Abstract
Objectives
The objective of this study was to assess serum histamine levels in patients with non–small cell lung cancer and investigate their association with tumor metastasis.
Methods
A retrospective study was conducted at Lu’an Hospital between 2020 and 2024, including 90 patients (70 with non–small cell lung cancer and 20 with benign pulmonary nodules) selected from an initial cohort of 416 patients after applying strict inclusion and exclusion criteria. Serum histamine levels were measured, and other clinical characteristics and laboratory parameters were obtained directly from the electronic medical record system. Receiver operating characteristic curves were used to evaluate the diagnostic performance. Non–small cell lung cancer patients were stratified by metastatic status for group comparisons and logistic regression analyses.
Results
Non–small cell lung cancer patients had significantly higher serum histamine levels than those with benign nodules (median: 8.58 vs. 5.97 µg/L, p = 0.024). A cutoff of 7 µg/L yielded a sensitivity of 65.7% and a specificity of 65.0% for distinguishing non–small cell lung cancer, with an area under the curve of 0.666. Among non–small cell lung cancer patients, those with metastasis (n = 35) had higher histamine levels than nonmetastatic cases (median: 9.39 vs. 7.18 µg/L, p = 0.001). Complement C3 levels and the neutrophil-to-lymphocyte ratio were also higher in metastatic patients. Multivariable logistic regression analyses showed that serum histamine (odds ratio: 1.213, 95% confidence interval: 1.011–1.455, p = 0.038) and complement C3 (OR: 1.488, 95% CI: 1.104–2.004, p = 0.009) were independently associated with metastasis in non–small cell lung cancer patients.
Conclusions
Serum histamine levels were elevated in non–small cell lung cancer patients and were independently associated with tumor metastasis. These findings provide preliminary evidence of a possible connection between histamine and the emerging field of allergo-oncology. Complement C3 was also found to be independently associated with metastasis. Further studies are warranted to validate these results and explore the underlying immunological mechanisms.
Introduction
Non–small cell lung cancer (NSCLC) is one of the leading causes of cancer-related death globally. 1 Although tumor metastasis is a key determinant of prognosis,2,3 increasing evidence suggests that immune tolerance plays a crucial role in contributing to tumor progression and immune escape in NSCLC.4–6
Histamine, a central mediator in allergic reactions, not only mediates classical inflammatory responses7,8 but also exerts diverse immunomodulatory effects. It can influence dendritic cell function 9 and modulate T-cell activity. 10 Histamine is primarily released by tissue-resident mast cells and circulating basophils. Notably, mast cell infiltration has been observed in lung cancer tissues. 11 However, the implications of histamine release in the tumor microenvironment and its systemic circulation in lung cancer patients remain to be fully elucidated.
The emerging field of allergo-oncology has recently drawn attention to the complex interplay between allergic responses and cancer biology. This interdisciplinary field explores how immune mediators, traditionally associated with hypersensitivity, may influence tumor development and immune surveillance. 12 Understanding this relationship may open new avenues for cancer diagnosis and therapy, particularly in malignancies such as NSCLC, where immune escape mechanisms are critical.
Our previous study indicated that histamine can alter the function of monocyte-derived dendritic cells and promote the differentiation of regulatory T cells. 13 These observations raise the question of whether histamine modulates immune responses in the tumor microenvironment, although this remains to be elucidated in the context of NSCLC.
Despite previous insights into histamine’s role in immune regulation, its clinical relevance in pulmonary diseases—particularly in distinguishing benign from malignant lung lesions and in relation to metastatic progression in NSCLC—remains uncertain. In this retrospective study, we measured serum histamine levels in patients with benign pulmonary nodules and NSCLC, including comparisons between metastatic and nonmetastatic cases. These observations suggest a possible connection between histamine levels and NSCLC metastasis. The findings may be relevant to future investigations within the emerging framework of allergo-oncology.
Materials and methods
Study participants
This was a retrospective observational study. The study was conducted in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 14 Between 2020 and 2024, 416 patients with pulmonary space-occupying lesions were initially identified through the electronic medical record system. After applying strict inclusion and exclusion criteria, 90 patients were included (70 with NSCLC and 20 with benign pulmonary nodules). Clinical data and all laboratory parameters, except for serum histamine, were extracted from the electronic medical records. The inclusion criteria were as follows: (a) all patients underwent comprehensive clinical evaluation, including imaging, hematology, and pathology, and had complete clinical data; (a) all patients had a first-time diagnosis; (c) patients had no recent pulmonary or acute infections and had not used antibiotics within the past week; and (d) patients had no concurrent primary tumors in other organs. According to the electronic medical records, no patients had documented recent drug allergies or antihistamine use prior to blood sampling. However, data on other allergic conditions (e.g. food or environmental allergies) were not available.
The study protocol was approved by the Ethics Committee of Lu’an Hospital affiliated to Anhui Medical University (Approval No. 2023LLKS011, 10 April 2024). A waiver of informed consent was granted due to the retrospective nature of the study and anonymized data handling. All patient data were anonymized. The study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024).
Samples collection and analysis
Venous blood samples (at least 6 mL) were collected from each patient upon admission, before conducting any treatments that could potentially affect the test indicators. Of the 6 mL, 2 mL was placed into an EDTA-2K anticoagulant tube for complete blood count (CBC), and the remaining 4 mL was collected in a plain tube without anticoagulant for serum separation. The serum was used to measure complement C3 and C4 levels.
Histamine levels were measured using human histamine ELISA (enzyme-linked immunosorbent assay) kits (catalog number: HB028-Hu; Shanghai Hengyuan Biotechnology Co, Ltd, Shanghai, China). Serum samples were separated within 1 h of collection by centrifugation at 3000 rpm for 10 min and stored in aliquots at –80°C until analysis. To preserve sample integrity, each aliquot was thawed only once to avoid repeated freeze–thaw cycles. All samples were processed and handled according to the standardized protocols to ensure consistency and stability. According to the manufacturer’s specifications, the detection range of the assay was 0.5–18 µg/L, with a minimum detectable concentration (sensitivity) of 0.125 µg/L. Quality control materials provided with the kit were included in each run to ensure assay reliability.
CBC parameters were analyzed using the Sysmex XN-2100™ automated hematology analyzer. This system uses hydrodynamic focusing for red blood cell and platelet assessment and semiconductor-based flow cytometry for analyzing other blood cells. Serum complement C3 and C4 levels were measured using the turbidimetric method on a Beckman Coulter AU-5831™ fully automated biochemical analyzer.
Statistical analysis
All statistical analyses were conducted using SPSS version 19.0. Continuous variables were expressed as median (Q1, Q3), and categorical variables as n (%). Group comparisons were performed using the Mann–Whitney U test for continuous variables and the chi-square or Fisher’s exact test for categorical variables. Receiver operating characteristic (ROC) analysis was used to assess the diagnostic value of serum histamine. To identify the risk factors for NSCLC metastasis, variables with p < 0.05 in univariate analysis were entered into multivariate binary logistic regression. The results were reported as odds ratios (ORs) with 95% confidence intervals (CIs). A p-value <0.05 was considered statistically significant.
Results
Patient selection
A total of 416 patients were initially identified from the hospital’s electronic medical record system. From this initial cohort, 93 were excluded due to a lack of pathological confirmation, 76 were excluded because they were first diagnosed outside our hospital or prior to sample collection, 107 were excluded for concurrent pulmonary or acute infections at admission, 24 for antibiotic use within 1 week before sampling, and 26 for coexisting or uncertain other primary tumors. No patients were excluded for recent drug allergy or antihistamine use. A total of 90 patients were included in the final analysis. The patient selection process is summarized in Figure 1.

Flowchart of patient selection.
Patient characteristics
A total of 90 patients participated in the study, including 44 men and 46 women. Of these, 20 patients had benign pulmonary nodules (median age of 62.0 years; interquartile range (IQR): 55.00–68.75), while 70 patients were diagnosed with NSCLC (median age of 67.0 years; IQR: 54.75–73.25). There were no significant differences between the two groups in terms of age (p = 0.262), sex (p = 0.910), smoking status (p = 0.861), or alcohol consumption (p = 0.936). The prevalence rates of hypertension and diabetes were higher in the NSCLC group (32.86% and 14.29%, respectively), but these differences were not statistically significant (p = 0.268 and p = 0.109, respectively).
Serum histamine levels were significantly higher in NSCLC patients than in those with benign pulmonary nodules (8.58 (5.92, 11.09) vs. 5.97 (4.61, 9.44) µg/L, p = 0.024). The patient characteristics and serum histamine levels are summarized in Table 1.
Comparison of basic clinical features and serum histamine between the two groups.

p values calculated by Fisher’s exact test due to small expected cell counts.
NSCLC: non–small cell lung cancer.
Diagnostic performance of serum histamine in differentiating NSCLC from benign pulmonary nodules
ROC analysis was performed to assess the diagnostic value of serum histamine in distinguishing NSCLC from benign pulmonary nodules. At the optimal cutoff value of 7 µg/L, determined using the Youden index, serum histamine demonstrated a sensitivity of 65.7%, a specificity of 65.0%, and an area under the curve of 0.666 (Figure 2).

ROC curve for serum histamine in differentiating benign and malignant pulmonary masses. ROC: receiver operating characteristic.
Among the 70 NSCLC patients, 45 had serum histamine levels ≥7 µg/L (true positives), while 25 had levels below this cutoff (false negatives). Of the 20 patients with benign pulmonary nodules, 7 had histamine levels ≥7 µg/L (false positives) and 13 had levels <7 µg/L (true negatives). Based on these results, the positive predictive value was 86.5% and the negative predictive value was 34.2%.
Serum histamine and its association with metastasis in NSCLC patients
Based on metastatic status, the 70 patients with NSCLC were divided into two groups: metastatic (n = 35) and nonmetastatic (n = 35). Laboratory parameter comparisons between the two groups are presented in Table 2.
Comparison of laboratory parameters in NSCLC patients with and without metastasis.
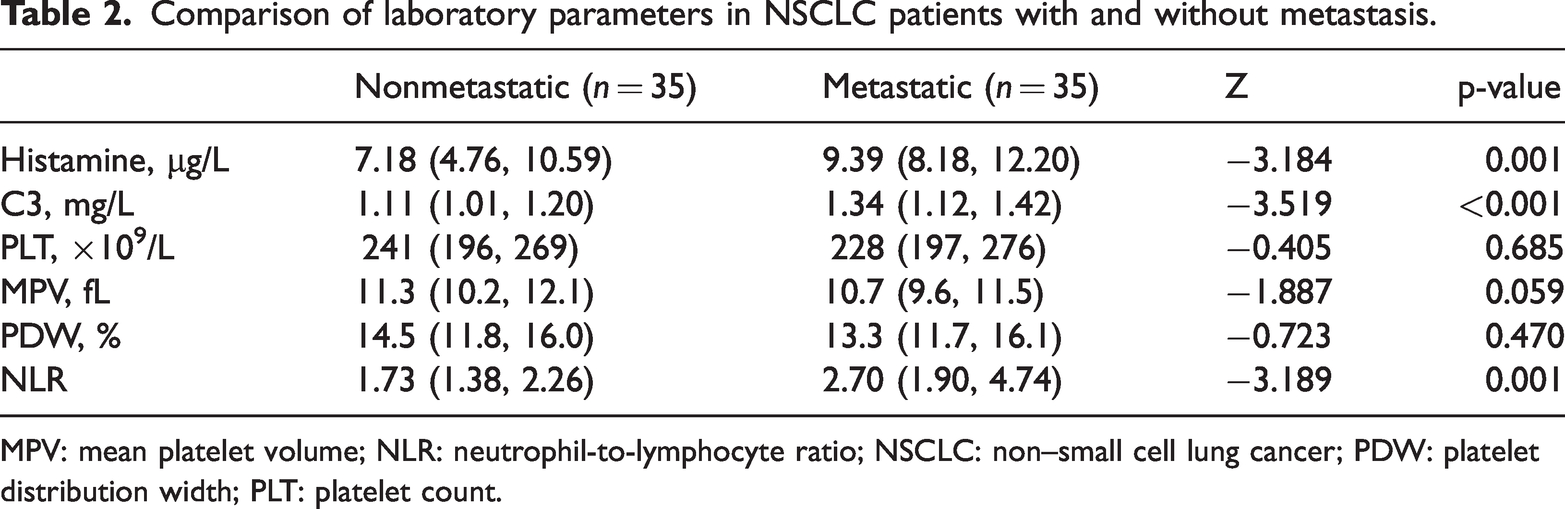
MPV: mean platelet volume; NLR: neutrophil-to-lymphocyte ratio; NSCLC: non–small cell lung cancer; PDW: platelet distribution width; PLT: platelet count.
Serum histamine levels were significantly higher in patients with metastatic NSCLC than in those without metastasis (median (IQR): 9.39 (8.18–12.20) vs. 7.18 (4.76–10.59) µg/L, p = 0.001). Complement C3 levels and the neutrophil-to-lymphocyte ratio (NLR) were also elevated in the metastatic group (C3: 1.34 (1.12–1.42) vs. 1.11 (1.01–1.20) mg/L, p < 0.001; NLR: 2.70 (1.90–4.74) vs. 1.73 (1.38–2.26), p = 0.001). The mean platelet volume (MPV) was lower in the metastatic group (10.7 (9.6–11.5) vs. 11.3 (10.2–12.1) fL), but the difference did not reach statistical significance (p = 0.059). No significant differences were observed in platelet count or platelet distribution width between the two groups. Detailed comparisons are presented in Table 2.
Logistic regression analysis of factors associated with metastasis in NSCLC patients
To further evaluate the potential association between laboratory parameters and metastasis in NSCLC patients, a binary logistic regression analysis was performed, including serum histamine, complement C3 (multiplied by 10 for scaling), and NLR as independent variables.
The results demonstrated that both histamine and C3 were independently associated with the presence of metastasis. Specifically, serum histamine was significantly associated with metastasis (p = 0.038; OR: 1.213; 95% CI: 1.011–1.455), indicating that each 1 µg/L increase in histamine level raised the odds of metastasis by 21.3%. Similarly, C3 (scaled ×10) was also significantly associated with metastasis (p = 0.009; OR: 1.488; 95% CI: 1.104–2.004). In contrast, NLR was not significantly associated with metastasis (p = 0.273) (Table 3).
Logistic regression analysis of factors associated with metastasis in NSCLC patients.

CI: confidence interval; NLR: neutrophil-to-lymphocyte ratio; NSCLC: non–small cell lung cancer; OR: odds ratio; SE: standard error.
Discussion
In this study, serum histamine concentrations were significantly elevated in NSCLC patients compared to those with benign nodules, demonstrating moderate diagnostic value. Moreover, histamine levels were higher in NSCLC patients with metastasis than in those without, and histamine was identified as an independent risk factor for metastasis in multivariate analysis.
Histamine has been reported to play dual roles in tumor regulation, promoting or inhibiting tumor growth in various malignancies, including cervical cancer, ovarian cancer, colorectal cancer, and melanoma.15–18 Given the widespread expression of histamine receptors (H1, H2, H3, and H4) on both immune and tumor cells,19–22 fluctuations in histamine levels may influence tumor behavior. Our previous work demonstrated that histamine induces indoleamine 2,3-dioxygenase (IDO) expression in dendritic cells via H2 receptor activation. 13 IDO-positive dendritic cells are well-established mediators of immune tolerance, capable of suppressing T-cell responses and promoting an immunosuppressive tumor microenvironment. These results may provide a possible context for the elevated histamine levels observed in metastatic NSCLC patients; however, the specific mechanisms and their implications for tumor progression require further investigation.
These findings align with growing evidence that histamine may influence the tumor microenvironment, with reported roles in vasodilation, 23 immune modulation, 24 and tumor invasion. 25 Although our results show an association between elevated serum histamine and metastasis in NSCLC, and histamine was identified as an independent risk factor in multivariate analysis, the causal role of histamine in facilitating tumor cell dissemination and immune escape remains to be established. Our results provide observational data relevant to the emerging field of allergo-oncology, which investigates the links between allergic inflammation and cancer biology.
NLR, 26 complement C3, 27 and MPV 28 have been reported to associate with tumor progression and metastasis in lung or other malignancies. In our study, serum histamine levels were significantly elevated in the metastatic NSCLC group, along with higher NLR and C3 levels, whereas MPV exhibited a downward trend that approached but did not reach statistical significance (p = 0.059). Multivariate analysis in our cohort identified only histamine and C3 as independent risk factors for metastasis.
Mechanistically, histamine is principally released by mast cells, which infiltrate lung cancer tissues, 11 and components C3 and C5 can be cleaved into the anaphylatoxins C3a and C5a, which, in turn, trigger mast cell degranulation,29–31 leading to histamine release and enhancing vascular permeability and leukocyte recruitment. This suggests a potential bidirectional interaction between histamine and complement activation, mediated by mast cells. Although direct mechanistic links in NSCLC remain unexplored, such crosstalk has been described in other malignancies and inflammatory contexts. For instance, C3a-driven mast cell activation induces histamine release, and mast cell–derived C3 may influence local immune regulation. 32 The concurrent elevations of histamine and C3 observed in metastatic NSCLC patients could therefore reflect a coordinated immunoregulatory loop, in which mast cells contribute to a metastasis-permissive microenvironment.
Despite these observed associations, this study has certain limitations. The cross-sectional nature of our study precludes any causal inference; thus, it remains unclear whether histamine elevation actively contributes to NSCLC progression or merely reflects tumor-related biological processes. Second, although logistic regression was performed, the relatively modest sample size—constrained by strict inclusion and exclusion criteria as well as the limited availability of eligible patients within our institutional database—limited our ability to conduct robust multivariable analyses adjusting for potential confounders such as age, comorbidities, and tumor burden. Third, non–drug allergic histories were not collected because the electronic medical record system only recorded drug allergies, limiting the assessment of patients’ overall allergic background.
Future research should consider longitudinal designs to track histamine dynamics over time and assess their prognostic relevance. Investigating the expression of histamine receptors and associated signaling molecules in tumor and immune cells may further clarify the biological role of histamine in NSCLC. Larger, multicenter studies are warranted to validate these preliminary findings and determine the clinical relevance of serum histamine in lung cancer.
In conclusion, this study demonstrates that serum histamine is elevated in NSCLC, particularly in metastatic cases, suggesting a potential role in tumor progression. These preliminary findings underscore the need for further research to explore the diagnostic and immunological relevance of histamine in lung cancer.
Footnotes
Acknowledgements
The authors thank all participants and staff involved in this study. We also acknowledge the assistance of AI language tools for language editing purposes.
Author contributions
Zhai Rongrong, Shu Wenyue, Hu Yanyun, Zhang Fengli, and Sun Lei contributed to the study conception and design. Data collection and analysis were performed by Zhai Rongrong, Shu Wenyue, and Hu Yanyun. The first draft of the manuscript was written by Zhai Rongrong and Shu Wenyue. All authors reviewed and approved the final manuscript.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest regarding the publication of this paper.
Funding
This study was supported by the Science and Technology Planning Project of Lu’an City, Anhui Province (Project No.2023lakj005).
