Abstract
Aerobic exercise training promotes cardiovascular, brain and cognitive health. Regular exercise is associated with higher cardiorespiratory fitness, commonly assessed by peak oxygen uptake (VO2peak) during maximal effort testing. Higher cardiorespiratory fitness has been linked to preserved brain health, particularly higher gray matter volume and perfusion. The brain relies heavily on oxidative metabolism, yet the relationship between cardiorespiratory fitness and brain oxidative metabolism remains underexplored. This study investigated the association between VO2peak and two key cerebral metabolic parameters: the cerebral metabolic rate of oxygen consumption (CMRO2) and oxygen extraction fraction (OEF), which represents the balance between cerebral blood flow (CBF) and CMRO2. Thirty-seven healthy adults aged ⩾50 underwent maximal cardiopulmonary exercise testing for VO2peak assessment. Neuroimaging included dual calibrated functional MRI (dc-fMRI) and quantitative susceptibility mapping (QSM). Higher VO2peak correlated positively with higher CBF across whole-brain gray matter but showed no relationship with CMRO2. Conversely, higher VO2peak negatively correlated with lower OEF from both dc-fMRI and QSM. These findings suggest that greater cardiorespiratory fitness enhances cerebral perfusion without changing resting metabolic rate in healthy older adults, resulting in a reduced oxygen extraction. These results are consistent with exercise yielding improved vascular–metabolic coupling, which would reduce the likelihood of transient hypoxic episodes.
Keywords
Introduction
Cardiorespiratory fitness naturally declines with age but can be maintained or improved through regular aerobic exercise.1,2 While the gold standard for assessing cardiorespiratory fitness is maximal oxygen uptake (VO2max), due to practical limitations in older adults, peak oxygen uptake (VO2peak)—which closely corresponds to VO2max—is often used instead. 3 Higher VO2peak is consistently linked to lower all-cause mortality, improved cardiovascular health, and better cognitive and physical health outcomes in aging populations.4–8
In the brain, regular aerobic exercise and higher cardiorespiratory fitness promote cognitive health, enhancing executive functions and memory in both younger and older adults.6,9,10 Structurally, higher cardiorespiratory fitness has been linked to greater cerebral volumes of gray and white matter, most notably in the hippocampus, but also in frontal and temporal regions.11,12 Recent quantitative syntheses suggest these volumetric effects are present but generally small and heterogeneous: aerobic exercise appears to help retain hippocampal volume, 13 and broader pooled evidence in older and at-risk populations often yields modest or neutral average effects on whole-brain GM and hippocampus. 14 Evidence from meta-analyses similarly points to small yet positive effects of exercise on cognition. 15 Functionally, cardiorespiratory fitness is thought to support cerebrovascular health by increasing cerebral blood flow (CBF), thereby improving oxygen and nutrient delivery to brain tissue.16–18
Potentially underlying these beneficial effects, sustained exercise has been shown in both animal and human studies to promote angiogenesis, neurogenesis, and synaptic plasticity—particularly in the hippocampus.19,20 Moreover, exercise has been demonstrated in preclinical models to enhance mitochondrial function within neurons, contributing to more efficient brain energy metabolism and resilience against oxidative stress.21–24 Conversely, lower cardiorespiratory fitness has been linked to mitochondrial dysfunction, 25 adversely impacting brain metabolism and overall brain health. Metabolic deficits and mitochondrial dysfunction may specifically manifest as a reduction in the cerebral metabolic rate of oxygen consumption (CMRO2), which represents the rate at which the brain consumes oxygen to support cellular metabolism.26,27 Another important biomarker sensitive to mitochondrial health is the oxygen extraction fraction (OEF), which reflects the balance between cerebral oxygen supply and utilization.
Improving cardiorespiratory fitness likely exerts beneficial effects on brain oxidative metabolism biomarkers, which can be non-invasively assessed using biomarkers such as CMRO2 and OEF. Investigating the relationship between VO2peak and these metabolic markers is clinically relevant because they provide unique insights into brain function beyond traditional vascular metrics like CBF. For instance, elevated OEF could indicate a mismatch between oxygen delivery and cellular metabolic demands, suggesting an increased risk of transient ischemia. 28 Similarly, reductions in CMRO2 might reflect compromised mitochondrial efficiency or neuronal loss.26,27 Thus, while higher fitness generally improves CBF, exploring CMRO2 and OEF specifically offers critical insights into the metabolic integrity and cellular health of the brain—dimensions not captured solely by vascular assessments. To date, only one study has investigated the relationship between whole-brain CMRO2 and VO2peak, reporting a negative association. 29 However, no studies have yet examined the regional associations between cardiorespiratory fitness and CMRO2 or the global and regional associations with OEF. Understanding how these parameters vary with cardiorespiratory fitness in healthy adults may offer insight into the mechanisms linking fitness to brain health and whether exercise can be used to improve brain metabolic health.
In this study, we aimed to examine how VO2peak relates to cerebral metabolic health in healthy adults aged 50 years and older. To do so, we assessed key vascular and metabolic biomarkers, including OEF, CBF, and CMRO2. OEF was measured using two complementary techniques: dual-calibrated functional MRI (dc-fMRI) and quantitative susceptibility mapping (QSM) to cross-validate our results, while CBF was measured using arterial spin labeling (ASL). We also derived estimates of CMRO2 by integrating these modalities. We then explored the associations between all brain parameters and VO2peak, offering novel insights into the links between fitness and brain metabolism.
We hypothesize that higher cardiorespiratory fitness, as measured by VO2peak, is positively associated with cerebral vascular health, reflected by higher CBF and better cerebral metabolic health reflected by lower OEF and higher CMRO2.
Methods
Participants
Inclusion criteria in this study included being older than 50 years and fluent in either English or French to ensure informed consent and valid administration of neuropsychological assessments.
Exclusion criteria included a history of cardiovascular, neurological, psychiatric or respiratory disorder, thyroid disease, potential cognitive impairment (mini mental state examination (MMSE) < 25), current tobacco use, high alcohol consumption (more than two drinks per day), contraindications to MRI (e.g. ferromagnetic implants, claustrophobia), having cardiometabolic risk factors known to affect brain vascular and metabolic health, including treated or untreated hypertension (BP > 140/90 mmHg) and diabetes (types 1 and 2). Participants were also excluded if they had undergone surgery under general anesthesia within the past 6 months, had malignant arrhythmias during exercise, arthritis or claudication, severe exercise intolerance, or excessive discomfort due to hypercapnia (>5 on the Banzett dyspnea scale). 30
Figure 1 indicates the flow diagram of the participant inclusion in this study. A total of 54 (females;
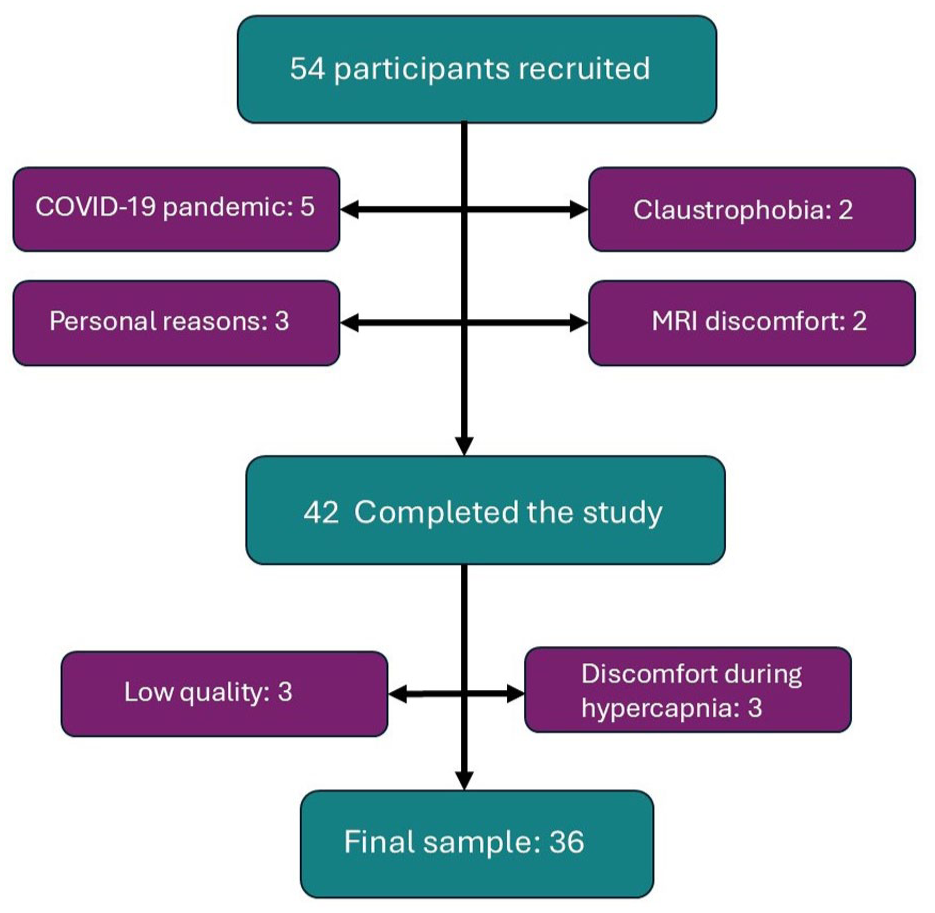
Flow diagram of participant inclusion. A total of 54 participants were screened and consented. Of these, 12 discontinued participation, leaving 42 who completed the study. Among these, three were excluded due to low-quality data and three were excluded due to discomfort during the hypercapnia challenge, resulting in a final analyzed sample of 36 participants.
Data was collected over three visits. Prior to the first visit, participants completed a medical history questionnaire to determine eligibility. Visit 1 included written informed consent, assessment of global cognitive function using the mini-mental state examination (MMSE) 31 and Montreal Cognitive Assessment (MoCA). 32 Additionally, participants underwent a 2-min hypercapnia test to ensure their ability to tolerate the respiratory manipulation during the subsequent MRI session (with two participants being excluded at this stage). During the next visit, participants completed a stress test on a cycle ergometer to determine their peak oxygen consumption. The final visit was dedicated to the MRI acquisition. Demographic information for all participants is in Table 1.
Demographic data on participants with respiratory data.
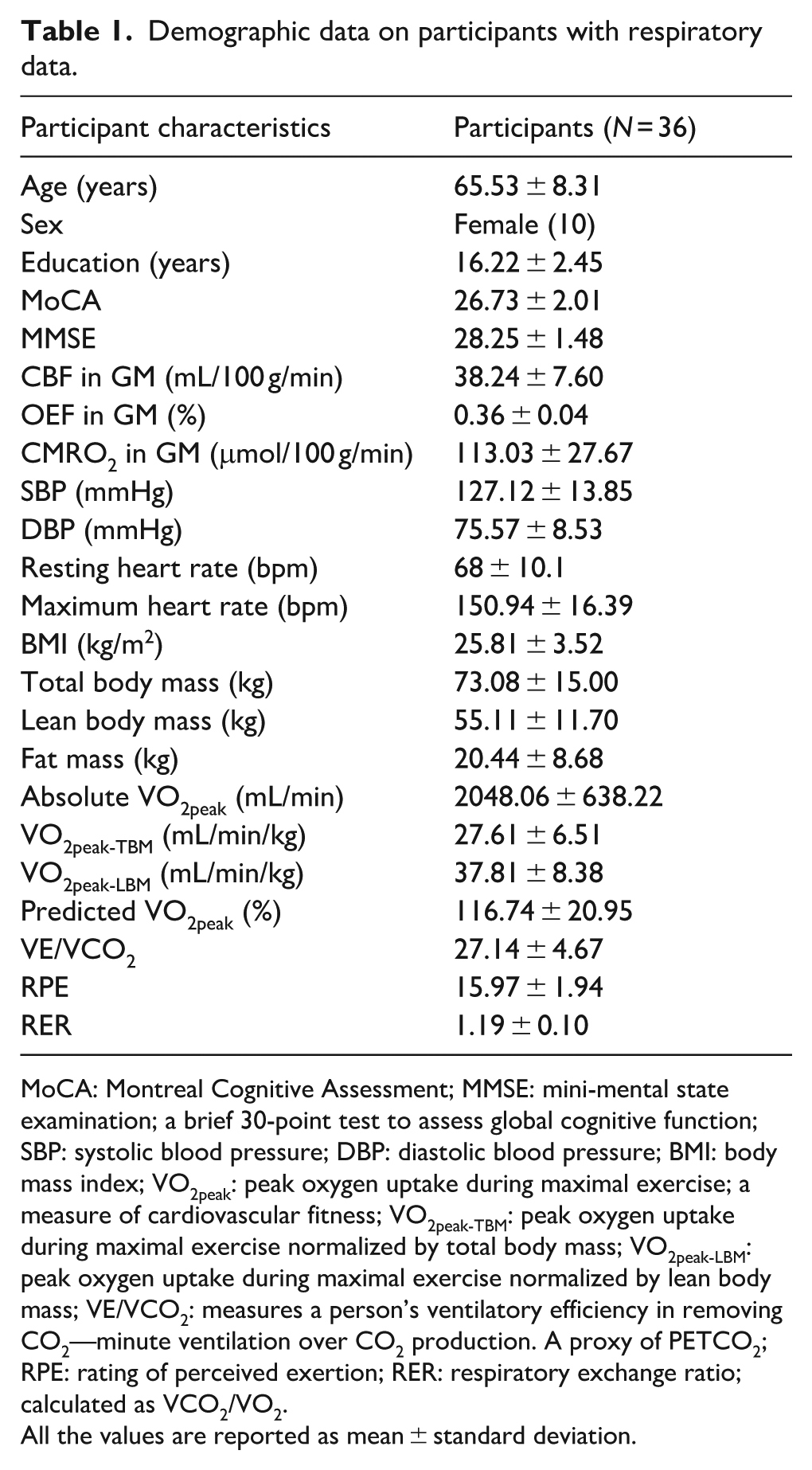
MoCA: Montreal Cognitive Assessment; MMSE: mini-mental state examination; a brief 30-point test to assess global cognitive function; SBP: systolic blood pressure; DBP: diastolic blood pressure; BMI: body mass index; VO2peak: peak oxygen uptake during maximal exercise; a measure of cardiovascular fitness; VO2peak-TBM: peak oxygen uptake during maximal exercise normalized by total body mass; VO2peak-LBM: peak oxygen uptake during maximal exercise normalized by lean body mass; VE/VCO2: measures a person’s ventilatory efficiency in removing CO2—minute ventilation over CO2 production. A proxy of PETCO2; RPE: rating of perceived exertion; RER: respiratory exchange ratio; calculated as VCO2/VO2.
All the values are reported as mean ± standard deviation.
Maximal cardiopulmonary exercise test (CPET)
Incremental CPET to volitional exhaustion was performed on a cycle ergometer (E100; Cosmed, Italy) according to the latest recommendations and as previously published.33,34 A 3-min warm-up phase at an initial workload of 20 W was followed by an incremental exercise test, with increases of 10–20 W/min depending on the participant’s physical capacity, performed at a pedaling cadence between 60 and 80 rpm. The recovery phase consisted of 2 min of active recovery at 20 W at pedaling speed between 50 and 60 rpm, followed by 3 min of passive recovery, during which participants remained seated quietly on the ergometer. Gas exchange parameters were continuously measured at rest, during exercise, and during recovery using a metabolic system (Cosmed Quark; Cosmed, Italy), capturing on a breath-by-breath basis and then averaged in 10 s increments as recently published. 33 There was continuous electrocardiogram (ECG) monitoring before, during the test and in the recovery (T12x; Cosmed, Italy). Diastolic (DBP) and systolic (SBP) blood pressures (Tango M2; Suntech, USA) and ratings of perceived exertion (RPE) were measured at rest and every 3 min throughout the test. The protocol interrupted if one of the following happened: (1) inability to maintain the required cadence of 60 rpm despite verbal encouragement, (2) clinical indications requiring interruption of the test, such as volitional exhaustion, (3) abnormal ECG findings or angina, and (4) excessive systolic blood pressure response (210 mmHg for men, 190 mmHg for women 35 ). Eighty percent of the participants could not exercise at maximal effort and therefore reach RPE > 17 due to muscle pain, knee issues, coughing, or dyspnea. Thus, the highest VO2 value reached during the exercise phase, averaged over a 10-s interval, was considered VO2peak and expressed in mL/min/kg of lean body mass. Lean body and fat mass were estimated using a body composition analyzer, TANITA bioelectrical impedance analysis device (model: TANITA BC-418; TANITA Corporation, Tokyo, Japan) immediately before the CPET, while participants were in a rested, hydrated condition. We chose normalization by lean body mass to better reflect an individual’s physiological capacity and metabolic efficiency, as it accounts for the oxygen-consuming, metabolically active tissue (muscle) and excludes non-metabolic fat mass. 36
MRI acquisition
Data were collected using a 3T Skyra MRI system with a 32-channel head coil. The acquisition protocol included structural MRI, dual-echo pseudo-continuous arterial spin labeling (pCASL) for simultaneous acquisition of perfusion and blood oxygen level dependent (BOLD) signals and a blood magnetization map (M0) for perfusion quantification. pCASL data were acquired with a voxel resolution of 3.4375 × 3.4375 × 7 mm3, TR/TE1/TE2/alpha: 4000/10/30 ms/90°, labeling duration of 1517 ms with a post-labeling delay (PLD) of 1300 ms. The M0 acquisition had identical parameters but with a TR of 10 s, to ensure a fully recovered magnetization. A 3D multi-echo gradient echo (ME-GRE; TR/TE1/TE2/TE3/TE4/flip angle = 20/6.92/13.45/19.28/26.51 ms/9°, 0.7 × 0.7 × 1.4 mm3 voxel size) phase and magnitude data were acquired for all the coils separately for the reconstruction of QSM maps. Flow compensation of the first TE was completed by nulling the gradient moment to ensure minimizing flow artifact effects for the venous mapping. 37
As the MRI data were acquired within two sub-sessions to allow the participant to take off the mask for the rest of the data acquisition, two T1-weighted acquisitions were collected to ensure registration accuracy of the pCASL slab. One lower resolution acquisition was performed right before pCASL while the participant had the mask on, and a higher resolution acquisition was performed during the second sub-session, without the mask. The low resolution T1-weighted acquisition was acquired using a magnetization prepared rapid gradient echo (MPRAGE) sequence with TR/TE/Flip angle = 15/3.81 ms/25° with a 1.5 mm isotropic resolution. The high-resolution T1-weighted structural images were also acquired with an MPRAGE sequence, with TR/TE/Flip angle = 2300/2.32 ms/8° with a 0.9 mm isotropic resolution.
Respiratory manipulation
The RespirAct™ system (RespirAct™; Thornhill Research, Toronto, Canada) was used during the breathing manipulation to target specific end-tidal partial pressures of CO2 and O2. RespirAct® RA-MR™ single-use subject masks were used for breathing manipulations in the MRI scanner. The masks were fixed on the participant’s face using transparent adhesive tape to prevent leakage. The top part of the masks is designed to fit inside the nose region of the 32 channel SIEMENS head coil without any discomfort. Three conditions were sequentially targeted for 2 min, preceded and followed by 2 min of room air inhalation: a hypercapnic condition (targeting 5 mmHg above baseline for end-tidal CO2), an isocapnic hyperoxic condition (targeting 150 mmHg above baseline for end-tidal O2), and a combined hyperoxia and hypercapnia condition (targeting 5 mmHg above baseline for end-tidal CO2 and 150 mmHg for end-tidal O2). A rebreathing face mask was used to deliver gases to the participants. The system delivered gas at a flow rate of 20 L/min, while the concentrations of exhaled CO2 and O2 were monitored. On their first study visit, a familiarization session was done with a 2-min hypercapnic manipulation to ensure participant comfort during the MRI. Breathing discomfort was assessed using the Banzett dyspnea scale and those with a score >5 were not invited to continue in the study (
Respiratory data analysis
CO2 and O2 were sampled continuously throughout the breathing manipulation by the RespirAct, which also generates a time series of end-tidal CO2 and O2 partial pressures. The end-tidal partial pressure is used as a proxy for arterial gas concentrations. 38 To combine these values with the MRI data, a MATLAB script was employed to smooth the data, remove outliers, and resize the data to match the durations of the BOLD and ASL signals.
Imaging data processing
T1 processing
All structural images underwent preprocessing with the Brain Extraction Tool (BET) in FSL to remove the skull. Subsequently, FSL’s FAST was employed to segment the structural images into gray matter (GM), white matter (WM), and cerebrospinal fluid (CSF).
Preprocessing of BOLD and pCASL
Both ASL and BOLD datasets were corrected for motion using MCFLIRT from FSL. Then, a mask was created using the motion corrected BOLD images and BET, and applied to both echoes to remove the scalp. The first echo of the dual-echo pCASL acquisition was used to compute the perfusion-weighted time-series, from which CBF was estimated using surround subtraction. Volumes that contained voxels with intensity values outside of three standard deviations from the mean were flagged. Then, volumes with more than 50% of voxels identified as outliers were excluded from further analysis, to ensure adequate tSNR. 39 The M0 image underwent skull stripping via BET and was utilized for calibrating the perfusion image. The second echo was used to extract the BOLD-weighted signal via surround addition, reflecting changes in blood oxygenation.
CBF maps using pCASL data
Perfusion was quantified from the preprocessed ASL time-series using FSL’s BASIL for kinetic modeling combined with the M0 acquisition. 39 Finally, partial volume correction was applied using a weighted average taking into account tissue contributions using the segmentation results of GM, WM, and CSF from the T1-weighted data as in Asllani et al. 40
OEF and CMRO2 maps using calibrated fMRI
Calibrated fMRI is a quantitative imaging approach that combines BOLD and ASL measurements during controlled respiratory manipulations (e.g. brief hypercapnia and/or hyperoxia) to calculate OEF using the general calibration model (GCM), based on the deoxyhemoglobin dilution model of the BOLD signal. 36 Following OEF estimation, CMRO2 was calculated using CBF maps and arterial oxygen content using Fick’s principle, as described in Gauthier and Hoge. 41 Calibrated fMRI uses measurements obtained during respiratory manipulation to yield baseline physiological measures (OEF, CBF, CMRO2). CMRO2 was assumed to remain constant during both hypercapnia and hyperoxia across all calculations. 41
QSM data reconstruction
To reconstruct the QSM maps, the magnitude data of the coils were combined by the square root of the sum of squares of each coil data for each TE. 42 The phase data of multi-coil ME-GRE data was combined and unwrapped using the ROMEO toolbox to ensure spatiotemporal coherence and temporal stability. 43 Then, the QSM (X) maps were reconstructed using the first echo data with total-generalized variation (TGV) QSM reconstruction method. 42 The TGV algorithm minimizes the noise propagation through consecutive QSM processing steps and preserving edges between different tissue types, especially the edges between veins and surrounding tissue. The QSM maps were zero-referenced by the mean values of the CSF in ventricles. 44 The veins were extracted using a multi-scale recursive ridge filtering method 45 from the QSM data reconstructed from the first TE data which was the only echo time with flow compensation. OEF is directly related to the concentration of paramagnetic deoxyhemoglobin present in veins using the following formula:
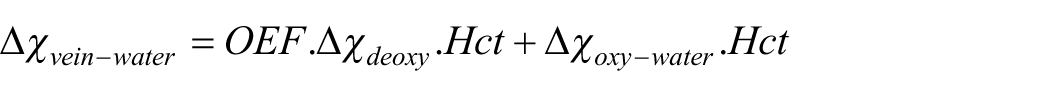
where the
Registration of QSM and calibrated fMRI to MNI space
The pCASL images were registered to MNI space in three separate steps. First, a rigid body registration with nearest neighbor interpolation was conducted using Advanced Normalization Tools (ANTs) 47 to align the pCASL mean images with the low-resolution (1.5 mm) T1-weighted image. Next, the low-resolution T1-weighted images were registered to the high-resolution (0.9 mm) T1 space using a multistage approach that combined rigid, affine, and nonlinear (SyN) transformations with a multi-resolution strategy. In the final step, the high-resolution T1 images were registered to MNI space using the same transformation method applied for the registration from low to high resolution. Finally, the pCASL images were registered to MNI space by applying the three transformation matrices obtained from the previous registration steps, utilizing three warp images. The QSM images were transformed to the high-resolution T1 space and then to the MNI space using the same methods.
After registering all images to MNI space, we extracted regional CBF, OEF, and CMRO2 from the pCASL data using the LPBA40 atlas. 48 Mean CBF values were calculated for each region; however, due to incomplete coverage and low signal quality in some areas, only 44 of the 56 atlas regions were retained for analysis. Regions such as the occipital, lingual, and fusiform cortices were excluded for these reasons. We also included overall gray matter CBF, resulting in a total of 45 regions analyzed. For the QSM data, because the number of veins detected require larger regions to be reliable, mean OEF values were extracted from veins in frontal, temporal, parietal, and occipital lobes using the MNI structural atlas.
Statistical analysis
Normality of regional data distributions was assessed using the Shapiro–Wilk test. For each biomarker—CBF from calibrated fMRI, OEF from both calibrated fMRI and QSM, and CMRO2 from calibrated fMRI—we performed linear regression analyses with VO2peak-LBM as the predictor, while controlling for age and sex as covariates. To facilitate comparison with the existing literature, we additionally performed the same analyses, but with VO2peak-TBM. Mean arterial pressure (MAP) was included as an additional covariate in the CBF analysis only. To account for multiple comparisons, we applied false discovery rate (FDR) correction to the resulting
Comparison of whole-brain OEF between dc-fMRI and QSM
To evaluate agreement between modalities, whole-brain OEF estimates obtained from dc-fMRI were compared with venous OEF derived from QSM. Two complementary approaches were applied: Bland–Altman analysis was used to assess absolute agreement and identify systematic bias between methods, and Pearson correlation analysis was used to examine the linear association between OEF values obtained from the two techniques.
Results
All regional data distributions passed the Shapiro–Wilk test for normality (
CMRO2 and VO2peak-LBM
We observed no significant associations between CMRO2 and VO2peak-LBM in any brain region (
CBF and VO2peak-LBM
We found widespread positive associations between CBF and VO2peak-LBM in whole gray matter, with regional effects in bilateral frontal, temporal, as well as subcortical structures (Figure 2(a)). A full list of regions is provided in Table 2.
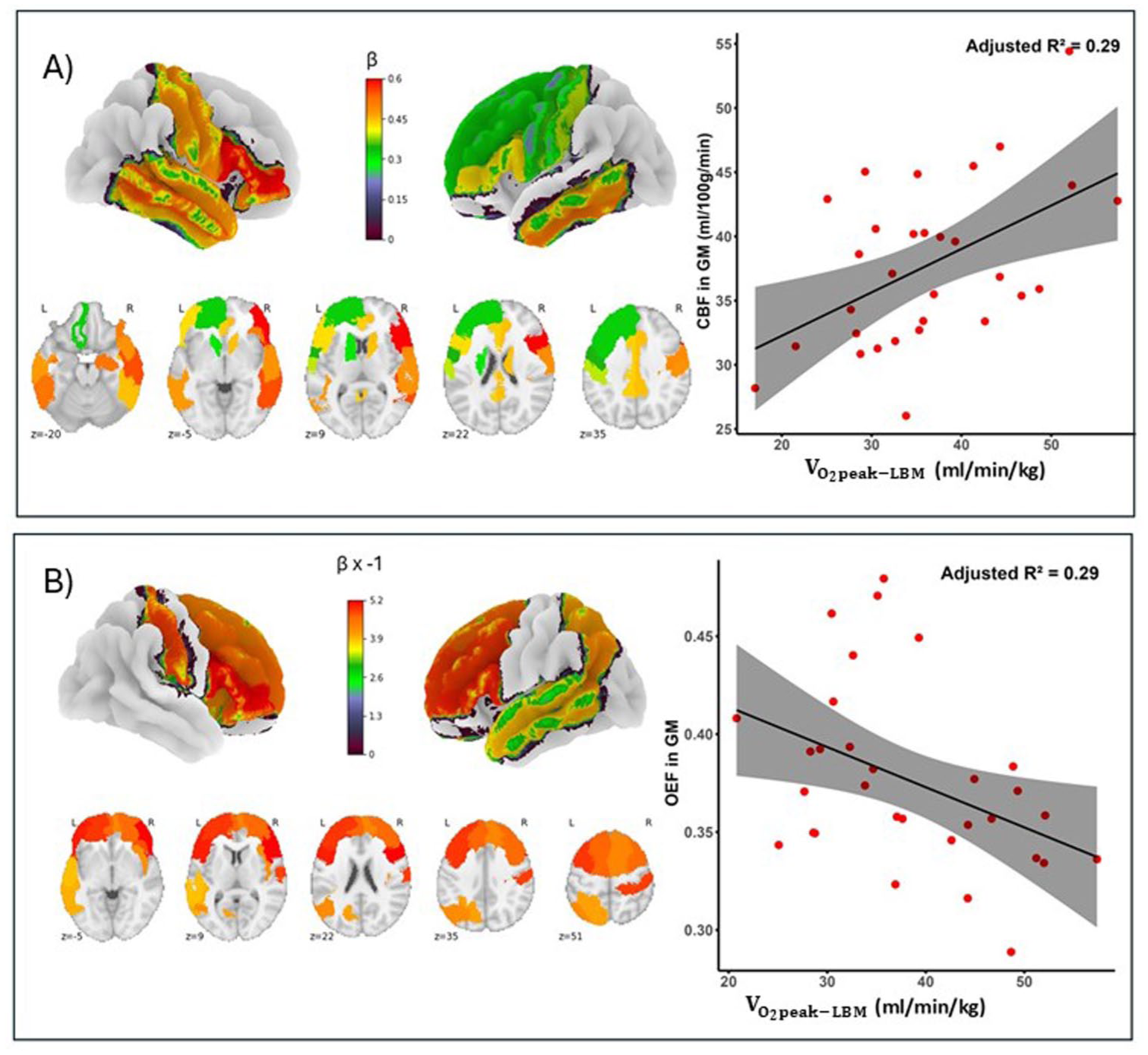
Correlations between VO2peak-LBM and CBF and between VO2peak-LBM and OEF. (a) Positive association between VO2peak-LBM and CBF measured using pCASL. β coefficient maps (multiplied by 10−3 for ease of visualization; left) are overlaid on 3D brain surface, and corresponding scatterplots (right) show linear relationship between CBF in GM and VO2peak-LBM, with 95% confidence intervals shaded. (b) Negative association between VO2peak-LBM and OEF estimated from dc-fMRI. β coefficient maps (multiplied by −1 for ease of visualization; left) are overlaid on 3D brain surface, and corresponding scatterplots (right) show linear relationship between OEF in GM and VO2peak-LBM, with 95% confidence intervals shaded.
Brain regions showing significant associations between CBF and VO2peak-LBM with age, sex, and MAP as covariates.
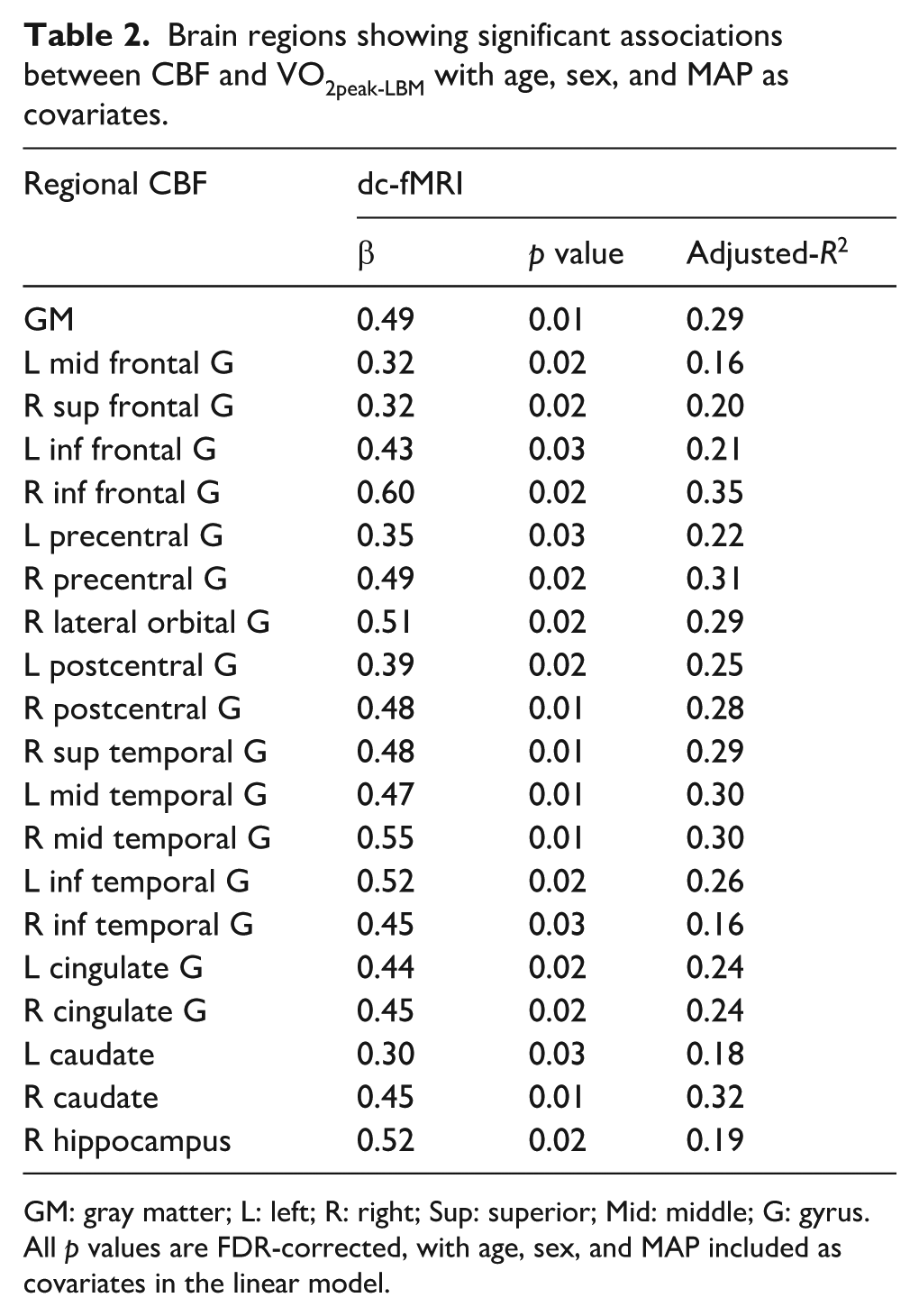
GM: gray matter; L: left; R: right; Sup: superior; Mid: middle; G: gyrus.
All
OEF and VO2peak-LBM
In our dc-fMRI data, we observed a negative association between OEF and VO2peak-LBM across multiple brain regions (
Brain regions showing a significant negative association between VO2peak-LBM and OEF in both dc-fMRI and QSM techniques.
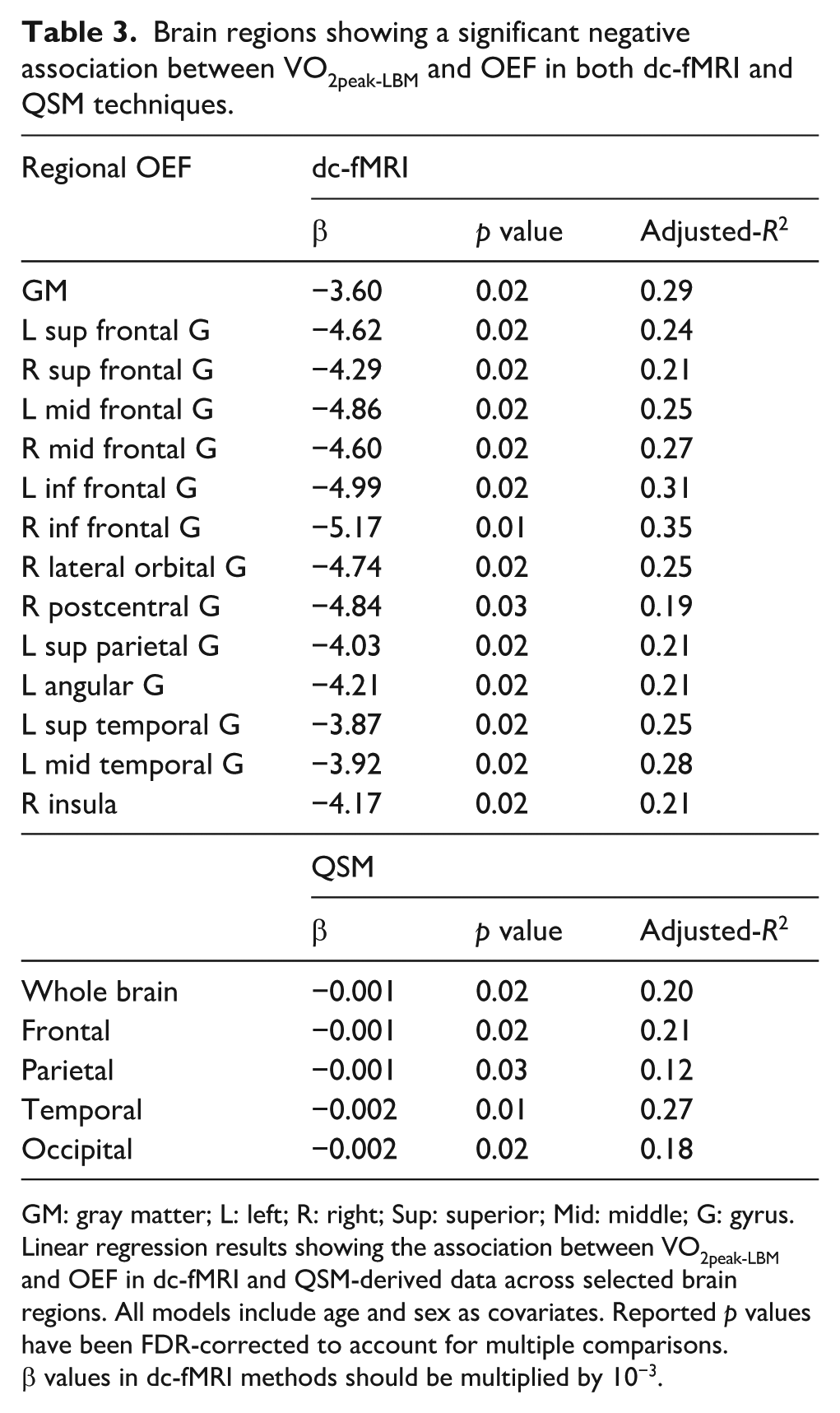
GM: gray matter; L: left; R: right; Sup: superior; Mid: middle; G: gyrus.
Linear regression results showing the association between VO2peak-LBM and OEF in dc-fMRI and QSM-derived data across selected brain regions. All models include age and sex as covariates. Reported
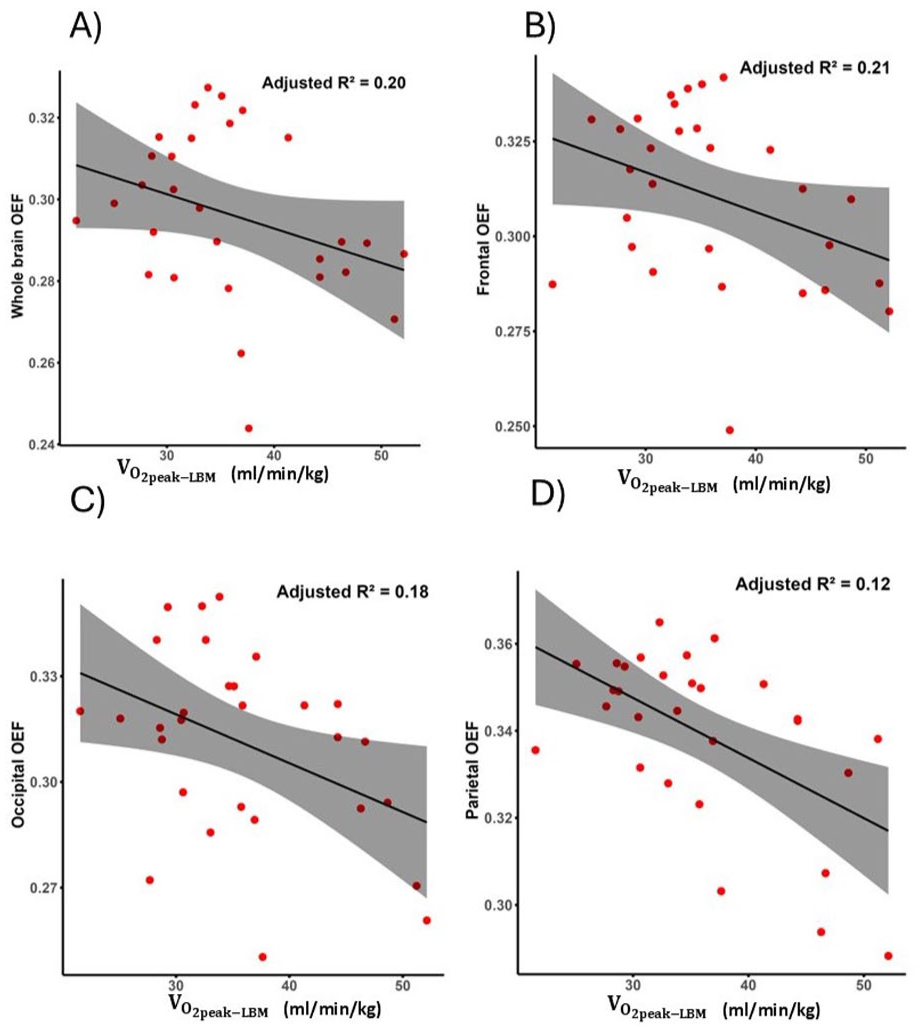
Associations between VO2peak-LBM and regional venous OEF derived from QSM. The scatter plots illustrate significant inverse relationships between VO2peak-LBM and OEF in the whole brain (a), frontal lobe (b), occipital lobe (c), and parietal lobe (d). Shaded areas represent 95% confidence intervals.
To facilitate visualization of these relationships, representative CBF, OEF, and CMRO2 maps are shown for two participants with low and high VO2peak-LBM (29.27 and 48.66 mL/kg/min, respectively) in the Figure S2.
CBF, OEF, CMRO2, and VO2peak-TBM
To facilitate comparison with other studies, we repeated the previous analysis, using VO2peak-TBM (see Supplementary Materials). The direction and statistical significance of CBF and VO2peak-TBM associations remained unchanged (Table S2 and Figure S3). Similarly for the relationship between VO2peak_TBM and OEF, the direction of all relationships remained the same, though some regions were no longer significant (Figures S4 and S5 and Table S3). As observed for VO2peak-LBM, there was no significant association between CMRO2 and VO2peak_TBM.
dc-fMRI OEF versus QSM venous OEF
For the whole brain OEF, dc-fMRI yielded systematically higher OEF than QSM. Mean ± SD values were 0.35 ± 0.05 for dc-fMRI and 0.3 ± 0.02 for QSM. Bland-Altman analysis showed a bias of +0.05 OEF units (
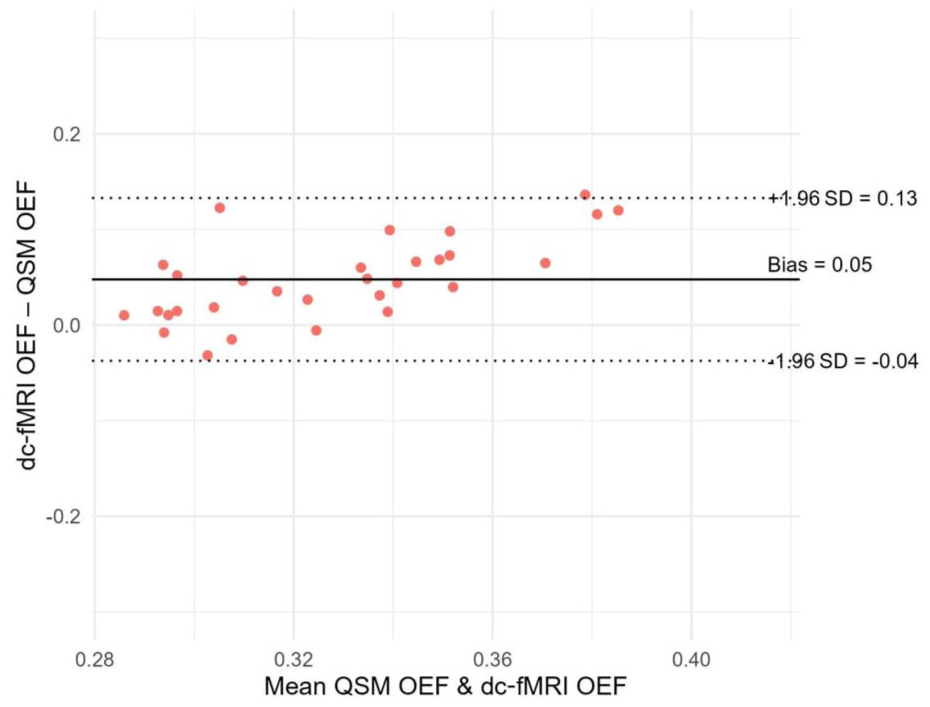
Bland–Altman agreement between GM dual-calibrated BOLD (dc-fMRI) and QSM OEF. Each point represents one participant (
Discussion
In this study, we investigated the relationship between VO2peak-LBM and key physiological brain biomarkers, including vascular (CBF) and metabolic (OEF and CMRO2) measures, in healthy older participants. To our knowledge, this is the first study to examine the association between VO2peak-LBM and regional brain metabolism biomarkers. Our results revealed a positive relationship between VO2peak-LBM and CBF across the whole gray matter, as well as in the frontal, temporal, and subcortical regions, including the hippocampus and caudate. In contrast, we found no significant relationship between VO2peak-LBM and CMRO2 in any brain region. Finally, negative associations between VO2peak-LBM and OEF measured using dc-fMRI were observed in the whole brain, gray matter, frontal and temporal lobes, and insula. With QSM, the negative associations extended to the entire brain and all major lobes, including the frontal, temporal, occipital, and parietal regions. This study demonstrates for the first time that, in healthy adults over 50, cardiorespiratory fitness is linked with higher oxygen availability, likely contributing to protecting the brain against transient ischemic episodes.
Our finding of a positive association between VO2peak and CBF is consistent with reports of greater perfusion in gray matter with higher fitness,16,49,50 but contrasts with some studies reporting null or inverse associations.10,19,51,52 Several factors could account for these differences, including differences in the way fitness was measured. Some studies directly measure VO2max, while others, like this study report a VO2peak-LBM. Here, since most of the participants could not meet the VO2max criteria (RER > 17), we instead report VO2peak. Furthermore, the values reported here are normalized by lean body mass (VO2peak-LBM) whereas many prior studies normalize by total body weight (VO2peak-TBM). Third, demographic composition differs across studies, particularly in sex distribution and age. Our sample included 28% females, whereas the cohort in Intzandt et al. comprised ~66% females, a disparity that matters given evidence for sex-specific cerebrovascular responses to physical activity.53–55
Taken together, these methodological and demographic differences likely contribute to the mixed findings across studies. Nevertheless, our findings align with the exercise-related vascular adaptations, such as increased capillary density—observed in animal models56,57—as well as exercise-induced upregulation of growth factors like brain-derived neurotrophic factor (BDNF) and vascular endothelial growth factor (VEGF), which are known to promote angiogenesis.58,59 However, future studies in larger cohorts are needed to disentangle the parameters that could affect this relationship, including sex, age, fitness level, and the presence of other factors known to affect CBF such as hypertension.
Conversely, we observed no significant relationship between CMRO2 and VO2peak-LBM, suggesting that cerebral metabolic demands remain relatively stable across fitness levels in healthy older adults. While there is a paucity of studies in the literature to contextualize our findings, functional imaging studies using techniques such as functional near-infrared spectroscopy (fNIRS) have indicated that higher fitness may positively influence brain oxygenation during cognitive tasks or exercise, primarily among older adults.60,61 Interestingly, one previous study reported a negative relationship between global CMRO2 and VO2peak. 29 However, participants in that study were generally older and were not selected to be free of hypertension and other vascular risk factors. Furthermore, higher global CMRO2 in that study was also related to fatigability and multi-morbidity, suggesting that there may be a nonlinear relationship whereby CMRO2 may increase as a compensatory mechanism in some conditions. This is, for example, consistent with findings relating higher fatigability to higher CMRO2 in multiple sclerosis. 62 While we did not collect fatigability information in this study, future studies should attempt to disentangle this potentially nonlinear relationship between VO2peak and CMRO2 depending on health status and fatigability.
Our study breaks new ground in documenting for the first time the relationship between OEF and VO2peak-LBM. We observed a negative relationship between VO2peak-LBM and cerebral OEF, suggesting that healthy individuals with higher cardiovascular fitness achieve greater cerebral oxygen delivery without increasing oxygen utilization, consistent with the absence of a corresponding relationship with CMRO2. To our knowledge, this is the first study to demonstrate this relationship, which we confirmed using two independent imaging techniques. This lower OEF, combined with unchanged CMRO2 and elevated CBF is consistent with a greater delivery of O2 to tissue, but since the same absolute amount of O2 is extracted from arterial blood, more O2 remains in venous blood. This improved vascular–metabolic coupling may be beneficial by ensuring that oxygen supply remains adequate even when the need for O2 transiently increases, thereby lowering the likelihood of transient hypoxic episodes. Clinically, elevated OEF has been associated with tissue at greater risk of further ischemic damage after stroke, 28 highlighting the relevance of optimal oxygen delivery for maintaining brain health.
Exploring the relationships between VO2peak and these cerebral vascular and metabolic biomarkers is clinically valuable, as changes in OEF and CMRO2 may offer sensitive indicators of mitochondrial dysfunction. Mitochondrial impairment is characterized by a reduced ability to utilize oxygen, 63 leading to concurrent reductions in CMRO2 and OEF if cerebral blood flow remains relatively stable. Conversely, elevated OEF values typically reflect vascular impairment, such as increased vessel stiffness or reduced endothelial function, often observed with aging or cardiovascular risk factors.64,65 Thus, understanding whether higher aerobic fitness associates with alterations in these biomarkers in healthy adults could shed light on the underlying mechanisms through which exercise interventions confer brain health benefits. These results could inform targeted preventive strategies in populations vulnerable to metabolic and cerebrovascular dysfunction.
By applying both calibrated fMRI and QSM, our study demonstrates consistent associations between VO2peak-LBM and OEF, reinforcing the robustness of our findings. Importantly, we found that QSM-derived OEF values are systematically lower by ~15% compared to dc-fMRI, with no evidence of proportional bias across the range. These lower values could be due to the greater sensitivity of QSM to partial volume effects and those small veins that cannot be detected with the vein segmentation technique used here. In addition, it has been shown that even in the presence of various flow compensation schemes, there is still artificial phase signal in the veins due to blood flow (even very slow like in the pial veins) and OEF is underestimated. 37 Additionally, the effect of vessel orientation on QSM data reconstruction could also bias estimates. It has been shown that veins oriented near the magic angle of the dipole kernel are more under sampled and as a result, the QSM reconstruction algorithm fails to reconstruct their complete susceptibility information resulting in OEF underestimation in such vessels. 46 Lack of proportional bias and relatively narrow limits shows that QSM is reliably tracking the same physiological effect as dc-fMRI OEF. Hence, QSM emerged as a practical alternative in settings where calibrated fMRI may be difficult to implement. The latter, while powerful, requires respiratory manipulations which can be uncomfortable and rely on expensive specialized equipment, as well as prolonged scan times, which can pose challenges—especially for older adults or clinical populations. There are multiple MRI-based OEF measurement techniques that do not require respiratory manipulations and specialized equipment such as T2-Relaxation-Under-Spin-Tagging (TRUST) MRI 66 and quantitative blood oxygen level dependent (qBOLD) MRI, 67 though these require specific pulse sequences that are not available on the commercial MRI scanners. One of the advantages of QSM-based techniques is that they rely on the GRE sequence, available on all MRI scanners, and benefit from a relatively short acquisition time, making them particularly well-suited for broader clinical applications.
As an initial exploration of fitness-related brain metabolic patterns, our study lays the groundwork for future investigations in patient populations, where understanding these relationships could inform more personalized and targeted interventions.
Limitations
The primary limitation of this study is the small sample size, which resulted from the difficulty of finding subjects who met our strict inclusion and exclusion criteria, and possibly from participant attrition due to discomfort experienced during the hypercapnia challenge. This limitation may reduce the generalizability of our findings. Future studies with larger cohorts and a wider range of health status are needed to further support our results.
The second limitation of this study is that lean body mass was derived using a bioelectrical impedance analysis system rather than dual-energy X-ray absorptiometry which is considered the reference method. 68 Previous studies on body composition analysis reported systematic underestimation of lean mass measurement using different bioelectrical impedance devices.69,70 Future studies should validate our findings with dual-energy X-ray absorptiometry. Importantly for the validity of our findings however, our results remained largely similar, though with reduced statistical significance in some cases, when using VO2peak-TBM. This is expected however, since normalizing by total body mass introduces additional physiological variability, that is, unrelated to the metabolic demand specific to the effort test. 36
ASL suffers inherently from a low tSNR, which can be exacerbated by prolonged arterial transit time in populations with vascular risk. A single PLD, as employed here, therefore makes ASL vulnerable to underestimation of CBF in individuals with delayed transit time. Future studies should use multi-PLD sequences to improve CBF quantification and use background suppression to improve SNR. Background suppression was not used here to maintain BOLD SNR in our dual-echo acquisition for OEF and CMRO2 quantification.
The accuracy of venous OEF values from QSM depends critically on the quality of the vein segmentation and on partial-volume effects—particularly in veins whose diameter is significantly smaller than the image resolution—which tend to cause systematic underestimation. Thus, future studies should develop vein segmentation techniques that are more sensitive to the veins with smaller diameter will help to decrease the effect of partial volumes. Furthermore, future studies should use susceptibility reconstruction techniques that better delineate between veins and surrounding tissue, such as X-separation techniques, which facilitates vein segmentation from paramagnetic and diamagnetic susceptibility maps.
Conclusion
This study provides new evidence that higher cardiorespiratory fitness, measured by VO2peak (normalized by lean body mass), supports better oxygen delivery to the brain in healthy adults. Participants with greater fitness had increased blood flow to the brain, enabling enhanced oxygen availability without raising the brain’s overall metabolic demand or the need for greater oxygen extraction. Additionally, we showed that QSM, an imaging method not requiring respiratory challenges, provides similar estimates of brain oxygen use compared to traditional calibrated MRI, making it a useful alternative, especially in populations who cannot comfortably tolerate breathing challenges. Together, these results offer new insights into the neurovascular effects of fitness and provide a foundation for future studies exploring these relationships in aging and clinical populations, where targeted interventions may benefit from individualized assessments of brain oxygenation and perfusion.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251413924 – Supplemental material for Greater cardiorespiratory fitness is associated with higher cerebral blood flow and lower oxygen extraction fraction in healthy older adults
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251413924 for Greater cardiorespiratory fitness is associated with higher cerebral blood flow and lower oxygen extraction fraction in healthy older adults by Safa Sanami, Ali Rezaei, Stéfanie A Tremblay, Zacharie Potvin-Jutras, Dalia Sabra, Brittany Intzandt, Christine Gagnon, Amélie Mainville-Berthiaume, Lindsay Wright, Mathieu Gayda, Josep Iglesies-Grau, Anil Nigam, Louis Bherer and Claudine J Gauthier in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
We would like to thank everyone who contributed to this project: Paule Samson, Thomas Vincent, Julie Lalongé, Hakima Benhalima, Milla Shakleva, Victoria D’Amours, Agathe Godet, Stephanie Beram, Roni Zaks, Robert Hovey, Alexandre Bailey, Catherina Medeiros, Amélie Mainville-Berthiaume, and Zineb Rouabah. Thank you also to the laboratories of Dr. Louis Bherer and Dr. Mathieu Gayda. Lastly, we would like to acknowledge our research participants without whom none of this would have been possible.
Author contributions
Safa Sanami and Ali Rezaei contributed equally to this work, performing the main analysis, data acquisition, and manuscript writing. Stéfanie A Tremblay, Zacharie Potvin-Jutras, and Dalia Sabra contributed to data acquisition, manuscript review, and editing. Brittany Intzandt, Christine Gagnon, Amélie Mainville-Berthiaume, Lindsay Wright, Mathieu Gayda, Josep Iglesies-Grau, and Anil Nigam contributed to manuscript review and editing. Louis Bherer and Claudine J Gauthier contributed significantly to conceptualization, main methodology, manuscript editing, and review.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding awarded to Claudine J Gauthier from the Natural Sciences and Engineering Research Council of Canada (NSERC Discovery Grant: RGPIN-2015-04665; 2024-06455), Fonds de recherche du Québec (FRQ 5232), the Heart and Stroke Foundation of Canada (G-17-0018336), the Heart and Stroke Foundation New Investigator Award, the Henry JM Barnett Scholarship and the Michal and Renata Hornstein Chair in Cardiovascular Imaging. Additional support was provided by the Vascular Training Platform (VAST; to Ali Rezaei), the Canadian Institutes of Health Research (FRN: 175862, to Stéfanie A Tremblay), the Heart and Stroke Foundation of Canada and Brain Canada (to Zacharie Potvin-Jutras), the Mirella and Lino Saputo research chair in cardiovascular health and the prevention of cognitive decline (to Louis Bherer) and the Alzheimer Society Research Program Postdoctoral Award (to Brittany Intzandt).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
