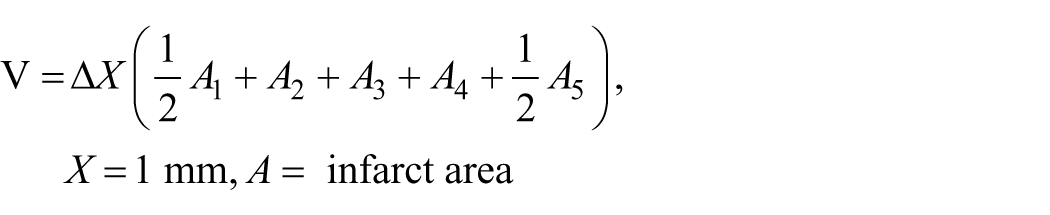Abstract
We investigated whether basement membrane (BM) laminins influence regional differences in the vasculature by performing single-cell RNA sequencing on cerebral blood vessels from mice lacking the major vascular laminins in endothelial and smooth muscle BMs, laminin α4 (Lama4 −/− ) and laminin α5 (Tek-cre:Lama5 −/− ), and wild-type littermates. Our dataset expands existing cerebral vascular transcriptomic profiles and reveals that Lama4 −/− endothelial cells exhibit increased arterial marker expression and reduced postcapillary venule identity. In vitro and in vivo studies indicated that compensatory upregulation of laminin α5 in Lama4 −/− vessels enhances expression of junctional proteins (Ocln, Cldn5) and promotes vessel contractility via increased expression of contractile molecules in mural cells. Additionally, loss of Lama4 upregulated expression of large artery markers (Gja4, Dll4, Tgfb2) and resulted in elevated autotaxin (Enpp2) levels, a key enzyme in lysophosphatidic acid production implicated in stroke. Accordingly, Lama4 −/− mice exhibited worsened stroke outcomes, driven not by immune infiltration or junctional defects, but by increased vascular permeability likely mediated by autotaxin and/or activation of resident myeloid cells. Our data suggest that laminin α4/α5 ratios in vascular BMs affect functional zonation between arterioles, capillaries and postcapillary venules by modulating metabolic pathways in endothelial and mural cells, and indirectly influencing resident myeloid cells.
Introduction
Vascular zonation refers to spatial differences in structure, function and molecular characteristics along the arteriovenous axis within an organ. 1 In the brain, vascular zonation shapes blood–brain barrier (BBB) integrity, nutrient and metabolite exchange and leukocyte entry points during neuroinflammation – factors that contribute to stroke severity and dementia risk.
Arterial identity is largely established by flow and VEGF-induced Notch signalling, while venous identity is driven by COUP–TFII, which represses arterial genes (reviewed in Trimm and Red-Horse 2 ). Beyond these developmental cues, vascular functional zonation is also reflected in the local cellular and extracellular environment. In the brain, the neurovascular unit (NVU) – a tightly interconnected system of blood vessels and neural cells that is essential for central nervous system (CNS) homeostasis 3 – differs between arterial and venous regions. In addition to endothelial heterogeneity, in vivo imaging shows that mural cells, including smooth muscle cells and pericytes, display zone-specific morphologies and PDGF receptor-β/smooth muscle actin expression patterns.4,5
All NVU components are further embedded in specialized extracellular matrices (ECMs), with basement membranes (BMs) being especially abundant due to the brain’s dense vasculature. 6 These BMs form structural and functional barriers between endothelial and mural cells, as well as between the vasculature and the CNS parenchyma, defining perivascular spaces that harbour long-lived myeloid and fibroblastic cells6–8 that are enriched around arteries.9,10 Although limited, studies examining perivascular macrophage populations in hypertension models suggest that these cells contribute to BBB maintenance 9 and are rapidly reprogrammed in pathological situations, like stroke, to an inflammatory phenotype that exacerbates neurological deficits. 11 Together, these findings indicate that resident myeloid populations contribute to arterial dysfunction and vascular–neuronal communication. However, the mechanisms governing their arterial localization and long-term maintenance remain poorly understood.
Like all BMs, those of cerebral blood vessels consist of a collagen type IV and laminin network, interconnected by proteoglycans, such as agrin and perlecan, and nidogens. 7 Both collagen IV and laminin are multi-isoform families, whose specific combinations create biochemically and functionally distinct BMs across different organs and vascular segments. 7 Laminins are heterotrimers composed of α, β and γ chains. In endothelial BMs, laminin-411 (α4β1γ1) and laminin-511 (α5β1γ1) are variably expressed along the vascular tree, forming a scaffold beneath endothelial cells and surrounding pericytes. Laminin α4 and α5 chains are also present in vascular smooth muscle cell BMs, typically in combination with laminin β2 and γ1. 12 Beyond the vessel wall, the endothelial tube with its associated pericytes and perivascular cells is further encased by an astroglial layer and a parenchymal BM, which demarcates the boundary with the brain parenchyma and contains laminins 211 and 111. 6
The molecular heterogeneity of vascular and parenchymal BMs in both development and adulthood suggests roles beyond structural support or barrier formation. In particular, laminin α4, the first laminin expressed in the vascular system and broadly distributed, has been implicated in vascular tube formation and branching.13–15 By contrast, laminin α5 expression begins perinatally and predominates in arterial endothelial and smooth muscle BMs, 6 with much lower levels in veins. Its expression decreases at postcapillary venules – the primary sites of leukocyte extravasation.16–18 Laminin α5 also stabilizes VE-cadherin at endothelial junctions, a process essential for resistance artery responses to shear stress. 19 Together, these findings suggest that distinct laminin isoforms play specialized roles along the vascular tree, particularly within the microvasculature. We therefore hypothesize that the vascular laminins contribute to the functional zonation of cerebral vessels.
We investigated this possibility by performing single-cell RNA sequencing (scRNA-seq) of cortical blood vessels from mice lacking laminin α4 (Lama4 −/− ) or laminin α5 (Tek-cre:Lama5 −/− ), major vascular BM components and wild-type littermates. In Lama4 −/− mice, compensatory upregulation of laminin α5 occurs.14,16 In vitro and in vivo analyses showed that laminin α5 enhances the expression of junctional proteins (Ocln, Cldn5), suppresses markers of postcapillary venules and endothelial activation (Vcam1, Icam1), and promotes contractile gene expression in mural cells – altogether contributing to an artery-like phenotype. Conversely, laminin α4 suppresses large artery endothelial markers (Gja4, Dll4, Edn1, Tgfb2), supports the quiescent state of resident myeloid cells and represses Enpp2 (autotaxin), a key enzyme in lysophosphatidic acid (LPA) production implicated in stroke pathology.20,21 Consistent with this, Lama4 −/− mice exhibited more severe outcomes after transient middle cerebral artery occlusion (tMCAO) compared to wild-type and an endothelial-specific Lama5 knockout (Tek-cre:Lama5 −/− ), a phenotype independent of immune cell infiltration and instead linked to elevated autotaxin and potentially also activated resident myeloid cells.
Together, our data indicate that the laminin α4/α5 balance in vascular BMs influences cerebral vessel zonation – particularly at arteriole-capillary and capillary-postcapillary venule transitions – by modulating endothelial and mural cell metabolism and indirectly affecting resident immune cell states.
Material and methods
Mice
Age- (8–14 weeks) and sex-matched Lama4 −/− 14 and Tek-Cre:Lama5 −/− 22 mice were used. As similar results were obtained for Lama4+/+ and Lama5 floxed/floxed mice, data were pooled and referred to as wild-type (WT) controls. Animal experiments were approved by the Landesamt für Verbraucherschutz und Ernährung, Nordrhein-Westfalen (LAVE; license numbers: 81-02.04.2023.A130 and 84-02.05.50.17.004), carried out according to German Animal Welfare Act guidelines and reported according to ARRIVE guidelines.
Single cell RNA sequencing
Mice were transcardially perfused with ice-cold PBS, brains removed, minced in ice cold DMEM (Sigma–Aldrich) and digested for 60 min at 37 °C in 30 U/ml papain (LK003153; Worthington Biochemical), 40 µg/ml DNase type IV (LS006331; Worthington Biochemical) and 0.125 mg/ml Liberase TM (5401119001; Sigma–Aldrich). Myelin was removed by 10 min 1000g centrifugation through 22% BSA. To maximize the yield of all cells associated with cerebral vessels, including those present in low numbers or embedded within the ECM, and to optimize cell assignment and reduce batch effects, brains from three WT and three Lama4 −/− mice were pooled/sample.
Single cells were encapsulated in gel bead emulsion using the 10× Chromium system (10× genomics). Libraries were prepared according to manufacturer’s instructions using Chromium Single Cell 3′ Library and Gel Bead Kit v3 (10× genomics) and sequenced in Illumina NextSeq 500 using High Output Kit v2.5 (150 cycles; Illumina).
The Cellranger (10× genomics) pipeline was followed to demultiplex the sequencing data, to align it to the mouse reference genome (mm10), to count the reads, and to aggregate the resulting cell-gene matrices according to sample replicates. Subsequent data analysis was performed with R package Seurat (version 3.1). Raw counts were normalized with SCTransform 23 and samples were integrated (reciprocal PCA) to correct for the batch effect. 24 Dimensional reduction, unsupervised clustering, visualization and differential expression analysis was performed using Seurat. Gene set enrichment analysis (GSEA) was performed using GSEA and Molecular signature database (MSigDB). 25 RNA-seq data generated for this paper have been deposited in the NCBI GEO (GSE308748).
Immunofluorescence
Brain preparations and immunofluorescence staining were as previously described. 6 Primary and secondary antibodies employed are in Supplementary Tables 1 and 2. Sections were analyzed using Zeiss confocal laser scanning (LSM 900 and LSM800) and Spinning-disk confocal microscopes. Images were analyzed using Volocity 6.3 (Perkin-Elmer), Zen Blue (Zeiss) and Imaris 10 (Oxford Instruments). Mean fluorescence intensity was quantified in ZEN Blue from maximum intensity projections after applying a manually adjusted threshold to eliminate background.
Automated pericyte morphology and coverage quantification was performed in ZEN Blue. After Gaussian smoothing and automatic three-sigma thresholding of raw images, pericyte and vessel areas were segmented to calculate pericyte coverage on the vascular surface. Outliers were manually curated post-batch analysis.
For 3D reconstruction of pericytes, vessel surfaces were rendered in Imaris 10 and pericytes were manually traced and colour-coded based on morphology.
Western blots
Brains were homogenized in RIPA buffer with protease inhibitors. Lysates (50 μg protein/lane) were separated under reducing conditions, except for PECAM-1 which was analyzed under non-reducing conditions. After transfer to nitrocellulose, membranes were probed with antibodies listed in Supplementary Tables 1 and 2. Band intensities were quantified using ImageJ; due to lower vessel density in Lama4 −/− brains, signal intensities of VCAM-1, claudin-5, prostacyclin synthetase and SMA were normalized to the PECAM-1 signal; autotaxin signal intensity was normalized to tubulin. GAPDH and tubulin served as loading controls.
Flow cytometry
Mice were transcardially perfused with ice-cold PBS, brains removed, minced in ice cold DMEM and digested at 37 °C for 60 min using papain (100 units/brain) and DNase (0.1 mg/ml). Myelin was removed by 10 min 1000g centrifugation through isotonic Percoll (Amersham). Endothelial cells were stained with the antibodies listed in Supplementary Table 1 and analyzed using a FACSCelesta (Becton Dickinson) and FlowJo.
Transient middle cerebral artery occlusion (tMCAO)
Transient focal ischaemia using the intraluminal filament model was performed on 12-week male mice. 26 The left middle cerebral artery was occluded for 60 min and reperfused for 24 h; ipsilateral (IL) brain hemispheres represent stroke samples, contralateral (CL) hemispheres represent non-stroke controls. Motoric scores were performed anonymously as described. 27 Mice exhibiting no deficit (score 0) or with rolling to the paretic side (score 4) were excluded.
For analysis of leukocytes after stroke, IL and CL hemispheres were dissociated, subjected to a discontinuous density gradient centrifugation using Percoll and total cells were counted. Leukocytes were stained with the antibodies listed in Supplementary Table 1 and acquired using a FACSCalibur (Becton Dickinson) and analyzed using FlowJo.
Infarct volume measurement
Infarct volumes were measured using 5 × 1 mm coronal sections incubated in 2% 2,3,5-triphenyltetrazolium chloride (TTC) for 20 min at 37 °C. Sections were fixed in 2% PFA and photographed. Infarct volume (V) was calculated as follows:
Swelling (S) of IL hemispheres was calculated and expressed as percent of the CL hemisphere:
S =
The oedema-corrected infarct volume (Vi) was: Vi =
Bulk RNA sequencing
Mice were perfused with PBS and brains were isolated. After removing the olfactory bulb and cerebellum, the remaining IL and CL hemispheres were homogenized in TRIzol (Life Technologies). Brains were pooled (two per group). Differential centrifugation was performed to remove myelin and RNA was extracted via chloroform/phenol extraction and purified with DNase and RNeasy columns (Qiagen) for bulk RNA sequencing (Illumina NextSeq500).
FASTQ files were generated using bcl2fastq (version v2.17.1.14; Illumina). Subsequent analysis was performed using the Galaxy program. Quality control was performed using FASTQC (version 0.72+galaxy1). The last 10 low-quality bases were trimmed away. The trimmed reads (70 bp) were mapped with RNA STAR (version 2.6.0-b1) to the mouse reference genome (mm10) with gencode vM22 gene annotation. Reads were counted with featureCounts tool (version 1.6.4). The raw counts were normalized using DESeq2 package in R program. Differentially expressed genes were detected using unadjusted p-value (Wald’s test) and log2 fold change. Functional analysis was performed using GSEA. Normalized bulk RNA-seq data (GSE309181) was deconvoluted using SCADEN (version 0.9.1) using existing single cell profiles. 28
LPA measurements
MALDI-2-MS imaging was performed on a modified timsTOF fleX mass spectrometer (Bruker). 29 A sublimated 2,5-dihydroxyacetophenone matrix was used to acquire positive-ion mode images (200–2000 m/z, 25 µm pixel size). Data was processed in SCiLS Lab (Bruker), with lipid assignments based on accurate mass (5 ppm window).
Statistics
Analyses used GraphPad Prism 9. Tests were selected based on normality (assessed by Q–Q plots and Shapiro–Wilk test). For the comparison of two groups, a t-test was applied if the data were normally distributed, and the Mann–Whitney test was applied if the data were not normally distributed (non-parametric). For comparisons involving multiple groups, one-way ANOVA or Kruskal–Wallis tests were used. The specific test used for each comparison is indicated in the corresponding figure legend. Significance was defined as p < 0.05. Data are means ± SD.
Results
Single cell RNA sequencing of Lama4 −/− cerebral vessels
To comprehensively investigate differences between Lama4 −/− and littermate WT cerebral blood vessels, single cell RNA sequencing (scRNA-seq) was performed on endothelial and associated mural and immune cell enriched samples isolated from the cerebral cortex of adult male animals; neuron contamination was reduced to 0.007% (130 transcripts) by depleting myelin positive populations 30 (sFigure 1(A)). 18,828 single cell transcriptomes were clustered and annotated based on previous studies31,32 (sFigure 1(B)). UMAP plots show the distribution of cell types identified, confirming high proportions of endothelial (47%), perivascular (15%) and myeloid cells including microglia (29%; sFigure 1(C)), which did not differ significantly between WT and Lama4 −/− samples. Additional clustering characteristics are shown in sFigure 1(D) and (E). This revealed low expression of most laminins in mature WT tissues and the absence of ECM expression in immune cells (sFigure 1(F) and (G)). Supplementary Files 1 and 2 contain the complete gene lists (GSE308748); full names of genes occur in Supplementary Table 3.
Endothelial laminins affect expression of arterial markers
Analysis of gene profiles of endothelial and mural cells showed a similar trend, with no changes from WT in absolute numbers of cells in Lama4 −/− samples but with a shift towards higher expression of genes that typically associate with arterial endothelium (Gja4, Edn1, DII4, Tgfb2, Gskn3 and Klf2; Figure 1(a)–(c) and sFigure 2(A) and (B)) and more contractile (Acta2, Cnn1, Tagln, Fbln5) or vasoactive mural cells (Ptgis; Figure 1(d)–(g) and sFigure 2(C) and (D)). Endothelial clustering revealed six groups (Figure 1(a) and sFigure 2(A)), with a slightly higher proportion of cells expressing a large artery gene profile (Gja4, Tgfb2, Edn1, DII4) and capillaries with high junctional tightness (Cldn5, Ocln) in Lama4 −/− compared to WT (Figure 1(b)), and a reduction in the proportion of cells expressing genes associated with postcapillary venules/activated endothelial cells (Icam1, Vcam1; Figure 1(c)). In addition, high expression of Enpp2 was observed in Lama4 −/− endothelial and mural cells (Figure 1(b)), which encodes autotaxin, required for lysophosphatidic acid (LPA) generation, and has been implicated in stroke.20,33 Tissue-resident macrophages (Figure 1(h)–(j) and sFigure 2(E) and (F)) were also reduced in Lama4 −/− mice, but had higher proportions of MHCII+ and phagocytotic populations (Lyve1 low , Mrc1, Wfdc17, Cd68; Figure 1(j)), despite absence of ECM expression by these cells (sFigure 1(F) and (G)). Additionally, high Ttr (8.45-fold) expression in Lama4 −/− endothelium and mural cells was observed (Figure 1(b) and (e)), which encodes transthyretin that binds T4 and T3 in the circulation. The higher expression of Enpp2 was confirmed using various methods (see below). By contrast, RNA-Scope showed that Ttr was highly expressed only in choroid plexus epithelium and was not detectable by Western blotting of brain samples lacking the choroid plexus. qPCR of isolated cerebral vessels could further not confirm its elevated expression in Lama4 −/− samples (data not shown). We therefore concluded that there was some contamination of the Lama4 −/− cerebral vessels sample by ambient RNA, probably from choroid plexus epithelium RNA; thus, further analysis of Ttr/transthyretin was discontinued.
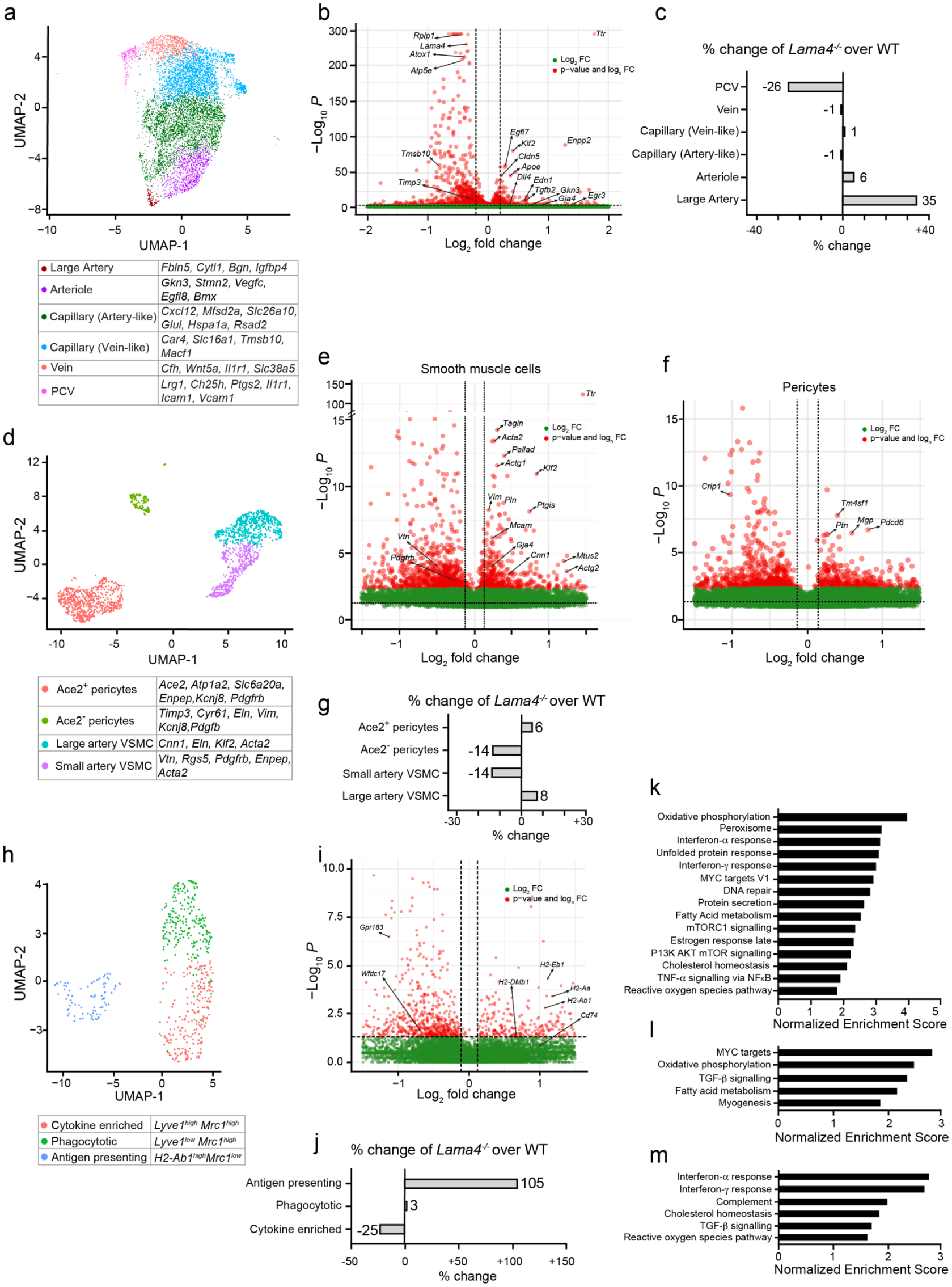
scRNA-seq data for endothelial cell populations (a–c), smooth muscle cells and pericytes (d–g) and resident immune cells (h–j) in enriched cerebral vessel samples from Lama4 −/− and WT littermates. UMAPs identified (a) six endothelial (d) two smooth muscle and two pericytes and (h) three myeloid cell populations. Different cells are colour coded based on their cluster affiliations shown in associated tables. Volcano plots of genes upregulated and downregulated in Lama4 −/− samples compared to WT littermates in (b) endothelial cells, (e) smooth muscle cells, (f) pericytes and (i) myeloid cells; (c, g, j) corresponding graphs of percentage changes in Lama4 −/− over WT littermates based on transcriptomic profiles. Unbiased GSEA analyses showing enrichment of pathways in Lama4 −/− samples compared to WT littermates in (k) endothelial cells, (l) all mural cells and (m) resident myeloid cells.
Unbiased gene set enrichment analysis (GSEA) revealed enrichment of metabolic gene sets, including oxidative phosphorylation and fatty acid metabolism, in Lama4 −/− endothelial and mural cells (Figure 1(k) and (l)), as well as enhanced proteolysis in Lama4 −/− endothelial cells (Figure 1(k)) and, in mural cells, genes associated with myogenesis and TGFβ signalling (Figure 1(l)). Similar analyses for tissue-resident macrophages revealed enrichment of IFNα and IFNγ responses, cholesterol homeostasis and TGFβ signalling (Figure 1(m)).
Compensatory Lama5 upregulation enhances junctional tightness and mesh-like pericyte phenotype
As Lama4 −/− mice show compensatory enhanced laminin α5 deposition in endothelial BMs,16,34 scRNA-seq was performed on isolated cerebral vessels from mice lacking endothelial laminin α5 expression (Tek-cre:Lama5 −/− ; sFigure 3). Sixty three thousand and fifty cells were analyzed, which showed the same clusters as for Lama4 −/− analyses. This revealed the opposite pattern to that observed in Lama4 −/− endothelium with regards to Vcam1, Ocln and Klf2 (sFigure 3(A)–(C)), that is, upregulation of the leukocyte adhesion molecule, Vcam1 (and Lama4), and downregulation of the tight junction molecule Ocln, and the shear response gene Klf2, suggesting that some results observed in Lama4 −/− endothelium are due to the elevated expression of laminin α5. Immunofluorescence staining confirmed more VCAM-1 positive vessels, in particular postcapillary venules, in Tek-cre:Lama5 −/− and fewer in Lama4 −/− brains (Figure 2(a)). A similar pattern was found for ICAM-1, another leukocyte adhesion molecule and marker of postcapillary activation 35 (sFigure 4(A) and (B)). Immunofluorescence staining for occludin revealed less intense staining at endothelial cell-cell junctions in Tek-cre:Lama5 −/− brains and more intense staining in Lama4 −/− brains compared to WTs (Figure 2(b)). Staining for another tight junction molecule, claudin 5, revealed more microvessels showing high intensity staining in Lama4 −/− brains and slightly fewer in Tek-cre:Lama5 −/− brains compared to WTs (Figure 2(c)). Western blots of brain extracts, normalized to PECAM-1+ endothelium, confirmed higher VCAM-1 on Tek-cre:Lama5 −/− and slightly lower levels on Lama4 −/− endothelium (Figure 2(d) and (e)), and higher claudin 5 on Lama4 −/− endothelium (Figure 2(d) and (f)). In addition, qPCR showed higher expression of both Ocln and Cldn5 in endothelial cells sorted from Lama4 −/− brains and lower expression in those from Tek-cre:Lama5 −/− brains compared to controls (sFigure 4(C)–(E)). Plating of a brain endothelial cell line (bEND.5) on laminin 411 versus 511 in vitro showed higher Cldn5 and Ocln expression on laminin 511 (sFigure 4(F)).
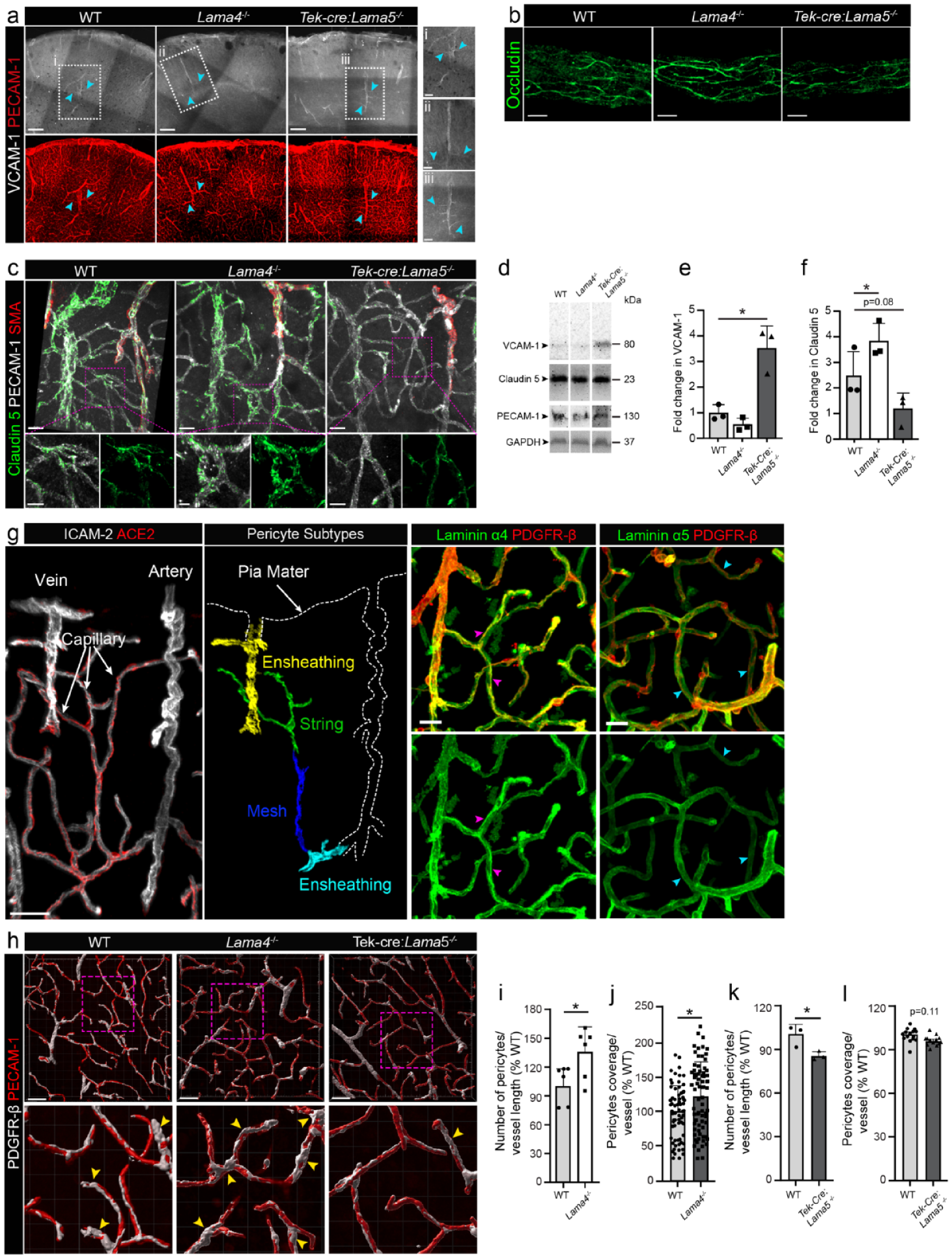
Enhanced endothelial junctional molecule expression and more mesh-like pericytes in Lama4−/− vessels due to compensatory upregulation of Lama5. (a) Immunofluorescence staining of Lama4 −/− , Tek-Cre:Lama5 −/− and representative WT control brain sections for VCAM-1. Higher magnifications are shown to the right. Immunofluorescence staining of Lama4 −/− , Tek-Cre:Lama5−/− and representative WT control brain sections for (b) occludin and (c) claudin 5. Higher magnifications show claudin 5 expression on capillary endothelial cells. (d) Representative Western blots for VCAM-1, Claudin 5 and PECAM-1 in Lama4 −/− , Tek-Cre:Lama5−/− and WT brain extracts and (e, f) corresponding quantification of signal intensities normalized to PECAM-1 (one-way ANOVA, three to five mice per genotype in at least two separate experiments). (g) Immunofluorescence staining for ACE2+ pericytes and ICAM-2+ endothelial cells identified ensheathing, string and mesh pericytes along the vascular tree. Immunofluorescence staining for PDGFRβ+ pericytes together with laminin α4 or laminin α5 in WT brain sections shows the presence of laminin α4 (magenta arrowheads) and lack of laminin α5 (cyan arrowheads) in string and mesh pericytes in representative WT control brain sections. (h) PDGFRβ+ string and mesh pericytes on PECAM-1+ vessels in WT, Lama4 −/− and Tek-Cre:Lama5 −/− mice (yellow arrowheads indicate mesh pericytes in the higher magnifications). Quantification of PDGFRβ+ pericyte number and coverage per vessel length in Lama4−/− (i,j) and Tek-Cre:Lama5 −/− (k, l) brains expressed as a percentage of the appropriate WT control (unpaired t-test, three to five mice per genotype in at least two separate experiments, in (j) and (l) each data point indicates an image analyzed); data are means ± SD. Scale bars: (a) 200 µm, (b) 10 µm, (c) 20 µm, (c, higher magnification) 10 µm, (g) 30 µm and (h) 40 µm.
Analysis of lung endothelial cells isolated from transgenic mice showed a similar pattern of results, indicating that laminin α5 induced enhanced expression of junctional molecules is not specific for the brain (sFigure 4(C)–(E)). As VCAM-1 and ICAM-1 are endothelial activation markers, expressed by postcapillary venules, 35 these data suggest that laminin α5 in the endothelial BM suppresses activation and that Lama4 −/− mice have a reduced proportion of vessels that function as postcapillary venules (Figure 1(c)), while Tek-cre:Lama5 −/− mice have more (sFigure 3(C)). This is consistent with the reduced extravasation observed in Lama4 −/− mice and enhanced extravasation in Tek-cre:Lama5 −/− mice in different inflammatory models.16,34,36
Interestingly, Tek-cre:Lama5 −/− vessels also showed the opposite gene expression profile to that observed in Lama4 −/− pericytes (sFigure 3(D)–(F)), which are embedded within the endothelial BM, suggesting that some of the effects observed in Lama4 −/− mural cells, in particular pericytes, result from compensatory enhanced expression of laminin α5, rather than loss of laminin α4. Staining for pericytes using ACE2 allowed identification of the three types of pericytes along the vascular tree: ensheathing, string and mesh (Figure 2(g)). 37 Staining for PDGFRβ together with laminins α4 or α5, revealed intense laminin α4 staining along pericyte processes of string and mesh pericytes, which was not the case for laminin α5 (Figure 2(g)), consistent with the higher Lama4 expression in pericytes relative to endothelium in scRNA-seq analyses of WT mice (sFigure 1(F)). PDGFRβ staining and quantification of pericyte cell bodies per blood vessel length revealed increased pericyte numbers and coverage in Lama4 −/− brains (Figure 2(h)–(j)), with high magnification revealing a more mesh-like phenotype 5 (Figure 2(h)). The opposite results were found in Tek-cre:Lama5 −/− vessels (Figure 2(h), (k) and (l)), which showed a 13% reduction in pericyte number (Figure 2(k)) and a 5% reduction in pericyte coverage (Figure 2(l)), reflecting a decrease in mesh-like phenotype and an increase in string pericytes. Mesh pericytes are smooth muscle actin negative; their branching processes allow them to interact with endothelial cells and other pericytes, potentially facilitating communication and coordination of blood flow regulation. 5 This is consistent with the elevated Klf2 expression in endothelium and mural cells and the exacerbated shear response in Lama4 −/− mice, 19 and the reduced Klf2 expression pattern (sFigure 3(E)) and almost complete absence of shear response in Tek-cre:Lama5 −/− mice. 19
No difference was noted in Enpp2 (autotaxin), Gja4, Edn1 or Tgfb2 expression in either endothelium or mural cells of Tek-cre:Lama5 −/− mice (sFigure 3(B)), nor were there major differences in resident myeloid populations (sFigure 3(G) and (H)), indicating that these changes in Lama4 −/− where due to the loss of laminin α4.
Laminin α4 suppresses some arterial characteristics
In vivo immunofluorescence staining for smooth muscle actin (SMA) and PECAM-1 was used to distinguish arteries from veins, revealing fewer and slightly shorter arteries in Lama4 −/− brains (Figure 3(a)–(c)) but with a 15% larger diameter compared to WT controls (Figure 3(d)); no significant differences were observed for Tek-cre:Lama5 −/− arteries (not shown). This is in accordance with previous data showing outward remodelling of Lama4 −/− resistance mesenteric arteries, probably due to chronic elevated shear stress. 19 Although the SMA+ vessels were shorter, staining intensities per vessel were higher in Lama4 −/− compared to WT brains (Figure 3(e)). Quantification of the number of SMC cells per vessel length revealed similar results for Lama4 −/− and WT vessels, confirming higher expression of SMA in Lama4 −/− VSMCs (Figure 3(f) and (g)) as suggested by the scRNA-seq data. In addition, Ptgis was one of the most highly upregulated genes in Lama4 −/− smooth muscle cells (Figure 1(e)), which encodes prostacyclin synthetase required for prostacyclin synthesis, a potent vasoactive lipid mediator that leads to vasodilation. Western blots of Lama4 −/− brain extracts confirmed higher amounts of SMA and prostacyclin synthetase (Figure 3(h)–(j)). The increased prostacyclin synthetase expression probably contributes to the exacerbated shear-induced vasodilation in Lama4 −/− mice. 19
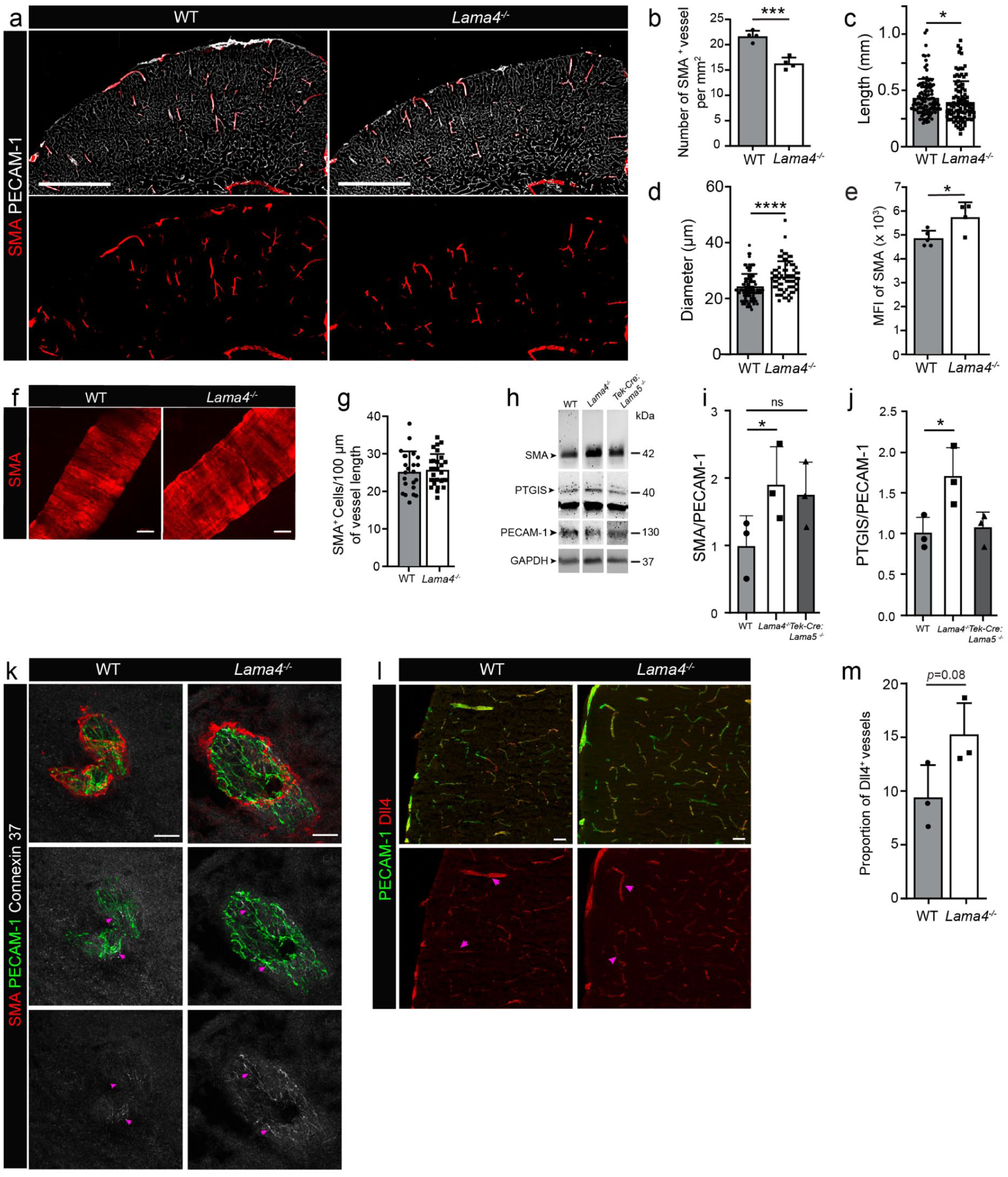
Laminin α4 suppresses some arterial endothelial markers. (a) Immunofluorescence staining of PECAM-1 and SMA to mark arteries in Lama4 −/− brains and corresponding quantification of (b) number, (c) length, (d) diameter and (e) MFI of SMA+ penetrating arteries (unpaired t-test for (b), (d) and (e) and Mann–Whitney for (c); at least two 200 µm sections from three to five brains per genotype were analyzed in at least two experiments; in (c) and (d), each data point represents an artery). (f) High resolution image of SMA+ arteries in Lama4 −/− and WT littermate brain sections and (g) corresponding quantification of SMA+ cells per vessel length (unpaired t-test, at least two 200 µm sections from three brains per genotype were analyzed in at least two experiments, each data point represents an artery). (h) Representative Western blots for SMA and PTGIS in Lama4 −/− , Tek-Cre:Lama5 −/− and WT brain extracts with (i, j) corresponding quantification of signal intensities normalized to PECAM-1 (one-way ANOVA, three to five mice per genotype in at least two separate experiments). (k) Immunofluorescence staining for connexin 37 at arterial endothelial junctions (magenta arrowheads) and (l) of Dll4 (magenta arrowheads) in Lama4 −/− and WT littermate brain sections and (m) corresponding quantification of Dll4+ vessels expressed as a percent of total vessels counted (unpaired t-test, at least three sections from three brains per genotype were analyzed in at least two experiments). Data are means ± SD. Scale bars: (a) 500 µm, (f) 10 µm, (k) 25 µm and (l) 50 µm.
Stainings for connexin 37 (encoded by Gja4) in arteries showed enhanced expression levels at endothelial junctions (Figure 3(k)), while staining for delta-like 4 (Dll4), a marker of arterial endothelium, showed slightly higher intensity and broader staining (10%) in Lama4 −/− arteries (Figure 3(l) and (m)).
To investigate potential differences in tissue perfusion resulting from differences in vessel size or density, magnetic resonance imaging (MRI) was performed. The basal cerebral blood flow (CBF), as measured by arterial spin labelling, was not different between WT and Lama4 −/− mice at the time points checked (6, 8 and 14 weeks; sFigure 5(A)). The microvessel density index, however, was lower in the Lama4 −/− mice (sFigure 5(B)), suggesting a slight reduction in vessel density as observed in stainings for ICAM-2 (sFigure 5(C) and (D)) and by flow cytometry (sFigure 5(E)). Tek-cre:Lama5 −/− mice showed no reduction in vessel density as shown by flow cytometry (sFigure 5(F)).
Hence, there is a slightly lower vessel density in Lama4 −/− brains but more vessels exhibit arterial characteristics and perfusion of the organ is normal.
Indirect effects of loss of vascular Lama4 expression
Despite the absence of expression of laminins by immune cells (sFigure 1(F) and (G)), resident myeloid populations associated with the cerebral arteries and the pia 38 showed clear differences between WT and Lama4 −/− brains (Figure 4(a)). Macrophages defined by Mrc1 (which encodes CD206), Ms4a7, Pf4 expression separated into three clusters (Figure 1(h) and sFigure 2(E) and (F)), as previously reported 10 ; however, with some differences. We identified macrophages enriched in phagocytosis markers (Mrc1, Wfdc17, Cd68, Ctsd) with low Lyve1 expression, a cytokine enriched population (Ccl7, Ccl3 Atf3, Ednrb) exhibiting high Lyve1 expression, and a Lyve1 negative population characterized by high expression of markers of antigen presentation (H2-Ab1 which encodes MHCII) and leukocyte infiltration (Ccr2; Figure 1(i) and (j)). We refer to these macrophage populations as phagocytotic, cytokine-enriched and MHCIIhigh antigen presenting populations, respectively. Lama4 −/− mice had higher proportions of the MHCIIhigh (H2-Ab1) population, which were not restricted to the choroid plexus as reported by others 10 but occurred both perivascularly and in the meninges as shown by immunofluorescence staining (Figure 4(a)). Immunofluorescence staining for CD206, MHCII and pan-laminin revealed an overall reduction in perivascular macrophages and confirmed that a higher proportion of CD206+ macrophages were MCHIIhigh in Lama4 −/− brains (Figure 4(a)); this was confirmed by flow cytometry of brains (without choroid plexus; Figure 4(b)). Hence, although there were fewer resident macrophages, they had an activated phenotype.
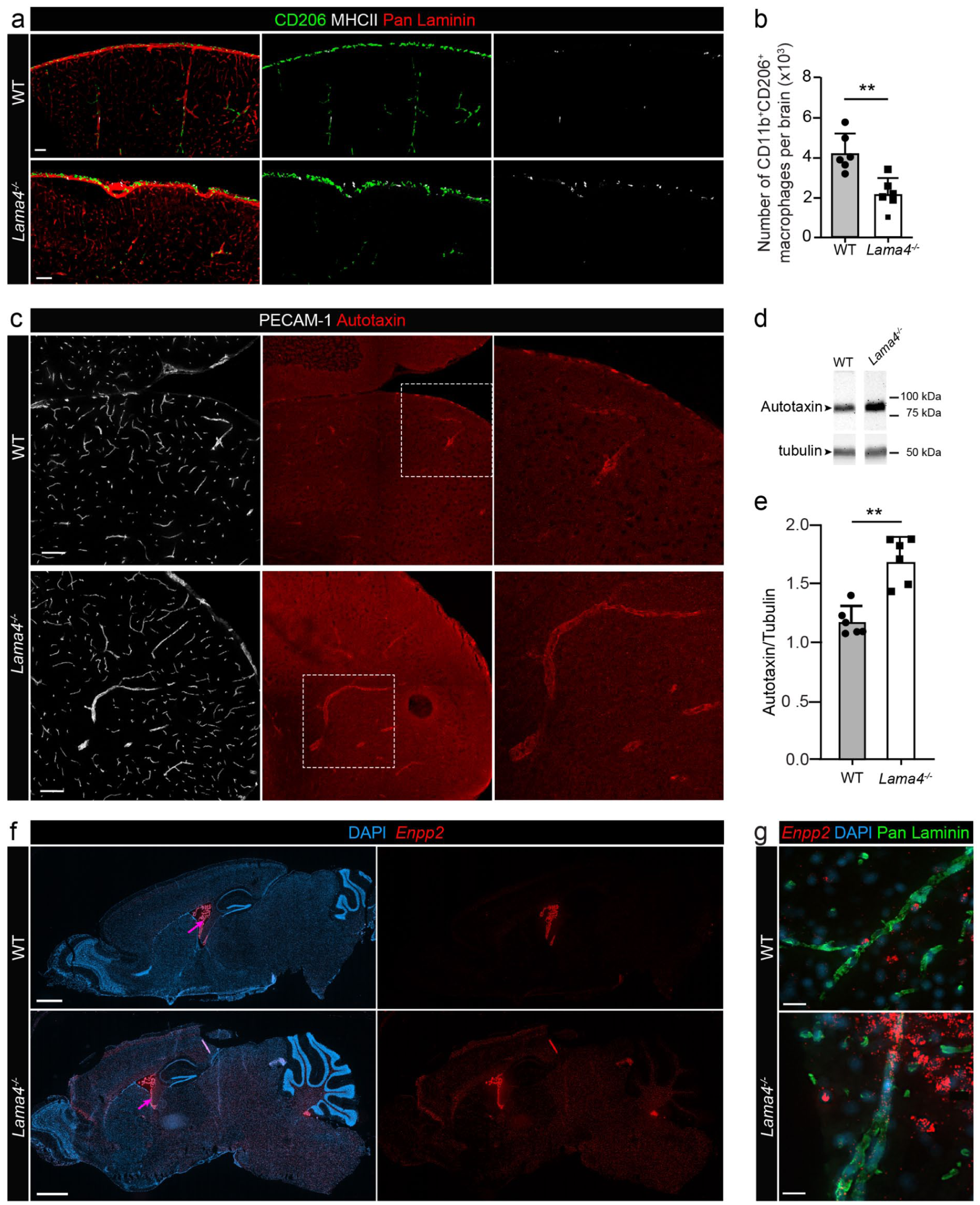
Indirect effects of loss of Lama4 expression on resident myeloid populations and autotaxin expression. (a) Triple immunofluorescence staining for CD206+ resident macrophages, MHCII and pan laminin to mark vascular and pial BMs in Lama4−/− and WT littermates brain sections. (b) Flow cytometry quantification of CD11b+CD206+ macrophages in Lama4 −/− or WT littermate brains (unpaired t-test, six brains per genotype were analyzed in at least two experiments). (c) Double immunofluorescence staining for PECAM-1 to mark blood vessels and autotaxin in Lama4 −/− and WT littermates brain sections; right panels show boxed areas at higher magnification. (d) Representative Western blot for autotaxin in brain extracts from Lama4 −/− and WT mice and (e) corresponding quantification of signal intensity normalized to the tubulin control (unpaired t-test, six mice per genotype in at least two separate experiments). (f, g) RNAscope together with immunofluorescence staining for DAPI as a nuclear marker and pan laminin to mark vascular BMs. Data are means ± SD. Scale bars: in (a) 100 µm, (c) 100 µm, (f) 1 mm and (g) 10 µm.
Although Ttr was highly expressed in all Lama4 −/− vascular cells analyzed, in vitro confirmation of this result using qPCR of isolated cerebral vascular cells, staining of tissue sections or Western blots for transthyretin in brain sections were inconclusive due to high variability. By contrast, elevated expression of autotaxin encoded by Enpp2 in Lama4 −/− brains was confirmed by immunofluorescence staining and Western blots of brain extracts (Figure 4(c)–(e)). RNAscope revealed Enpp2 expression mainly by perivascular cells, probably pericytes and fibroblasts, and by choroid plexus epithelium in WT brains, as previously reported, 39 and enhanced expression in vascular and brain parenchymal cells in Lama4 −/− brains (Figure 4(f) and (g)). Furthermore, immunofluorescence staining and RNAscope both suggested enhanced expression of autotaxin protein (Figure 4(g)) by brain parenchymal cells (sFigure 1(F)). This suggests that the elevated autotaxin expression in Lama4 −/− brains may in part be due to indirect effects, such as the activated status of resident myeloid cells or prolonged enhanced shear stress, both of which promote autotaxin expression.40,41
tMCAO as a test of arterial function
To further test arterial functions in Lama4 −/− mice and to assess the functional consequence of elevated autotaxin, which is known to worsen stroke outcome,21,33 a transient middle cerebral artery occlusion (tMCAO) model was employed. 60 min tMCAO and 24 h reperfusion was performed in Lama4 −/− and age- and sex-matched WT controls and in Tek-Cre:Lama5 −/− mice as a control for effects specifically due to loss of laminin α4. Lama4 −/− mice exhibited more severe stroke symptoms as assessed by motoric activity, swelling and lesion volume compared to WT littermates and Tek-Cre:Lama5 −/− mice (Figure 5(a)–(c)). However, flow cytometry revealed similar numbers of CD45+ leukocytes in the ipsilateral (IL) brain hemispheres in Lama4 −/− , Tek-Cre:Lama5 −/− and WT mice (Figure 5(d)), and similar ratios of total CD45+, neutrophils, macrophages, monocytes or microglia in IL compared to contralateral (CL) hemispheres (sFigure 6(A)), confirming our earlier observations that stroke severity at early stages after ischaemia does not correlate with leukocyte extravasation into the parenchymal tissue. 26 Immunofluorescence staining of brain sections for pan-laminin and CD45 revealed most leukocytes within or closely associated with arterioles located close to the surface of the brain in all mice (Figure 5(e) and sFigure 6(B)), as previously shown for WT mice. 26 The enhanced stroke severity observed in Lama4 −/− mice, therefore, was not determined by the extent of immune cell infiltration, nor can it be explained by junctional differences given the lack of a stroke phenotype in Tek-cre:Lama5 −/− mice but is consistent with the higher autotaxin levels in these mice (Figure 4(c)–(e)).
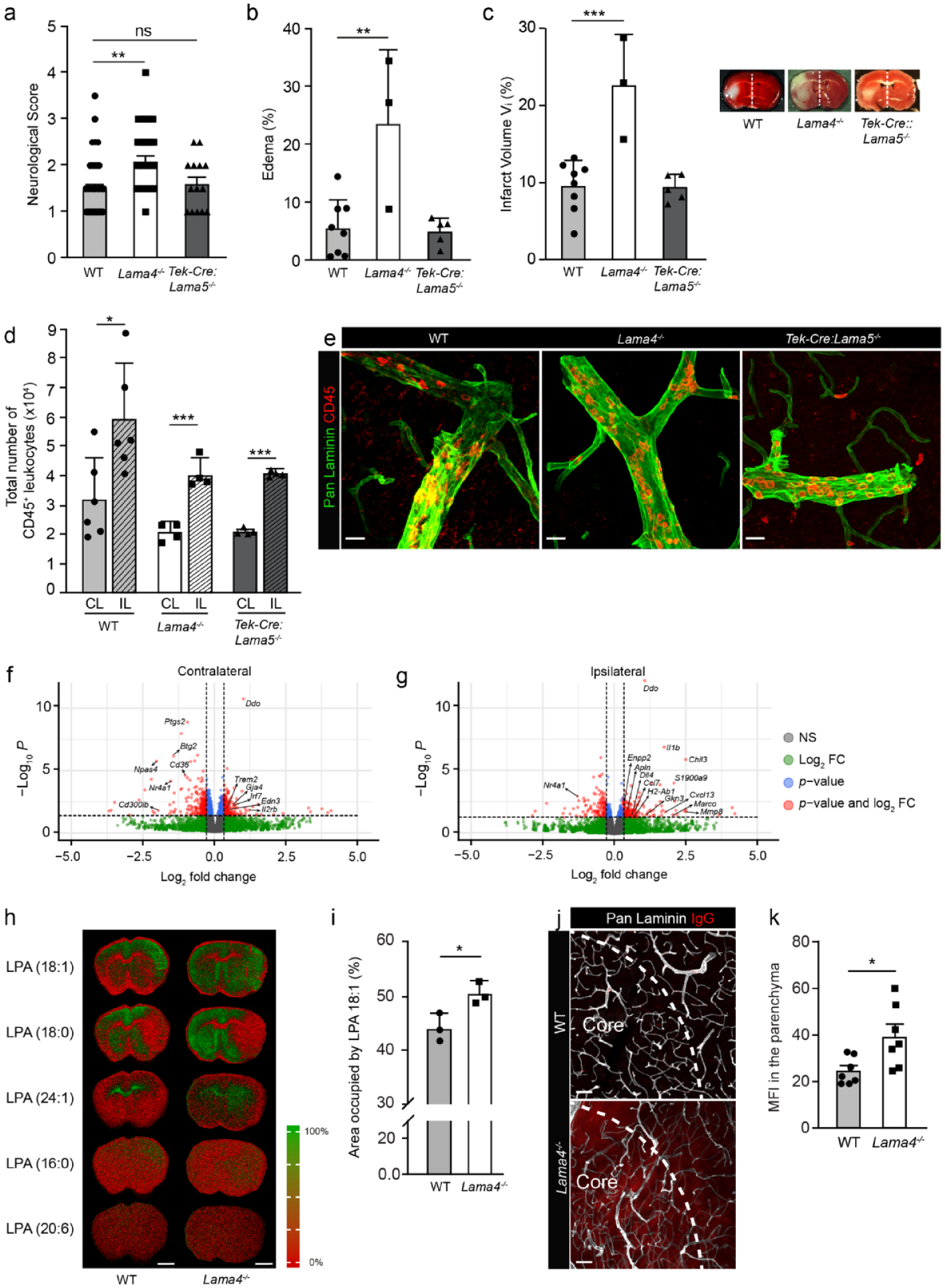
More severe tMCAO (60 min/24 h reperfusion) in Lama4 −/− correlates with higher LPA levels. (a) Neurological scores, (b) swelling in IL hemispheres and (c) infarct volumes in Lama4 −/− , Tek-Cre:Lama5 −/− and WT controls (ANOVA followed by post hoc Tukey; for (a) at least 14 mice/genotype and, for (b, c), at least three brains per genotype in at least three experiments). (d) Flow cytometry quantification of total CD45+ leukocytes in IL and CL hemispheres of Lama4 −/− , Tek-Cre:Lama5 −/− and WT controls (ANOVA followed by post hoc Tukey, three to six brains per genotype from at least three experiments). (e) Representative immunofluorescent staining for pan-laminin to mark the border to the CNS parenchyma and CD45+ leukocytes in brain sections from Lama4 −/− , Tek-cre:Lama5−/− and WT controls. (f) Volcano plots showing relative differences in expression levels of all DEGs between WT and Lama4 −/− mice for CL (f) and IL (g) hemispheres (total number of genes analyzed = 20346); p < 0.05 and −0.7 ⩾ Log2 fold change ⩽ 1.3 are shown in red (DEGs = 204 (CL) and 213 (IL)). (h) Representative mass spectrometry imaging visualizing the signal intensity distribution of m/z: 437.27 tentatively assigned to LPA in the post stroke brain. Oedema enlarged hemisphere is the IL side. LPA (18:1) is increased on the IL side relative to the CL in both Lama4 −/− and WT mice. (i) % Area occupied by LPA (18:1) signal (unpaired t-test, three mice per genotype in at least two experiments). (j) Representative immunofluorescent staining for pan-laminin and IgG in Lama4 −/− and WT brain sections and (k) corresponding quantification of IgG immunofluorescent signal in the CNS parenchyma (unpaired t-Test, seven brains per genotype were analyzed in three experiments). Data are means ± SD. Scale bars: (e) 25 µm, (h) 2 mm and (j) 60 µm.
Bulk RNA sequencing
Bulk RNA sequencing performed on the cerebral cortex from IL and CL brain hemispheres from Lama4 −/− and WT controls at 24 h after ischaemic stroke, confirmed the higher degree of neuron damage in Lama4 −/− mice and demonstrated that stroke-induced transcriptomic differences dominated over differences due to genotype (sFigure 7(A) and (B)). Volcano plots show fold changes in gene expression and p values in CL and IL (Figure 5(f)) brain hemispheres from Lama4 −/− compared to WT mice. Supplementary Files 3 and 4 contain the complete gene lists (GSE309181).
Despite the prominent stroke effects, comparison of IL hemispheres from WT and Lama4 −/− mice also revealed upregulation of several arterial markers in Lama4 −/− including Gja4, Dll4, Gkn3, Edn1, as well as inflammatory parameters such as Il1b (interleukin-1b), Ccl7, Mmp8, Chil3, Cxcl13 and Marco and Enpp2 (autotaxin; Figure 5(f) and (g)), substantiating the scRNA-seq data. The heatmap in sFigure 7(C) shows the top genes using a cut-off of p ⩽ 0.05, listed according to fold-change. GSEA revealed enrichment of metabolic gene sets (oxidative phosphorylation), ribosomal genes, and gene sets involved in muscle in CL Lama4 −/− brain hemispheres (sFigure 7(D)), consistent with the scRNA-seq data. In IL Lama4 −/− brain hemispheres, GSEA showed enrichment for metabolic processes, cytokine signalling, Toll-like receptor and NOD-like receptor signalling (sFigure 7(E)), suggesting a proinflammatory status. Given the absence of enhanced infiltration of immune cells in the IL hemisphere of Lama4 −/− mice compared to WT, changes in cytokine signalling and Toll-like receptor genes are likely to represent differences in resident myeloid populations.
Enpp2 encodes autotaxin, a phosphodiesterase that converts lysophosphatidylcholine (LPC) to lysophosphatidic acid (LPA), a lipid mediator that acts through several G-coupled protein receptors (LPAR1–6) with diverse effects including ones on vascular integrity.42,43 As LPA levels exacerbate stroke severity, 21 we employed in situ mass spectrometry to analyze the LPA species in IL and CL hemispheres of Lama4 −/− and WT brains after tMCAO, revealing elevated levels of several LPA species (18:1, 18:0, 24:1) in Lama4 −/− brains compared to WT controls, with enrichment in the IL hemispheres (Figure 5(h) and (i)). 33
As autotaxin affects vessel permeability, 44 the presence of serum proteins, including IgG, in the CNS parenchyma of Lama4 −/− mice was assessed using immunofluorescence staining, revealing higher intensity and broader staining in the lesion core and penumbra (Figure 5(j) and (k)). To determine whether blood vessels of Lama4 −/− mice are generally more permeable than those of WT or Tek-Cre:Lama5 −/− mice, a modified Miles permeability assay was performed. 45 Extravasation of intravenously injected Evan’s blue dye in response to intradermal injection of PBS into the back skin of age- and sex-matched mice revealed no differences between Lama4 −/− , Tek-Cre:Lama5 −/− or WT mice (sFigure 8), indicating similar basal permeabilities. However, an approximately two-fold higher extravasation of Evans Blue into the dermis of Lama4 −/− mice was measured following intradermal injection of the vascular endothelial cell permeability factor (VEGF) compared to Tek-Cre:Lama5 −/− or WT mice (sFigure 8), indicating enhanced permeability to soluble molecules but only under stress conditions, which is consistent with the generally higher levels of autotaxin in these mice.
The more severe stroke phenotype in the Lama4 −/− mice compared to Tek-cre:Lama5 −/− mice is consistent with the elevated levels of autotaxin (and consequently LPA), given that this was the major difference between these two phenotypes. However, the fact that autotaxin is upregulated in several cell types in the brain parenchyma of Lama4 −/− mice, which do not express laminin α4, suggests that the proinflammatory phenotype, resulting from changes in resident myeloid populations or exacerbated shear response probably contribute to elevated autotaxin and higher stroke severity in these mice.
Discussion
Our data demonstrate that BM laminins exert segment-specific effects along the vascular tree. Notably, Lama4 −/− mice exhibited reduced vascular density and a shift toward an arterial phenotype, primarily at the expense of postcapillary venule identity. This was accompanied by increased expression of the shear-responsive gene Klf2, elevated levels of contractile and vasoactive molecules in smooth muscle cells, and a higher number of mesh-like pericytes per vessel. 5 These observations suggest that the balance between laminin α4 and α5 in the microvascular BM plays a role in establishing the functional zonation of cerebral vessels. High levels of laminin α5 in endothelial BMs appear to promote an arterial phenotype, not only in endothelial cells but also in pericytes enclosed within the same BM. Conversely, laminin α4 suppresses the expression of key arterial markers, such as connexin 37, Delta-like 4 and TGF-β2, and is essential for maintaining the quiescent state of perivascular myeloid cells, which preferentially associate with arteries.
Elevated expression of autotaxin in Lama4 −/− brains likely results from chronic shear stress – evidenced by outward arterial remodelling 19 – and/or activation of resident myeloid cells. This is consistent with worsened outcomes following transient middle cerebral artery occlusion (tMCAO) in Lama4 −/− mice.
Gene expression profiling and immunofluorescence staining of Lama4 −/− and Tek-Cre:Lama5 −/− tissues, along with qPCR of sorted cells and in vitro studies using bEND.5 cells, support the conclusion that compensatory upregulation of laminin α5 in Lama4 −/− vessels underlies the enhanced expression of junctional proteins (claudin-5 and occludin), consistent with our previous studies,19,46 and suppresses the expression of the endothelial activation markers, ICAM-1 and VCAM-1. In Lama4 −/− vessels, the higher number of pericytes and altered pericyte morphology are also likely due to this compensatory laminin α5 upregulation, given that Tek-Cre:Lama5 −/− vessels exhibit the opposite phenotype. Specifically, the rise in mesh pericytes, which are α-smooth muscle actin-negative and associated with the arteriole-to-capillary transition, 5 may enhance vascular permeability under VEGF stimulation and contribute to the exacerbated shear response in the Lama4 −/− mice. 19 These mesh pericytes have been implicated in blood flow regulation and BBB maintenance,4,47 and possess branching processes that facilitate interactions with endothelial cells and neighbouring pericytes, supporting coordinated vascular function. This is consistent with elevated Klf2 expression in both endothelial and mural cells in Lama4 −/− mice and aligns with the heightened shear sensitivity. Interestingly, increased pericyte coverage correlated with reduced VCAM-1 and ICAM-1 expression in Lama4 −/− mice, as previously reported in other mouse strains, 48 while Tek-Cre:Lama5 −/− mice showed the opposite trend. This suggests that pericytes embedded in laminin α5-rich endothelial BMs may contribute to maintaining endothelial quiescence.
As expected, Tek-Cre:Lama5 −/− vessels did not show significant changes in SMC gene expression or Western blots; however, a potential role of laminin α5 in enhancing contractile gene expression in SMCs in Lama4 −/− mice cannot be excluded, and requires analysis of a vascular smooth muscle specific Lama5 knockout mouse.
Importantly, elevated arterial marker expression (e.g. Gja4, Dll4 and Tgfb2) was observed exclusively in endothelial cells lacking laminin α4, supporting the notion that laminin α4 negatively affects arterial differentiation. Interestingly, the arterial expansion in Lama4 −/− mice was at the expense of postcapillary venules, not venular endothelium per se. To our knowledge, this is the first report of a factor defining postcapillary venule identity that is not simply related to vessel diameter.
We have previously shown that laminin α5 expression begins in arterial vessels during embryonic development and increases postnatally. 49 Laminins 411/511 modulate Notch1 signalling during retinal angiogenesis, 15 and laminin α5 has been reported to inhibit proliferation of various cell types, including epithelial and haematopoietic cells.50–52 Our pathway analyses reveal a shift toward enhanced oxidative phosphorylation and lipid metabolism in Lama4 −/− mice, consistent with an arterial phenotype and a quiescent state. 2 Our data therefore suggest that vascular laminins do not determine arterial fate during development but rather maintain arterial identity – laminin α5 promotes, while laminin α4 inhibits this phenotype. This model is supported by the diminished shear responses in resistance arteries in Tek-Cre:Lama5 −/− mice 19 – opposite to the Lama4 −/− phenotype.16,19
Some of the changes observed in Lama4 −/− vessels are likely to be indirect. For example, the elevated endothelial expression of TGF-β2, known to upregulate expression of smooth muscle actin and other contractile proteins,53–55 may contribute to the enhanced contractile phenotype of SMCs in Lama4 −/− vessels. Along the same lines, both bulk and scRNA-seq analyses revealed an inflammatory phenotype in Lama4 −/− mice with clear increases in cytokine expression, and an associated reduction in resident macrophages localized along arteries, despite the fact that immune cells do not express ECM molecules, as confirmed here. These results suggest a role for vascular BMs in either the seeding and/or maintenance of perivascular myeloid populations, but potentially also their activation status, topics that are currently under investigation. It cannot be determined here whether changes in these resident perivascular macrophages also contributed to the more severe stroke in Lama4 −/− mice. However, their reduced numbers in Lama4 −/− brains, albeit enhanced activation status, suggest that the resident perivascular macrophages could have a suppressive effect either on endothelial activation and/or exacerbated vessel dilation in response to increased shear (after vessel reperfusion).
Interestingly, elevated autotaxin expression in Lama4 −/− brains by cells not normally expressing laminin α4 may result from chronic shear stress and/or inflammation as suggested by others.56,57 Autotaxin has been implicated in various inflammatory conditions and modulates purinergic signalling. 58 Elevated autotaxin levels are also associated with worse stroke outcomes,21,33 and endothelial-specific deletion of Enpp2 (autotaxin) reduces stroke severity. 33 The worsened stroke phenotype in Lama4 −/− mice is consistent with these findings, and supports the hypothesis that laminin α4 loss indirectly drives autotaxin upregulation via inflammatory and potentially also biomechanical cues.
Finally, our scRNA-seq analysis extends existing profiles of cerebral vascular cells. We identified a likely postcapillary venule endothelial population (marked by Icam1, Vcam1, Il1r1) that was diminished in Lama4 −/− mice. This may represent the activated endothelial subset previously described in WT EAE, 30 potentially contributing to the reduced EAE severity observed in Lama4 −/− mice. 16 Furthermore, we distinguish large arteries and arterioles by smooth muscle gene expression, with high Klf2 and Acta2 (αSMA) levels in arteries versus Vtn and Pdgfrb in arterioles.
Together, these data indicate that the ratio of laminin α4 to α5 in vascular BMs contributes to the functional zonation of cerebral vessels by influencing metabolic pathways in endothelial and mural cells and by modulating the activation and maintenance of resident myeloid populations. This finely tuned balance underlies the specialization of arterioles, capillaries and postcapillary venules in the cerebral vasculature. Although our findings point to direct roles for vascular laminins in establishing zonation, we cannot exclude the possibility that some effects arise indirectly, for example, through their influence on the localization or maintenance of perivascular macrophages.
Supplemental Material
sj-pdf-5-jcb-10.1177_0271678X251410040 – Supplemental material for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels
Supplemental material, sj-pdf-5-jcb-10.1177_0271678X251410040 for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels by Tushar Deshpande, Kishan Kapupara, Melanie-Jane Hannocks, Jula Huppert, Sai-Kiran Samawar, Devika Rag Thulichery, Sigmund Budny, Sharang Ghavampour, Jian Song, Lingzhang Meng, Ralf H Adams, Hyun-Woo Jeong, Lydia Wachsmuth, Cornelius Faber, Jens Soltwisch, Rupert Hallmann and Lydia Sorokin in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-1-jcb-10.1177_0271678X251410040 – Supplemental material for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels
Supplemental material, sj-xlsx-1-jcb-10.1177_0271678X251410040 for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels by Tushar Deshpande, Kishan Kapupara, Melanie-Jane Hannocks, Jula Huppert, Sai-Kiran Samawar, Devika Rag Thulichery, Sigmund Budny, Sharang Ghavampour, Jian Song, Lingzhang Meng, Ralf H Adams, Hyun-Woo Jeong, Lydia Wachsmuth, Cornelius Faber, Jens Soltwisch, Rupert Hallmann and Lydia Sorokin in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-2-jcb-10.1177_0271678X251410040 – Supplemental material for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels
Supplemental material, sj-xlsx-2-jcb-10.1177_0271678X251410040 for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels by Tushar Deshpande, Kishan Kapupara, Melanie-Jane Hannocks, Jula Huppert, Sai-Kiran Samawar, Devika Rag Thulichery, Sigmund Budny, Sharang Ghavampour, Jian Song, Lingzhang Meng, Ralf H Adams, Hyun-Woo Jeong, Lydia Wachsmuth, Cornelius Faber, Jens Soltwisch, Rupert Hallmann and Lydia Sorokin in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-3-jcb-10.1177_0271678X251410040 – Supplemental material for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels
Supplemental material, sj-xlsx-3-jcb-10.1177_0271678X251410040 for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels by Tushar Deshpande, Kishan Kapupara, Melanie-Jane Hannocks, Jula Huppert, Sai-Kiran Samawar, Devika Rag Thulichery, Sigmund Budny, Sharang Ghavampour, Jian Song, Lingzhang Meng, Ralf H Adams, Hyun-Woo Jeong, Lydia Wachsmuth, Cornelius Faber, Jens Soltwisch, Rupert Hallmann and Lydia Sorokin in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-xlsx-4-jcb-10.1177_0271678X251410040 – Supplemental material for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels
Supplemental material, sj-xlsx-4-jcb-10.1177_0271678X251410040 for Vascular basement membrane laminins modulate functional zonation of cerebral microvessels by Tushar Deshpande, Kishan Kapupara, Melanie-Jane Hannocks, Jula Huppert, Sai-Kiran Samawar, Devika Rag Thulichery, Sigmund Budny, Sharang Ghavampour, Jian Song, Lingzhang Meng, Ralf H Adams, Hyun-Woo Jeong, Lydia Wachsmuth, Cornelius Faber, Jens Soltwisch, Rupert Hallmann and Lydia Sorokin in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
We thank Dirk Reinhardt (EIMI) for stroke surgeries; Laura Schnell and Melissa Chmara for animal breeding, genotyping and tissue collection.
Author contributions
TD performed the scRNA-seq and bulk RNA-seq analyses and verified data with immunofluorescence. KK verified a large part of the scRNA-seq and bulk RNA-seq data using flow cytometry, immunofluorescence and Western blots. SB contributed to Western blots. JH performed stroke experiments and bulk RNA-seq. SG and LM contributed to the analyses of junctional proteins. S-KS and DRT analyzed resident macrophage populations. M-JH contributed to verification of the scRNA-seq data using immunofluorescence and contributed to writing of the manuscript. JS, RHA and H-WJ contributed to planning and execution of the scRNA-seq experiments. LW and CF performed the MRI. JS performed the in situ mass spectrometry for LPA species. RH contributed to the project design and LS conceived the project, acquired funding and wrote the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The work was supported by funding to LS from the German Research Foundation (DFG; SO285/14-1), the European Commission (SVD@targets, ERC grant number 101054805) and the Federal Ministry for Education and Research (BMBF; DeCoDis) and the core unit PIX of the Medical Faculty and the IZKF Münster.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical considerations
Experiments on mice were approved by the Landesamt für Verbraucherschutz und Ernährung, Nordrhein-Westfalen (LAVE; animal permit license numbers: 2018.A068 and Histo4), carried out in accordance with the German Animal Welfare Act guidelines and reported according to Animals in Research: Reporting In Vivo Experiments (ARRIVE) guidelines.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.