Abstract
Cerebral small vessel disease (cSVD) impairs the physiological mechanisms that continuously match cerebral haemodynamics to metabolic need. We have monitored this impairment non-invasively employing in-house developed broadband near-infrared spectroscopy (bNIRS) and an animal model of cSVD, the spontaneously hypertensive stroke-prone rat (SHRSP). We also assessed the vasodilating agent, nimodipine, as a potential protective treatment. Male SHRSPs were randomly allocated at 3 months of age to receive either a placebo or nimodipine diet. Both before and after 3 months on diet, the changes in the concentration of oxyhaemoglobin (HbO2), deoxyhaemoglobin (HHb) and oxidised cytochrome-c-oxidase (oxCCO) in the somatosensory cortex was obtained from the bNIRS recordings, and changes in blood oxygenation (HbDiff = HbO2 − HHb) and blood volume (HbT = HbO2 + HHb) were calculated. Metabolic-haemodynamic coupling was assessed by analysing the intrinsic slow wave oscillations of metabolic (oxCCO) and haemodynamic signals (HbO2, HHb, HbT and HbDiff) at 0.02–0.06 Hz, using wavelet coherence and semblance. Coherence and semblance were significantly (p < 0.001) reduced in aged SHRSPs, indicating impaired metabolic-haemodynamic coupling, but these measures were significantly (p < 0.001) protected by treatment with nimodipine. We reveal dysregulation of cerebral metabolic-haemodynamic coupling in SHRSP, and, importantly, demonstrate the protective effect of nimodipine, a drug suitable for clinical use in cSVD.
Keywords
Introduction
Cerebral small vessel disease (cSVD) describes pathologies affecting the small vessels of the brain, including arteriolar stiffness, 1 deposition in the vessel wall, 2 capillary constriction and dysfunction 3 and vascular rarefaction. 4 cSVD is responsible for increased risk for stroke and dementia 5 largely due to impaired physiological mechanisms of the cerebral vasculature that constantly adjust blood flow to meet the changing metabolic demand from neurons. 6 MRI studies have reported reduced haemodynamic responses upon visual7,8 or somatosensory stimulations9,10 in patients with cSVD and in model animals. However, the dysregulation between direct neuronal metabolism and haemodynamic response, especially in the resting state, is difficult to monitor with commonly used functional MRI techniques, but broadband near infrared spectroscopy (bNIRS) provides a solution.11–14 Specifically, the wavelength spectrum covered by bNIRS not only differentiates oxyhaemoglobin from deoxyhaemoglobin, but also distinguishes the oxidised cytochrome c oxidase (oxCCO) from its reduced state based on their extinction coefficients.12,13,15 Cytochrome c oxidase is a key component (complex IV) in the mitochondrial electron transport chain involved in aerobic oxygen metabolism, and cerebral metabolism can thus be indicated by the change in concentration of oxCCO. 11 During neuronal activation, the oxCCO signal increases, together with HbO2, indicating a vascular response to increase oxygen delivery to meet the new metabolic demand, while the HHb signal decreases accordingly. 13 Further, in the resting state, the cerebral metabolic and haemodynamic signals undergo spontaneous oscillations, termed as slow wave oscillations, with those ranging between 0.02 and 0.06 Hz considered as of neurogenic origin 16 triggered by spontaneous electrophysiological activity. 17 These intrinsic oscillations of HbO2, HHb and oxCCO and the high temporal resolution of bNIRS enable the detection of uncoupling between cerebral metabolism and haemodynamics in the resting state. Indeed, by analysing intrinsic slow wave oscillations, previous studies have confirmed an uncoupling of cerebral metabolism with haemodynamic response in patients with subarachnoid haemorrhage, 18 and with blood pressure in newborns with hypoxic ischaemic encephalopathy. 14
Here, we used an established animal model of cSVD, the spontaneously hypertensive and stroke prone rat (SHRSP), which mimics the epidemiological, pathological and pathophysiological features of cSVD.19,20 We aim to explore if metabolic-haemodynamic uncoupling can be detected from slow wave oscillations measured with bNIRS in SHRSP. Further, we have established that chronic treatment with nimodipine, a potent vasodilator of CNS vessels, restores vascular function in SHRSP 10 and we also aim to study if the protective effect of nimodipine manifests as improved metabolic-haemodynamic coupling in the resting state.
Materials and methods
Male SHRSPs were bred in-house and maintained in a 12-h light/dark cycle, with food and water ad libitum. The animals were randomly allocated at 3 months of age to receive either a control diet (placebo group), or a diet containing 200 mg/kg nimodipine (nimodipine group; equivalent to a daily intake of 12 mg nimodipine per kilogram body weight; Ssniff Spezialdiäten GmbH, Germany), both of which continued for 3 months. Measurements of metabolic-haemodynamic coupling were made using the bNIRS system (see below) when the animals were 3 months of age (i.e. before diet), and at 6 months of age (after 3 months on diet). The potential metabolic-haemodynamic uncoupling caused by cSVD pathology was indicated by comparing SHRSPs at 3 months of age (i.e. before the development of cerebral pathologies 19 ) with animals 6 months of age. The potential therapeutic effect of nimodipine was indicated by comparing SHRSPs at 6 months old after being fed either a placebo or nimodipine diet. All experiments were performed in accordance with the UK Home Office Animals (Scientific Procedures) Act (1986), and were approved by the Ethics Committee of University College London and the UK Home Office, following the ARRIVE 2 guidelines (Animal Research: Reporting of In Vivo Experiments).
To measure metabolic and haemodynamic signals, animals were anaesthetised with 2% isoflurane in room air, maintaining body temperature at 37°C using a homeothermic heating pad (Harvard Apparatus, Cambridge, UK), and monitoring pulse oximetry, heart rate and breathing rate with a pulse oximeter (MouseOx; STARR Life Sciences, Oakmont, USA) sensor positioned on the foot. The head was stabilised using a customised stereotaxic frame, the skin overlying the somatosensory cortices 4 mm lateral and 0.2 mm rostral to bregma on both sides 21 was shaved. The source and detector fibres of the bNIRS system were placed in close contact with the skin over the right and left somatosensory cortical regions, respectively, to account for the small size of rodent head and to obtain information from both hemispheres. A spectrometer (Ventana VIS-NIR) detected alterations in the spectrum of light across 780–900 nm emitted from a light source (HL-2000-HP) and transmitted through the somatosensory cortex between the source and the detector fibres, and relative changes in the concentration of oxyhaemoglobin (HbO2), deoxyhaemoglobin (HHb) and oxidised cytochrome-c oxidase (oxCCO) were calculated, as described previously. 15
The signals of the aforementioned parameters were recorded at 0.5 Hz across a 30-min period and were processed with an automated wavelet de-noising function in MATLAB (MathWorks, MA, USA), before calculation of HbDiff (i.e. the change in oxygenation; [HbO2] − [HHb]) and HbT (i.e. the total blood volume; [HbO2] + [HHb]) to represent overall haemodynamic response. Thus, four haemodynamic parameters, namely HbO2, HHb, HbDiff and HbT, were used to analyse their coupling with the metabolic signal (i.e. oxCCO). The metabolic-haemodynamic coupling was determined by wavelet coherence and semblance, based on a continuous wavelet transform with the complex Morlet wavelet. 22 Wavelet coherence represents the similarity in spectral power between the metabolic and haemodynamic signals, and varies between 0 and 1, with 1 indicating a complete dynamic synchronisation. Wavelet semblance 23 indicates the instantaneous phase difference between the two signals, and ranges between −1 to +1, with 0 suggesting a complete absence of relationship and +1 and −1 suggesting high alignment in an in-phase and an anti-phase pattern, respectively. The average values of wavelet coherence and semblance at a frequency band between 0.02 and 0.06 Hz were calculated and compared between the three groups, namely the 3-month-old animals, the placebo group and the nimodipine group. Statistical significance was determined by individual t-test using MATLAB (MathWorks, MA, USA), and results indicated as mean ± SD.
Results
As illustrated by representative images in Figure 1(a), 3-month-old animals showed high coherence and semblance between oxCCO and HbO2, especially in the frequency band of interest (0.02–0.06 Hz), distinguished by the rectangle and also illustrated in Figure 1(b), while placebo-treated animals demonstrated significantly lower values in coherence (Figure 1(c)) and semblance (Figure 1(d)) between oxCCO and the four haemodynamic parameters (HbO2, HHb, HbDiff and HbT) compared with 3-month-old animals. Further, the animals treated with nimodipine showed significantly preserved coherence (Figure 1(c)) and semblance (Figure 1(d)) between the metabolic and haemodynamic signals compared with placebo animals of the same age. Values of metabolic-haemodynamic coupling of the three groups and statistical significance are listed in Table 1.
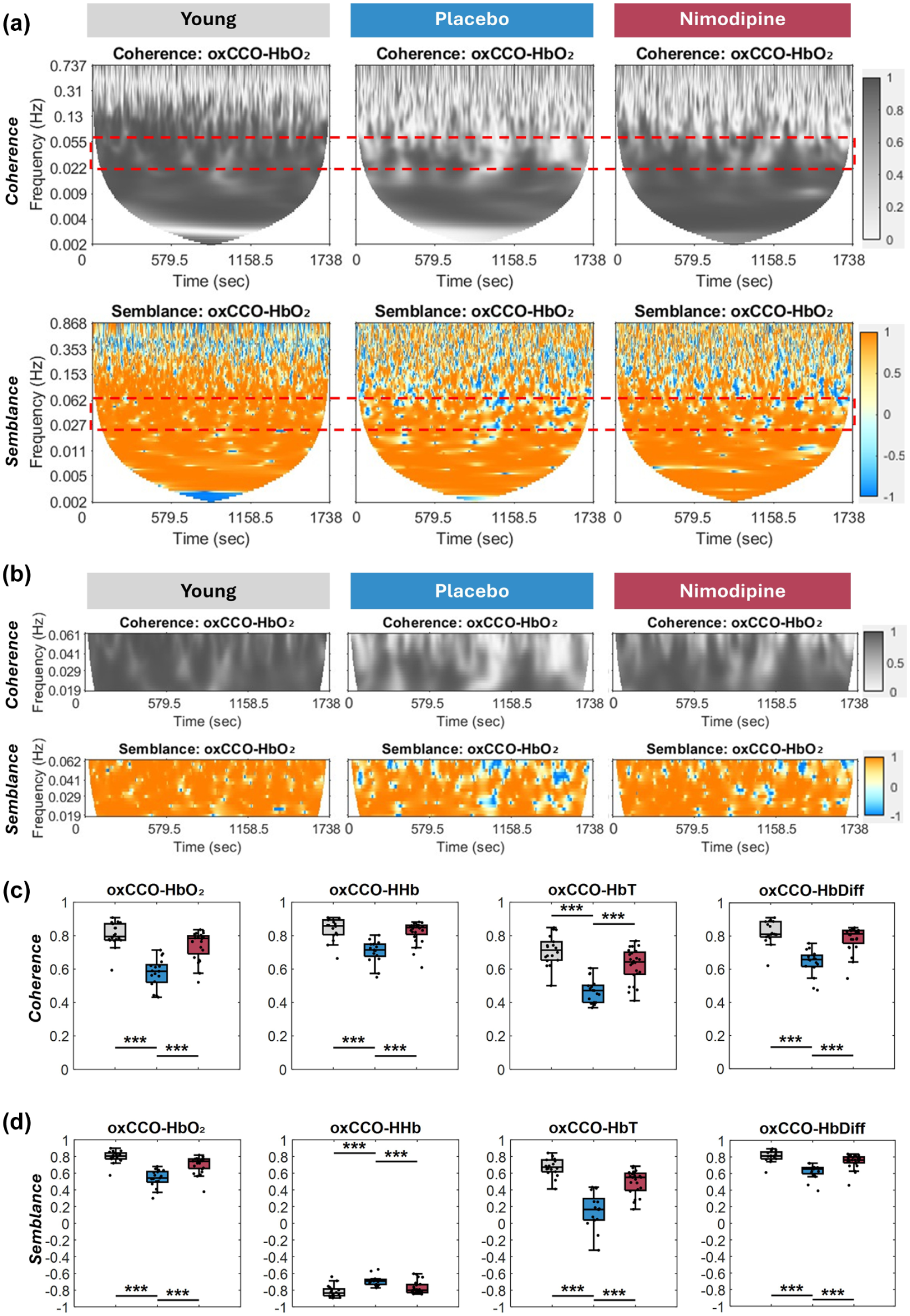
Metabolic-haemodynamic coupling is decreased in SHRSP, but protected by treatment with nimodipine. (a) Graphs illustrate coherence and semblance derived across the entire frequency range from the recorded signals in a representative animal from each of the groups (young, placebo- and nimodipine-treated), with the frequency band of interest (0.02–0.06 Hz) distinguished by the rectangle, and also separately illustrated in (b). Statistical analysis between groups showed that SHRSPs at 6 months of age (blue) showed significantly lower coherence (c) and semblance (d) between oxCCO and haemodynamic signals (HbO2, HHb, HbT and HbDiff) compared with young animals 3 months of age (grey). However, animals treated with nimodipine (red) showed significantly improved coherence (c) and semblance (d) in all four parameters compared with the placebo group.
Wavelet coherence and semblance between metabolic and haemodynamic signals in three groups of SHRSPs.
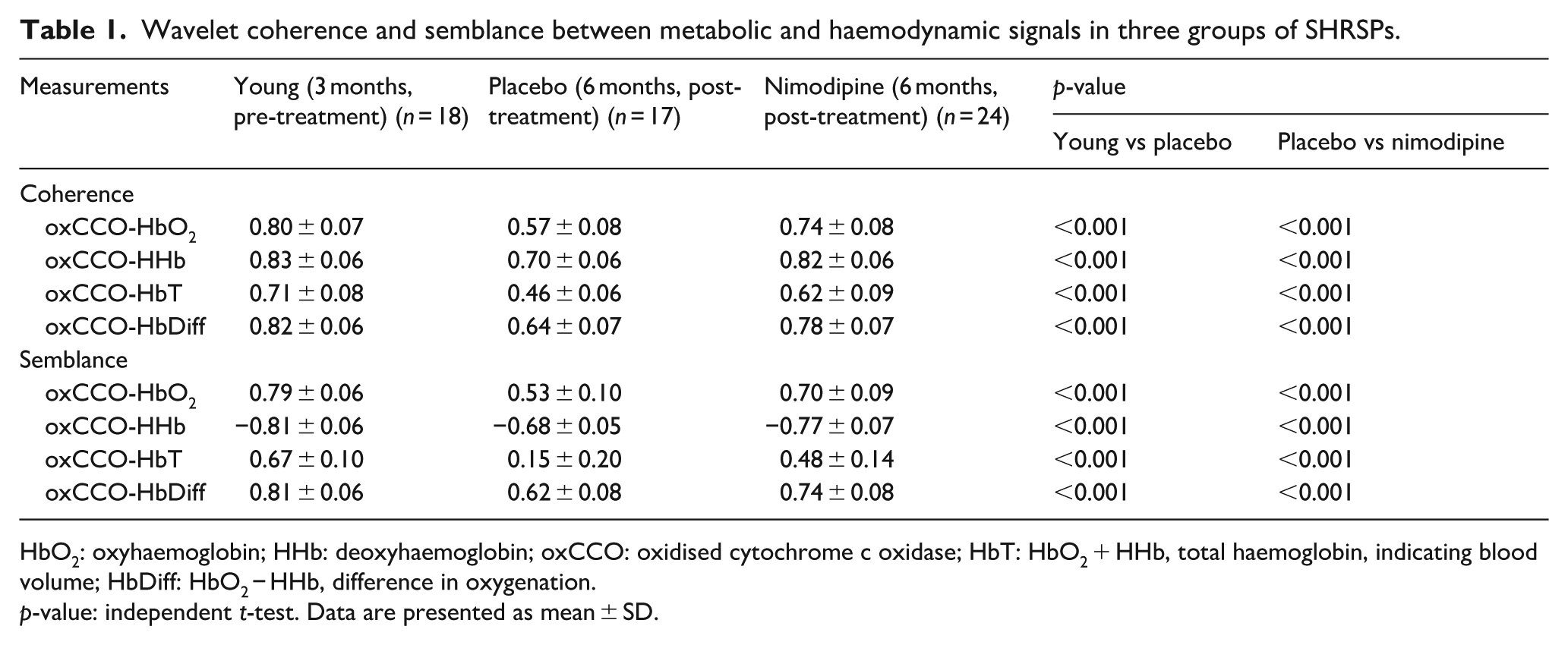
HbO2: oxyhaemoglobin; HHb: deoxyhaemoglobin; oxCCO: oxidised cytochrome c oxidase; HbT: HbO2 + HHb, total haemoglobin, indicating blood volume; HbDiff: HbO2 − HHb, difference in oxygenation.
p-value: independent t-test. Data are presented as mean ± SD.
Discussion
We demonstrate that metabolic-haemodynamic coupling is impaired in the SHRSP model of human cSVD, as measured by wavelet coherence and semblance detected by bNIRS. Further, we show that chronic treatment with nimodipine, an L-type calcium channel blocking agent with potent cerebrovascular activity,24,25 protects from the uncoupling between metabolism and haemodynamics that otherwise occurs with age.
The metabolic-haemodynamic uncoupling shown in SHRSP indicates an impairment in the ability of the diseased vasculature to match the delivery of oxygenated blood to meet the changing metabolic demand from neuronal activity, which can be detected even in the resting state. This impairment substantiates the lower haemodynamic response upon neurological stimulation (termed as neurovascular coupling, NVC) that has been extensively reported in patients with cSVD7,8,26–30 and in animal models.9,10,31,32 It is reasonable to speculate that this persistent and severe imbalance between oxygen demand and supply may initiate downstream pathological events in cSVD, including hypoxia33–35 and mitochondrial failure, 36 which eventually lead to lesion formation 35 and cognitive decline. 37 Our study demonstrates the metabolic-haemodynamic uncoupling in prolonged disease by comparing aged SHRSP with young animals, and future studies examining normotensive animals may provide additional valuable insights.
Importantly, our findings show that the impaired metabolic-haemodynamic coupling in SHRSP can be at least partly prevented by chronic treatment with nimodipine. This protection by nimodipine observed in the resting state is an advance from our previous study which reported restored NVC in the treated animals, 10 and from studies in other cerebral diseases.38,39 Observations made in the resting state address a limitation of NVC in which changes in haemodynamic response are recorded upon artificial external stimulation to activate neurons. In the case of NVC, it is not clear whether a higher haemodynamic signal is due to stronger neuronal stimulation (i.e. more surviving neurons), or a better vascular response. However, the fact that the effect of nimodipine can be observed in the resting state indicates that the protection is mainly in the vasculature, because if the vasculature remained diseased it would still show uncoupled haemodynamic signals even if tissue metabolism was enhanced by greater neuronal survival.
The observed protection of metabolic-haemodynamic coupling by nimodipine is in line with reported evidence of long-term treatment of nimodipine in preserving cognitive function (which is closely related with cerebral metabolism 40 ) in patients with cerebrovascular disease 41 and in the same model of cSVD. 10 The mechanism underlying the protection remains unclear, not least because nimodipine has several potentially beneficial actions. The vasodilating action of the drug will reduce blood pressure (not measured in this study), and clinical studies have shown that controlling blood pressure improves vascular reactivity in patients with hypertension. 42 However, nimodipine crosses the blood-brain barrier and it has recognised direct neuroprotective actions43,44 which may also be involved. Not least, nimodipine can act directly on the neurovascular unit 45 and studies have reported a therapeutic effect of nimodipine in relaxing pericytes 46 and preventing calcium influx into glial cells and neurons.47,48 Thus, although the exact mechanism(s) responsible for the protective effect of nimodipine in our study remains to be determined, the results indicate a novel therapeutic strategy for neuroprotection in cSVD, achieved in association with the preservation of mechanisms that dynamically adjust the blood flow to meet the metabolic need.
The application of bNIRS in measuring metabolic-haemodynamic coupling in the resting state, as illustrated in our study, overcomes limitations faced by common methods using functional MRI, namely the complexity of the procedure and variation caused by stimulation. 49 Indeed, bNIRS systems boast convenience due to their portability and low-cost, and proven ability to be useful in situations difficult to cover by MRI. 14 Further, signals obtained from functional MRI indicate changes in haemodynamics, rather than actual neuronal activity or subsequent metabolism. 50 On the other hand, bNIRS directly measures the change in concentration of oxCCO, and thus the coupling between metabolism and haemodynamics can directly be performed with bNIRS. However, future studies are required to validate the clinical importance of metabolic-haemodynamic coupling measured from slow wave oscillations using bNIRS, and it is likely that oscillations at other frequencies, or signals obtained from other brain regions, might be a better biomarker in other neurological diseases. 16
In conclusion, we reveal that imbalance between oxygen supply and demand in SHRSP can be indicated by measurement of metabolic-haemodynamic coupling using slow wave oscillations of bNIRS signals, preparing this protocol for future clinical use. Further, we show that nimodipine protects metabolic-haemodynamic coupling, suggesting a therapeutic value in cSVD.
Footnotes
Acknowledgements
None.
Author contributions
Conceptualisation, Z.Y., I.T. and K.J.S.; methodology, Z.Y., F.L., I.T. and K.J.S.; data curation, Z.Y. and Y.X.; writing – original draft preparation, Z.Y. and K.J.S.; writing – review and editing, all authors; visualisation, Z.Y. and K.J.S.; supervision, I.T. and K.J.S.; funding acquisition, I.T. and K.J.S. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Prof. Tachtsidis is the CEO and founder of Metabolight Ltd. The work presented in this publication was conducted entirely as part of his academic role at UCL.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by funding to KS from the Multiple Sclerosis Society (United Kingdom), the International Progressive Multiple Sclerosis Alliance, and the Fondation Leducq. IT acknowledges funding from Innovate UK HYPERPROBE 10048387; fastMOT 10063660; and EPSRC EP/W035154/1.
