Abstract
Objectives
To analyze the evidence comparing the benefits and risks of continuous versus intermittent milk feeding in low birth weight (LBW) infants.
Methods
Three electronic databases were searched and screened to identify randomized controlled trials of continuous and intermittent milk feeding of LBW infants up to October 2016.
Results
Eight trials were included in this meta-analysis. Continuous feeding had no effect on days to full feeds and time to regain birth weight. There were no significant differences in the number of apneas, invasive ventilation, changes in body length, occipitofrontal circumference, skinfold thickness, and total protein, and in the number of stools between the two feeding strategies. Continuous feeding was associated with higher gastric residual volume, noninvasive ventilation, weight gain, increase in bilirubin, and longer nil by mouth. There were no significant differences in adverse events and confounders between the two feeding strategies.
Conclusions
Continuous milk feeding was superior to intermittent feeding in LBW infants in terms of weight gain. However, continuous feeding was also associated with increased nil by mouth duration, increased bilirubin, increased noninvasive support, and increased gastric residuals. Continuous feeding thus confers advantages in terms of weight gain, but also has disadvantages compared with bolus feeding.
Keywords
Introduction
Preterm birth is the most common cause of neonatal morbidity and mortality worldwide. The incidence of infants delivered before 37 weeks gestation ranges from 12% to 13% in the USA and from 5% to 11% in Europe and other developed countries.1–3 Birth before 37 weeks is associated with a higher incidence of neonatal death and serious adverse events. In addition, despite a survival rate of >85% in preterm infants with a gestational age <28 weeks, the incidence of neurobehavioral disabilities ranges from 5% to 15%, resulting in cognitive, behavioral, and social delays. These dysfunctions often persist into adulthood and affect health outcomes.4–6
Human milk has essential nutrients and immunologic factors for preterm infants. However, tube feeding is necessary for very low birth weight (LBW) infants because they are unable to coordinate sucking, swallowing, and breathing.7,8 Gastrointestinal immaturity, including lactase activity, gastrointestinal motor function, and pancreatic function, also affect the provision of enteral nutrition in LBW infants, delaying full feeding. 9 Previous studies have demonstrated the impacts of continuous and intermittent milk feeding in preterm infants and suggested associations between continuous feeding and energy efficiency, duodenal motor function, nutrient absorption, and splanchnic oxygenation. However, intermittent bolus feeding results in a more-physiological release pattern of gastrointestinal tract hormones, stimulates gastrointestinal tract development, and enhances protein accretion.10–14 Given the potential advantages of different feeding strategies in clinical practice, we examined previous randomized controlled trials (RCTs) to compare continuous feeding with intermittent bolus feeding in LBW infants. We conducted a systematic review and meta-analysis of RCTs comparing continuous versus intermittent bolus feeding in LBW infants to explore recent evidence for the benefits and risks of these feeding strategies.
Materials and Methods
Data sources, search strategy, and selection criteria
This review was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis statement issued in 2009. 15 Ethical approval and patient consent were not required because this was an analysis of previously published studies. Any RCTs that investigated the benefits and risks of continuous versus intermittent tube feeding in LBW infants were eligible for inclusion in this meta-analysis. Electronic databases including PubMed, Embase, the Cochrane Library, and ClinicalTrials.gov were systematically searched using the combination of search terms (“Infant” OR “neonatal” OR “newborn”) AND (“low birth weight” OR “preterm” OR “premature”) AND (“milk” OR “enteral nutrition”) and restriction of ‘‘randomized controlled trial’’ (the deadline was October 2016). The reference lists of all the relevant original and review articles were searched manually to identify additional eligible studies. Unpublished studies and updated information on some included trials that could provide useful data were also identified. Only studies published in English were included. The article title, study design, infant status, intervention, and reported outcome variables were used to identify potential studies for inclusion.
The titles and abstracts of the studies were reviewed independently by two reviewers to exclude unrelated studies, and the full texts of the relevant studies were retrieved. Any inconsistencies between the two authors were settled by group discussion. Studies were considered eligible if they met the following criteria: (1) randomized controlled design; (2) participants were LBW infants; (3) infants received continuous feeding or intermittent bolus feeding; and (4) the investigated outcomes included days to full feeds, time to regain birth weight, number of apneas, invasive ventilation, change in body length, change in occipitofrontal circumference, change in skinfold thickness, change in total protein, number of stools, gastric residual volume, noninvasive ventilation, weight gain, change in bilirubin, nil by mouth, necrotizing enterocolitis, sepsis, deaths, patent ductus arteriosus, intraventricular hemorrhage, respiratory distress syndrome, mechanical ventilation, bronchopulmonary dysplasia, small-for-gestational-age, antenatal steroids, and preeclampsia/hemolysis elevated liver enzymes and low platelets. In the event of overlapping reports, only the most recent outcomes were included. Trials that did not meet these inclusion criteria were excluded.
Data extraction and quality assessment
Two reviewers independently extracted the following data from the identified studies: first author’s name, publication year, country, sample size, gestational age, birth weight, percentage of boys, percentage small-for-gestational-age, percentage using antenatal steroids, percentage patent ductus arteriosus, interventions, controls, definition of LBW, duration of intervention, and the outcomes investigated. Disagreements were resolved by consensus or by consultation with a third reviewer. The quality of each included RCT was assessed according to the Jadad scale. 16 Briefly, the studies were assessed based on the following aspects: randomization, allocation concealment, blinding, baseline comparability, and loss to follow-up, and were then graded and scored from 0 (poor quality) to 5 (good quality).
Statistical analysis
The data were extracted from the individual studies and the effect estimate and 95% confidence intervals (CIs) were calculated. Relative risk (RR) was determined for dichotomous data and weighted mean differences (WMDs) were used for continuous data. All summary analyses were performed using random-effects models.17,18 Statistical heterogeneity among the included studies was calculated using the I2 and Q statistics. P values <0.10 were considered indicative of significant heterogeneity.19,20 Subgroup analyses were conducted for days to full feeds, time to regain birth weight, and necrotizing enterocolitis based on publication year, country, gestational age, birth weight, and percentage boy. Sensitivity analyses were performed by removing each individual study to evaluate the influence of a single study. 21 Funnel plots were employed to evaluate qualitative publication bias, and Egger’s 22 and Begg’s tests 23 were used to assess quantitative publication bias. All reported P values were two-sided, and P values <0.05 were considered statistically significant for all included studies. Statistical analyses were performed using Stata software (version 10.0; Stata Corporation, TX, USA).
Results
Literature search
A systematic literature search of PubMed, Embase, and the Cochrane Library identified 912 studies that met the search criteria. After reviewing the titles and abstracts of these studies, 894 studies were discarded due to irrelevant topics and duplicates, while the remainder were considered to potentially fulfill the inclusion criteria and their full texts were reviewed. Full-text review excluded 10 studies for the following reasons: study reporting the same infants, insufficient data for endpoints of interest, and study with no appropriate control. Eight trials were finally included in the meta-analysis.24–31 These eligible studies were published between 1987 and 2015. The detailed study selection process is shown in Figure 1.
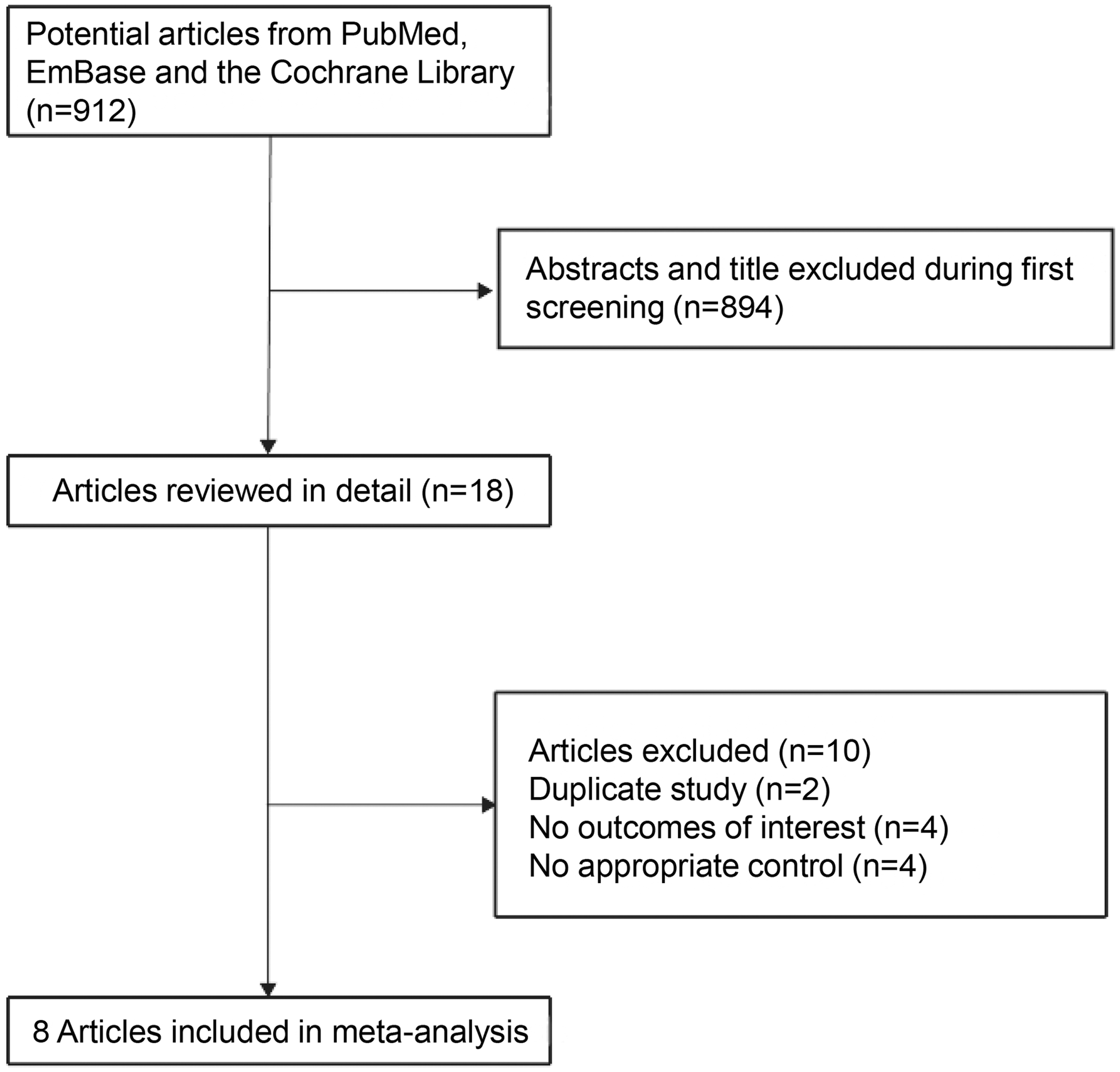
Study selection process.
Baseline characteristics of included studies
The baseline characteristics of the included trials are presented in Table 1. All eight trials involved a total of 728 LBW infants. Four trials were conducted in the USA,26,27,30,31 three in Europe,24,28,29 and the remaining one in Asia. 25 The gestational ages ranged from 26.8 to 30.7 weeks, the birth weight ranged from 849 to 1219 g, and the percentage of boys ranged from 42.7% to 55.3%. Study quality was evaluated using the Jadad score. Overall, two trials had a score of 3, two trials a score of 2, and the remaining four trials a score of 1 (Table 1).
Characteristics of included studies.

BW, birth weight; LBW, low birth weight; NA, not available; PDA, patent ductus arteriosus; SGA, small-for gestational age.
Feeding tolerance and weight gain
Six of the eight included trials provided data for days to full feeds. Significant heterogeneity was observed (I2 = 91.4%; P < 0.001). The pooled estimate of the WMD of days to full feeds was 1.41 days (95% CI –1.03 to 3.85), indicating no significant difference between continuous and intermittent bolus feeding (Figure 2). Sensitivity analysis showed that the conclusion was not affected by excluding any individual trial (Table 2).

Influence of continuous versus intermittent feeding methods on days to full feeds.CI, confidence interval.
Sensitivity analysis for days to full feeds.

aDays to full feeds according to birth weight 700–1000 g; bdays to full feeds according to birth weight 1001–1250 g. CI, confidence interval; WMD, weighted mean difference.
Six of the eight included trials provided data for time to regain birth weight. Continuous feeding was not associated with time to regain birth weight (WMD: –0.55; 95% CI: –1.21 to 0.11) (Figure 3), and this conclusion was also not affected by the exclusion of any specific trial (Table 3).

Influence of continuous versus intermittent feeding methods on time to regain birth weight.CI, confidence interval.
Sensitivity analysis for time to regain body weight.

aTime to regain body weight according to birth weight 700–1000 g; btime to regain body weight according to birth weight 1001–1250 g; ctime to regain body weight according to human milk; dtime to regain body weight according to preterm formula. CI, confidence interval; WMD, weighted mean difference.
A summary of feeding tolerance and weight gain outcomes is presented in Table 4. Continuous feeding was associated with significantly higher levels of gastric residual volume (P < 0.001), noninvasive ventilation (P = 0.001), weight gain (P < 0.001), increase in bilirubin (P < 0.001), and nil by mouth (P < 0.001). However, there were no significant differences in the number of apneas, invasive ventilation, change in body length, change in occipitofrontal circumference, change in skin fold thickness, change in total protein, and number of stools between the continuous and intermittent feeding strategies.
Summary of other outcomes.

CI, confidence interval; GRV, gastric residual volume; IV, invasive ventilation; NV, noninvasive ventilation; OC, occipitofrontal circumference; SFT, skin fold thickness; TP, total protein; WMD, weighted mean difference.
Adverse outcomes and confounders
Seven of the eight included trials provided data for necrotizing enterocolitis. Heterogeneity assessment of these studies revealed an I2 value of 0.0%, suggesting no significant heterogeneity. Continuous feeding had no significant effect on the risk of necrotizing enterocolitis (RR: 1.06; 95% CI: 0.64 to 1.75) (Figure 4). Sensitivity analysis indicated that the conclusion was not changed by the exclusion of any specific trial (Table 5). The results for other adverse events and confounders are shown in Table 6. There were no significant differences in sepsis, patent ductus arteriosus, intraventricular hemorrhage, respiratory distress syndrome, mechanical ventilation, bronchopulmonary dysplasia, small-for-gestational-age, antenatal steroids, and preeclampsia/hemolysis elevated liver enzymes and low platelets between the continuous and intermittent bolus feeding strategies.

Influence of continuous versus intermittent feeding methods on necrotizing enterocolitis.CI, confidence interval.
Sensitivity analysis for necrotizing enterocolitis.

CI, confidence interval; RR, relative risk.
Summary of other adverse outcomes.

CI, confidence interval; HELLP, hemolysis elevated liver enzymes and low platelets; PDA, patent ductus arteriosus; RDS, respiratory distress syndrome; RR, relative risk; SGA, small-for gestational age.
Subgroup analysis
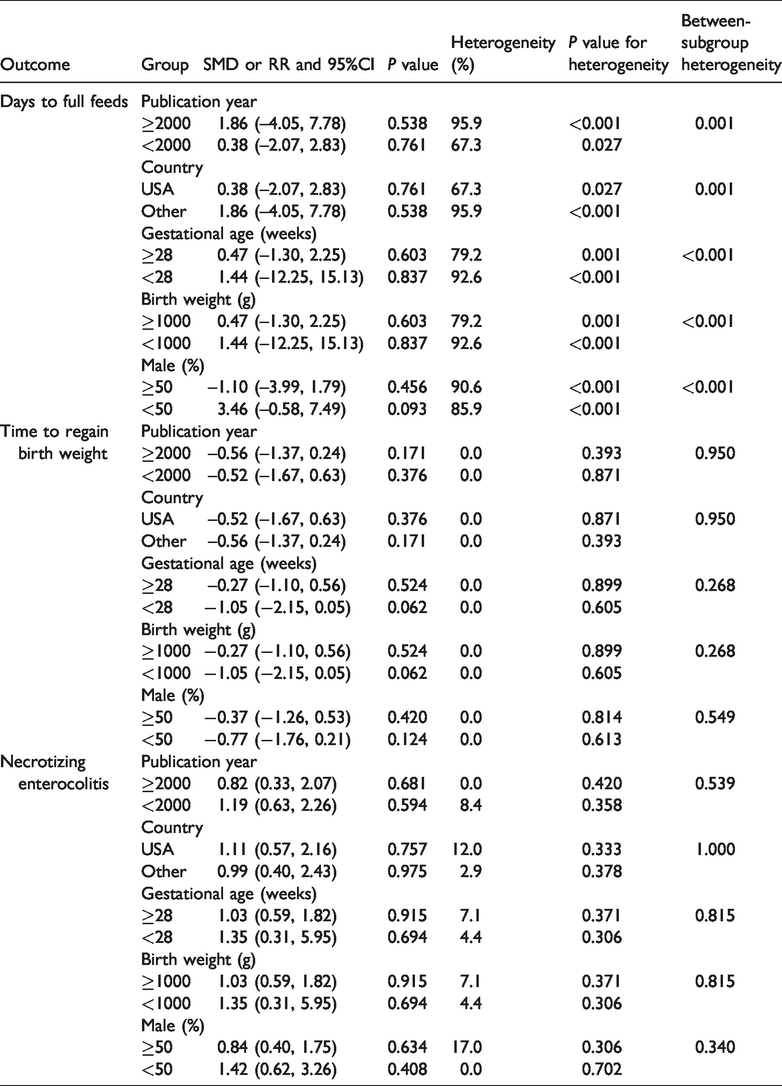
The results of subgroup analyses for days to full feeds, time to regain birth weight, and necrotizing enterocolitis are shown in Table 7. The results were consistent with the overall analysis. However, LBW infants who received continuous feeding had longer to full feeds in studies published from the year 2000 compared with studies published before 2000 (P = 0.001). LBW infants in the USA also had a shorter time to full feeds compared with those in other countries (P = 0.001). Higher gestational age and birth weight were associated with shorter times to full feeds compared with the corresponding subsets (both P < 0.001), and a higher percentage of boys was also associated with fewer days to full feeds (P < 0.001). There were no other significant differences between the subgroups.
Subgroup analyses.
Publication bias
A review of the funnel plots could not rule out potential publication bias for days to full feeds and time to regain birth weight (Figure 5). However, Egger’s and Begg’s test results showed no evidence of publication bias for these outcomes.

Funnel plots for (a) days to full feeds and (b) time to regain birth weight.WMD, weighted mean difference.
Discussion
The present meta-analysis was designed to compare the benefits and risks of continuous feeding with those of intermittent bolus feeding in LBW infants. Compared with intermittent feeding, continuous feeding was associated with higher levels of gastric residual volume, noninvasive ventilation, weight gain, increase in bilirubin, and nil by mouth. However, there was no significant difference in terms of days to full feeds, time to regain birth weight, number of apneas, invasive ventilation, changes in body length, occipitofrontal circumference, skinfold thickness, or total protein, number of stools, and any potential adverse events. These findings might clarify the impacts of these two feeding strategies in LBW infants, and might also assist clinicians in selecting the appropriate feeding method for these infants. These findings were similar to those of another meta-analysis carried out in parallel with the present analysis, which reached similar conclusions using a slightly different set of studies. 32 These two meta-analyses are therefore complementary.
A previous meta-analysis conducted in 2001 found that continuous feeding in very LBW infants was associated with a longer time to reach full feeds, and had no significant effect on somatic growth, days to discharge, or the incidence of necrotizing enterocolitis. 33 Furthermore, their updated meta-analysis 34 in 2003 reported a similar conclusion, and also reported a trend towards earlier discharge for infants weighing <1000 g who received continuous feeding. That meta-analysis was further updated in 2011, and the findings were consistent with the results of the previous meta-analysis, which concluded that infants weighing <1000 g, with a birth weight of 1000 to 1250 g and fed via continuous nasogastric tube feeding, had higher weight gains. 35 The inherent limitations of this previous review were its sample size, methodologic limitations, conflicting results, especially for several indexes available in only a few trials, and summary outcomes with broad CIs, leading to a lack of statistically significant differences due to the small sample size and lack of statistical precision. We therefore conducted an updated meta-analysis to evaluate the benefits and risks of the two feeding strategies.
The current meta-analysis found that continuous feeding had no effect on days to full feeds and time to regain birth weight, though several of the included trials reported inconsistent results. Dollberg et al. 25 performed an RCT based on two centers, which indicated that infants receiving intermittent gastric bolus reached full feeds earlier with less delay than infants receiving continuous gastric infusion. They pointed out that intermittent feeding could induce faster gut maturation in very LBW infants.
Schanler et al. 31 indicated that bolus tube feeding provided the best advantage in premature infants. The results of individual trials were consistent with the summary result. The potential reason for this could be a need for stomach distention by a minimum volume of feeds to optimize gut peristalsis. The time to regain birth weight was similar between infants fed with continuous and intermittent feeding strategies because the infants received complementary parenteral nutrition, thus accelerating growth in LBW infants.
There was no significant difference in the incidence of necrotizing enterocolitis between the two feeding methods, consistent with the findings of a previous meta-analysis. 32 Individual trials reported similar conclusions. Rovekamp-Abels et al. 24 indicated that continuous feeding reduced the risk of necrotizing enterocolitis by 41%, but the result was not significant, and Schanler et al. 31 similarly found that continuous feeding reduced the risk by 51%. Other included trials28–30 reported a harmful effect of continuous feeding in terms of necrotizing enterocolitis, but the difference was not significant because there were fewer necrotizing enterocolitis events than expected. In addition, the incidence of necrotizing enterocolitis after achieving full enteral feeding might have been underreported, leading to bias in the summary result. Further large-scale RCTs are needed to verify this conclusion.
The findings of subgroup analyses were consistent with the overall results. Publication year, country, gestational age, birth weight, and percentage of boys were identified as significant confounders for days to full feeds (P-value between subgroups < 0.05). In addition, although several significant differences were detected between the two methods, these conclusions for other outcomes might have been affected by a smaller number of trials included in the corresponding outcomes or subgroups. The present study thus provided relative results and a synthetic review.
The present meta-analysis had some limitations. First, the number of included trials was small, with low to moderate study quality. Second, there might have been language bias in study selection. Third, several of the adverse events, such as suspected necrotizing enterocolitis, might have had different definitions among the studies. However, suspected necrotizing enterocolitis was not included as an outcome in this systematic review.26,29,30 In addition, variable definitions, detection methods, and reporting prevented the analysis of outcomes such as length of stay, growth failure, and infections. Fourth, the included trials had different baseline characteristics, including gestational age, birth weight, and percentage of boys, thus influencing the data and introducing bias. Fifth, in the planning stage, data on the incidence of infections in the infants administered the two feeding methods were not available due to the long-term use of nasogastric tubes. Sixth, fewer trials reported other outcomes, and subgroup analyses were therefore not conducted. Finally, the broad time span of the studies that were included and evaluated (1987–2015) suggests that the studies were likely to cover different patient populations. Combined with the low quality of the studies, this decreased the clinical utility of the conclusions of this meta-analysis.
In conclusion, the present meta-analysis demonstrated that continuous feeding was superior to intermittent feeding for LBW infants in terms of weight gain. However, there was no difference in body length, occipitofrontal circumference, skin folds, and proteins, and increased time spent nil by mouth, increased bilirubin, increased noninvasive support, and increased gastric residuals. Thus although continuous feeding confers some advantages in terms of weight gain, it also has some disadvantages compared with bolus feeding. The goal of feeding in LBW infants is to reduce the length of time to full feeds and the risk of potential adverse events. Further large-scale trials are therefore needed to evaluate the benefits and risks of different feeding strategies in these infants.
