Abstract
Objective
Liver biopsy is the gold standard test for assessment of liver pathology. This study was performed to assess the predictive value of spleen thickness for liver pathology and the role of routine follow-up procedures in significant liver pathology for patients with chronic hepatitis B (CHB) with persistently normal alanine aminotransferase (PNALT) or minimally raised alanine aminotransferase (ALT).
Methods
Patients with CHB who underwent percutaneous liver biopsy were retrospectively reviewed. The relationship of liver pathology with age, ALT, hepatitis B e-antigen, and spleen thickness was statistically analyzed, and the predictive accuracy of spleen thickness was evaluated.
Results
In total, 80.65% of patients had significant necroinflammation and/or fibrosis. Nearly 60% of patients had splenomegaly, of which 89.12% had a histopathological grade of ≥G2 and/or S2. Spleen thickness was predictive of liver pathology, and significant histological findings increased as the hepatitis B virus (HBV) DNA level increased.
Conclusions
Spleen thickness is an effective predictor of liver pathology in patients with PNALT or minimally raised ALT. Additionally, the prevalence of significant histological findings tended to increase as the HBV DNA level increased. Patients with CHB and splenomegaly and a high HBV DNA level should be treated early with antivirals to improve liver pathology.
Keywords
Introduction
Approximately one-third of the world’s population has serological evidence of a past or present infection with hepatitis B virus (HBV), and 350 to 400 million people are chronic HBV surface antigen (HBsAg) carriers. 1 Once infected with HBV, a patient can develop chronic hepatitis B (CHB) and gradually develop cirrhosis or hepatocellular carcinoma.2,3 More than 500,000 deaths are attributable to cirrhosis and liver cancer annually (World Health Organization and Centers for Disease Control and Prevention fact sheets are available at www.who.int and www.cdc.gov). Hepatic fibrogenesis is a dynamic process, and removal of the virus may lead to the reversal of fibrosis.4–6 Therefore, identification of the best time for antiviral therapy is very valuable for the management of CHB. There are specific indications for the treatment of patients with an alanine transaminase (ALT) level of twice the upper limit of normal (2×ULN) or higher; however, there is greater subjectivity for patients with persistently normal ALT (PNALT) or minimally raised ALT according to the current 2012 guidelines of the European Association for the Study of the Liver (EASL) and the 2010 guidelines of the Chinese Society of Hepatitis, among others.1,7–9 Liver biopsy should be based on liver pathology or fibrosis by indirect indicators; however, liver biopsy is an invasive procedure with major complications, including bleeding and bile leakage. 10 This, together with sampling error, 11 leaves many patients afraid to undergo liver biopsy compared with other noninvasive inspection techniques. However, no generally accepted noninvasive inspection method is currently available. The following two procedures are typically applied for a comprehensive assessment. First, routine follow-up data are collected, such as age, ALT, hepatitis B e-antigen (HBeAg), and HBV DNA. Second, imaging that can assess hepatic fibrosis is performed, including transient elastography (TE), magnetic resonance elastography (MRE), main portal vein (MPV) diameter measurement, and ultrasonography.
Among patients who do not comply with antiviral indications, researchers and guidelines currently pay close attention to patients aged ≥40 years;6,7,12 however, the 2012 EASL guideline 8 and another group of researchers 13 recommended advancing this age to 30 years. Although some studies have shown that the necroinflammatory grade and fibrotic stage were significantly associated with the ALT level,14–16 HBV viral load, and HBeAg status,14,17–19 others have shown no association with routine follow-up data such as ALT, HBeAg, HBV DNA, and liver pathology.20,21 In short, no unified conclusion has been reached.
In terms of noninvasive imaging used to assess hepatic fibrosis, both MRE22,23 and TE 24 have excellent diagnostic performance for the detection of fibrosis. However, some meta-analyses have shown that TE is not ideal for the detection of stage F2–F4 fibrosis compared with its ability to identify cirrhosis.25,26 Both MRE and TE have limitations. 27 Patients who are obese (or have an elevated body mass index) have narrow intercostal spaces, making TE difficult to perform.28,29 Additionally, Coco et al. 30 suggested that TE may not be as reliable in patients with CHB. Currently, MRE cannot be performed in patients with an increased hepatic iron content because of signal-to-noise limitations that prevent wave visualization, and it is a costly procedure. 22 In summary, regardless of the sensitivity, specificity, cost, or risk perspective, no ideal tool that can assess hepatic fibrosis is yet available. In China, ultrasonography is often used as a routine follow-up imaging tool to assess the dynamic changes in the liver and spleen among patients with CHB. Furthermore, the Chinese Society of Hepatitis 2010 CHB guidelines 7 indicated that patients with splenomegaly should be considered for treatment. Ultrasonography is superior to CT and MRI in regard to safety as well as real-time and spatial resolution,31,32 and it is often the first-line imaging modality used to assess changes in the liver, spleen, and other organs. Ultrasonography reportedly might show the progression of CHB through progressive changes in liver or spleen echogenicity, 33 indicating that it is particularly important for patients with PNALT or a minimally raised ALT to undergo early clinical interventions to prevent further serious consequences. Ultrasonography is considered a trustworthy tool because of its convenient operation, inexpensiveness, and stability of the results. However, this noninvasive imaging technique is not suggested as a routine follow-up procedure for patients with CHB in the current guidelines, including the 2012 EASL guidelines, 2012 Asian Pacific Association for Study of Liver guidelines, and others.6–9
In the present study, we observed changes in spleen thickness and other laboratory parameters and compared the differences in clinical and histopathological features of patients with CHB with PNALT versus minimally elevated ALT. We aimed to determine the predictive value of the spleen thickness and the MPV diameter in liver pathology as well as the role of routine follow-up procedures in significant liver pathology for patients with CHB with PNALT or minimally raised ALT.
Materials and methods
Patients
This study included patients with CHB who underwent a percutaneous liver biopsy from August 2009 to October 2013 in the Department of Infectious Disease, Sir Run Run Shaw Hospital, Hangzhou, China. These patients had been serum-positive for HBsAg for at least 6 months but had not undergone antiviral, glycyrrhizin, or thymosin therapy. Patients were excluded if they had concomitant liver diseases, including acute hepatitis (HAV, HCV, HEV, EBV, or CMV), hepatic carcinoma, chronic hepatitis C or D infection, Wilson’s disease, autoimmune liver disease, drug-induced hepatitis, alcoholic liver disease, fatty liver, anemia such as that caused by thalassaemia or glucose-6-phosphate dehydrogenase deficiency, Budd–Chiari syndrome, hematological malignancies, and other diseases that could cause splenomegaly. Patients with an ALT level of ≥2×ULN were also excluded. Prior to liver biopsy, all patients were informed about the procedure, including alternatives, risks, benefits, and limitations, before they provided written consent. The study was approved by the Medical Ethics Committee of Sir Run Run Shaw Hospital, Zhejiang University, School of Medicine.
Study design and patients
Demographic, clinical, and laboratory data (age, sex, ALT level, HBeAg status, HBV DNA, and changes in ultrasound findings at the time of liver biopsy) were recorded during the week prior to the liver biopsy. All patients were divided into two groups (PNALT and mildly elevated ALT) and then further divided into four subgroups (<0.5×ULN, 0.5–1.0×ULN, ≤1.5×ULN, and 1.5–2.0×ULN).1,12 According to the literature, 40 years of age is the clinical cutoff for evaluation of patients with CHB; therefore, we divided the total patient population into two age groups (<40 and ≥40 y).6,7,12,34 We then further divided them into four subgroups, with every 10 years defined as an age group (18–29, 30–39, 40–49, and 50–59 y). Similar to the Reveal study, the patients were then divided into three groups according to their HBV DNA level (<1 × 103, 1 × 103–9.99999 ×105, and ≥1 × 106 copies/mL).35–40 The examined ultrasonography findings of patients with simple chronic hepatitis were the spleen thickness (the spleen was considered normal if its thickness was ≤4 cm on ultrasonography), width of the splenic vein, and intensity of the hepatic echo.41,42 We observed changes in the spleen size using these analyses.
Materials
The laboratory tests were measurement of the serum ALT level, HBV markers, and the HBV DNA level. The serum ALT level was determined with an automated Beckman Access Immunoassay system (UniCel DxI 800; Beckman Coulter, Brea, CA, USA) and is expressed as ×ULN. HBV markers were detected by a chemiluminescent microparticle immunoassay (ARCHITECT i2000 SR; Abbott, Wiesbaden, Germany). The HBV DNA level was quantified by the Applied Biosystems real-time PCR system (ABI 7500; Applied Biosystems, Foster City, CA, USA), with the lowest detection limit at 1000 copies/mL. Percutaneous liver biopsies were guided by a Philips ultrasonography system (iU22; Philips, Andover, MA, USA), with a fine needle and a 18G sheathed cutting needle (Bard Max-Core Disposable Core Biopsy Instrument; Bard Biopsy Systems, Tempe, AZ, USA).
Liver biopsy and histopathology
Liver biopsies were performed under routine ultrasound guidance with a minimum length of 1.0 cm (range, 1.5–2.5 cm) of the liver biopsy sample and at least 11 portal tracts required for histopathological diagnosis.11,43,44 Histopathological grades of necroinflammation and stages of fibrosis were determined using the Scheuer system.43,45 Significant histology was defined as necroinflammation of grade ≥G2 or fibrosis of stage ≥S2.7,11,17,43,46
Splenic thickness measurement
After the patients had fasted for about 8 hours, ultrasonographic examination for measurement of the splenic thickness was performed with the patients in the supine position or with the right arm recumbent. The minimum distance from the depressed surface of the spleen to the contralateral convex surface was measured through the intercostal oblique section. 47
Statistical analysis
Normally distributed variables are presented as mean ± standard deviation, and skewed variables are presented as median and interquartile range. Data were analyzed using SPSS for Windows, version 13.0 (SPSS Inc., Chicago, IL, USA). Statistical analyses of the relationship between the inflammation grade or fibrosis stage and age, ALT, HBeAg, and spleen thickness were performed using the chi-square test. The change trends between liver histology and the incremental HBV DNA level, ALT level, and age were performed using the Cochran–Armitage trend test. All significance tests were two-tailed, and a P value of <0.05 was considered statistically significant. The predictive accuracy of the spleen thickness and the MPV diameter was evaluated using receiver operating characteristic curves.
Results
Histological findings
This study included 62 patients with CHB with either PNALT or minimally raised ALT who underwent liver biopsy. The patients’ characteristics are shown in Table 1. The mortality and major complication rates were 0% in our series. Of all 62 patients, 50 (80.65%) had significant necroinflammation and/or fibrosis, and 12.90% had established cirrhosis. Thirty-nine patients had ≥G2 histopathological changes, 44 patients had ≥S2 histopathological changes, and 8 (12.90%) patients had established cirrhosis (Table 2). Most of the patients in this study had significant histological changes.
Characteristics of the 62 patients with chronic hepatitis B.

Data are presented as n (%), mean ± standard deviation, or median (25th–75th percentile). ALT, alanine aminotransferase; HBV, hepatitis B virus; HBeAg, hepatitis B e-antigen; PNALT, persistently normal alanine aminotransferase.
Inflammation grades and fibrosis stages of 62 patients.
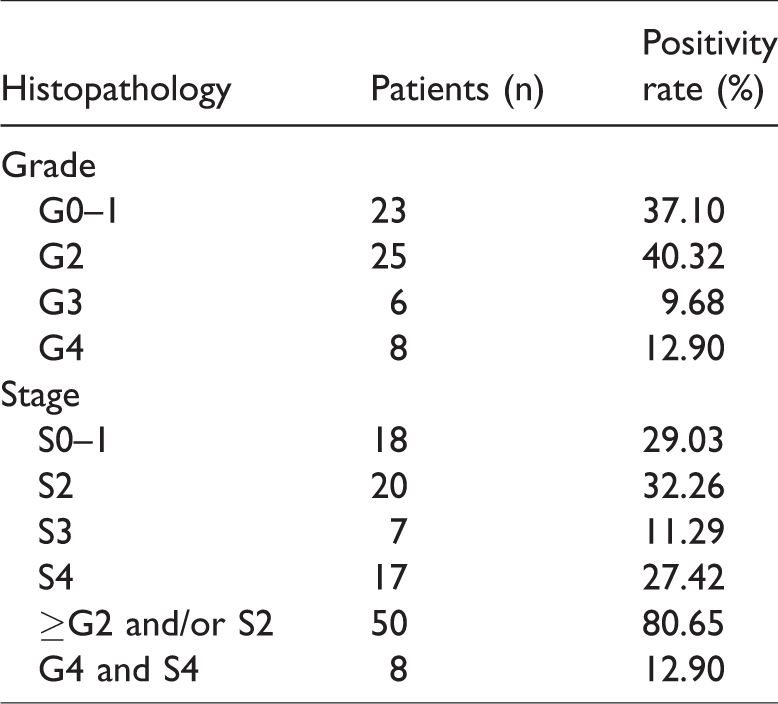
Relationship between inflammation grade or fibrosis stage and spleen thickness, age, ALT, and HBeAg.

ALT, alanine aminotransferase; HBeAg, hepatitis B virus e-antigen.
Correlation between liver histopathology and spleen thickness, MPV diameter, ALT, HBeAg status, and HBV DNA (Table 3) (Figure 1)
Statistical analysis (chi-square test) confirmed a close and statistically significant correlation between significant hepatic fibrosis (≥S2) and splenomegaly (P = 0.01). Significant necroinflammation (≥G2) alone had no correlation with splenomegaly, but of the 37 patients with splenomegaly, 89.12% (33) had histopathology of ≥G2 and/or S2. Additionally, the area under the curve for the MPV diameter and spleen thickness was 0.667 and 0.782, respectively. In comparison, the spleen thickness was more predictive of the liver pathology in patients with CHB with ALT of <2×ULN; that is, the spleen thickness was one of the best predictors of liver pathology. The receiver operating characteristic curve analysis (Figure 1) suggested that the most useful cutoff value of the spleen thickness was 4.58 cm, where the sum of the positive likelihood ratio (4.56) was the highest. The sensitivity and specificity of the cutoff value of ≥4.58 cm were 38.0% and 91.7%, respectively.

The area under the curve (AUC) of spleen thickness was 0.782 (95% CI 0.609–0.954). The AUC of the MPV diameter was 0.667(95% CI 0.485–0.848). The curves were constructed using the data of 62 CHB patients, 50 patients had significant necroinflammation and/or fibrosis (≥G2 or ≥S2), 37 patients had splenomegaly.
Our study also showed no correlations between the HBeAg status, age, or ALT level and significant necroinflammation (≥G2) or fibrosis (≥S2).
Cochran–Armitage trend test between liver pathology and age, ALT, or HBV DNA
We further analyzed the change trends between liver histology and the incremental HBV DNA level, ALT level, and age with the Cochran–Armitage trend test in patients with PNALT and mildly elevated ALT. The prevalence of significant histological findings increased with incremental increases in the HBV DNA level from 1 × 103 copies/mL to 1 × 103–9.99999 × 105 copies/mL and ≥1 × 106 copies/mL (df = 2, P = 0.03, z = 2.21) (Table 4). Although these differences were statistically significant, it seems that more patients had significant necroinflammation and/or fibrosis in the 1 × 103–9.99999 × 105 copies/mL group than in the ≥1 × 106 copies/mL group. The prevalence of significant histological findings did not increase with incremental changes in the ALT level from ≤0.5×ULN, 0.5–1.0×ULN, and ≤1.5×ULN to 1.5–2.0×ULN (df = 3, z = 0.88). Additionally, there was no clear trend across the age groups (df = 3, z = 0.97) (Table 4).
Cochran–Armitage trend test between liver pathology and different age, ALT, and HBV DNA groups.

ALT, alanine aminotransferase; HBV, hepatitis B virus.
Patients’ dispositions
Fifty patients had histopathology of ≥G2 and/or S2, among whom 47 patients chose antiviral therapy. One of these 47 patients (G4/S4 histopathology) developed liver cancer 2 years later and underwent radical surgery. The remaining 46 patients were still undergoing follow-up at the clinic, with normal liver function, a negative HBV DNA status, and no worsening of the imaging findings before liver puncture. Twelve patients had histopathology of <G2 and/or S2. Eight patients underwent no antiviral treatment and continued outpatient follow-up, and their clinical situation remained basically the same as before. Three patients chose antiviral therapy and continued follow-up at the clinic; they maintained normal liver function, a negative HBV DNA status, and no worsening of the imaging findings before liver puncture.
Discussion
Based on current guidelines and reports,6–8,12,13 there is a dispute regarding at what age antiviral therapy should be initiated for patients with PNALT or minimally raised ALT. Some studies have shown that the presence of PNALT or minimally raised ALT could predict fibrosis in patients with chronic liver disease together with laboratory test results and age, among other parameters.34,48,49 In the present study, we found that the average age of patients with PNALT or minimally raised ALT was 39.03 years and that the results of significant necroinflammation (≥G2) or fibrosis (≥S2) between the two age groups (<40 and ≥40 y) was not significantly different. The prevalence of significant histological findings did not appear to increase with increasing age (groups divided by 10-year age increments), indicating that 40 years is not a pivotal age. More research is needed to identify the prime time for treatment when patients with PNALT or minimally raised ALT are divided into high-risk and low-risk populations.
The present study did not reveal a correlation between significant necroinflammation (≥G2) or fibrosis (≥S2) and the HBeAg status, which is consistent with most previous reports.14,17–19,50 In contrast, other studies have revealed different findings.21,22,35,51 HBeAg is related to many complex mechanisms, and its relationship with liver pathology requires further study.
We found no correlation between significant necroinflammation (≥G2) or fibrosis (≥S2) and the ALT level when patients were divided into normal ALT and minimally raised ALT groups. Furthermore, there was no trend in the liver histology or incremental ALT level, consistent with a report by Alam et al. 13 and in agreement with the suggestion to lower the ULN for ALT to 30 IU/L for men and 19 IU/L for women.15,16
Significant necroinflammation and/or fibrosis (≥G2, 62.90%; ≥S2, 70.97%) was found in 80.65% of the patients in our study, similar to a study showing that significant fibrosis is not rare in Chinese patients with CHB exhibiting PNALT. 52
We also found that the prevalence of significant histological findings increased with incremental increases in the HBV DNA level, similar to the results of several previous studies.17–20 Conversely, antiviral therapy should improve liver pathology.4–6 In our study, more patients had necroinflammation and/or fibrosis in the HBV DNA 1 × 103–9.99999 × 105 than ≥1 × 106 copies/mL group. Presumably, patients with a moderate virus load have repeated viral immune clearance, and after every immune clearance, the viral load is likely lower than before, resulting in repeated liver cell damage and gradual changing liver pathology. 53
Our analysis confirmed that the spleen thickness is one of the best predictors of liver pathology and showed that this parameter has high specificity at 4.58 cm. Judging from the analysis of the pathophysiology of fibrosis, injury and subsequent inflammation are prominent in most diseases that lead to fibrosis. 50 The MPV diameter has great significance in judging the degree of esophageal varices and cirrhosis, but it has little significance for patients with CHB with mild liver dysfunction.54,55 Therefore, fibrosis is a dynamic process, and routine follow-up protocols that include changes in B-mode or Doppler ultrasonography findings that are associated with fibrosis should be recognized and further applied and studied.
Conclusion
Splenomegaly is a good predictor of significant necroinflammation (≥G2) and fibrosis (≥S2) in patients with PNALT or minimally raised ALT, and it has very high specificity for hepatic pathological changes when the spleen thickness is >4.58 cm. Additionally, we observed that the prevalence of significant histological findings increased with incremental increases in the HBV DNA level. Observation of splenomegaly and identification of HBV DNA levels that are associated with fibrosis can prompt antiviral treatment, which improves liver pathology. It is important for patients with PNALT or minimally raised ALT to undergo analysis of the spleen thickness by ultrasonography as a follow-up procedure.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was funded by the General Research Plan for Medical and Health of Zhejiang Province (Class A) x 2015KYAl32).
