Abstract
Objective
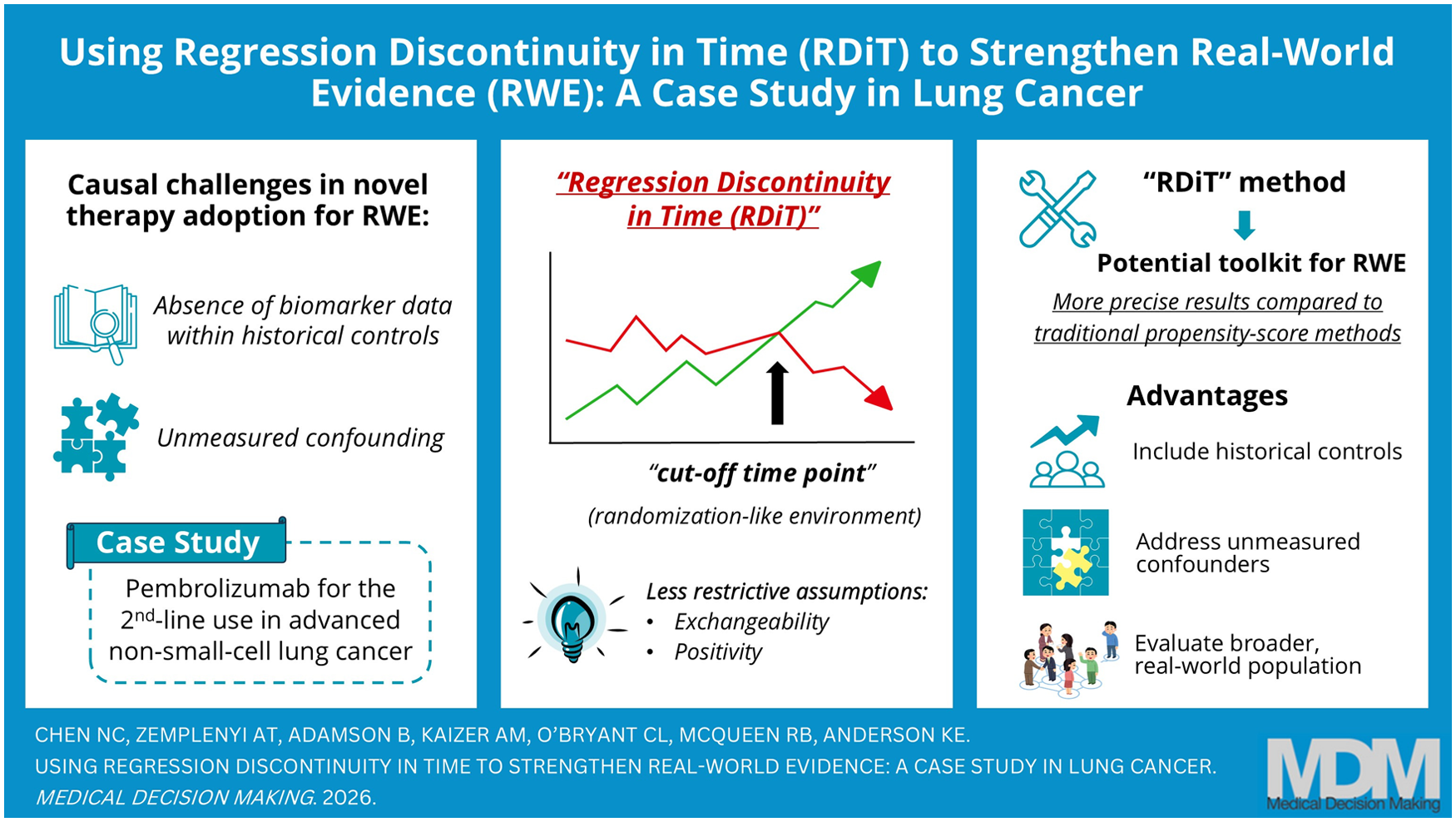
Real-world evidence is increasingly leveraged to assess treatment effectiveness outside of clinical trials, yet unmeasured confounders and missing data pose challenges to causal inference, which is particularly problematic when incorporating historical controls that lack recent prognostic factors. We applied the regression discontinuity in time (RDiT) design, a quasi-experimental approach, in a real-world case study of second-line pembrolizumab versus docetaxel for advanced non-small-cell lung cancer (aNSCLC). We compared results from the RDiT method with time-stratified inverse probability treatment weighting (ts-IPTW), benchmarking results against long-term trial data.
Methods
We conducted a retrospective cohort study of patients who received second-line pembrolizumab or docetaxel after platinum-based chemotherapy between 2011 and 2023. The introduction of pembrolizumab (Q2 2016) served as the discontinuity threshold in an RDiT framework, with treatment probabilities estimated via logistic regression. Survival outcomes, including hazard ratios (HRs), median overall survival, and restricted mean survival time (RMST), were compared across RDiT, ts-IPTW, and reconstructed trial estimates.
Data Sources
This study used the US-based, electronic health record–derived deidentified Flatiron Health Research Database.
Results
Among 1,975 patients (1,014 pembrolizumab, 961 docetaxel), RDiT estimated an adjusted median survival of 11.5 mo for pembrolizumab versus 6.9 mo for docetaxel (HR 0.65, 95% confidence interval [CI]: 0.48, 0.89), compared with ts-IPTW (HR 0.52, 95% CI: 0.42, 0.64) and 5-y trial data (HR 0.70, 95% CI: 0.61, 0.80). RDiT produced smaller survival gains that were better aligned with trial results relative to ts-IPTW, suggesting it may help mitigate unmeasured confounding in real-world studies.
Conclusions
The RDiT may provide effect estimates more consistent with trial data, particularly when confounding is a concern. More research is required to examine its performance in other applications.
Highlights
The regression discontinuity in time (RDiT) method incorporates historical controls and addresses unobserved confounders to strengthen causal inference.
Compared with traditional propensity score–based approaches, RDiT accommodates historical and concurrent controls and reduces reliance on comprehensive measurement of observed confounders when treatment practices or biomarkers change over time.
As real-world evidence increasingly informs regulatory, coverage, and pricing decisions, rigorous analytic methods are essential to produce credible and decision-relevant estimates.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
