforator and thus depict the flaps intraoperatively based only on documented anatomic markers. The ALT flap elevation guidelines established by Adler et al. 8 and the flap designs described by Lin et al. 9 might facilitate great progress in surgical manipulation.
In the present study, the ALT flap was generally designed with its center at the midpoint of the ASIS and lateral superior border of the patella. The proximal line along the anterolateral intermuscular septum and the flap bound medial to the septum were incised together. The perforator was identified by dissecting the distal part laterally, and the pedicle of the flap (the lateral circumflex femoral artery) was found in the proximal part. Some surgeons preferred to incise the distal part lateral to the anterolateral intermuscular septum and to dissect the tissue medially, which facilitated identification of the perforator because it provided full exposure with no tension of the severed covering skin or fascia lata. 12 Retrograde dissection with tracing of the perforator may help to establish a suitable length and diameter of the perforator for anastomosis at the recipient site. 13 However, with the lateral incision technique described herein, the intramuscular course of the initially identified perforator cannot be accurately estimated; 14 it usually courses through the middle part of the vastus lateralis muscle, which could make dissection of the intramuscular vessel more difficult. In 2008, Adler et al. 8 published a description of their medial incision starting technique for harvesting of complex lateral femoral circumflex chimeric free tissue transfers. We used this technique in 15 cases and found the initially identified septocutaneous perforator described by Adler et al. 8 did not consistently appear and was easy to injure. However, the uppermost lateral perforator from the descending branch of the lateral femoral circumflex vessel was easy to find and was usually covered by a smaller amount of muscle than other lateral perforators. Despite the tension from the intact skin and fascia lata, locating the perforator site emerging from the muscle was not difficult. By conceptualizing and estimating the perforator course before it entered the muscle and separating the fascia from the muscle, we found a bundle of dense tissue with the vessel piercing its upper fascia. This was very useful when the flap width was about 6 cm. The medial incision was about 1.5 cm medial to the septum to assure that the perforator entered the flap. When the flap width was more than 7 cm, we attempted to find a septocutaneous perforator or another lateral perforator to form two flaps. The donor site was closed directly and a less severe scar resulted when using a split-skin graft. This could not be achieved by the two classic and conventional procedures. In some cases, the descending branch of the lateral femoral circumflex vessel coursed under the medial partial vastus lateralis muscle and was sometimes mistaken for the perforator. Therefore, we modified the procedure described by Adler et al. 8 to more efficiently locate the perforator, which might have prevented flap failure in all of our patients. The lateral branch (perforator) from the lateral femoral circumflex vessel should be tracked to the point at which it enters the vastus lateralis muscle, and a small amount of overlying muscle can sometimes be dissected to assure its course. The vascular orientation can provide accurate guidance to estimate the area in which the perforator comes off the muscle.
With the later (medial) technique, we could shorten the procedure by about 1 hour and form several chimeric flaps, although other factors also likely affected the operative time. The second lateral perforator had a large amount of overlying muscle, which could be the reason for the lack of a significant difference in the operator’s subjective view of the difficulty between the two procedures. Harvesting was fully successful in all 28 consecutive patients. The anatomical variance may become clear as more cases are accumulated. With the previous two procedures, each of which involves its own method of designing the flap, the perforator may not appear in the field of the flap, especially if its width is <6 cm. Therefore, we have herein presented some surgical modifications to reduce the risk. Some reports have described the absence of cutaneous perforators arising from the descending branch of the lateral circumflex femoral artery in a small proportion of cases involving the ALT flap. 15 Identification of an available perforator might be facilitated by much more carefully searching for and dissecting the perforator as well as meticulously dissecting and separating the semitranslucent connective membrane encapsulating the stem vessel bundle. Although anatomical variations were encountered in the present study, planning the ALT flap based on established anatomical markers was still valid. When using these anatomical markers to design the ALT flap, the donor site scar becomes less clinically relevant with a split-skin graft. Moreover, direct visualization of the nutritional vessel allows for evaluation of its characteristics such as pulsation, elasticity, and flexibility of the vascular wall, while angiography provides only images. In some cases, the vascular wall is inflexible and chain-like, and although the vessel usually seems adequate on filling images, its wall may be damaged and unable to pulsate efficiently.
Classic procedures for ALT flap harvesting with or without assistance by preoperative perforator location instruments are still practicable, and starting with a medial incision is more efficient than starting with a lateral incision. Moreover, starting with a medial incision may facilitate an easier change to another type of flap when the ALT flap is not feasible. 16
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
