Abstract
Objectives
Acute gastroenteritis, including illness caused by norovirus, is sometimes transmissible among susceptible persons who experience close contact, including those within a household, and it disturbs social activities of patients and their family. However, epidemiological assessment of the transmissibility and its heterogeneity has not been conducted. The present study aimed to quantify the frequency of household transmission that was likely caused by norovirus, and characterize its determinants.
Methods
A household survey was conducted, analyzing the history of suspected norovirus infection from January to March, 2017. Noro-like illness was clinically defined as a patient with either: (i) diarrhea or vomiting multiple times a day; or (ii) diarrhea or vomiting persisting for 2 or more days.
Results
Among 380 households, 132 households (34.7%) were eligible for epidemiological analysis, with an estimated secondary attack risk of 13.8% (38/276). Age-specific secondary attack risk was highest among index case patients aged 0 to 14 years (25.8%). The prevalence of vomiting in this age group was higher than in other age groups, with an odds ratio of household transmission estimated at 4.3.
Conclusions
Age-dependent heterogeneity was successfully identified, offering critical insights into future considerations for norovirus control at various social settings.
Introduction
Noroviruses are single-stranded positive-sense RNA viruses that belong to the Caliciviridae family. 1 The virus is transmitted via fecal contamination of food or water, through human-to-human direct contact, and by aerosolization of vomited virus. Norovirus has been recognized as the leading cause of food poisoning incidents in Japan, which is classically represented by eating raw oysters, but more recently, the infection has involved a larger fraction of human-to-human transmission among all infection events. 2
Acute gastroenteritis caused by norovirus is highly infectious and not necessarily sporadic. Norovirus infection can occur across all ages and is transmitted via close contact, such as in households, schools, and hospitals. 3 Because of acute symptoms including diarrhea and vomiting that last for a few days,4,5 the infection disturbs social activities of patients and their household contacts during the symptomatic period.
However, epidemiological assessment of norovirus transmissibility and its heterogeneity has been conducted. For example, clinical symptoms may vary with age, but age specificity of infection and symptoms has not been well studied. Moreover, the serial interval (SI; the time from illness onset in an index case patient to illness onset in the secondary case patient6,7) and the secondary attack risk (SAR; the risk of secondary transmission in a household given the index case patient) are key epidemiological indicators to plan prevention measures in household settings, but they have yet to be fully quantified in the household setting.
The present study aimed to estimate the frequency of acute gastroenteritis household transmission that was likely caused by norovirus, and characterize its determinants. Because of the limited practicality of confirming all suspected cases of norovirus infection, the present study explores the household transmission of acute gastroenteritis that is highly attributable to norovirus.
Materials and methods
Case definition
Clinical symptoms of norovirus infection are characterized by high frequency of diarrhea and vomiting.4,5 Thus, a noro-like illness (NLI) case was defined as a patient with either (i) diarrhea or vomiting multiple times a day; or (ii) diarrhea or vomiting that persisted for 2 or more days. Although nausea, abdominal pain, fever, and upper respiratory tract symptoms are sometimes observed during norovirus infection, 8 we did not include these symptoms as part of our case definition because sensitivity and specificity of these symptoms for NLI are known to be lower than those of diarrhea and vomiting. To identify the norovirus infection more specifically, we imposed the following exclusion criteria: (i) patients with pale feces, which is more likely indicative of rotavirus infection than a norovirus infection; 9 and (ii) patients with bloody stool, which is possibly suggestive of Escherichia coli infection or other bacterial infections. 10 Patients whose illness had a high likelihood of being attributed to other causes, such as pregnancy, and patients who were diagnosed with an influenza virus infection by a doctor were also not included in the study. These entry criteria do not immediately discriminate norovirus from other causes, and we do not intend to fully identify norovirus-associated acute gastroenteritis, but rather, examine household transmission of NLI.
Data collection
A retrospective household survey was conducted, collecting the history of NLI and its transmission in households from January to March 2017. The study period extended from October 2016 to March 2017 so that the norovirus epidemic period was fully covered. Norovirus is the leading cause of food poisoning in Japan, and its frequency during the study period was more than 5-fold higher than acute gastroenteritis of bacterial origin, which increases the prior probability of norovirus among patients with NLI. The survey was based on an internet-based questionnaire with convenient sampling. Household respondents were recruited from a Japanese community of research monitors using social surveys via the internet, which is maintained by a commercial research company in Tokyo. Although the size of the research monitors varies over time, there are consistently over 10,000 monitors from different households. The invitation conditions for our survey were as follows: (i) the respondent or his/her household members experienced multiple episodes of diarrhea or vomiting within one day; or (ii) the respondent or his/her household members experienced symptoms of diarrhea or vomiting for 2 or more days within the period from 1 October 2016 to 31 March 2017. A total of 380 households responded to our invitation, comprising 1578 household members. All participants were interviewed twice. The respondent did not receive any financial remuneration but was given points toward a reward system, as organized by the research company. To minimize recall bias, the survey was conducted during the norovirus epidemic season, which is in winter in Japan.
To estimate the SAR and the NLI SI, which is suggestive of norovirus infection, households were further selected using the following inclusion criteria: (i) the illness onset of the index case patient occurred between 1 October 2016 and 31 March 2017; (ii) all exposed individuals, such as the household contacts, lived together with the index case patient; and (iii) the time interval from illness onset in the index case patient to that of secondary patient(s) was less than or equal to 7 days. Whereas it is known that norovirus can be excreted in some exceptional cases from healthy individuals for more than 100 days 11 and can persist in a room environment for 20 to 60 days,12,13 the number of days in our selection criteria (7 days) reflects the most frequently observed length of SI in closed or semi-closed settings. 14
The existence of co-primary case patients (two or more patients who transmitted an infection as an index case at the same time), 15 was also taken into account in the present study, because it could reflect simultaneous exposures such as a foodborne infection event (e.g. food poisoning at a school). Hereafter, co-primary case patients were defined as individuals whose symptom onset took place on the same day. Considering that the shortest incubation period of norovirus infection has been reported to be 12 hours, 16 two or more patients with illness onset on the same day could also reflect a secondary transmission event. While we still calculated the SAR and SI assuming that one of co-primary case patients was the primary case patient, we also calculated these metrics assuming that there were actually two primary cases (results not shown).
Demographic variables including household size, age, and sex of each household member were analyzed for their relevance to the secondary transmission risk. When a participant met the definition for NLI, dates of illness onset for the participant and suspected household secondary case patients were also surveyed. Moreover, clinical datasets of symptoms including dehydration, abdominal pain, nausea, respiratory symptoms, and fever were also surveyed among all suspected cases in households.
Statistical analysis
Demographic and epidemiological characteristics of index case patients and their household contacts were analyzed. The SI was estimated from all possible pairs of observed secondary transmission. SAR was also calculated with the 95% confidence interval (CI) using the Wilson score method. To explore the age-specificity of infection, patients were stratified into three age groups: (i) infants and school-age children (0–14 years old); (ii) adults (15–59 years old); and (iii) elderly people (≥60 years old). These age groupings were used because of frequent occurrence of norovirus outbreaks in school settings, and to reflect the current official retirement age in Japan. Age-specific infectiousness and susceptibility were examined by stratifying the index case patients and household contacts by these age-groups. Possible association between secondary transmission and clinical symptom was also explored. Such association was examined using the univariate odds ratio (OR) and Fisher’s exact test, and SAR was compared by household size. The level of statistical significance was set at p=0.05. All statistical data were analyzed using R version 3.3.2.
Ethical considerations
All participants were provided with a written explanation in advance of the survey, and they were explicitly informed that their enrolment into this study was voluntary and that they had the right to withdraw at any time. Participants were also informed of the way in which the data obtained in the present study would be used, ensuring the confidentiality of personal information. Written informed consent was obtained from all participants. The study was approved by Ethics Board of Medical Research at Hokkaido University Graduate School of Medicine (reference number 16-057).
Results
Demographic characteristics of participants are shown in Table 1. Among the 380 households, 132 (34.7%) were eligible for analysis, including 132 index cases and 276 household contacts that involved five co-primary cases (i.e., five of the 276 contacts developed illness on the same day of the index case). The mean age of the index case patients was 44.7 years and 69 (52.3%) were male, while the mean age of household contacts was 42.4 years and 134 (48.6%) were male. A subgroup with diarrhea or vomiting two or more times a day included 126 index case patients and 251 contacts, including co-primary case patients, within the contacts. In another subgroup with diarrhea or vomiting for 2 or more days, 84 index case patients and 174 household contacts with no co-primary case patients were selected. No significant difference was observed in age and sex between the entire group and each subgroup. For the entire population, 38 out of 276 contacts, or a SAR of 13.8% (95%CI: 13.6–15.8), resulted in suspected secondary transmission with NLI.
Demographic characteristics in index case patients and household contacts.

SD, standard deviation.
†Eligible households including those with co-primary case patients.
SAR stratified by the index case patient age groups showed age-dependent patterns, with the highest estimate among patients aged 0 to 14 years (Figure 1a). The estimated SAR in children aged 0 to 14 years acting as the source of infection was 58.1% (95%CI: 49.2–66.9) (Table 2). The univariate OR of this age group was 15.4 (95%CI: 6.6–36.0), indicating that children aged 0 to 14 years were relatively more infectious than other age groups. Similarly, SAR by household contact age groups revealed a significant age-dependency (Figure 1b). Susceptible household members aged 0 to 14 years (when children aged 0–14 years act as exposed individuals in households) yielded the highest estimate at 25.8% (95%CI: 25.2–33.7) and the OR of a secondary attack was 2.5 (95%CI: 1.0–6.1) (Table 2). When SAR was stratified by household size, the estimate had greater values for larger household sizes, e.g., SAR was estimated at 25.0% (95%CI: 16.8–33.2) and 20.0% (95%CI: 12.0–28.0) in households comprising of five and six members, respectively. However, no significant difference was identified in the relationship between secondary transmission and household size. Sex was not associated with the risk of secondary transmission.

Secondary attack risk (SAR) of noro-like illness in households in Japan. (a) SAR stratified by three age groups for index case patients, reflecting age-dependent infectiousness and (b) SAR stratified by three age groups for the household contacts reflecting age-specific susceptibility. Black circles represent the mean SAR values and whiskers represent the lower and upper 95% confidence intervals.
Secondary attack risk of noro-like illness in Japan.

SAR, secondary attack risk; OR, odds ratio; CI, confidence interval.
*indicates a significant difference in the effect size.
Table 3 shows the frequency of different clinical symptoms among NLI cases. The prevalence of prolonged diarrhea for two or more days was more frequently seen in children aged 0 to 14 years compared with other groups. Similarly, observation of vomiting more than twice a day, prolonged vomiting for 2 or more days, and nausea were significantly more frequent in children aged 0 to 14 years compared with other age groups. Among those exposed to index case patients with diarrhea two or more times a day or prolonged for 2 or more days (n=259), 35 (13.5%) experienced secondary transmission. Otherwise, three out of 15 (20%) patients with exposure to patients with frequent or prolonged diarrhea experienced secondary transmission. The OR of secondary transmission given frequent or prolonged diarrhea was 0.7 (95%CI: 0.2–2.7). Among those exposed to index case patients with vomiting two or more times a day or prolonged for 2 or more days (n=106), 26 (24.5%) developed NLI. Similarly, among 170 contacts without frequent or prolonged vomiting, transmission occurred to 12 (7.1%) contacts. The OR of secondary transmission given frequent or prolonged vomiting was 4.3 (95%CI: 2.1–8.9).
Prevalence of with noro-like illness symptoms.

*indicates significantly high frequency compared with all other age groups.
The mean SI (and standard deviation (SD)) was 3.0 ± 2.0 days (Figure 2). The variance-to-mean ratio was thus 1.33. This indicates that the SI distribution was dispersed within the sample distribution shape, which was bimodal. This means that there was one peak on day 1 and 2 and another on day 5, which may indicate that the sample distribution could have included tertiary cases, but it is difficult to verify this notion only from questionnaire-based sample data.
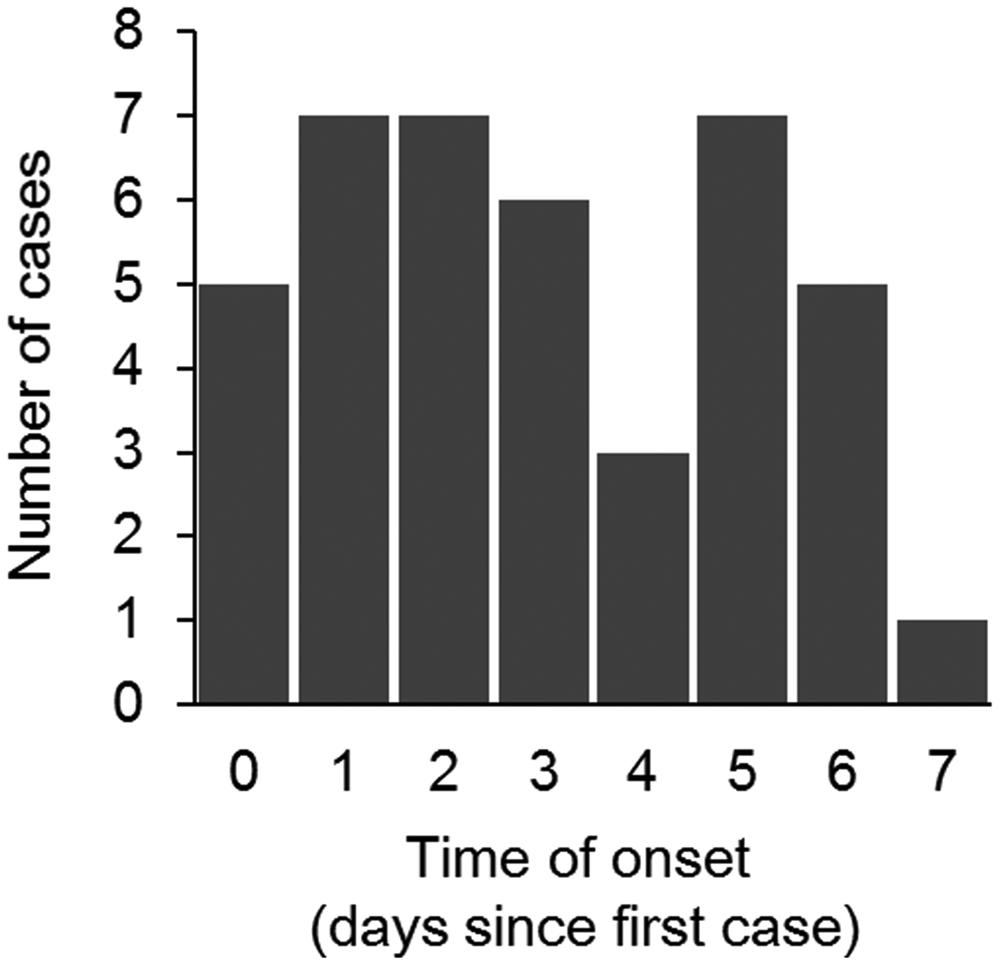
Serial interval distribution of noro-like illness in households in Japan. Distribution of the serial interval (the time from illness onset in the primary case patient to illness onset in the secondary case patient) for 38 households with fully recorded illness onset data. Five counts of illness onset on day 0 were co-primary case patients.
Discussion
We defined NLI as the presence of diarrhea or vomiting and conducted an epidemiological study collecting NLI household transmission data during the norovirus epidemic season, which is from October to March, in Japan. Recruiting more than 130 households eligible for analysis, the SAR of NLI was explicitly estimated, revealing that children aged 0 to 14 years are significantly more susceptible and infectious compared with other age groups. Children aged 0 to 14 years were more likely to experience prolonged diarrhea, and frequent and prolonged vomiting and nausea compared with other age groups. If the index case patient experienced frequent or prolonged vomiting, the risk of secondary transmission was 4.3 (95%CI: 2.1–8.9)-times higher than those not exposed. The mean SI was estimated to be 3.0 days, and the distribution appeared to be dispersed.
The present study is not the first to indicate that children are more likely to experience vomiting. Götz et al. 4 showed that vomiting is more frequent in younger individuals, and vomiting is more likely to contaminate the household environment, possibly contributing to successful household transmission.3,17 The present study presents the novel information that vomiting is positively associated with household transmission events and that vomiting was most frequently observed among children aged 0 to 14 years. Infectiousness and susceptibility were both found to be greater among children aged 0 to 14 years, suggesting that age-dependent contact characteristics and development of acquired immunity are likely to determine the fate of household transmission.
Although the mean SI of 3.0 days was consistent with published evidence, 18 the serial interval distribution shown in our study had a bimodal shape. We have determined the cutoff for the time interval between successive patients to be 7 days, based on published data, 4 and assumed that more than 95% of the secondary transmission arising from close contact occurs within 7 days. A similar timeline to the present study has been shown in household studies of influenza. 19 However, the shape of the SI could indicate that tertiary cases may have contaminated the distribution, and the actual SI may be less than 3 days. Because of the small sample size, we cannot judge whether the distribution was actually bimodal, and more detailed future observation is required including tracing of contact and, more preferably, comparison of the virus genome between a secondary case patient and candidates for primary case patients.
Additionally, we found that larger-sized households (e.g. five or six persons) yielded a higher estimate of the secondary transmission risk. While this could indicate that the household size has an effect on the transmission dynamics, a possible underlying explanation may be that larger households involve a higher number of susceptible individuals (children) who are more likely to acquire infection and cause a secondary transmission event.
There are several important limitations in this study. First, the sampling strategy in the present study fully relied on clinical symptoms, especially non-specific symptoms including diarrhea and vomiting, which were used to define NLI. Although sampling took place during the norovirus epidemic season, which is in winter in Japan, and the number of norovirus cases reported as part of public health surveillance is greater than that of other similar types of viral gastroenteritis, it is possible that our enrolled patients represent a small number of patients with other etiologies that include other pathogens (e.g., rotavirus, adenovirus, and sapovirus), which could cause similar symptoms as those of the norovirus infection. 20 However, a large-scale sampling of patients with confirmed norovirus is challenging, considering that most visits to a doctor do not result in confirmatory diagnostic testing, and there is no rapid diagnostic test that is covered by insurance for this specific infectious disease. A comparative clinical study of the symptomatology between norovirus and acute gastroenteritis resulting from other causative agents needs to be conducted. Some specific points, e.g. earlier recovery from norovirus compared to other viruses, may be identified through systematic collection of clinical data.
Second, we observed five possible co-primary case patients, and we regarded those five patients as having their illness caused by secondary transmission that led to illness onset of both index case patients and secondary case patients within the same day. As a sensitivity analysis, we also conducted all analyses assuming that all five co-primary case patients were actual co-primary case patients who brought the infection from the community to the households; the results of this analysis were not different from those shown in the Results section. As mentioned above, an analysis of the virus genome among all diagnosed cases could help to overcome the relevant difficulty in determining the identity of the first patient. Third, while our patients were clinically diagnosed, more than 30% of norovirus infections have been shown to remain asymptomatic. 21 Several patients with mild NLI were missed in the present study.
While an epidemiological study based on the clinical definition of patients involves several technical limitations, we believe that the present study showed a substantial number of previously unknown aspects in characterizing the household transmission of norovirus. Our study shows that children aged 0 to 14 years would act as the maintenance host of norovirus transmission, and outbreaks in elderly patients in nursing homes that frequently occur in Japan might not necessarily be the main driver of the norovirus epidemic at a population level. Moreover, we clarified that vomiting is likely associated with the elevated risk of secondary transmission, suggesting areas upon which to focus future interventions.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
HN received funding from the Japan Agency for Medical Research and Development (AMED), Japanese Society for the Promotion of Science (JSPS) KAKENHI (grant numbers 16KT0130, 16K15356 and 17H04701), Japan Science and Technology Agency (JST) CREST program (JPMJCR1413), and RISTEX program for Science of Science, Technology and Innovation Policy. RM, ST, and FM received funding support from JSPS KAKENHI (16H06581, 17H06487, and 17J07706, respectively). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
