Abstract
[18F]FDG–PET and β-amyloid-PET are established tools for assessing biomarker status in Alzheimer’s disease. In this study, we evaluated the potential of early-phase [18F]florbetaben (FBB) PET as a functional proxy for [18F]FDG–PET in preclinical Alzheimer’s disease models by examining regional perfusion and glucose metabolism in two transgenic mouse lines. Ninety-two APPPS1 (n = 17), APPSAA (n = 56), and age- and sex-matched wild-type mice (n = 19; 3–12 months, 40% female) underwent static [18F]FDG–PET (30–60 min p.i.) and dynamic [18F]FBB–PET (0–60 min p.i.). Standardized uptake values were derived for both [18F]FDG–PET and [18F]FBB–PET for the whole brain and 14 Ma–Benveniste–Mirrione atlas regions. We identified the 1–3 min p.i. time window as optimal, yielding the highest concordance with [18F]FDG (R = 0.53, p < 0.0001) across all regions. Both APPPS1 and APPSAA mice exhibited significant increases in perfusion (both p < 0.0001) and glucose metabolism (APPPS1: p = 0.0028; APPSAA: p < 0.0001) compared to wild-type controls. These findings demonstrate that early-phase [18F]FBB–PET not only mirrors [18F]FDG–PET-derived metabolic changes but also enables a single-scan assessment of β-amyloid pathology and brain function, thereby reducing the number of required scans and potentially the number of animals per study, and strengthening the translational value of preclinical PET research.
This is a visual representation of the abstract.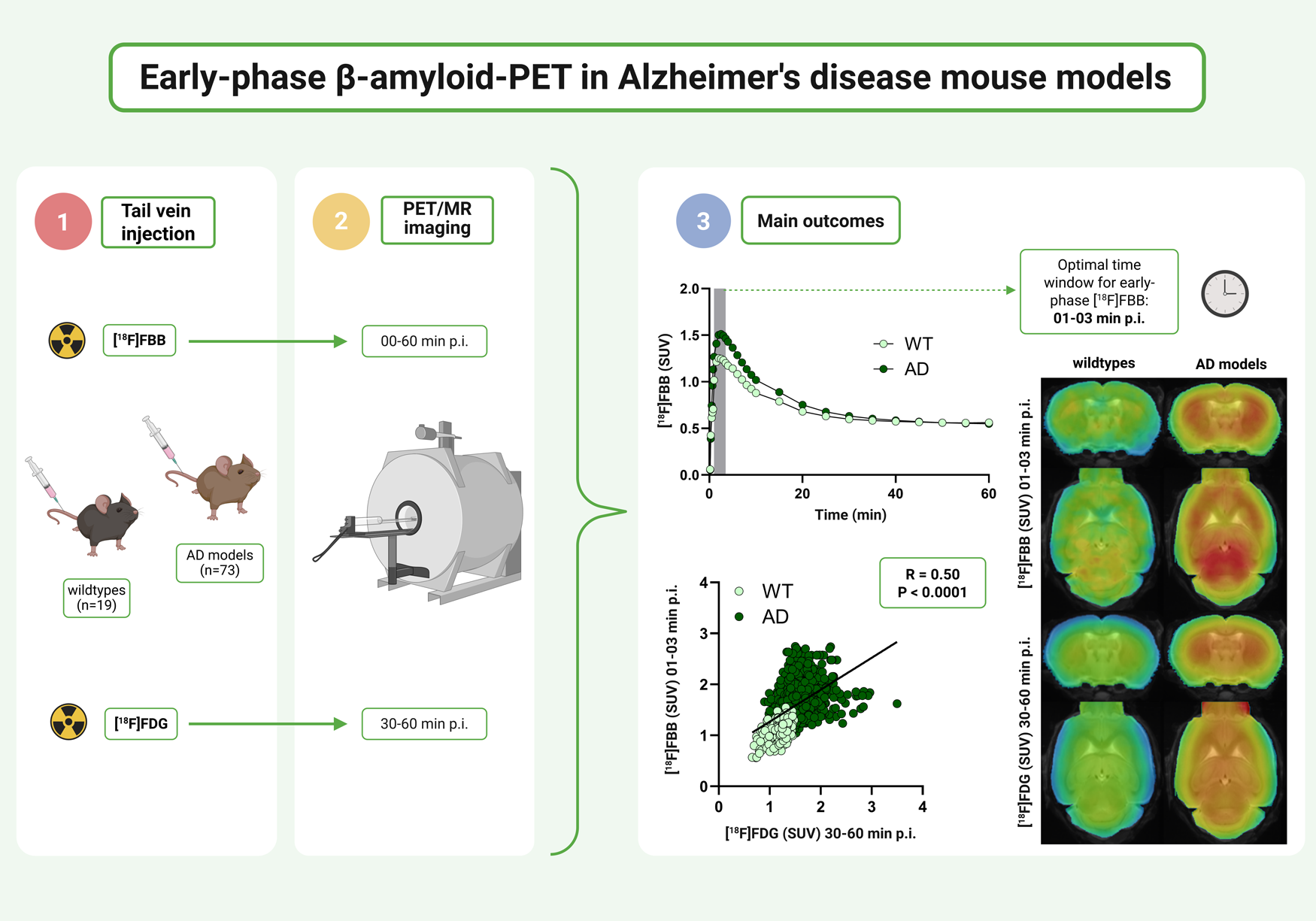
Introduction
Alzheimer’s disease (AD) remains a major medical and societal burden in the 21st century. Recent advances in biomarker research have transformed the diagnosis and monitoring of AD by introducing biologically grounded classification systems. The A/T/N framework categorizes AD pathology based on β-amyloid accumulation (A), tau pathology (T), and neurodegeneration (N). 1 Molecular imaging, particularly PET, plays a central role in detecting these biomarkers in vivo, even before clinical symptoms emerge. β-amyloid-PET and tau-PET identify pathological protein aggregates, while [18F]fluordeoxyglucose (FDG) PET provides a functional measure of glucose metabolism and neuronal integrity.2,3 Although these modalities offer complementary insights, they typically require separate imaging sessions, increasing patient burden and resource use. Recent human studies have demonstrated that early dynamic frames of β-amyloid-PET contain perfusion-related information that correlates well with metabolic patterns derived from [18F]FDG–PET. These early-phase images, acquired within minutes post-injection (p.i.), may thus serve as a proxy for cerebral metabolism and reduce the need for additional scans.4–6
These previous findings from human studies are promising, but they highlight a key back-translational challenge: determining whether early-phase β-amyloid-PET can reliably reflect cerebral metabolism in preclinical models of AD. Given the widespread use and established role of [18F]FDG and [18F]florbetaben (FBB) in preclinical PET imaging, assessing the potential of early-phase [18F]FBB to serve as a substitute for [18F]FDG is becoming increasingly relevant for diagnostic and therapeutic research. Moreover, there is an unmet need to enhance the efficiency of preclinical imaging protocols by reducing scan duration, minimizing animal stress, and adhering to the ethical principles of the 3Rs, replacement, reduction, and refinement. 7 Furthermore, while hypometabolism is a hallmark of neurodegeneration in patients, several AD mouse models exhibit cerebral hypermetabolism, likely driven by plaque-associated and activated microglia rather than neurons. 8
While early-phase β-amyloid-PET has shown promise as a proxy for glucose metabolism in humans,6,9,10 its validity in preclinical models remains uncertain, particularly in light of the divergent metabolic signatures observed in mice. Clarifying whether early-phase β-amyloid-PET signals can serve as a proxy for glucose metabolism is therefore essential for interpreting readouts in preclinical imaging of transgenic models of AD. Importantly, while perfusion and glucose metabolism represent distinct physiological processes, blood flow primarily reflecting substrate delivery and metabolism reflecting intracellular energy turnover, they are closely coupled through neurovascular and neurometabolic mechanisms.11–13 Accordingly, early-phase β-amyloid-PET does not measure metabolism per se but rather provides a perfusion-weighted signal that spatially and functionally correlates with metabolic activity. This study therefore investigates whether such coupling can be leveraged in preclinical models to approximate FDG-derived patterns.
To address this, we conducted a systematic comparison of early-phase [18F]FBB–PET and [18F]FDG–PET in two widely used AD mouse models. We aimed to evaluate the extent to which early-phase β-amyloid-PET captures functional brain changes associated with glucose metabolism. By doing so, we sought to assess its potential as a proxy imaging marker in preclinical research, with implications for improving efficiency, adhering to 3R ethical standards, and narrowing the translational gap between small-animal and clinical neuroimaging.
Materials and methods
Animals and study design
All animal experiments were approved by the local animal care committee of the Government of Upper Bavaria (Regierung Oberbayern; ROB-55.2-2532.Vet_02-22-125). They were overseen by a veterinarian and in compliance with the ARRIVE guidelines 2.0 and were carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments. Animals were housed in a temperature- and humidity-controlled environment with a 12 h light-dark cycle, with free access to food (Sniff Spezialdiäten GmbH) and water. Anesthesia prior to tracer application was induced with 3.0% isoflurane and maintained during PET scanning with isoflurane 1.5% delivered via a mask at 3.5 L/min. During all PET acquisitions, animals were positioned on a heated imaging bed to maintain body temperature, and individual physiological parameters (body temperature, heart rate, and respiration rate) were continuously monitored to ensure physiological comparability between groups. Following the experiment, the mice were transferred to fresh temporary cages equipped with food, water, and heating mats for warming. The animals were returned to their home cages only after they had fully regained consciousness. [18F]FBB (Neuraceq) precursor was provided by LIFE Molecular Imaging GmbH and the synthesis was performed as reported previously. 14 [18F]FDG was purchased commercially from Advanced Accelerator Applications (AAA).
PET/MRI imaging
All rodent PET procedures followed an established standardized protocol for radiochemistry, acquisition times, and post-processing which was transferred to a novel PET/MRI system.15,16 All mice were imaged with a 3 T Mediso nanoScan PET/MR scanner (Mediso Ltd., Hungary) using a triple-mouse imaging chamber. 17 Prior to [18F]FDG imaging, animals were fasted for at least 4 h to minimize baseline glucose levels. Two 2-min anatomical T1 MR scans (sagittal and axial) were performed before tracer injection and PET scan (head receive coil, matrix size 96 × 96 × 22 mm3, voxel size 0.24 × 0.24 × 0.80 mm3, repetition time 677 ms, echo time 28.56 ms, flip angle 90°).
Injected dose was 12.9 ± 2.2 MBq for [18F]FBB and 16.8 ± 3.1 MBq for [18F]FDG delivered in 200 μL saline via intravenous injection. [18F]FDG–PET data were acquired in a static 30–60 min p.i. window, whereas [18F]FBB–PET was recorded dynamically over a 0–60 min period. List-mode data within a 400–600 keV energy window were reconstructed using a 3D iterative algorithm (Tera-Tomo 3D, Mediso Ltd., Hungary) with the following parameters: matrix size 55 × 62 × 187 mm3, voxel size 0.3 × 0.3 × 0.3 mm3, eight iterations, six subsets. Decay, random, and attenuation corrections were applied. The T1 image was used to create a body-air material map for the attenuation correction.
Image preprocessing and analysis
All images were performed using PMOD (v3.5; PMOD Technologies, Basel, Switzerland). Unified spatial normalization (nonlinear warping, transient 0.6 mm3 Gaussian smoothing of the input image, 16 iterations, frequency cutoff 3, no thresholding) of all original Aβ- and [18F]FDG–PET images to a previously established template was performed.17,18 Reconstructed PET data were aligned to individual MR images and analyzed using the whole-brain and predefined regions of interest based on the Mouse Brain VOI Atlas (Ma–Benveniste–Mirrione). 19 For preliminary analysis of [18F]FBB images, data from the first 1–3 min p.i. were aggregated to generate early-phase images. These were then used for correlation with [18F]FDG-derived metabolic maps obtained from the static acquisitions. Tracer uptake was quantified using standardized uptake values (SUV) for both [18F]FDG and [18F]FBB for each region.
Statistical procedures
In order to evaluate the relationship between early phase [18F]FBB and [18F]FDG readouts, Pearson correlation coefficients were computed for regional SUV across 14 brain regions. To assess whether the relationship between early-phase [18F]FBB and [18F]FDG uptake followed a linear or non-linear trend, data were fitted using both linear and exponential models. Model performance was compared using the corrected Akaike Information Criterion (AICc), which accounts for model complexity and sample size to identify the best-fitting curve. The model with the lowest AICc value was considered to provide the best fit. Comparisons between the experimental groups APPPS1, APPSAA, and wild-type (WT) were conducted using one-way analysis of variance (ANOVA). Post-hoc tests were applied where appropriate to identify significant group differences. All analyses were performed in GraphPad Prism 10.1.2 and IBM SPSS Statistics 29, with significance defined as p < 0.05. Data are reported as means ± standard deviation, unless indicated otherwise.
Results
Demographics
Dynamic [18F]FBB–PET scans (0–60 min p.i.) and static [18F]FDG–PET scans (30–60 min p.i.) were successfully acquired in all animals (n = 92), including 17 APPPS1, 56 APPSAA, and 19 age- and sex-matched WT mice.20,21 A small subset of [18F]FBB–PET and [18F]FDG–PET scans acquired in APPSAA mice (n = 8) originated from a previous investigation conducted by our research group, while all remaining data were newly acquired and have not been published to date. 22 Differences in group sizes reflect colony availability. Animals ranged in age from 3 to 12 months, with 40.2% being female. Each mouse underwent both PET/MRI scans on separate days, with an interval of 9 ± 12 days between sessions. Detailed demographic data are displayed in Table 1.
Detailed demographic data.
Optimal time window for early-phase [18F]FBB imaging
Time-activity curves derived from dynamic [18F]FBB–PET data showed a rapid increase in signal within the first minutes p.i., followed by a distinct uptake peak within the first 5 min (Figure 1). WT animals reached their peak uptake at 2.1 ± 0.9 min p.i. with a SUV of 1.25 ± 0.28. APPSAA mice exhibited a peak at 2.2 ± 0.7 min p.i. with a SUV of 1.50 ± 0.31. APPPS1 mice showed a later peak at 3.6 ± 1.3 min p.i. with a SUV of 1.45 ± 0.16. After reaching peak values, the signal declined steadily over the remaining 60-min scan.
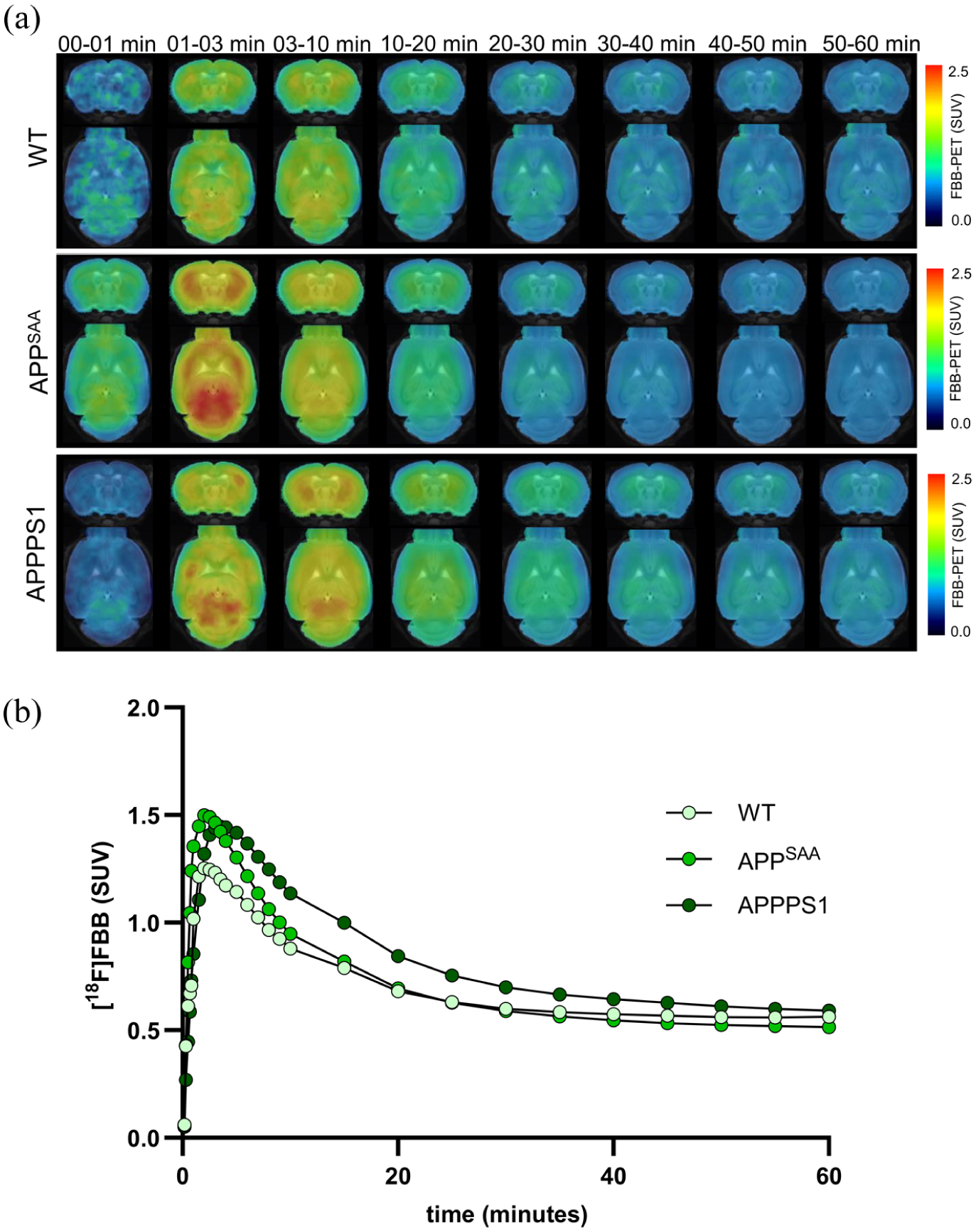
Dynamic β-amyloid-PET imaging: (a) representative averaged axial and coronal [18F]FBB–PET SUV maps of WTs, APPSAA, and APPPS1 during dynamic acquisition (0–60 min) projected upon a T1 MRI template and (b) averaged time-activity curves of whole-brain [18F]FBB–PET across the three groups during the dynamic acquisition.
Comparison of [18F]FDG–PET (30–60 min p.i.) with individual frames from dynamic [18F]FBB–PET (p.i.: 6 × 10, 6 × 30, 6 × 60, 10 × 300 s) demonstrated the highest agreement across the whole-brain for frames 7–10 corresponding to 1–3 min p.i., considering all three mouse models (Figure 2(a)). Similarly, the highest agreement in the cortex (frames 7–10), hippocampus (frames 7–10), and subcortical regions including the thalamus (frames 7–10) was observed consistently within the first 5 min p.i., with markedly weaker correlations at later time points. Together with the observed averaged perfusion peak at (t = 2.4 ± 1.0 min p.i.), these findings defined the optimal trade-off between correlation and count statistics within the 1–3 min p.i. window. Detailed statistics, including single frame correlation coefficients for all regions, are provided in Supplementary Table 1.
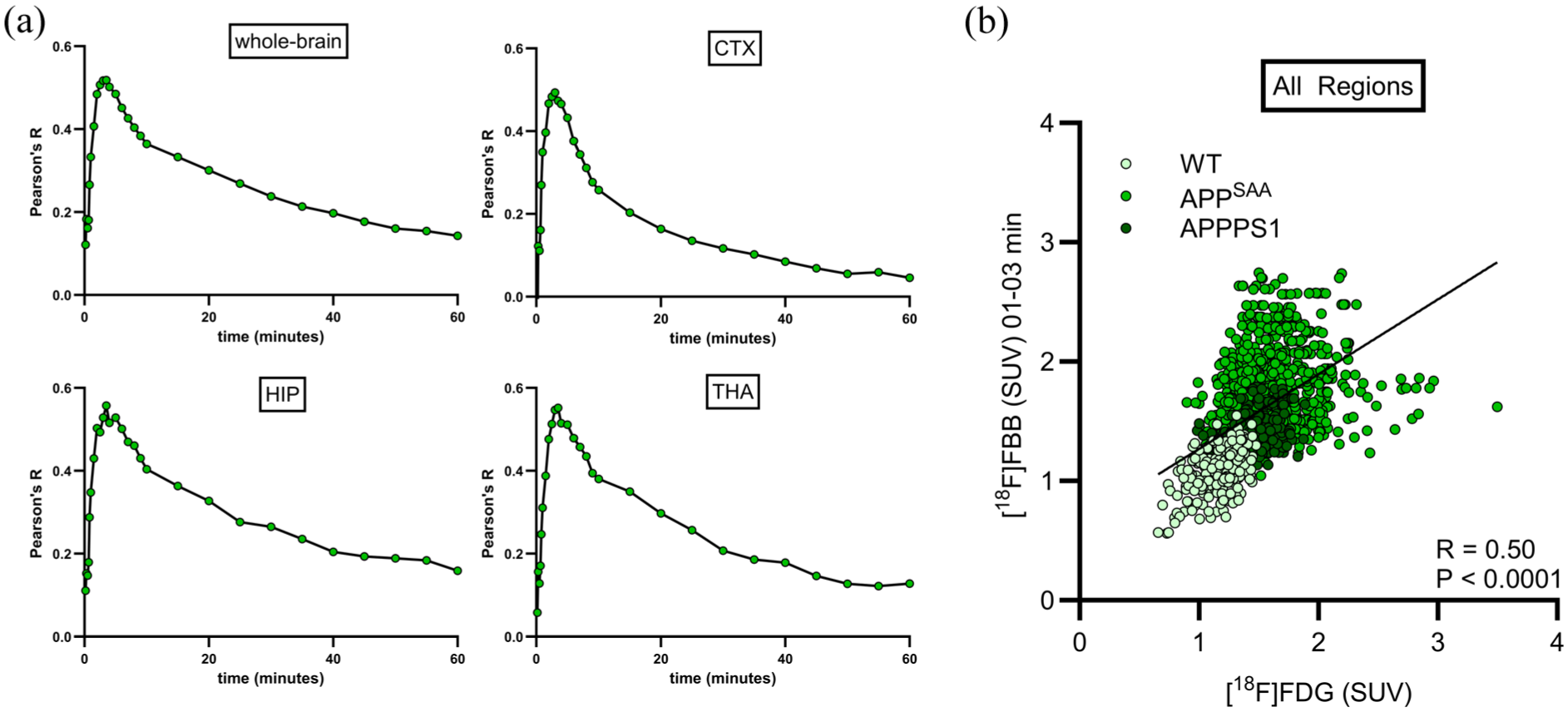
Agreement between early-phase β-amyloid–PET and FDG–PET: (a) Pearson correlation coefficients (R) between dynamic [18F]FBB–PET and static [18F]FDG–PET were calculated for all mice and plotted over time in four representative brain regions and (b) scatter plot depicts association between early-phase [18F]FBB SUV (1–3 min) and [18F]FDG SUV (30–60 min) in the 14 predefined brain regions. Correlation coefficients were computed using partial correlation, accounting for genotype as a cofactor to control for potential group-specific effects.
Relationship between early-phase [18F]FBB and [18F]FDG uptake
After identifying the optimal time window, correlations between early-phase [18F]FBB (1–3 min p.i.) and [18F]FDG scans were performed. Significant positive associations were observed across target brain regions, including the whole-brain (R = 0.53, p < 0.0001), cortex (R = 0.50, p < 0.0001), hippocampus (R = 0.49, p < 0.0001), and thalamus (R = 0.49, p < 0.0001; Supplementary Figure 1). Furthermore, the combined assessment of the predefined 14 target regions exhibited a strong correlation between early-phase [18F]FBB and [18F]FDG uptake (R = 0.50, p < 0.0001), consistently accounting for genotype as a covariate (Figure 2(b)). To evaluate whether this relationship deviated from linearity, we compared linear and non-linear (exponential) fits, which confirmed the linear model as the best fit (ΔAICc = 53.5, R 2 = 0.25 vs 0.22, F(1,1286) = 425.4, probability >99.9%).
Concordant perfusion and glucose metabolism in AD mouse models
Group comparisons revealed clear genotype-dependent alterations in both perfusion and metabolism. APPPS1 and APPSAA mice exhibited both visually and quantitatively significantly increased early-phase [18F]FBB uptake compared to WT controls across all regions examined, including the whole-brain (APPPS1: p < 0.0001; APPSAA: p < 0.0001), cortex (APPPS1: p = 0.0003; APPSAA: p < 0.0001), hippocampus (APPPS1: p < 0.0001; APPSAA: p < 0.0001), and thalamus (APPPS1: p = 0.0004; APPSAA: p < 0.0001; Figure 3(a) and (b) and Supplementary Figure 2). A similar pattern was observed in the [18F]FDG–PET-derived metabolism, with consistently elevated uptake in the whole-brain (APPPS1: p = 0.0028; APPSAA: p < 0.0001), cortex (APPPS1: p = 0.0027; APPSAA: p < 0.0001), hippocampus (APPPS1: p = 0.0049; APPSAA: p < 0.0001), and thalamus (APPPS1: p = 0.0012; APPSAA: p < 0.0001) in both transgenic mouse models (Figure 3(a) and (c) and Supplementary Figure 3).
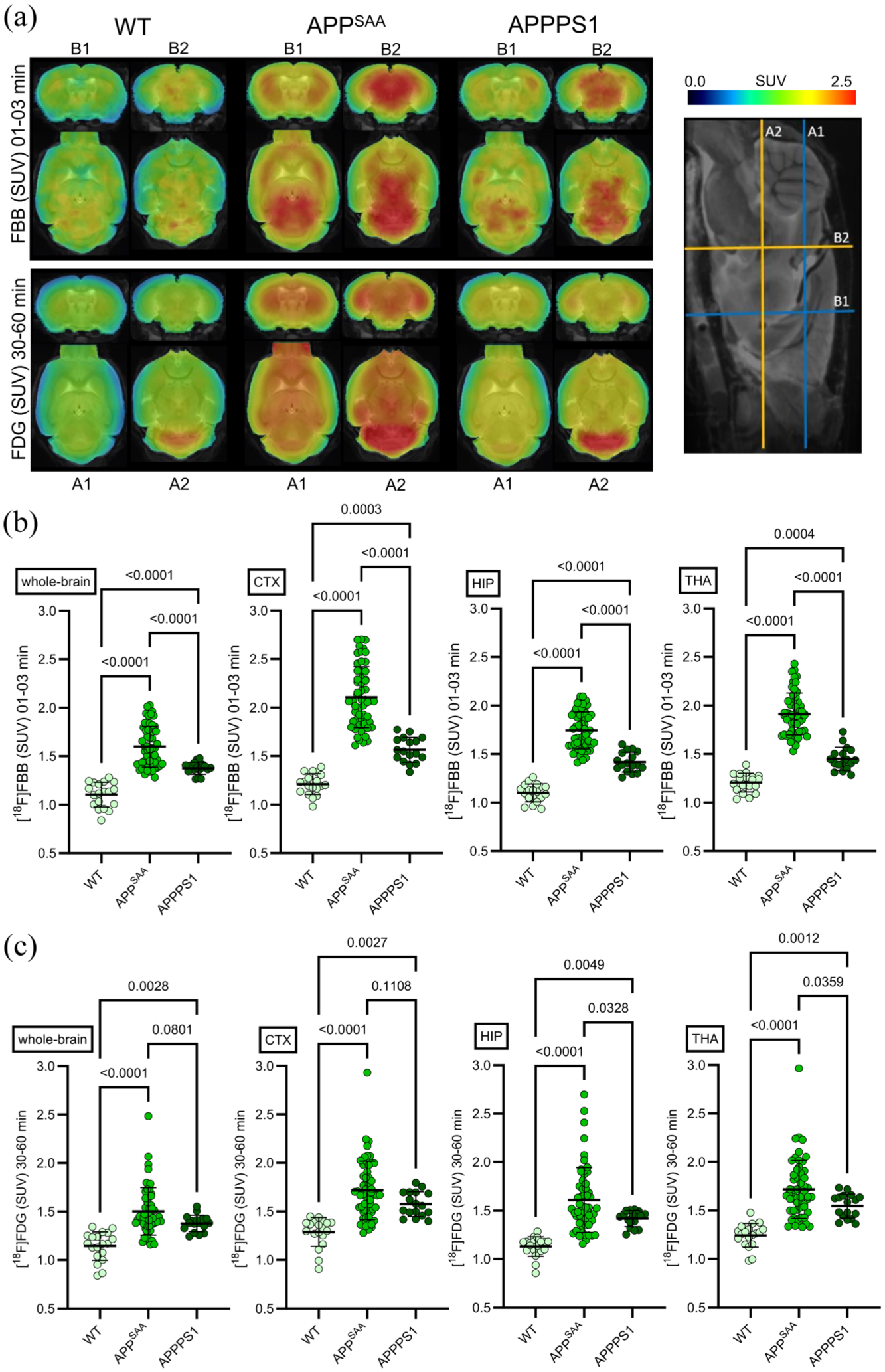
Comparison of cerebral perfusion and metabolism across AD mouse models: (a) averaged SUV-scaled axial and coronal maps show early-phase [18F]FBB and [18F]FDG uptake in both transgenic AD-models (APPSAA and APPPS1) and WT mice and (b, c) dot plots represent group-wise comparisons of early-phase [18F]FBB and [18F]FDG SUV in four representative target regions using repeated measures ANOVA, followed by post hoc pairwise comparisons with Tukey’s test for multiple comparisons.
Discussion
This study demonstrates that early, perfusion-weighted [18F]FBB–PET phases can serve as a proxy measure of cerebral metabolism in preclinical models of AD. By directly comparing dynamic early-phase [18F]FBB–PET and conventional [18F]FDG–PET in two widely used transgenic mouse models of amyloidosis (APPPS1 and APPSAA), 20 we successfully demonstrated a strong and regionally consistent correlation between early β-amyloid-PET tracer uptake and glucose metabolism. Notably, we identified the highest agreement between both modalities within the 1–3 min p.i. window, coinciding with peak perfusion-weighted values, and indicating that this time interval optimally captures perfusion-related signals that mirror metabolic brain changes. Finally, both transgenic models exhibited significantly increased early-phase [18F]FBB and [18F]FDG uptake relative to WT controls, reflecting a concordant pattern of elevated perfusion-weighted and metabolic signals.
The overarching aim of this preclinical study was to determine whether dynamic β-amyloid PET provides insights beyond plaque deposition and can serve as a proxy marker of brain metabolism through early-phase imaging. In human studies, the close relationship between metabolism and cerebral perfusion has been well established in regards to AD.23,24 Building on this, early-phase acquisitions of β-amyloid-PET have been proposed as functional biomarkers that reflect cerebral perfusion-weighted patterns.6,23 Consistent with these findings in humans, our results in transgenic AD mouse models demonstrate that early-phase β-amyloid-PET can similarly serve as a proxy marker for glucose metabolism, as measured by in vivo [18F]FDG–PET.
In particular, our findings demonstrate that both [18F]FDG–PET and early-phase [18F]FBB–PET revealed consistently elevated uptake across all examined brain regions in both APPPS1 and APPSAA mouse models compared to WT controls, which aligns with recent literature.20,25 Of note, among the two AD mouse models, APPSAA mice exhibited greater variability and higher peak perfusion-weighted values, reflecting the broader age range and higher average age in this group. In addition, the slightly longer and more variable time intervals between both scan timepoints within this cohort may have contributed to increased measurement variability.
Biologically, the Austrian (T714I) and Arctic (E693G) mutations in APPSAA mice promote aggregation-prone and proteolysis-resistant Aβ species, leading to earlier and more extensive β-amyloid deposition compared to the Swedish mutation (KM670/671NL) alone. APPSAA mice develop complex β-amyloid plaque morphologies with both diffuse and cored plaques, accompanied by dystrophic neurites, tau phosphorylation, lysosomal alterations, and robust neuroinflammatory activation, including increased microglial density and elevated TREM2 expression. 20 Moreover, the combination of Austrian and Arctic mutations alters the temporal trajectory of disease progression and the age of cognitive decline relative to other APP knock-in models, potentially contributing to broader phenotypic heterogeneity. 26
Similarly, a recent study using arterial spin labeling MRI reported increased cerebral blood flow in young and middle-aged APPPS1 mice across both cortical and subcortical brain regions. 27 This concordant pattern of increased perfusion and glucose metabolism reflects the well-established coupling between cerebral blood flow and metabolic demand, and suggests that early-phase β-amyloid-PET can reliably capture functional alterations associated with β-amyloid pathology in these models.
Currently, [18F]FDG–PET represents the gold standard in metabolic imaging, providing direct insight into glucose consumption and neuronal or astroglial activity.8,15,28,29 However, interpreting these results in preclinical models requires careful consideration of the underlying cellular mechanisms contributing to PET signals. Recent studies have demonstrated that [18F]FDG–PET signal is strongly influenced by microglial glucose consumption, with Trem2-deficient mice showing reduced [18F]FDG signal due to attenuated microglial activity.8,16 Conversely, aging WT mice and β-amyloid models with activated microglia exhibit hypermetabolism, indicating that [18F]FDG uptake in these animals may reflect neuroinflammation more than neuronal function.8,18,30 These findings are bolstered by data showing that microglial activation state influences network-level metabolic connectivity in mouse brains. 15
In addition to microglia, astrocytes substantially contribute to FDG uptake. Activation of astroglial glutamate transport has been shown to substantially affect [18F]FDG signal intensity, indicating that astrocytic glucose metabolism contributes to the coupling between synaptic transmission and cerebral glucose utilization. 28
Moreover, human studies have reported reductions in circulating lactate and pyruvate levels with preserved ketone-body concentrations in AD, consistent with impaired glycolytic and oxidative metabolism rather than adaptive substrate switching. 31 In this context, the elevated [18F]FDG uptake observed in AD mouse models may also indicate altered glycolytic activity associated with glial activation. These observations highlight that alterations in both energy substrate utilization and cellular metabolism should be considered when interpreting [18F]FDG–PET findings in preclinical models and patients.
However, not only astroglial activation but also neuronal hyperactivity during early stages of AD can contribute to [18F]FDG hypermetabolism, as demonstrated in human studies.32,33 Moreover, Aβ-dependent increases in functional connectivity have been shown to accelerate the spread of tau pathology from epicenters to connected brain regions via neuronal hyperactivity, ultimately resulting in transient increases of glucose metabolism during early AD. 34 This concept is further supported by our recent preclinical data of combined β-amyloid-PET and SV2A-PET, revealing localized increases in synaptic density surrounding Aβ plaques. 35
As dual-purpose β-amyloid-PET is increasingly leveraged for both pathological and functional imaging, defining an optimal early-phase acquisition window was a key objective of this study to ensure the translational value of early-phase PET metrics. In clinical settings, optimal early-phase windows for β-amyloid-PET tracers align with perfusion peaks: for [18F]FBB and [ 11 C]PiB, this typically occurs within the first 6–10 min.10,36–39 The substantially faster bolus transit and mean transit times observed in murine cerebral vasculature, relative to humans, mandate an earlier window to accurately capture peak tracer delivery in preclinical studies and account for species-specific hemodynamic differences.40–43 Given rodents’ significantly smaller brain volumes and faster circulatory dynamics, our 1–3-min window effectively mirrors the perfusion peak in mice, while maintaining translational consistency.
In addition to selecting an appropriate time window, the choice of quantification method is critical to ensure an accurate interpretation of PET signals in transgenic models. Our decision to use SUV instead of SUVR or kinetic modeling was guided by key practical and physiological considerations specific to small-animal PET. Arterial input function sampling is technically challenging in mice and ethically constrained. 44 Moreover, [18F]FDG–PET quantification via SUV has been shown to yield stable and physiologically meaningful results in both anesthetized and awake mice. 45 In contrast, SUVR requires the presence of a stable reference region, an assumption, that is, difficult to meet in transgenic AD models due to global metabolic alterations. Our approach is consistent with previous [18F]FDG–PET studies in APPPS1 and APPSAA mice, which also employed SUV-based quantification.20,46 In our cohort, we observed pronounced genotype-dependent increases in both perfusion and metabolism across all brain regions examined, reflecting a global effect. These alterations align with the underlying β-amyloid pathology characteristic of the two models used, APPPS1 and APPSAA. Both, while differing substantially in plaque kinetics and associated physiological responses, are well-established models of progressive amyloidosis, enhancing the generalizability of our findings.20,47 The widespread nature of these changes further challenges the validity of reference-based normalization approaches and reinforces the suitability of SUV as a robust and unbiased quantification method in this preclinical setting.
Nevertheless, some limitations of this study should be acknowledged. First, the cross-sectional study design may not fully capture the dynamic temporal changes in perfusion and metabolism that occur throughout disease progression. Longitudinal studies will be essential to validate the stability and predictive value of early-phase β-amyloid-PET as a metabolic proxy across different disease stages. Of note, our cohort did not include very old mice or models with advanced neurodegeneration. Therefore, according to the A/T/N framework, we cannot definitively classify the observed metabolic and perfusion-weighted changes as markers of neuronal injury (N). While we demonstrated a strong correlation between early-phase perfusion and glucose metabolism, both parameters were elevated in transgenic mice, likely reflecting a combination of inflammation and compensatory responses rather than neurodegeneration. Future studies should include mouse models exhibiting pronounced neuronal loss and synaptic degeneration, such as aged 5XFAD 48 or Niemann–Pick type C (NPC) knockout mice, 49 to further clarify the validity of early-phase amyloid-PET as a true marker of neurodegenerative processes. While our work focused on imaging-based biomarkers aligned with the A/T/N framework, 1 it is important to note that this classification does not include cognitive or behavioral dimensions. Cognitive decline and behavioral alterations are key clinical manifestations of AD, yet they are seldom systematically assessed in preclinical imaging studies. Indeed, behavioral phenotypes such as anxiety-like behavior and memory deficits can emerge early in certain AD models, as reported in 3xTg-AD mice. 50 The potential mismatch between biomarker-defined stages and cognitive outcomes highlights an important translational challenge. Future studies integrating longitudinal PET imaging with behavioral testing could therefore provide a more comprehensive view of disease progression and therapeutic efficacy in preclinical models.
Conclusion
Early-phase [18F]FBB β-amyloid-PET captures functional brain alterations with strong regional concordance to [18F]FDG–PET. Combining pathological and functional information in a single scan enhances the efficiency of preclinical imaging while reducing costs, scan time, and animal burden in line with 3R principles. This integrated approach is particularly valuable for longitudinal preclinical studies and enables a more precise phenotyping of disease models with fewer resources.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X261420082 – Supplemental material for Assessment of early-phase [18F]florbetaben images as a proxy for brain metabolism in mouse models of Alzheimer’s disease
Supplemental material, sj-docx-1-jcb-10.1177_0271678X261420082 for Assessment of early-phase [18F]florbetaben images as a proxy for brain metabolism in mouse models of Alzheimer’s disease by Robin Gröger, Amelie L Englert, Manvir Lalia, Lis de Weerd, Matteo Rovere, Lea Kunze, Giovanna Palumbo, Karin Wind-Mark, Sonja Fixemer, Sebastian N Roemer-Cassiano, Nicolai Franzmeier, Günter Höglinger, Andreas Zwergal, Sabina Tahirovic, Rudolf A Werner, Matthias Brendel and Johannes Gnörich in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
We thank Rosel Oos and Karin Bormann-Giglmaier for excellent technical support during small animal PET imaging. We thank Mathias Jucker for providing the APPPS1 mice.
Author contributions
Conceptualization: ST, MB, JG. Methodology: KW-M, RAW, MB, JG. Validation: NF, GH, AZ, ST, RAW, MB, JG. Formal analysis: RG, ALE, ML, LK. Investigation: RG, ALE, ML, LdW, MR, LK, GP, KW-M, SF. Resources: LdW, MR, SF, ST, RAW, MB, JG. Data curation: SNR-C, NF, MB. Writing—original draft: RG, ALE. Writing—review and editing: NF, AZ, ST, MB, JG. Visualization: RG, ALE. Supervision: ST, RAW, MB, JG. Project administration: JG.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: JG is funded by the Munich Clinician Scientist Program (MCSP). NF and MB were funded by the Deutsche Forschungsgemeinschaft (DFG) under Germany’s Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy, ID 390857198). MR thanks the Hector Fellow Academy for support.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MB is a member of the Neuroimaging Committee of the EANM. MB has received speaker honoraria from Roche, GE Healthcare, Iba, and Life Molecular Imaging; has advised Life Molecular Imaging and GE healthcare; and is currently on the advisory board of MIAC. AZ has received speaker honoraria from Dr. Willmar Schwabe GmbH, AstraZeneca and Pfizer. He got research support from Dr. Willmar Schwabe GmbH; and is currently on the advisory board of Vertigenius.
Ethical considerations
All animal experiments were approved by the local animal care committee of the Government of Upper Bavaria (Regierung Oberbayern; ROB-55.2-2532.Vet_02-22-125).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
