Abstract
The thalamus has been shown to undergo secondary degeneration after cerebrocortical ischemia. However, little is known about the time course of the retrograde thalamic degeneration. The present study was designed to investigate time-dependent changes in the morphology, protein synthesis and calcium metabolism of thalamic neurons in middle cerebral artery (MCA)-occluded spontaneously hypertensive stroke-prone rats that showed primary focal ischemia in the temporoparietal cortex after permanent occlusion of the left distal MCA. In the histologic study by light and electron microscopy, swelling of the nucleus and shrinkage of the perikarya were seen in some neurons of the ventroposterior (VP) thalamic nucleus on the lesioned side at 5 days after ischemia. At the same time, the incorporation of radiolabeled leucine in VP thalamic neurons began to decrease significantly with concomitant a decrease in the number of polyribosomes in the neurons. Conspicuous 45Ca accumulation was noted at 3 days after ischemia and persisted up to 1 month in the VP thalamic nucleus on the lesioned side. These findings suggest that the secondary thalamic degeneration after cortical infarction starts with disruption of calcium homeostasis in situ at the third day after MCA occlusion, followed by a decrease in polyribosomes but not by disaggregation of polyribosomes as seen in hippocampal CA1 neurons subjected to transient forebrain ischemia.
Keywords
Focal cerebral ischemia is known to cause irreversible neuronal degeneration in brain areas distant from the primary ischemic lesion without any reduction of regional CBF (rCBF) in the areas (Barron et al., 1973; Kataoka et al., 1989; Fujie et al., 1990; Hanyu et al., 1991; Tokuno et al., 1992). For example, permanent occlusion of the unilateral middle cerebral artery (MCA) in rats yields not only infarction in the ipsilateral cerebral cortex and caudoputamen but also secondary degeneration in the ipsilateral thalamus and substantia nigra (Tamura et al., 1981b). Because the cerebral cortex and caudoputamen receive extensive input from the thalamus and substantia nigra, respectively (Iizuka et al., 1989), the secondary neuronal damage as seen in MCA-occluded rats may involve retrograde degenerative processes (Iizuka et al., 1990; O'Gorman and Sidman, 1985a; O'Gorman, 1985b). Thalamic atrophy has also been observed clinically in patients with cerebral infarction in the territory supplied by the MCA, and this atrophy may lead to sustained dementia (Tamura et al., 1991). Thus, protection against ischemia-induced thalamic degeneration, if successfully conducted, would be of clinical value.
The morphologic features of neuronal degeneration in brain areas that are remote from but connected synaptically with the primary infarction site have been studied mainly by conventional light and electron microscopy, and it is indicated that the retrograde degeneration in the areas distant from the primary ischemic lesion is slowly progressive. However, little is known about the time course of the retrograde neuronal degeneration associated with cerebral infarction or about metabolic changes in the sites undergoing retrograde degeneration.
Spontaneously hypertensive stroke-prone (SH-SP) rats with permanent occlusion of the MCA above the rhinal fissure and distal to the striate branches invariably exhibit a certain cortical infarct and subsequent thalamic degeneration (Coyle et al., 1983; Okuyama et al., 1991; Kumon et al., 1996), and they thus appear to be an appropriate subject for the study of secondary neuronal damage in the thalamus. The present study was designed (1) to investigate the time course of thalamic degeneration in SH-SP rats with MCA occlusion by light and electron microscopy, (2) to examine the ischemia-induced changes in the protein synthesis within the thalamus by [14C]-leucine autoradiography, and (3) to detect the calcium accumulation in the thalamus of MCA-occluded rats by 45Ca autoradiography. The results of these experiments should shed light on the starting time of the treatment of secondary thalamic degeneration.
MATERIALS AND METHODS
Animal model
Male SH-SP rats weighing 280 to 320 g were subjected to focal cerebral ischemia with modifications of the method of Brint (1988). Tail systolic pressure in each conscious animal was measured just before MCA occlusion with a rat tail manometer-tachometer system (KN-21.0; Natsume, Tokyo, Japan). During the MCA occlusion procedure, animals were anesthetized with 1.5% halothane in a 2:1 mixture of nitrous oxide and oxygen. The rectal temperature was monitored by a digital thermometer and maintained at 37.0±0.5°C by a heating lamp. A small scalp incision was made at the midpoint between the left outer canthus and the pinna. The temporal muscle was separated and retracted to expose the zygoma and the squamosal bone. With a dental drill, a burr hole was made 1 mm rostral to the anterior junction of the zygoma and the squamosal bone, and the dura was carefully incised. The left MCA was exposed and coagulated at the point distal to the perforating arteries with a bipolar coagulator, and cut to ensure permanent occlusion.
Light microscopy
Rats were anesthetized with pentobarbital and perfused transcardially with 10% formalin in 0.1 mol/L phosphate-buffered saline (PBS) (pH 7.4) at 12 hours; 1, 3, and 5 days; 1 and 2 weeks; and 1, 2, and 3 months after MCA occlusion (four animals each). The brain was removed and embedded in paraffin, and serial coronal sections 2-μm thick were cut. The specimens were stained with hematoxylin and eosin or with 0.1% cresyl violet.
The area of the thalamus in sections at the level 3.6 mm posterior to bregma was measured by a planimeter, and the ratio of the thalamic area on the MCA-occluded side to that on the contralateral side (left/right) was calculated. The number of cells in the ventroposterior (VP) thalamic nucleus was evaluated by measuring the size of each cell with a computerized image processing system (Nexus 6400 System; Kashiwagi, Tokyo, Japan). A square of 0.33 times 0.3 mm (510 times 478 pixels) was designated as a region of interest on the video image. The size of each cell within the square was measured and expressed by its pixel size. Cell number and size were obtained from five regions of interest. Cells larger than 250 pixels (20 μm in diameter) were tentatively regarded as the neuron-rich group, and those smaller than 150 pixels (12 μm in diameter), except for endothelial cells and pyknotic neurons, were regarded as the glia-rich group. Although the cells larger than 250 pixels were considered to be mostly neurons, one could not exclude the possibility that the smaller cells included a few neurons. Therefore, we defined the cells larger than 250 pixels as the neuron-rich group. Although it was not always easy to distinguish between glial cells and degenerating pyknotic neurons, the majority of the pyknotic neurons were as large as 150 to 250 pixels (12 to 20 μm in diameter) and thus could be distinguished from morphologically intact neurons. Moreover, glial cells markedly outnumbered endothelial cells and pyknotic neurons in the rat thalamus. Thus, although pyknotic neurons and endothelial cells were included in the glial-rich group, the overestimation of the glial cell number appeared to be trivial.
Electron microscopy
Rats were anesthetized with pentobarbital and perfused transcardially with 2.5% glutaraldehyde-2% paraformaldehyde in 0.1 mol/L PBS (pH 7.4) at 12 hours; 1, 3, and 5 days; 1 week; and 1 month after MCA occlusion (four animals each). After removal of the brain, a tissue block containing the VP thalamic nucleus was dissected, postfixed with 1% osmium tetroxide for 2 hours, dehydrated, and embedded in epoxy resin. Neurons in the VP thalamic nucleus were identified on semi-thin sections stained with 1 % toluidine blue, and then ultrathin sections 70-nm thick were cut on an ultramicrotome. The ultrathin sections were examined with a transmission electron microscope (H-800; Hitachi, Tokyo). The number of polyribosomes (aggregation of more than four ribosomes) per 1 μm2 in the basal side of a perikaryon was assessed in 40 randomly chosen neurons of the VP thalamic nucleus by using a micro computer imaging device (MICD) image analyzer (Imaging Research, St. Catherine, Ontario, Canada).
Measurement of rCBF
rCBF was measured by [14C] iodoantipyrine autoradiography at 1, 3, 6, and 12 hours; 1 and 3 days; 1 week; and 1 month after MCA occlusion (three animals each). The femoral vein was cannulated under pentobarbital anesthesia, and a bolus of 50 μCi/kg of 4-iodo [N-methyl-14C] antipyrine (specific activity, 67.5 mCi/mmol; New England Nuclear, Boston, MA) was injected intravenously over a 30-second period with a microsyringe (Hamilton, Reno, NV). The animal was decapitated approximately 30 seconds after the start of isotope injection. The brain was promptly removed and frozen with dry-ice-cold isopentane. Coronal sections 20-μm thick were cut with a cryostat and mounted on poly-L-lysine—coated slide glasses. Coronal sections for macroautoradiography were exposed to Kodak NMC-1 film (Eastman Kodak, Rochester, NY) for 2 weeks to detect regional radioactivity in the thalamus and in the cerebral cortex. The mean optical density of five boxed areas (containing 100 pixels in 0.01 mm2 each) in the VP thalamic nucleus and in the cerebral cortex on both sides were calculated by using a MICD imaging analyzer and the standard optical densities of coexposed 14C-Microscales (Amersham, Arlington Heights, IL).
[14C]-leucine autoradiography
Amino acid incorporation in the thalamus was investigated by [14C]-leucine autoradiography at 12 hours; 1, 3, and 5 days; 1 and 2 weeks; 1 and 2 months after MCA occlusion (four animals each). The femoral vein was cannulated under pentobarbital anesthesia, and a bolus of 150 μCi/kg of L-[14C]leucine (C1-labeled; specific activity, 58.0 mCi/mmol; New England Nuclear) was injected intravenously with a Hamilton microsyringe. The animal was decapitated 45 minutes after the injection. The brain was promptly removed and frozen with dry-ice-cold isopentane. Coronal sections of 10- and 20-μm thickness were cut with a cryostat in alternate order, and mounted on poly-L-lysine—coated slide glasses. All sections were incubated with 10% trichloroacetic acid overnight, and rinsed with water for 5 minutes to remove nonincorporated [14C]-leucine. Twenty-μm sections for macroautoradiography were exposed to Kodak NMC-1 film for 4 weeks to detect regional radioactivity in the thalamus. The mean optical density of five boxed areas (containing 100 pixels in 0.01 mm2 each) in the VP thalamic nucleus on both sides were calculated and converted to radioactivity per tissue weight (nCi/g) using the MICD imaging analyzer and the standard optical densities of coexposed 14C-Microscales (Amersham).
Ten-μm sections were used for microscopic autoradiography after dehydration (Stumpf et al., 1991). NR-M2 (Konica, Tokyo) was applied to the sections which were then exposed for 4 weeks at 4°C. The incorporated [14C]-leucine, manifested as black deposits, was observed under a light microscope at x400 magnification.
45Ca autoradiography
Calcium accumulation was investigated by 45Ca autoradiography at 1 and 12 hours; 1, 3, and 5 days; 1 and 2 weeks; and 1 month after MCA occlusion (four animals each). Animals were anesthetized with pentobarbital and the femoral vein was cannulated. A bolus of 1 mCi/kg of 45Ca (Ca-labeled; specific activity, 6.75 Ci/g; ICN Biomedicals Inc, CA, USA) was injected intravenously with a Hamilton microsyringe 6 hours before decapitation. The brain was promptly removed and frozen with dry-ice-cold isopentane. Coronal sections of 10- and 20-μm thickness were cut with a cryostat in alternate order, and mounted on poly-L-lysine—coated slide glasses. Twenty-micrometer sections were exposed to Kodak NMC-1 film for 4 weeks. The mean optical density of five boxed areas (containing 100 pixels in 0.01 mm2 each) in the VP thalamic nucleus on both sides were calculated by the MICD imaging analyzer.
Ten-micrometer sections were used for microscopic autoradiography after dehydration. NR-M2 (Konica) was applied to the sections which were then exposed for 4 weeks at 4°C. The accumulated 45Ca, manifested as black deposits, was observed under a light microscope at x400 magnification.
Statistical analysis
All values were given as mean±SD. Statistical comparisons among experimental groups were performed by the two-factor analysis of variance, followed by Scheffé's test for multiple comparisons. Statistical comparisons among data in the left and right hemispheres of the same animals were performed by paired t-test.
RESULTS
Light microscopy
In SH-SP rats with occlusion of the left MCA above the rhinal fissure and distal to the striate branches, the infarction site was confined to the left temporoparietal cortex, and the size of infarction was similar in all rats. A significant reduction in the ratio of the ipsilateral thalamic area to the contralateral one was noted 1 month after MCA occlusion, and it was further enhanced 3 months after ischemia (Fig. 1). Among the thalamic nuclei, the left VP thalamic nucleus, which projects nerve fibers to the infarcted area, specifically showed progressive shrinkage.
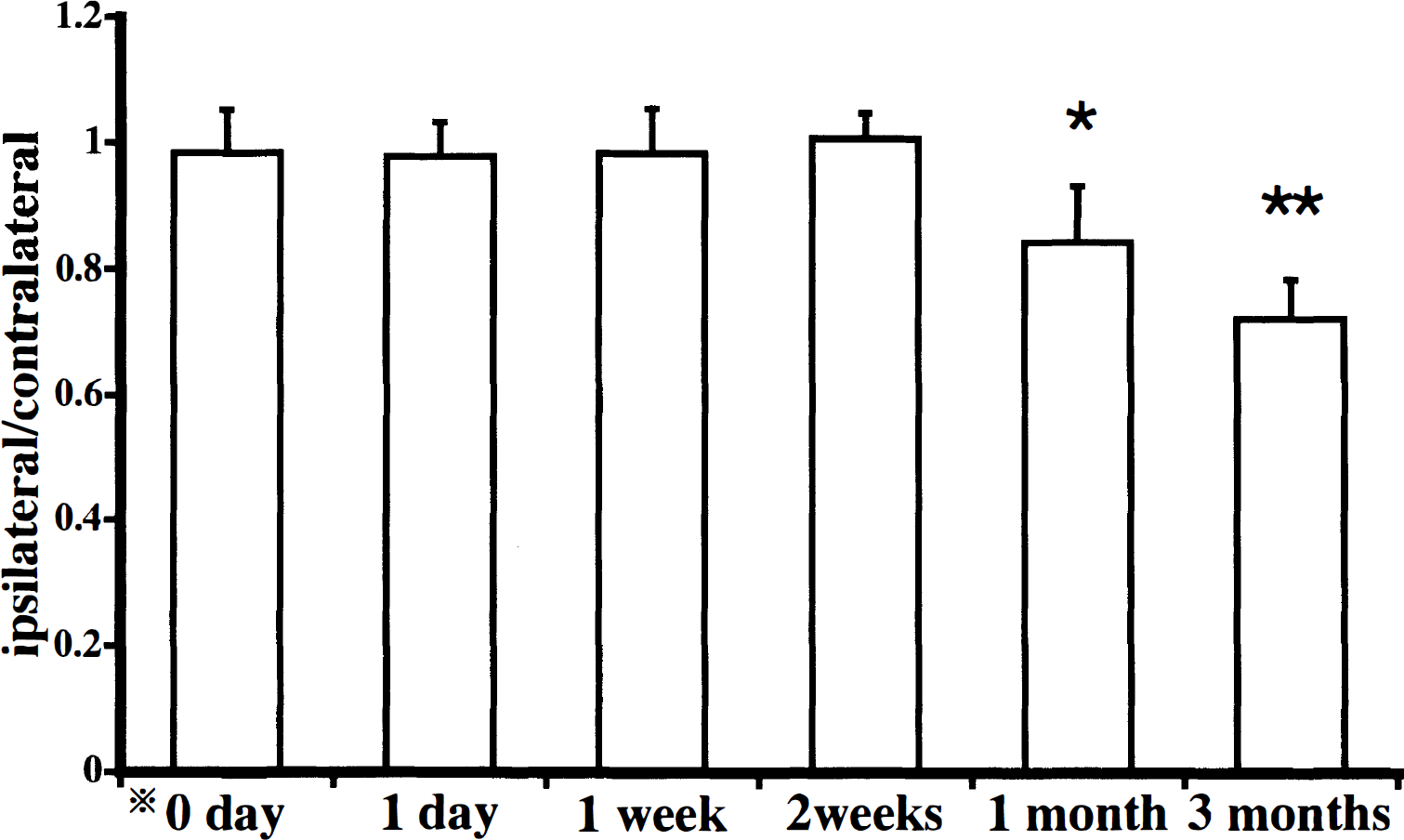
Time-dependent changes in the ratio of the left thalamic area to the right thalamic area after left middle cerebral artery occlusion. There were significant decreases in the ratio at 1 and 3 months after ischemia. Bars represent the SD. *0 day indicates normal control. Significant differences (*P < .05, **P < .01) are noted between ipsilateral and contralateral thalamus (paired t- test).
At high magnification, swelling of the cell nuclei and shrinkage of the neuronal perikarya were observed in the left but not right VP thalamic nucleus at the fifth day after MCA occlusion (Fig. 2A, B). One month after MCA occlusion, many neurons in the left VP thalamic nucleus had dark-stained pyknotic nuclei and shrunken perikarya, and glial cells were increased in number within the thalamic nucleus (Fig. 2C). Cell counting by the computerized image processing system revealed that neurons in the left VP thalamic nucleus were less numerous than those in the right VP thalamic nucleus at the seventh day after MCA occlusion and decreased gradually in number thereafter, and that the left VP thalamic nucleus contained more glial cells than the right one at the fifth day after MCA occlusion and exhibited a time-dependent increase in glial cells (Fig. 3).
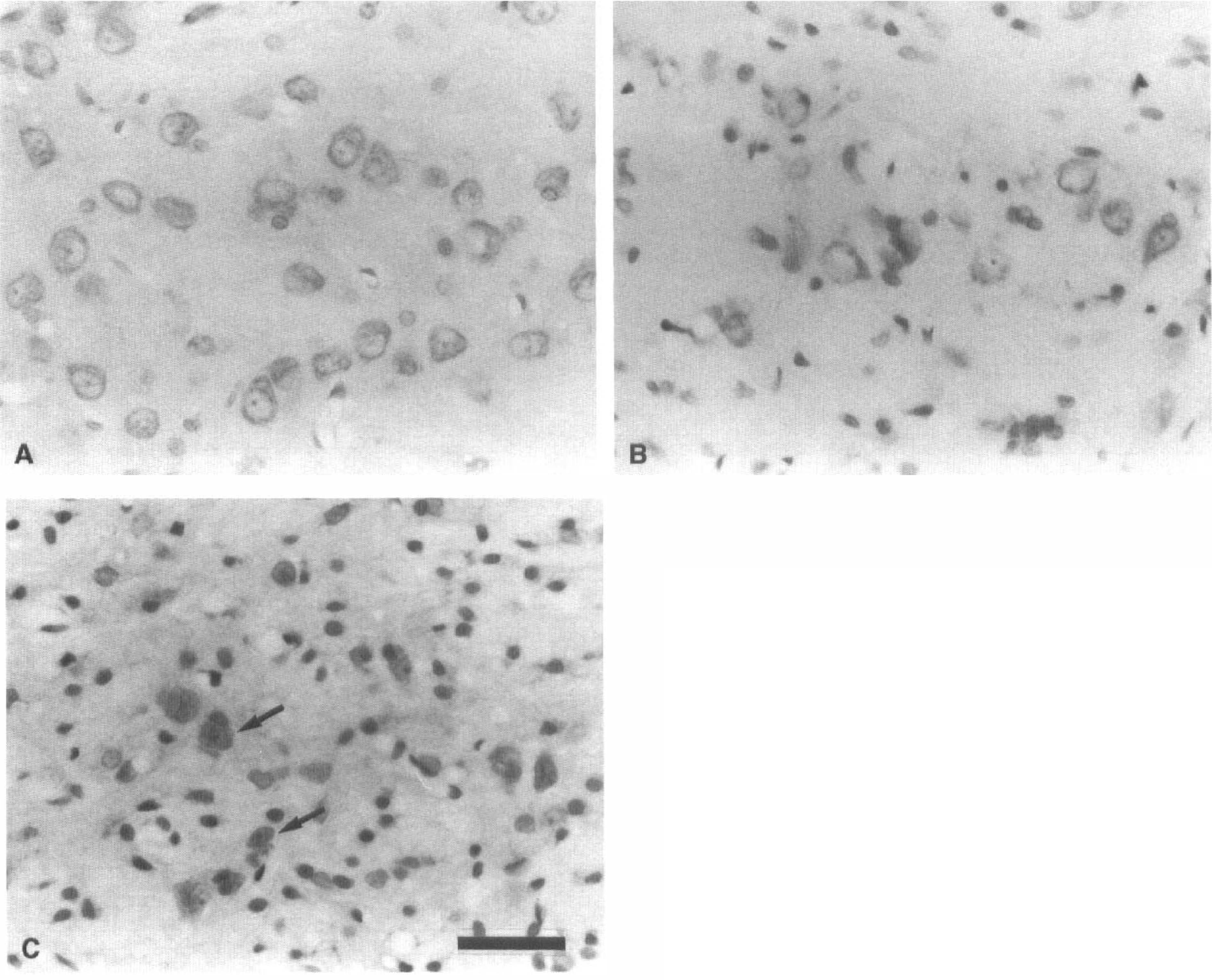
Photomicrographs of the ventroposterior thalamic nucleus in 1–μm paraffin sections stained with cresyl violet.
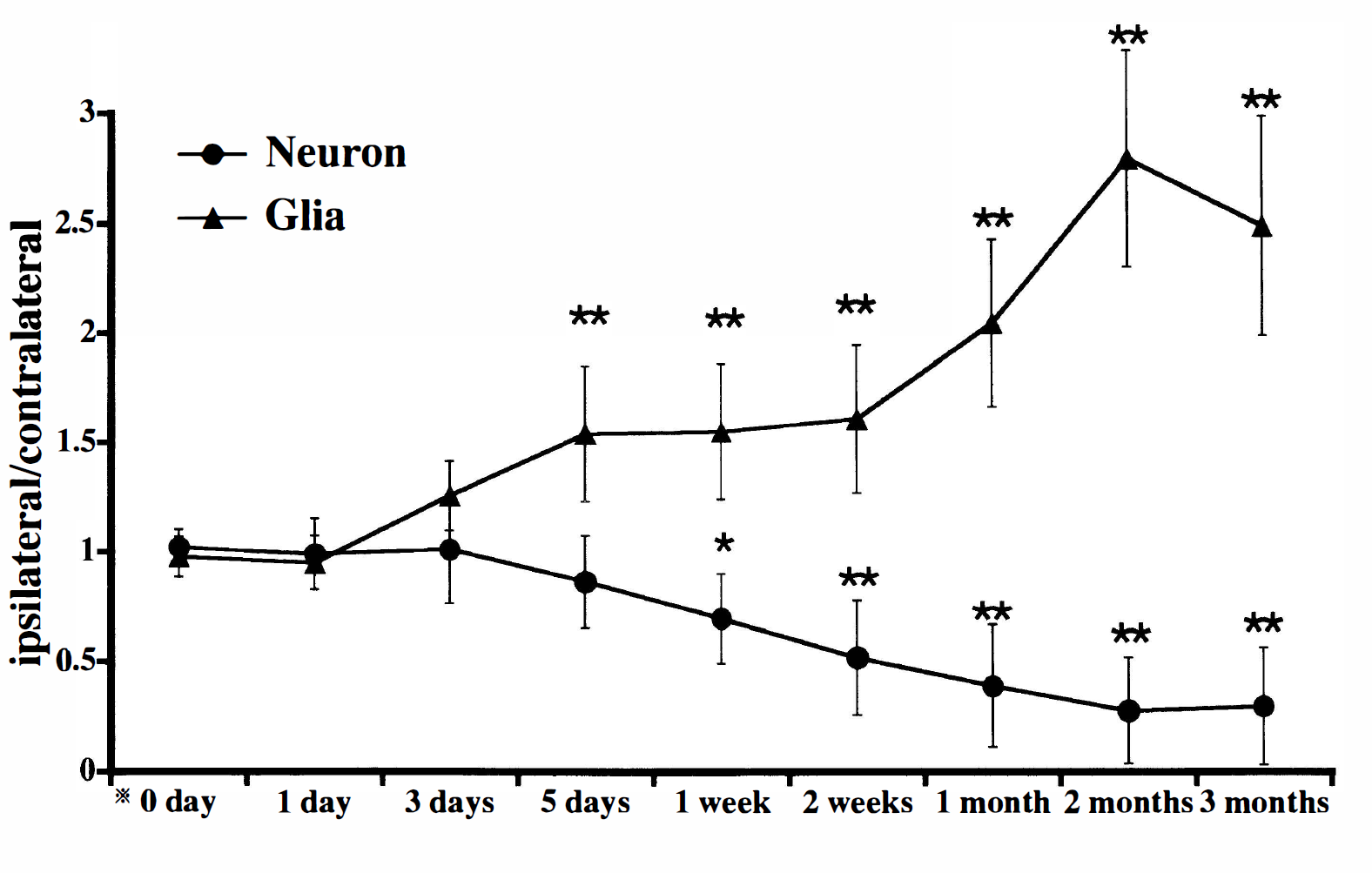
Time-dependent changes in the ratio of the number of ventroposterior (VP) thalamic neurons (closed circles) or glial cells (closed triangles) on the lesioned side to that of VP thalamic neurons or glial cells on the control side. There were significant decreases in the left-to-right ratio of neurons during the period 1 week to 3 months after middle cerebral artery (MCA) occlusion. In contrast, the left-to-right ratio of glial cells began to increase at the fifth day after MCA occlusion. Bars represent the SD. *0 day indicates normal control. Significant differences (*P < .05, **P < .01) are noted between ipsilateral and contralateral VP thalamic nuclei (paired t-test).
Electron microscopy
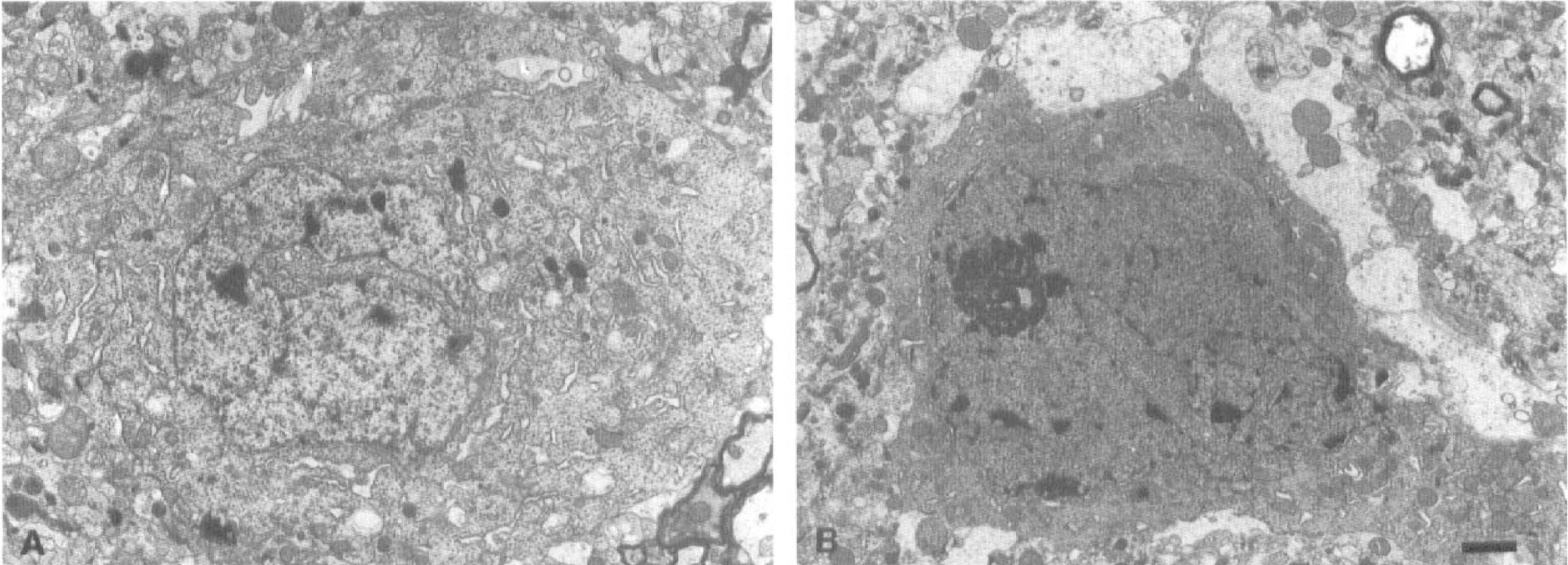
Electron micrographs of surviving neurons in the left VP thalamic nucleus are shown in Figure 4. Many ribosomes formed rosette-like aggregations, i.e., polyribosomes, in the perikarya of normal neurons (Fig. 4A). Perikaryal polyribosomes began to decrease in number at 5 days after the ischemic insult without showing any disaggregation of individual ribosomes (Fig. 4B, Table 1). At this period, thalamic neurons in the course of secondary degeneration exhibited partial vacuolation of the cytoplasm (Fig. 4B). During the period from 7 days to 1 month after MCA occlusion, the perikaryal polyribosomes gradually decreased in number (Fig. 4C, D, Table 1), and cytoplasmic vacuolation and mitochondrial swelling became conspicuous in a time-dependent manner. Besides the above changes in the cytoplasm, a deep indentation of cell nucleus and cell shrinkage associated with an abnormal high electron density were noted in a number of thalamic neurons at more advanced stage of degeneration (Fig. 5A, B).
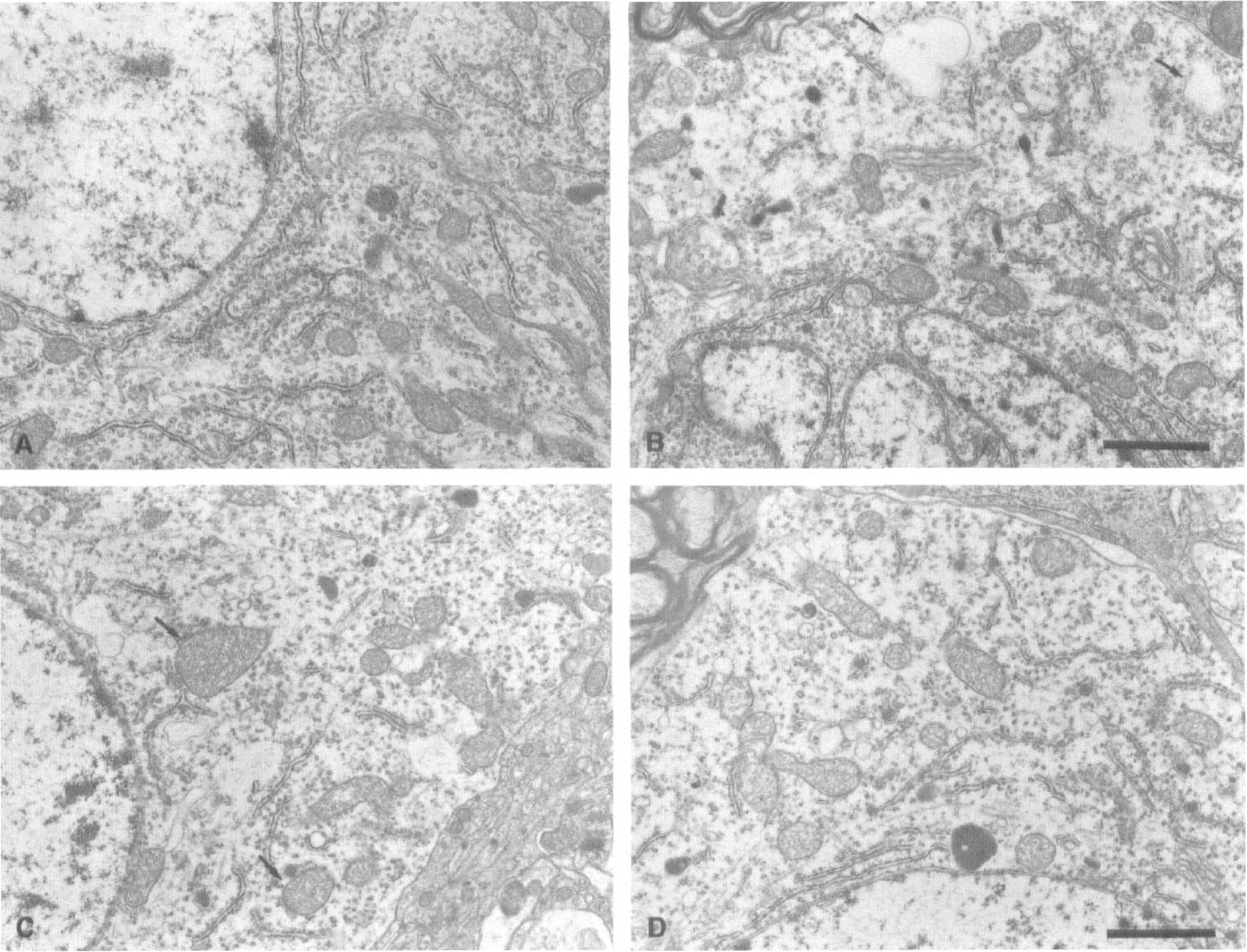
Electron micrographs of neuronal perikarya in the left ventroposterior thalamic nucleus.
Electron micrographs showing a deep indentation of the cell nucleus of a thalamic neuron at the fifth day after middle cerebral artery (MCA) occlusion
Time-dependent changes in polyribosome number in the ventroposterior thalamic nucleus after MCA occlusion in SH-SP rats
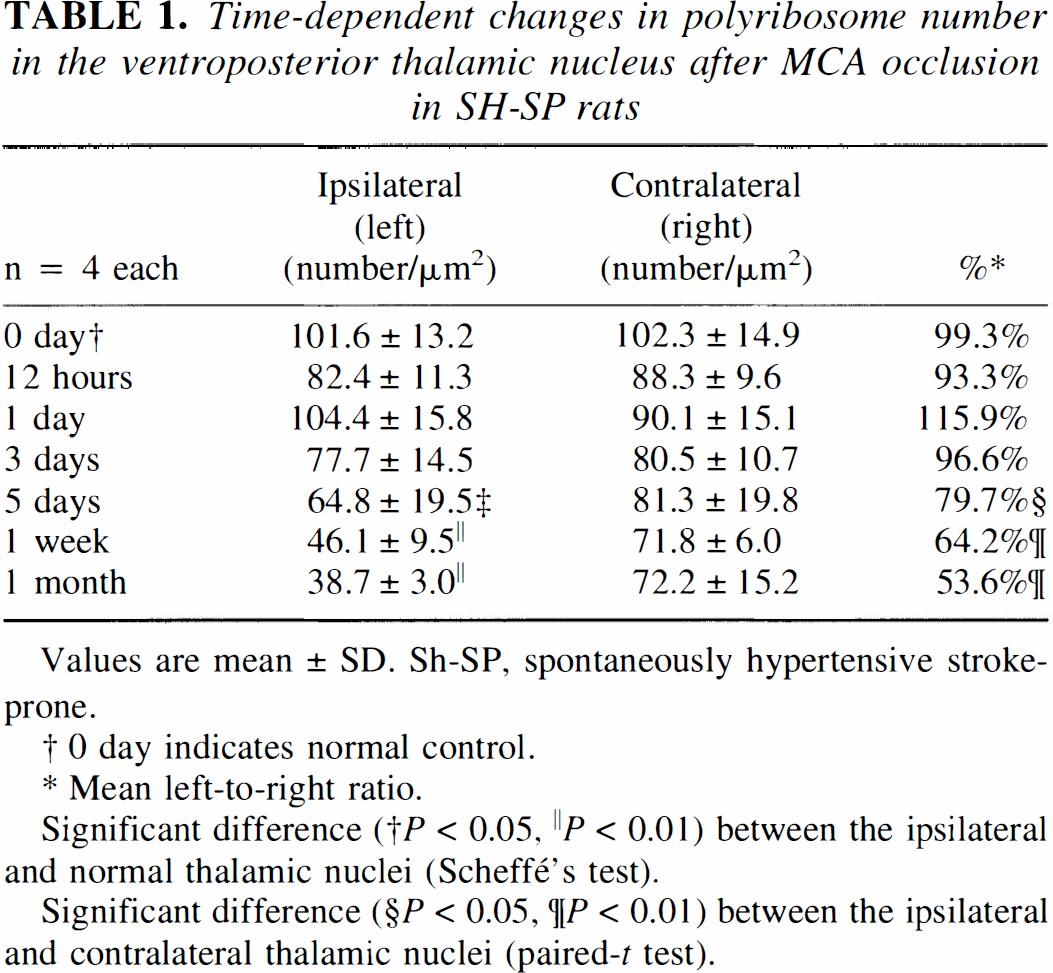
Values are mean ± SD. Sh-SP, spontaneously hypertensive stroke-prone.
0 day indicates normal control.
Mean left-to-right ratio.
Significant difference († P < 0.05, ∥P < 0.01) between the ipsilateral and normal thalamic nuclei (Scheffé's test).
Significant difference (§P < 0.05, ¶P < 0.01) between the ipsilateral and contralateral thalamic nuclei (paired-t test).
[14C] iodoantipyrine autoradiography
Table 2 shows the left-to-right ratio (light/right) of [14C] iodoantipyrine radioactivity in the cerebral cortex and in the VP thalamic nucleus. [14C] iodoantipyrine autoradiography showed that there was no significant difference in rCBF between the right and left thalamus until 1 month after the ischemic insult, whereas in the left temporoparietal cortex, rCBF severely decreased during 1 hour to 1 week after MCA occlusion (Table 2).
Time-dependent in accumulated [14C] iodoantipyrine radioactivity in the ventroposterior thalamic nucleus and cerebral cortex after MCA occlusion in SH-SP rats
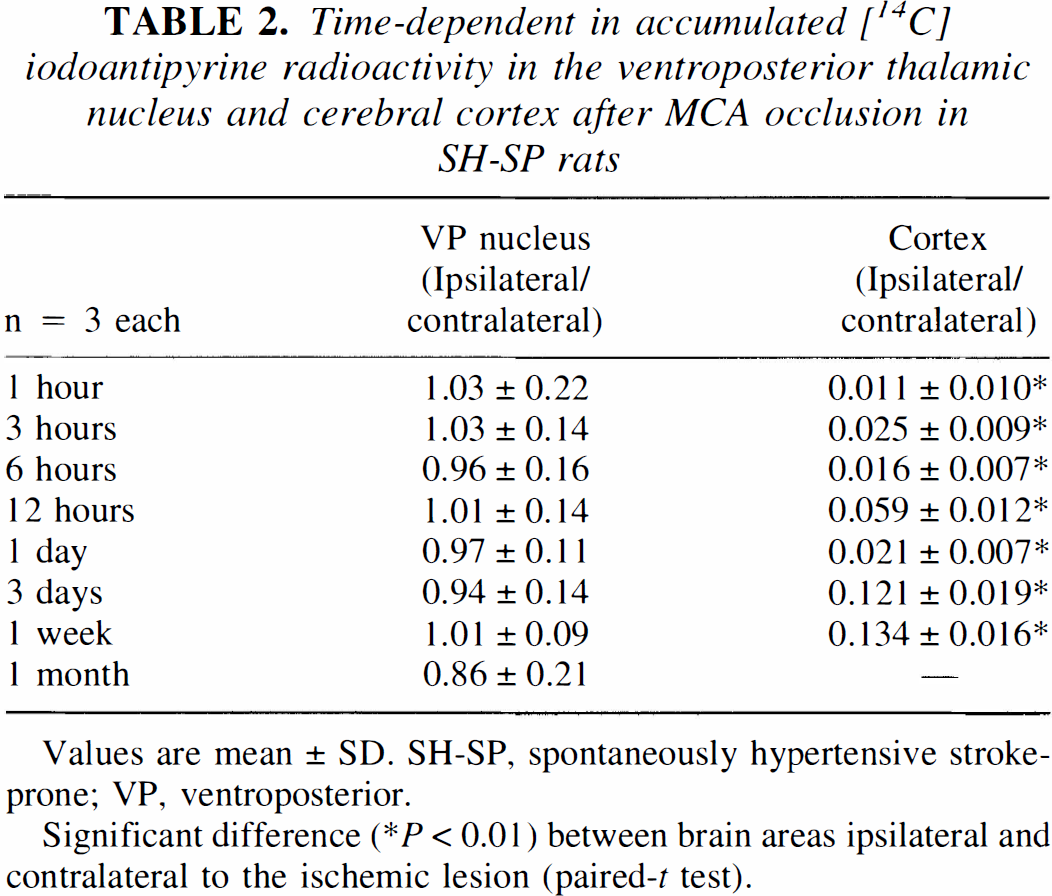
Values are mean ± SD. SH-SP, spontaneously hypertensive stroke-prone; VP, ventroposterior.
Significant difference (* P < 0.01) between brain areas ipsilateral and contralateral to the ischemic lesion (paired-t test).
L-[14C]-leucine autoradiography
The macroscopic L-[14C]-leucine autoradiography showed that there was no difference in amino acid incorporation between the right and left thalamus at the first day after the ischemic insult, while the amino acid incorporation was severely suppressed in the infarcted temporoparietal cortex at this time (Fig. 6A, Table 3). At the fifth day after the MCA occlusion, the amino acid incorporation in the ipsilateral VP thalamic nucleus was significantly reduced in comparison with that in the contralateral thalamus (Fig. 6B, Table 3). However, it returned close to the value of the contralateral thalamus during the period 1 to 2 weeks after ischemic insult (Fig. 6C, Table 3). At 1 month after the MCA occlusion, the amino acid incorporation in the VP thalamic nucleus on the infarction side was reduced again (Fig. 6D, Table 3). Such a transient postischemic resumption of amino acid incorporation as seen in the left VP thalamic nucleus was also noted in the infarcted temporoparietal cortex (Fig. 6).
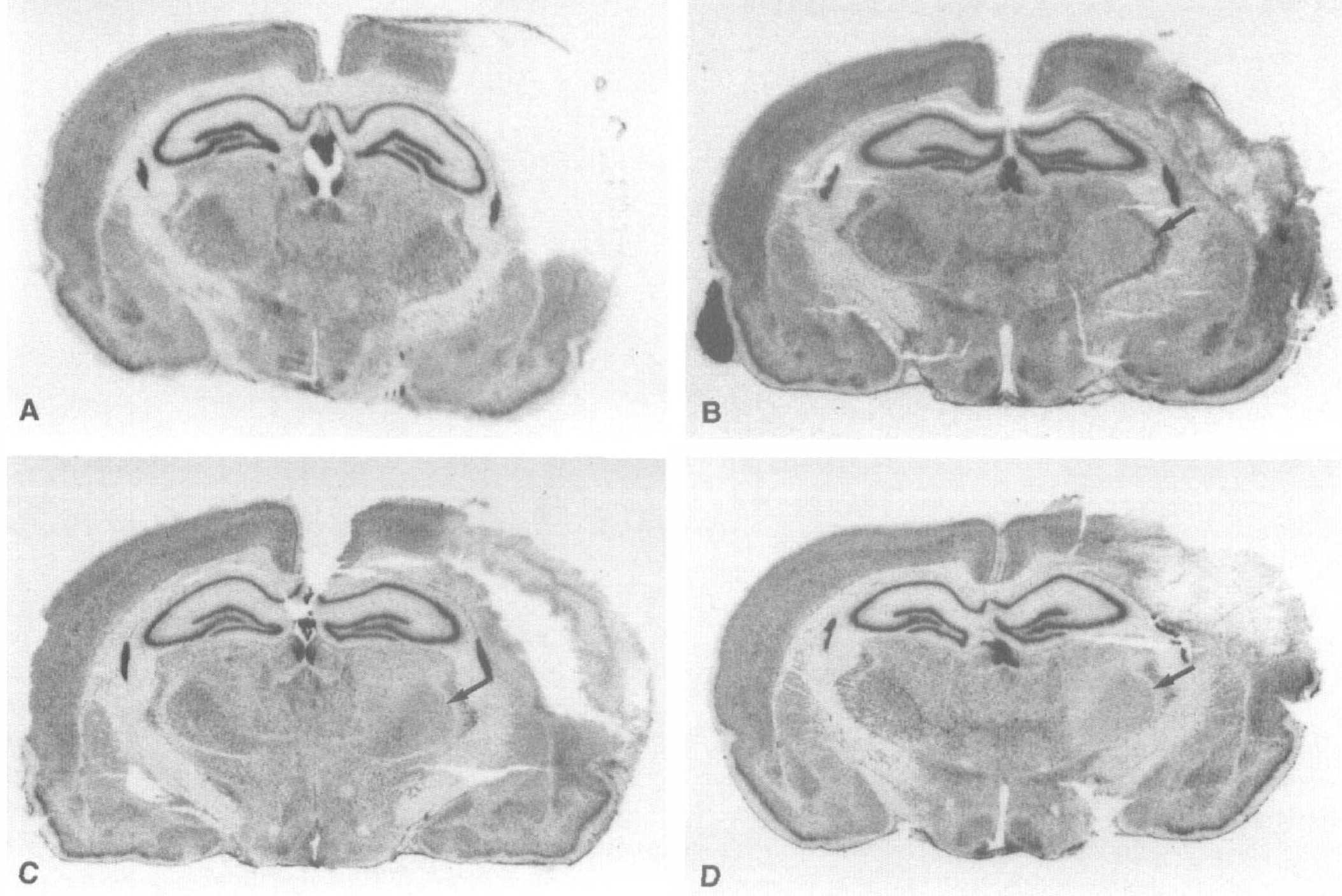
L-[14C]-leucine macroautoradiographs.
L-[14C]-leucine microscopic autoradiographs are shown in Figure 7. One day after MCA occlusion, deposits representing L-[14C]-leucine incorporation were seen mainly in and around thalamic neurons (Fig. 7A). They decreased in number at the fifth day after ischemia (Fig. 7B). During the period 1 to 2 weeks after MCA occlusion, the deposits were observed mainly in and around glial cells (Fig. 7C). At 1 month after MCA occlusion, L-[14C]-leucine incorporation was scarcely observed in neurons or glial cells (Fig. 7D).
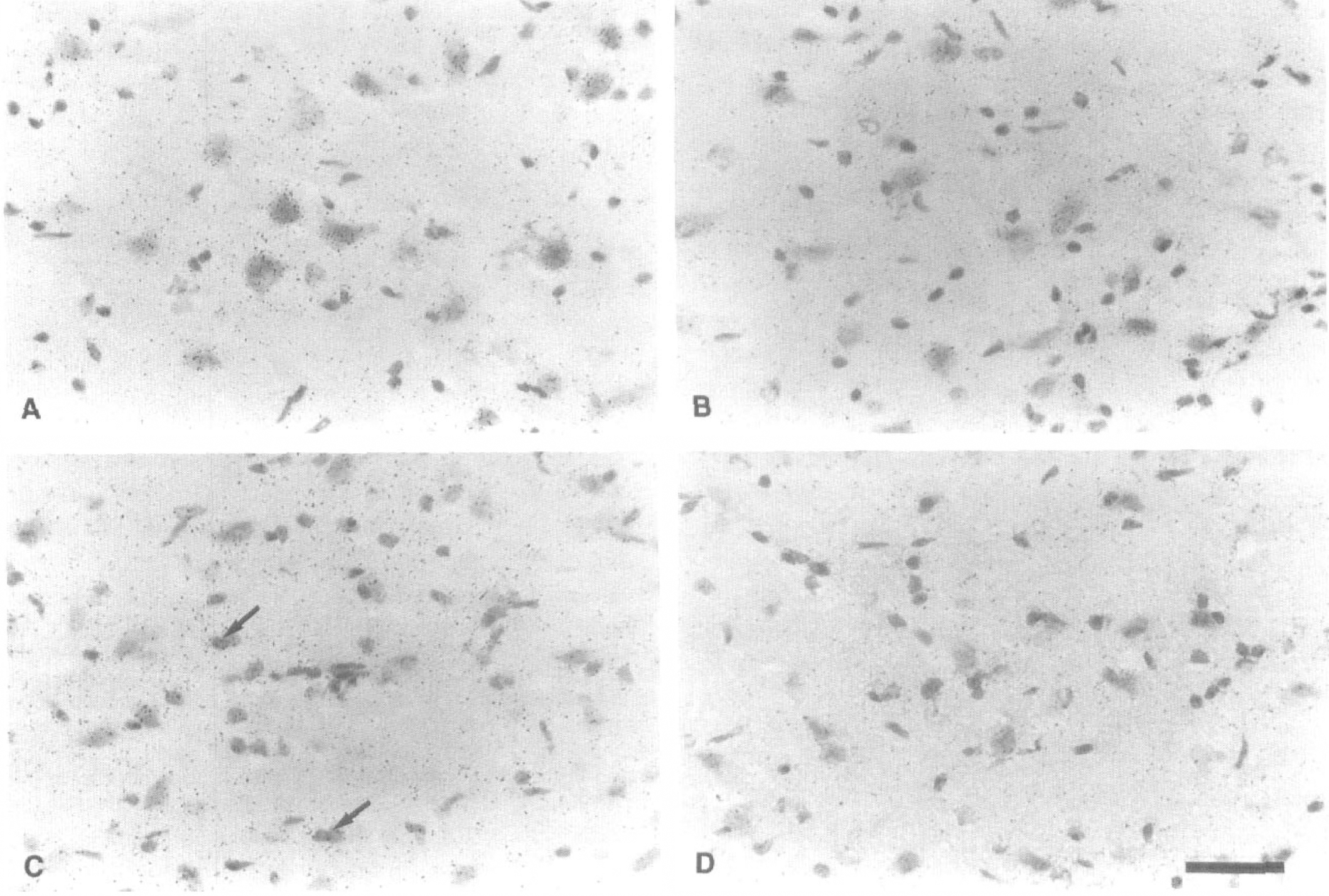
L-[14C]-leucine microautoradiographs in the ventroposterior (VP) thalamic nucleus on the lesioned side.
Time-dependent changes in incorporated [14C] leucine radioactivity in the ventroposterior thalamic nucleus after MCA occlusion in SH-SP rats
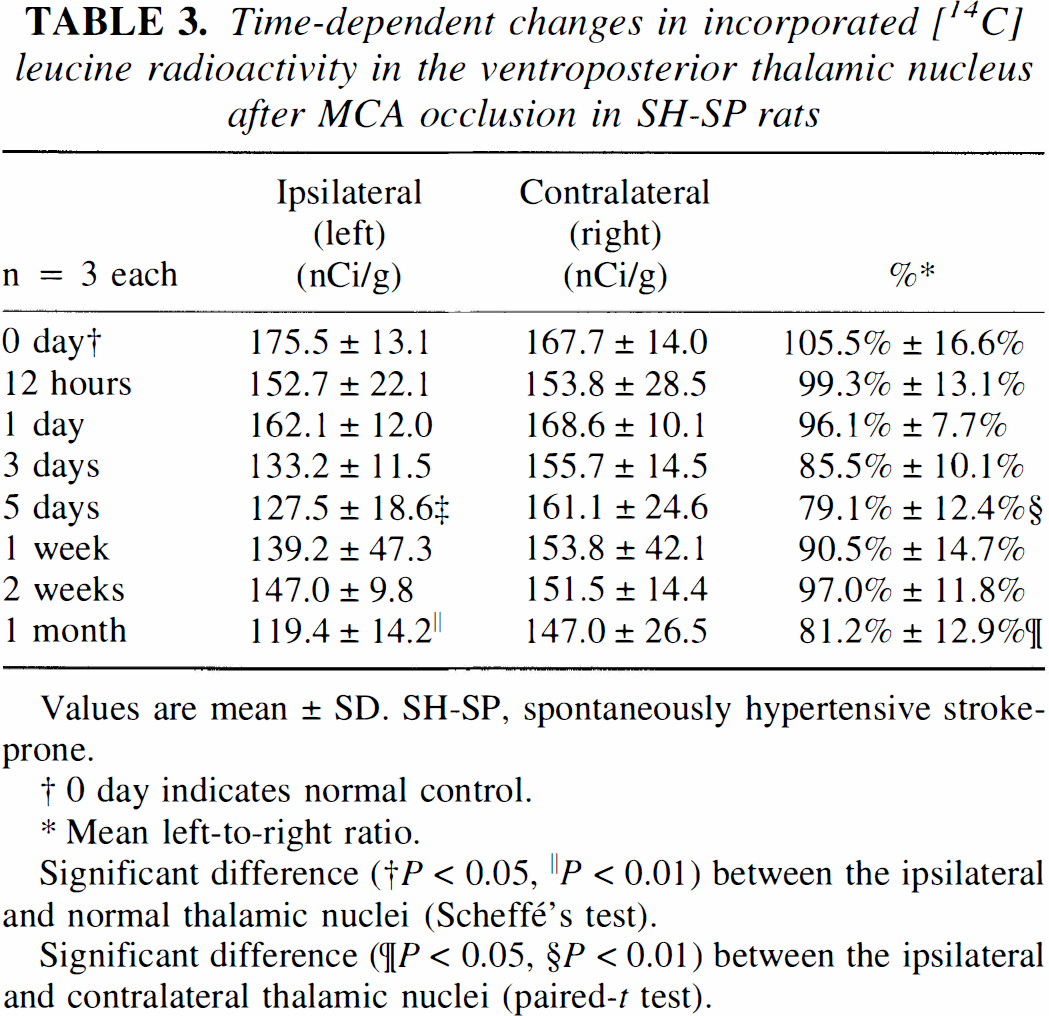
Values are mean ± SD. SH-SP, spontaneously hypertensive stroke-prone.
0 day indicates normal control.
Mean left-to-right ratio.
Significant difference († P < 0.05, ∥ P < 0.01) between the ipsilateral and normal thalamic nuclei (Scheffé's test).
Significant difference (¶P < 0.05, §P < 0.01) between the ipsilateral and contralateral thalamic nuclei (paired-t test).
45Ca autoradiography
The macroscopic 45Ca autoradiography showed 45Ca accumulation only in the infarcted temporoparietal cortex at 1 hour after MCA occlusion (Fig. 8A, Table 4). 45Ca accumulation occurred in the VP thalamic nucleus on the lesioned side by 3 days after MCA occlusion, and extended markedly into the entire temporoparietal cortex (Fig. 8B, Table 4). The accumulation of 45Ca in the left VP thalamic nucleus persisted even after an abrupt decrease in 45Ca accumulation in the infarcted temporoparietal cortex on postischemic day 14 (Fig. 8C, Table 4).
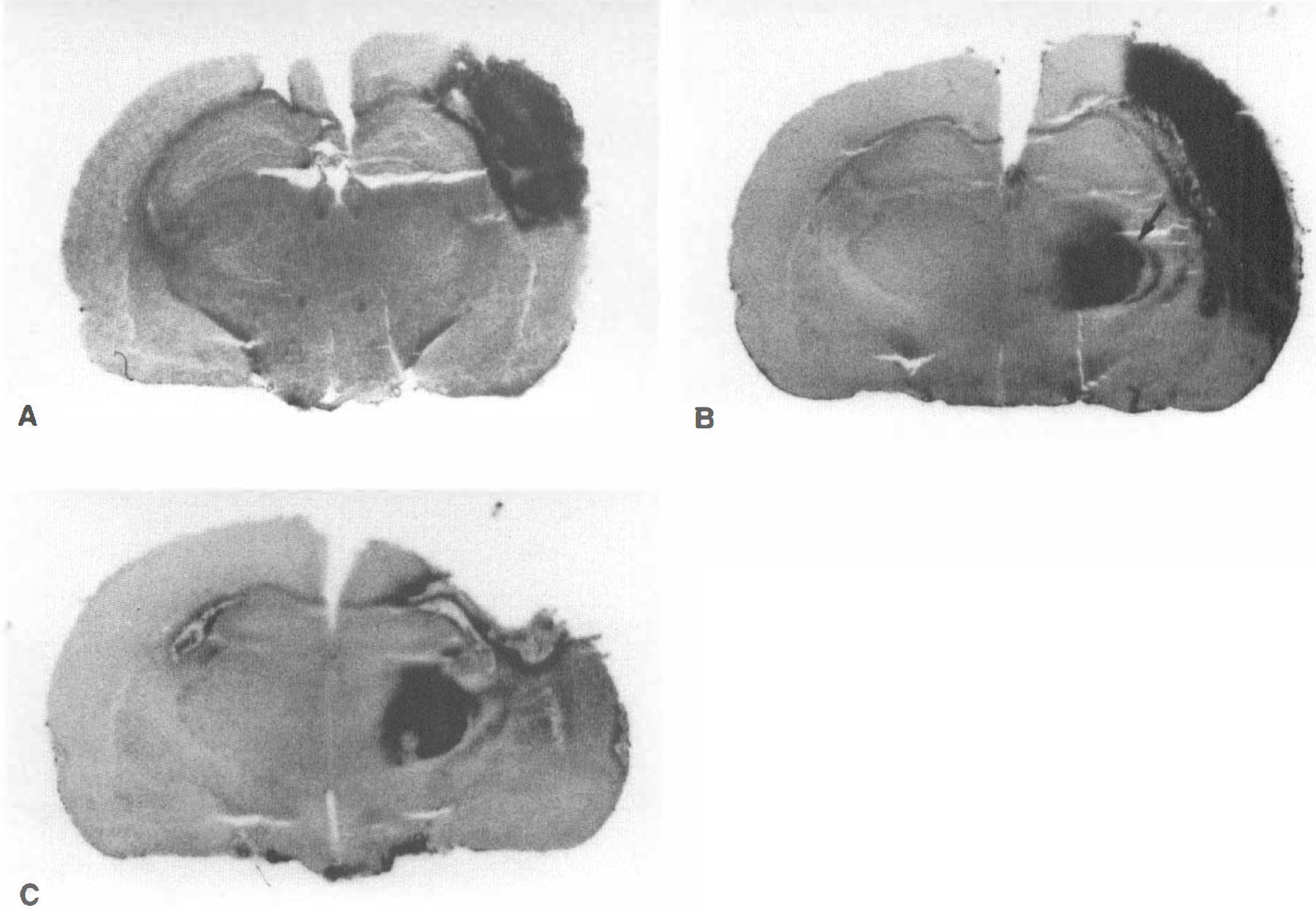
45Ca macroautoradiographs.
Time-dependent in accumulated [45Ca] radioactivity in the ventroposterior thalamic nucleus and cortex after MCA occlusion in SH-SP rats
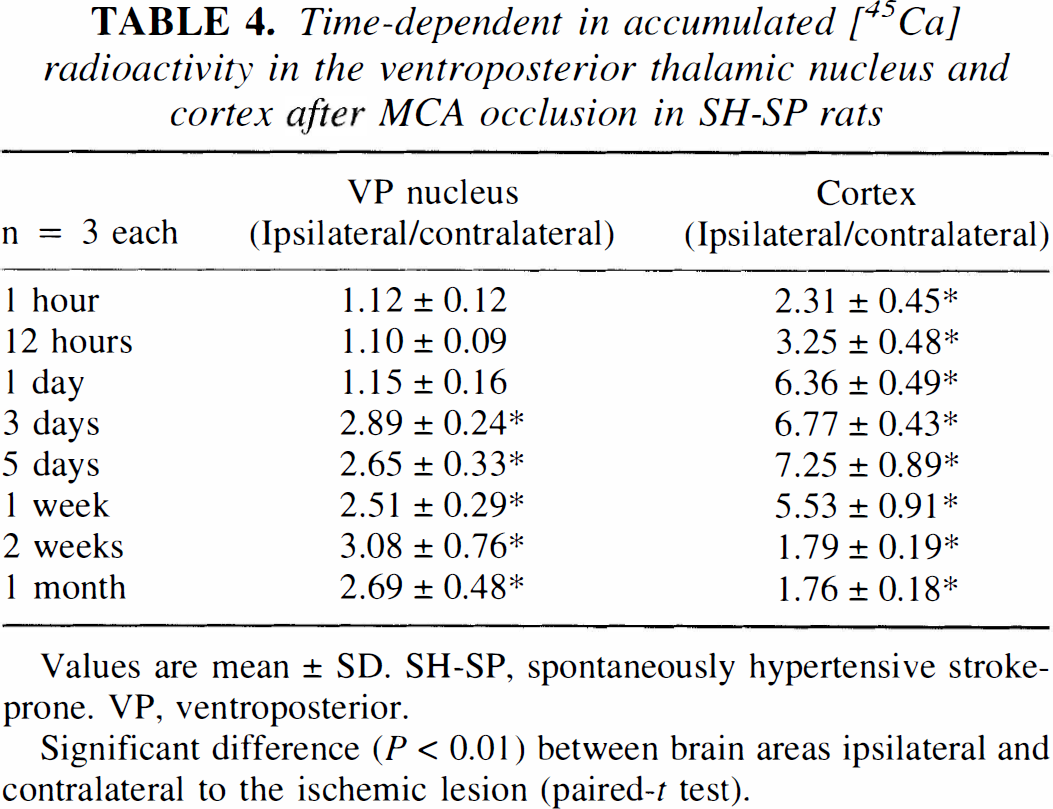
Values are mean ± SD. SH-SP, spontaneously hypertensive stroke-prone. VP, ventroposterior.
Significant difference (P < 0.01) between brain areas ipsilateral and contralateral to the ischemic lesion (paired-t test).
The microscopic 45Ca autoradiography performed at the third day after MCA occlusion showed that more deposits representing 45Ca radioactivity accumulated mainly in and around the neurons of the left and not right VP thalamic nucleus (Fig. 9).
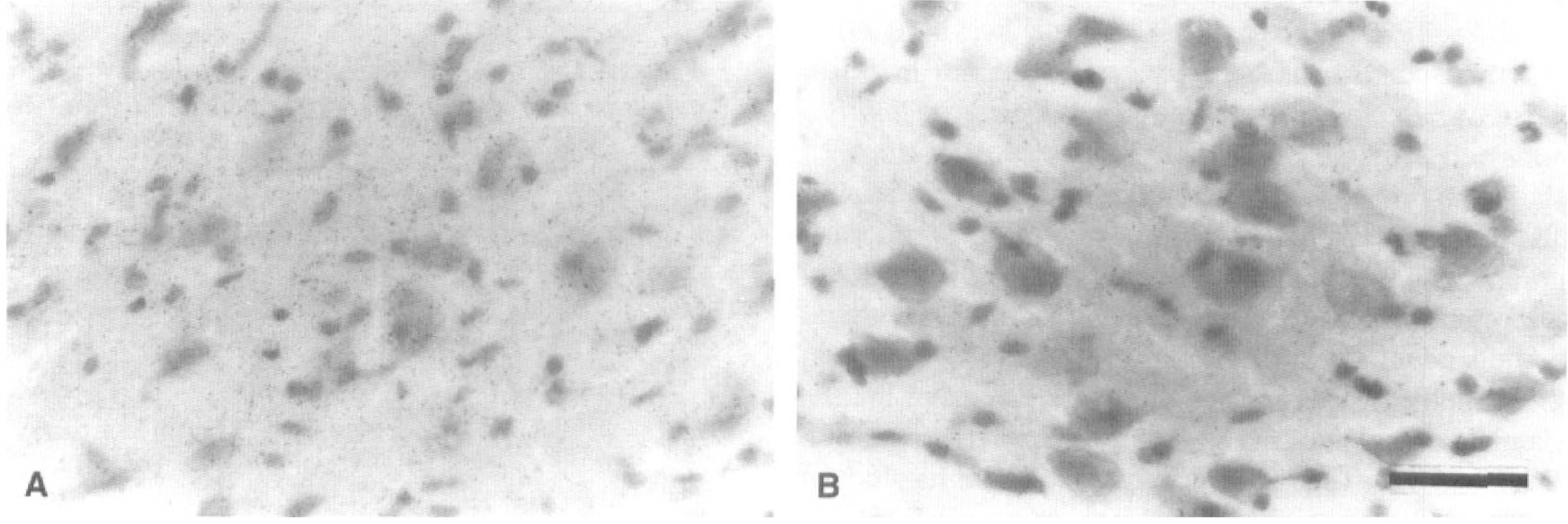
45Ca microautoradiographs in the bilateral ventroposterior (VP) thalamic nucleus at the third day after middle cerebral artery occlusion.
DISCUSSION
In the present study, SH-SP rats with occlusion of the MCA distal to the striate branches were used as a model to evaluate degeneration of the thalamus, which is remote from but connected synaptically with the primary ischemic lesion, because these rats exhibit a uniform and reproducible infarction localized only in the temporoparietal cortex, possibly due to little collateral pial anastomosis. We found that the blood flow of the thalamus was not affected in MCA-occluded SH-SP rats. This enabled us to investigate secondary thalamic degeneration without the effect of blood flow changes on the thalamus. In contrast, the conventional MCA-occlusion model using a different rat strain involves infarction not only in the temporoparietal cortex but also in the caudoputamen (Tamura et al., 1981a). Because both infarcted regions have synaptic connections with the thalamus and the size of the primary infarcted area varies considerably from rat to rat in the conventional MCA-occlusion model, it is difficult to quantitatively analyze the primary ischemic neuronal damage and subsequent thalamic degeneration. Taken together, the results of the present study suggest that our ischemic model using SH-SP rats may be more suitable for the investigation of thalamic degeneration after cortical ischemic damage than is the model previously reported.
Although there are several reports depicting thalamic degeneration associated with cerebral infarction, they refer only to relatively short-term morphologic alterations such as shrinkage of the thalamus 2 weeks after ischemia (Barron et al., 1973) and the development of neuronal abnormalities 1 week after ischemia (Iizuka et al., 1989, 1990; Yamada et al., 1991). In the present study, neuronal changes including cellular swelling and a decrease in polyribosomes in the VP thalamic nucleus on the lesioned side were observed 5 days after the MCA occlusion, and a significant reduction in the thalamic area was noted 1 month later. These morphologic changes are in agreement with the findings of previous studies (Iizuka et al., 1989, 1990; Fujie et al., 1990; Yamada et al., 1991) and reminiscent of the chromatolysis observed in neurons of the dorsal root ganglia after their axon injury (Evans and Gray, 1961; Meller, 1989). However, the VP thalamic neurons undergoing secondary degeneration, unlike the dorsal ganglion neurons subjected to process injuries, did not exhibit any disaggregation of individual ribosomes forming polyribosomes in harmony with a time-dependent decrease in the number of polyribosomes. In other words, chromatolysis did not occur in the thalamic neurons in response to the cerebral infarction in SH-SP rats. This finding is in accord with the results of previous studies using other focal ischemia models (Holmes, 1906; Iizuka et al., 1990).
In this study, however, we did not find nuclear chromatin fragmentation or apoptotic bodies throughout the experimental period; only a deep indentation of cell nucleus and cell shrinkage were noted in thalamic neurons at more advanced stages of degeneration. Although some of the degenerating thalamic neurons are known to exhibit DNA fragmentation as evaluated by deoxyribo-nucleotidyl transferase—mediated deoxyuridine triphosphate (dUTP)-biotin nick-end labeling (TUNEL) (Watanabe et al., 1997), they don't show all ultrastructures characteristic of apoptosis. We speculate that the secondary thalamic degeneration after cereberocortical infarction is a thalamus-specific event different from apoptosis or necrosis.
There have been a few studies showing metabolic or functional changes in the degenerating thalamus after cortical and striate infarction. In these study, reduction of glucose metabolism and suppression of somatosensory-evoked potentials were recognized at the first day after MCA occlusion (Kataoka et al., 1989; Tokuno et al., 1992). However, no report has focused on changes in protein synthesis in the degenerating thalamus after a selective cortical infarct. We examined radiolabeled amino acid incorporation to investigate the ability of protein synthesis in VP thalamic neurons after MCA occlusion, using L-[14C]-leucine as a tracer. It is adsorbed by the brain tissue at a high ratio (Oldendorf, 1971) and incorporated exclusively into newly synthesized proteins without remaining in the free radioactive form (Ingvar et al., 1985). The possibility of erroneous overestimation of the amino acid incorporation due to an increase in the specific activity of precursor pool must be considered under pathologic conditions when we interpret the data obtained from tracer-dose autoradiography because tracer-dose autoradiography with labeled amino acids cannot determine the specific activity of the precursor pool (Bodsch and Hossmann, 1983; Furuta et al., 1993). Nevertheless, a reduction in focal radioactivity as determined by tracer-dose amino acid autoradiography appears to reflect inhibition of protein synthesis in situ (Bodsch and Hossmann, 1986; Yoshimine et al., 1987).
In the present macroautoradiography, the amino acid incorporation into the VP thalamic nucleus on the lesioned side was reduced at the fifth day after cortical infarction, and it transiently returned close to the level of the control VP thalamic nucleus during 1 to 2 weeks after MCA occlusion. The microautoradiography showed that while deposits representing radioactivity were recognized mainly in and around neurons of the VP thalamic nucleus at the first and fifth day after MCA occlusion, they began to localize to glial cells at the first and second weeks after MCA occlusion. Therefore, the transient resumption of amino acid incorporation as detected in the macroautoradiography during 1 to 2 weeks after MCA occlusion is likely to reflect an increase in [14C]-leucine incorporation by reactive astrocytes, and amino acid incorporation in the thalamic neurons appears to decrease gradually after MCA occlusion. The suppression of amino acid incorporation in the neurons took place in accordance with the time-dependent decrease in the number of polyribosomes. These findings suggest that protein synthesis in the thalamic neurons decreased gradually after ipsilateral cortical infarction. In the gerbil hippocampal CA1 region, in which delayed neuronal death occurs after transient forebrain ischemia, polyribosomes in the perikarya of CA1 pyramidal neurons are abruptly diaggregated and amino acid incorporation is severely impaired within several hours after ischemic loading (Furuta et al., 1993). However, we observed that in rat VP thalamic neurons in the course of secondary degeneration, protein synthesis was maintained until 3 days after MCA occlusion and it gradually decreased thereafter. The question remains as to whether the decrease in protein synthesis is an epiphenomenon concurrent with the process of cell death or the cause of cell death. Protein synthesis appears to be crucial for cell survival on the basis of the finding that the application of protein synthesis inhibitors induces cell death in various organs (Cidlowski, 1982), but protein synthesis inhibitors prevent delayed neuronal death after transient global ischemia (Shigeno et al., 1990). Perhaps, inhibition of protein synthesis has diverse actions on damaged cells, depending on types, degrees, and stages of cell damage.
Among several methods for the measurement of the intracellular calcium level (Harris et al., 1981; Krnjevic et al., 1986; Deshpande et al., 1987), we selected 45Ca autoradiography to evaluate changes in Ca2+ in the VP thalamic nucleus because this technique reflects the in vivo distribution of calcium in tissue and tissue fluids at the time of 45Ca administration, despite the technique's poor resolution and difficulty in quantification (Nakamura et al., 1993). 45Ca accumulated in the cortical infarcted area as early as 1 hour after the MCA occlusion, whereas the VP thalamic nucleus on the lesioned side began to exhibit 45Ca accumulation at the third day after MCA occlusion, before the significant decrease in protein synthesis within the brain nucleus. Microautoradiographs showed that the 45Ca accumulated mainly in neurons of the VP thalamic nucleus. An overaccumulation of intracellular Ca2+ can induce cell death, acting occasionally as a setpoint of programmed cell death (Cohen, 1993). It can also exacerbate neuronal death in cases of cerebral ischemia. When hippocampal CA1 neurons vulnerable to brain ischemia show delayed neuronal death, an overload of intracellular Ca2+ is considered to inhibit protein synthesis, leading to cell death. It is plausible that the increase in the intracellular Ca2+ concentration before the decrease in protein synthesis in thalamic neurons of MCA-occluded SH-SP rats accelerates the slowly progressive thalamic degeneration. Presumably, the early Ca2+ increase in cases of brain ischemia leads to a decrease in polyribosomes in thalamic neurons or to a disaggregation of polyribosomes in hippocampal CA1 neurons, thereby inhibiting protein synthesis in different manners within the thalamic and CA1 neurons. The mechanisms by which Ca2+ regulates protein synthesis differentially remain to be elucidated.
Based on the present experimental results, the secondary thalamic degeneration is concluded to be a slowly progressive event after the primary cortical lesion has been established. The possibility of prevention of the thalamic degeneration is apparently greater than that of the prevention of the primary ischemic damage. Indeed, there is a time difference between the beginning of calcium accumulation and the suppression of protein synthesis in the thalamus after the onset of ischemia. It is tempting to speculate that the secondary thalamic degeneration could be prevented if neuroprotective agents are administered before the increase in the Ca2+ concentration in the thalamus. This speculation is supported by the finding that thalamic degeneration after cortical infarction is precluded by the intracisternal administration of basic fibroblast growth factor (Yamada et al., 1991) or by continuous infusion of ciliary neurotrophic factor into the lateral ventricle (Kumon et al., 1996) at early periods after ischemia. Further studies are needed to determine whether the prevention of secondary neuronal damage is beneficial together with treatment of the primary ischemic lesion.
Footnotes
Acknowledgments
The authors thank Professor T. Suzuki and Dr. Y. Ohta (Kinki University) for the generous provision of SH-SP rats, and Mr. M. Sudoh, Mr. S. Masuda, and Mr. D. Shimizu for their technical assistance.
