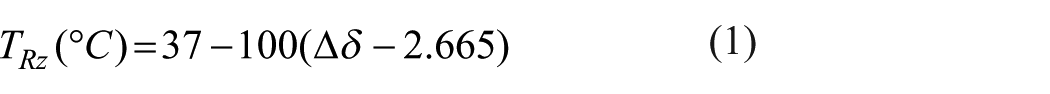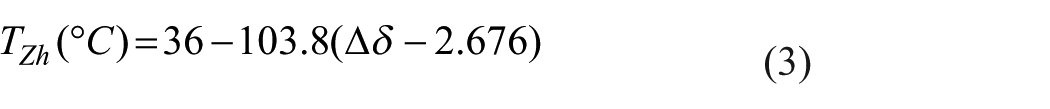Abstract
Cephalad fluid shift is strongly implicated in the brain-structural and craniovascular disturbances observed after long-duration spaceflight. It is part of what has been termed the cranial microgravity response (CMR). However, the physiological mechanisms underlying these changes remain unclear. We employed a ~2-h, −10° head-down tilt (HDT) paradigm to investigate the immediate effects of cephalad fluid shift on cranial venous outflow, cephalic venous vasculature, and brain temperature. Twenty-one adults (26.6 ± 2.4 years) underwent four MRI sessions combining 4D-flow measurements, susceptibility-weighed imaging, Time-of-Flight angiography, and MR Spectroscopy. HDT resulted in a significant increase in internal jugular vein cross-sectional area (p < 0.001), decreased jugular vein flow velocity (p < 0.001) and normalized mean flowrate (p < 0.001), a volumetric expansion of the main cervical arteries (p < 0.001), and a reduction in superior sagittal sinus flow velocity (p = 0.03). We also found a significant increase in global brain temperature after HDT (0.33°C ± 0.28°C; p < 0.001). These findings provide converging evidence for compromised cranial venous outflow in simulated microgravity, suggesting an upstream impact on the cerebral venous vasculature and thermal homeostasis of the head. Our setup now offers a comprehensive and fast-turnaround framework for the testing of countermeasures against the CMR.
Introduction
Despite comprising only 2% of body weight, the brain is one of the most highly perfused organs, receiving upwards of 20% of the total cardiac output regardless of body position. 1 This makes it particularly susceptible to the effects of microgravity, given that the human cardiovascular system is evolutionarily adapted to the gravitational hydrostatic pressure gradient on Earth. During weightlessness, this gradient disappears and fluid redistributes more uniformly throughout the body, producing a net “cephalic” fluid shift toward the upper torso and head. 2 This shift has been increasingly implicated in a range of neurovascular changes observed during long-duration spaceflight (LDSF), including disruptions to arterial structure and function, increased microbleed risk, volumetric expansion of the superior sagittal sinus (SSS), internal jugular vein (IJV) distension, and a rare case of obstructive venous thrombosis in the left IJV.3–8 Further investigations of the IJVs have revealed strong evidence of stagnant or reversed flow patterns and reduced peak flow velocities.9–11 Our group has also previously reported an expansion of cerebrospinal fluid (CSF) spaces and perivascular spaces (PVS) in several astronauts after LDSF, coupled with reductions in gray- and white-matter (GM and WM) volume.12–14 Collectively, these observations have been termed the Cranial Microgravity Response (CMR).15,16 A fluid shift-induced impairment of cerebral venous drainage may correspondingly inhibit CSF absorption capacity through the PVS and SSS, offering a potential explanation for the CMR as well as fluid-based ophthalmic disturbances observed in astronauts with the Space-Associated Neuro-ocular Syndrome (SANS).17–21
Given the close coupling of cerebral blood flow (CBF) and brain temperature (TBr) through cerebral autoregulation, 22 any disruption to the arterial or venous circulation during LDSF is likely to influence temperature homeostasis.23–26 In fact, the TBr would theoretically increase by 0.28°C/min without an adequate heat offloading system (i.e., intracranial venous pathways). 27 Elevated head temperatures have been previously reported in astronauts at rest and during exercise, with both rising faster and reaching higher levels than on Earth.28,29 However, aside from one study measuring suborbital skin temperature during ultra-short dry immersion, 30 the impact of microgravity on TBr remains largely unexplored. Progress has been partly limited by methodological constraints, as traditional thermometry methods (e.g., intracerebral thermistors) are invasive and restricted to focal measurements.31,32 This has encouraged the adoption of Magnetic Resonance (MR) Spectroscopy thermometry (MRS) as a non-invasive alternative. MRS can estimate TBr by measuring the chemical-shift difference between water and certain metabolite peaks and has already been validated across several clinical and research settings.25,32–35
Much of the current evidence on hemodynamic changes during spaceflight comes from Doppler ultrasound, which records real-time flow and pressure data but is constrained by poor spatial resolution, inconsistent velocity measurements, and operator-dependent expertise. 36 4D-flow MR Imaging (MRI) is a phase-contrast (PC) imaging technique that implements three-directional velocity encoding in addition to a time-resolved acquisition using electrocardiogram (ECG)-gating. Thus, it can provide time-resolved flow data from multiple vessels in a single acquisition that can be analysed post-hoc to derive complex hemodynamic parameters. 37 While 4D-flow MRI has previously been used in a ground-based experiment to investigate cardiovascular deconditioning in the aorta, 38 no 4D-flow study has examined the cervical or cerebral veins in the context of spaceflight.
MR-based approaches are currently limited to pre- and post-flight measurements due to logistical difficulties in performing imaging aboard the International Space Station (ISS). Head-down tilt (HDT) bed-rest offers a reliable ground-based solution, reproducing key physiological effects of spaceflight such as cephalad fluid shifts and lower body offloading without the cost or difficulty of in-flight experiments. 39 Indeed, several MR-based HDT studies have directly replicated astronaut findings from the ISS in healthy populations, including venous and ocular changes.38,40–47 However, most bed-rest studies suffer from long protocols requiring significant time and financial investment, limiting throughput and feasibility for speedy countermeasure development. 48
Here, we sought to address these gaps by using a multimodal neuroimaging approach to comprehensively characterise the structural and functional changes of the cephalic vasculature under HDT and supine positioning. We additionally aimed to investigate the effect of HDT on brain temperature. We hypothesised that acute HDT would result in (i) an increased area and/or volume of the IJVs and other cranial veins, (ii) reduced flow velocity and/or flowrate through these vessels, and (iii) a slight elevation in TBr, particularly within superficial cortical regions. To our knowledge, this study is the first to concurrently evaluate venous outflow and cerebral thermodynamics under simulated microgravity, as well as the first to include multiple supine control sessions.
Material and methods
Participants and study design
An initial cohort of 30 healthy adults (i.e., without any chronic medical illness) was recruited for this study. All subjects were screened for standard MR-related exclusion criteria through self-reports, for example, MRI-incompatible implants, pregnancy, unstable cardiovascular or neurological conditions, claustrophobia, etc. Additional exclusion criteria included age ⩾ 18 years, a body-mass-index (BMI) < 30 (due to the limited size of the scanner bore during HDT) and head size fitting within the field-of-view (FOV) such that the first four cervical vertebrae were clearly visible. This experiment was carried out in accordance with the Declaration of Helsinki and approved by the ethics committee of the Ludwig-Maximilians-University Hospital. All participants gave written informed consent and received a payment for participation. After initial screening, 27 subjects were deemed suitable for the experiment. Each subject underwent four MRI scanning sessions, divided into two conditions or session-pairs (Supine-Supine and Supine-HDT) to explore the effects of supine and HDT positioning on the variables of interest (Figure 1(a)). The −10° HDT was achieved using a custom-made laser-cut foam incline placed directly onto the scanner bed. Subjects were instructed to rest with their back upon the incline, extended head-down at a >5° orientation with respect to the infraorbitomeatal line cushioned within the head and neck coil for the entire duration of the HDT manipulation. The 10° elevation of the body and legs was the maximum achievable for a scanner with a 60 cm bore. A detailed description of the experimental design can be found in the Supplemental material. Of the screened subjects, four did not complete all sessions and two dropped out due to adverse effects during HDT. Thus, the final cohort consisted of 21 subjects, 10 males, and 11 females aged 24–34 (median ± IQR: 26.6 ± 2.4) with BMI ranging from 16.8 to 26.5 (21.3 ± 2.5).
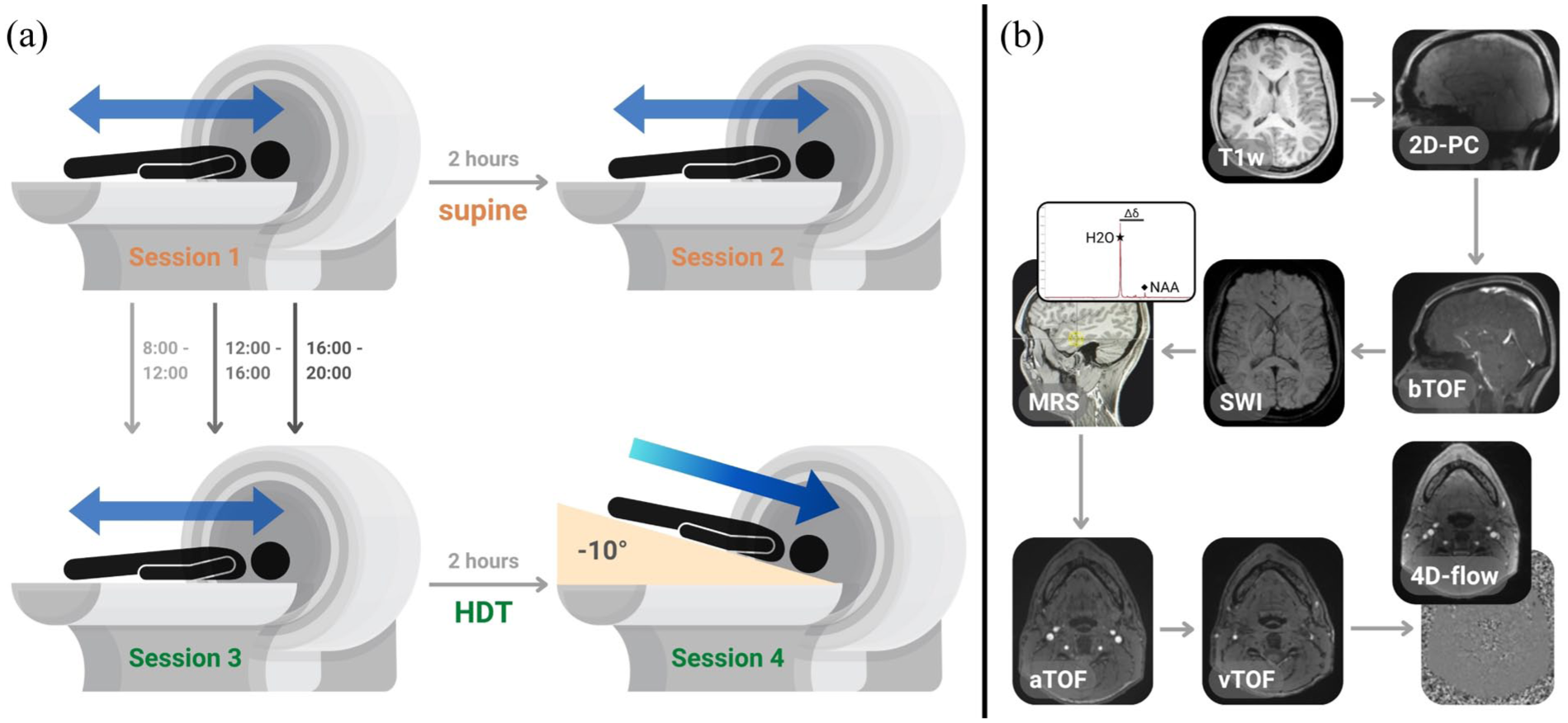
Overview of the experimental design and experimental protocol. (a) Participants underwent four MRI sessions in total: three in the supine position (sessions 1–3) and one after 2 h of −10° head-down tilt (session 4), split into two conditions (Supine-Supine and Supine-HDT). Each condition consisted of a baseline and follow-up session; sessions 1 and 2 were part of the Supine-Supine condition while sessions 3 and 4 were part of Supine-HDT. Both baseline sessions occurred at the same time of day (e.g., morning, afternoon, evening) to control for circadian variation. (b) Eight different scans were collected in each session (total scan time ~1.5 h). A high-resolution T1-weighted (T1w) scan served as the anatomical reference for all subsequent scans. Blood flow and vessel geometry were assessed using 2D phase contrast (2D-PC), Time-of-Flight (across the intracranial veins, cervical arteries, and cervical veins—bTOF, aTOF, and vTOF, respectively), Susceptibility-weighted imaging (SWI), as well as 4D-flow MRI. Brain temperature (TBr) was quantified from magnetic resonance spectroscopy (MRS) data.
Alongside demographic variables, we considered additional subject-independent factors that could act as potential confounds. To evaluate changes in core body temperature (TBo) within conditions, tympanic temperature was measured at the start and end of each scanning day using an infrared (IR) ear thermometer (ThermoScan® 7 IRT6520, Braun, Kronberg, Germany). Another laser-based IR thermometer recorded the ambient temperature in the scanning room, inside the scanner bore between same-day sessions, and the facial surface temperature (TSu) from the cheek (Raynger ST, Raytek, Santa Cruz, USA). Outdoor temperature, interscan time, and total scanning duration were also noted down.
MRI data acquisition
MRI data acquisition was conducted on a 3T MAGNETOM Prisma Scanner (Siemens Healthineers, Erlangen, Germany) with a 64-channel head and neck coil. Each scanning session consisted of 11 separate image acquisitions, spanning six different imaging modalities and lasting approximately 90 min per session (Figure 1(b)). Each session began with the acquisition of a high-resolution T1-weighted (T1w) anatomical image (repetition time (TR) = 2060 ms, echo time (TE) = 2.17 ms, flip angle (FA) = 12°, bandwidth = 230 Hz, voxel size = 0.51 mm3 isotropic).
Structural angiography/venography
To create structural angiograms and venograms of the cervical blood vessels, 3D arterial (TE/TR = 3.42/21 ms, FA = 18°, voxel size = 0.3 × 0.3 × 0.5 mm3) and venous (TE/TR = 4.17/23 ms, FA = 18°, voxel size = 0.3 × 0.3 × 0.5 mm3) time-of-flight (TOF) sequences were obtained (aTOF and vTOF, respectively). The middle slab of both scans was centered on the C2/C3 boundary across all subjects. A susceptibility-weighted image (SWI) of the brain was separately acquired to characterize the small venous vasculature (i.e., venules) of the brain (TR = 28 ms, TE = 20 ms, FA = 15°, voxel size = 0.6 mm × 0.6 mm × 1.2 mm, FOV = 220 mm). Lastly, an intracranial venous TOF or bTOF (TE/TR = 4.56/19 ms, FA = 60°, voxel size = 1.0 mm × 1.0 × 2.5 mm) and a 2D-PC-MRI (FOV = 240 mm2, TR = 81.7 ms, TE = 9.89 ms, FA = 15°, velocity-encoding (venc) = 10 cm/s) sequence were performed to investigate structural and functional changes of the main cerebral veins, including the SSS and rectal sinus.
4D-flow MRI
To evaluate time-resolved changes in the blood flow of the major cervical veins, 4D-flow MRI was carried out using a kt-GRAPPA accelerated dual-venc sequence. 49 Dual-venc imaging mitigates the effects of velocity aliasing by combining low- and high-venc acquisitions to accurately capture a broader range of velocities. 49 The scan was also positioned at the C2/C3 level, with a FOV = 175 mm2, TR/TE = 7.5/4.6 ms, slice thickness = 0.9 mm, FA = 15°, venclow = 20 cm/s and venchigh = 40 cm/s. For each cardiac cycle, 6–10 timepoints were reconstructed using prospective ECG-gating; the total acquired number of cardiac time frames depended on the individual heartrate (HR), which ranged from 39 to 100 beats-per-min, leading to scan times between 20 and 7 min (i.e., longer scan times for slower heartrates).
Magnetic resonance spectroscopy
To investigate variations in brain temperature, four single-voxel 1H MRS scans were obtained using a Point RESolved Spectroscopy (PRESS) pulse sequence (TR = 1500 ms, TE = 135 ms, voxel size = 15 mm3 isotropic, FA = 90° and 170 excitations). Each scan was performed in a different region of interest (ROI) to characterize regional temperature gradients. We positioned one ROI in the deep brain (right thalamus, rTha) and three near the cortical surface (left temporal pole (lTPC), left occipital cortex (lOcC), and right prefrontal cortex (rPFC)).
Data analysis
A general quality assessment of all T1w structural scans was conducted using MRIQC and can be found in the Supplement.50,51 All 4D-flow magnitude, SWI, 2D-PC-MRI, and TOF images were registered in native space to the corresponding T1w image using ANTs linear registration. 52
4D-flow MRI
4D-flow MRI data was analyzed using a recently developed semi-automated pipeline in MATLAB. 53 After preprocessing (including eddy current correction, noise masking, phase unwrapping, and velocity anti-aliasing), the low and high-venc data were combined to reconstruct a dual-venc dataset and a corresponding 3D-PC-MR angiogram (PC-MRA). 49 A maximum intensity projection (MIP) of this 3D-PC-MRA image served as the anatomical basis for segmentation of the cervical veins.
An overview of the segmentation pipeline can be seen in Figure 2(a). Briefly, pre-processed dual-venc 4D-flow MR images underwent region growing, centreline identification, and cut-plane extraction to obtain hemodynamic information for the left and right IJVs (lIJV and rIJV, respectively). Six hemodynamic variables were quantified based on previous work9,42: cross-sectional area (CSA, mm2), stroke volume (SV, mL per cardiac cycle), mean flowrate (MFR, mL/s), peak and mean velocity over the cardiac cycle (vpeak and vmean, cm/s), and the time-averaged volumetric flow per unit area, dubbed normalized MFR (nMFR, mL/s per mm2). Color-coded peak systolic 3D streamlines were generated from velocity data in MATLAB to visualize overall flow pattern changes across sessions.
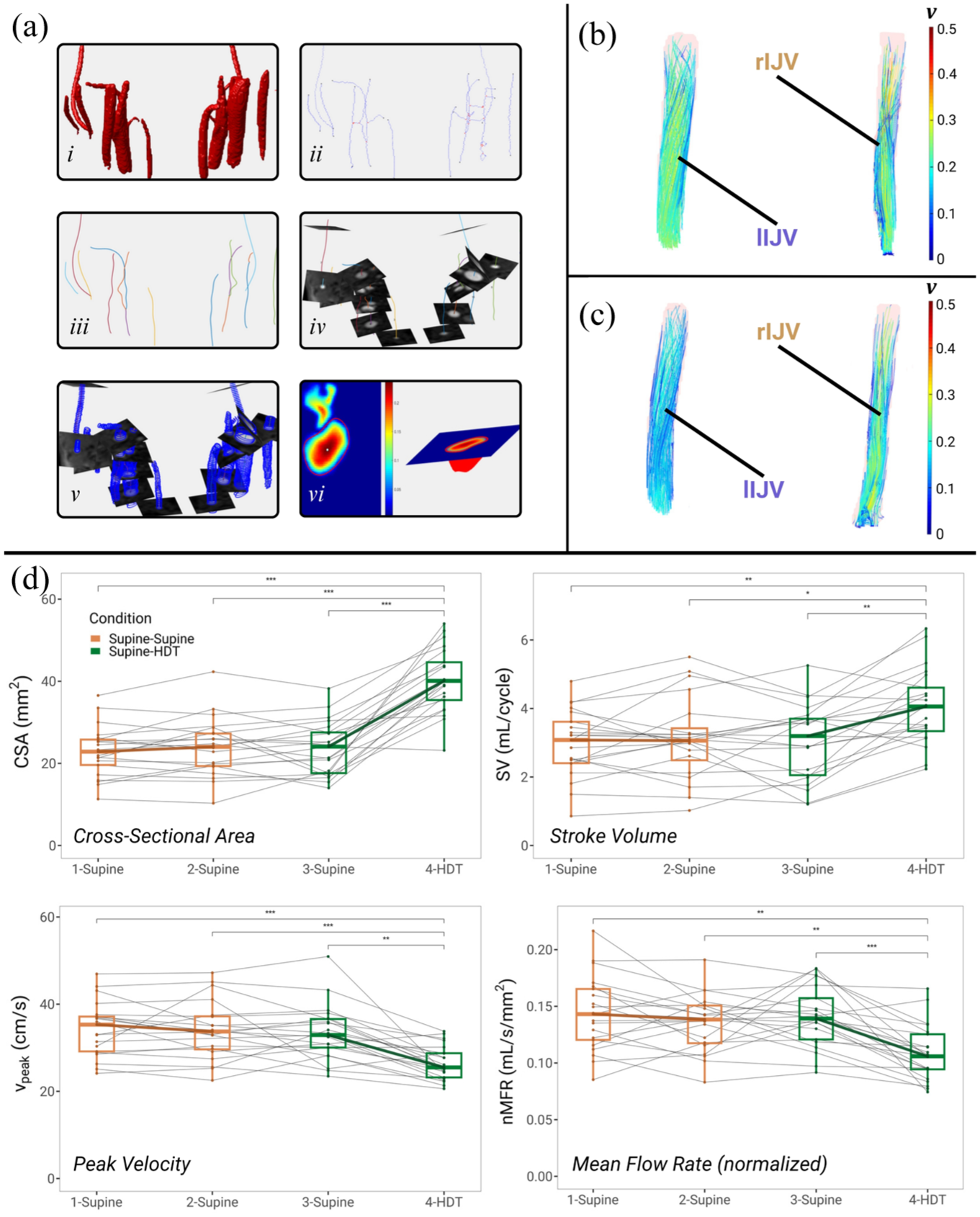
Segmentation, streamline visualization, and selected flow metrics from the internal jugular veins. (a) Representative pipeline for IJV segmentation and 4D-flow analysis, showing: (i) PCMRA-derived 3D vascular geometry, (ii) region growing and centerpoints after skeletonization, (iii) centerline identification (iv) cut-plane overlays, (v) ROI overlay, and (vi) sample flow profile. (b–c) Streamline visualization of velocity vectors from the right (rIJV) and left (lIJV) IJVs of a representative subject. A clear reduction in flow velocity can be appreciated during (b) HDT (session 4) compared to (a) supine baseline (session 3). (d) Group-level changes across all sessions for the cross-sectional area (CSA), stroke volume (SV), peak velocity (vpeak), and normalized mean flow rate (nMFR), derived from time-averaged 4D-flow data. Data are plotted as paired values per subject with boxplots overlaid for group metrics.
2D-PC and SWI
Both 2D-PC-MRI and SWI images were analyzed using a custom pipeline in Python v3.12.7, starting with N4 bias field correction, non-local means denoising, registration, and skull-stripping using SynthStrip. 54 After intensity normalization within a pre-defined window, Otsu thresholding and morphological operations were applied to the 2D-PC data to isolate the cranial venous sinuses of interest. The resulting binary mask was quantified to obtain a total combined volume (in cm3) of the SSS and rectal sinus followed by a pairwise analysis using an in-house MATLAB-based tool. The maximum velocity of the SSS (vsss) was calculated from the masked phase image based on the pre-determined venc (10 cm/s). A modified version of Braincharter in Python v3.12.7 was used for segmentation of the SWI data, 55 specific details on which can be found in the original publication and Supplemental material. The binary vessel masks created by this pipeline were similarly quantified to obtain a total volume (in cm3) and voxel-wise statistics for the cortical venules and veins.
TOF angiography
All TOF images were bias field corrected, denoised, and smoothed with a 1 mm Gaussian filter. A contour-based morphological approach removed extraneous skin and subcutaneous fat signals for better vessel identification and segmentation. The resulting stripped images were intensity-thresholded to obtain MIPs of the major cervical veins and arteries in the imaging slab. These vessels included the facial, jugular, and vertebral veins in the vTOF, and the vertebral and carotid arteries in the aTOF. For the bTOF, we used SynthStrip to remove extraneous signals from the skull before thresholding and MIP creation to obtain a mask of the cerebral veins. All TOF images were quantified in a similar fashion to the SWI and 2D-PC images to obtain a total volume (in cm3).
MRS thermometry
After conversion into NIfTI format using spec2nii, 56 eddy-current-corrected scans were batch processed using jMRUI v7.0. 57 All spectra underwent a visual quality inspection for motion artifacts, spurious peaks, excessive noise, and/or insufficient water suppression. 58 Preprocessing consisted of signal conjugation, phase and frequency correction, zero-filling, and apodization denoising of the of the free induction decay signal. 59 To determine the frequency shift difference (Δδ, ppm) between water (H2O) and N-acetylaspartate (NAA), time-domain fitting of the NAA and H2O resonance with a Gaussian lineshape model was carried out using the in-built jMRUI AMARES algorithm. 60 The algorithm was fitted using 5 peaks such that the H2O, NAA, Cho, and both Cr signals were captured; if this failed, fitting was attempted with additional peaks. Temperature per ROI (TlOcC, TlTPC, TrPFC, TrTha) was calculated using the equation most recently described by Rzechorzek et al. 34 calibrated in human brain tissue (TRz):
As an additional validation step, temperatures were estimated with two other commonly used equations in the literature, the original proposed by Cady et al. 61 (TCa) and the default jMRUI formula from Zhu et al. 62 (TZh).
Global TBr was calculated by taking the simple average of the temperature readings from the four ROIs. Spectral linewidth and signal-to-noise ratio (SNR) were recorded as quality checks.
Statistical analysis
Data visualization and preliminary descriptive statistics were performed in R v4.4.163,64; final statistical analyses were performed using IBM SPSS Statistics v29.0. 65 All data were first assessed for normality using the Shapiro–Wilk test, followed by a repeated-measures analysis-of-variance (rmANOVA) on the normally-distributed primary measures of interest, with session as the main within-subject factor. The ROI was considered as an additional factor in the analysis of 4D-flow variables (lIJV or rIJV) and temperature (lOcC, lTPC, rPFC, rTha). In instances where the assumption of sphericity did not hold, Greenhouse-Geiser-corrected F-statistics were reported instead.
To account for between-subject variance, age, HR, and BMI were considered as covariates in secondary analyses of most variables. Correlations between the covariates and variables of interest were assessed through Pearson and Spearman correlations, with strong correlations (r > 0.5) further analyzed using simple linear regressions. Follow-up pairwise comparisons within and across conditions were investigated with paired-sample t-tests for each variable of interest. The Wilcoxon Signed Rank-test was used as the non-parametric equivalent. Normally distributed continuous variables are reported with mean ± SD; non-normal variables are reported as median ± IQR. Statistical significance was defined as p < 0.05 (two-sided), with trends considered as 0.05 ⩽ p ⩽ 0.1. Correction for multiple comparison testing was performed using the Benjamini–Hochberg False Discovery Rate (FDR) procedure. 66
Results
Subjects were randomly split based on start time, with seven subjects per time block (morning, afternoon, evening); there were no differences in age or BMI between blocks (p > 0.5). No significant changes in tympanic (TBo), scanner, or ambient room temperature were observed across sessions (Table 1). However, there was a significantly larger increase in TSu in the Supine-HDT condition compared to Supine-Supine (+2.4°C vs +1.6°C, difference: 0.7°C ± 0.9°C). An overview of the rmANOVA and baseline versus follow-up pairwise comparisons for all major variables of interest can be found in Table 2. Further demographic characteristics, comparisons with Supine-Supine sessions, and covariate analyses can be found in the Supplemental material.
External temperature and scanning-related variables (n = 21).
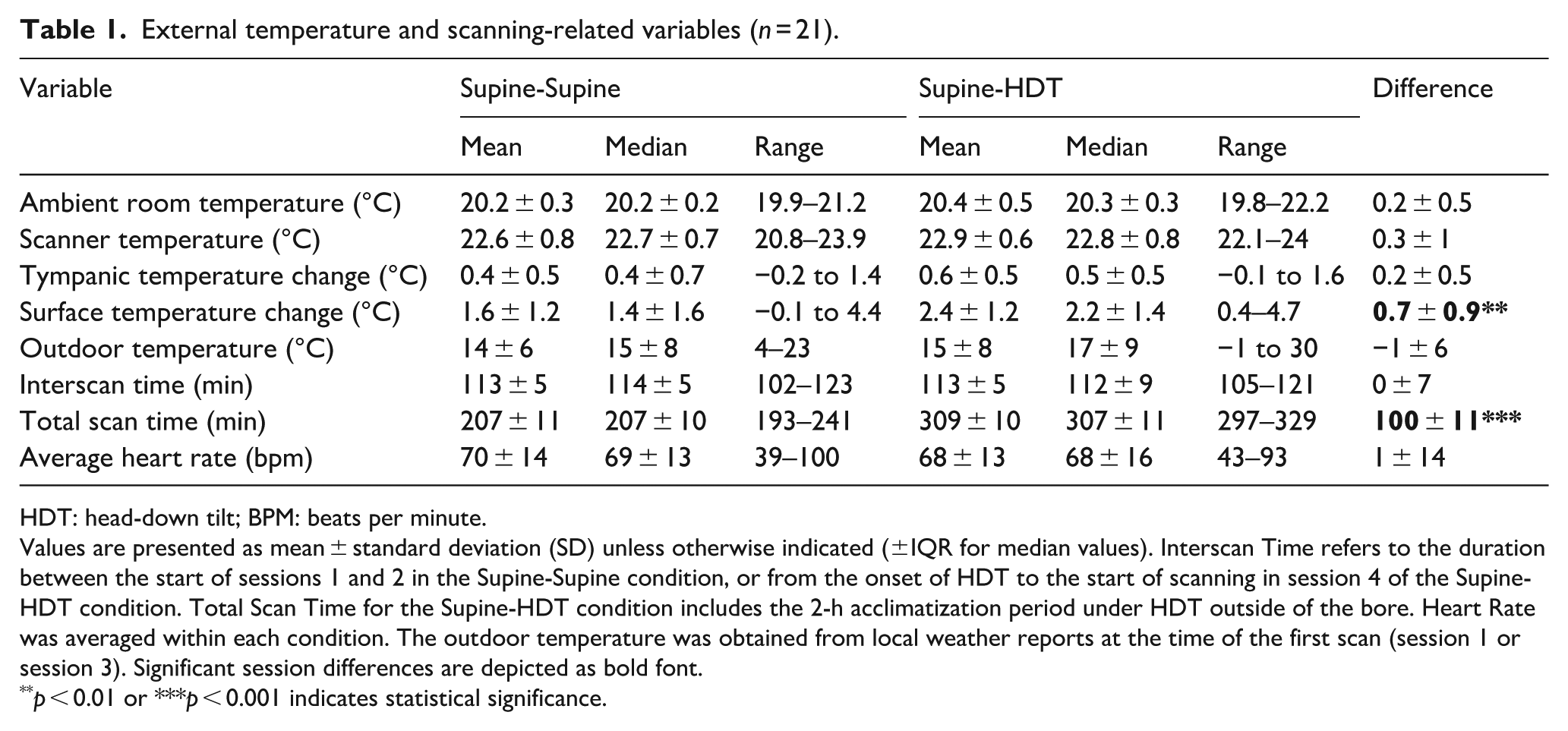
HDT: head-down tilt; BPM: beats per minute.
Values are presented as mean ± standard deviation (SD) unless otherwise indicated (±IQR for median values). Interscan Time refers to the duration between the start of sessions 1 and 2 in the Supine-Supine condition, or from the onset of HDT to the start of scanning in session 4 of the Supine-HDT condition. Total Scan Time for the Supine-HDT condition includes the 2-h acclimatization period under HDT outside of the bore. Heart Rate was averaged within each condition. The outdoor temperature was obtained from local weather reports at the time of the first scan (session 1 or session 3). Significant session differences are depicted as bold font.
p < 0.01 or ***p < 0.001 indicates statistical significance.
Group-level means and statistical comparisons for venous flow and temperature metrics.
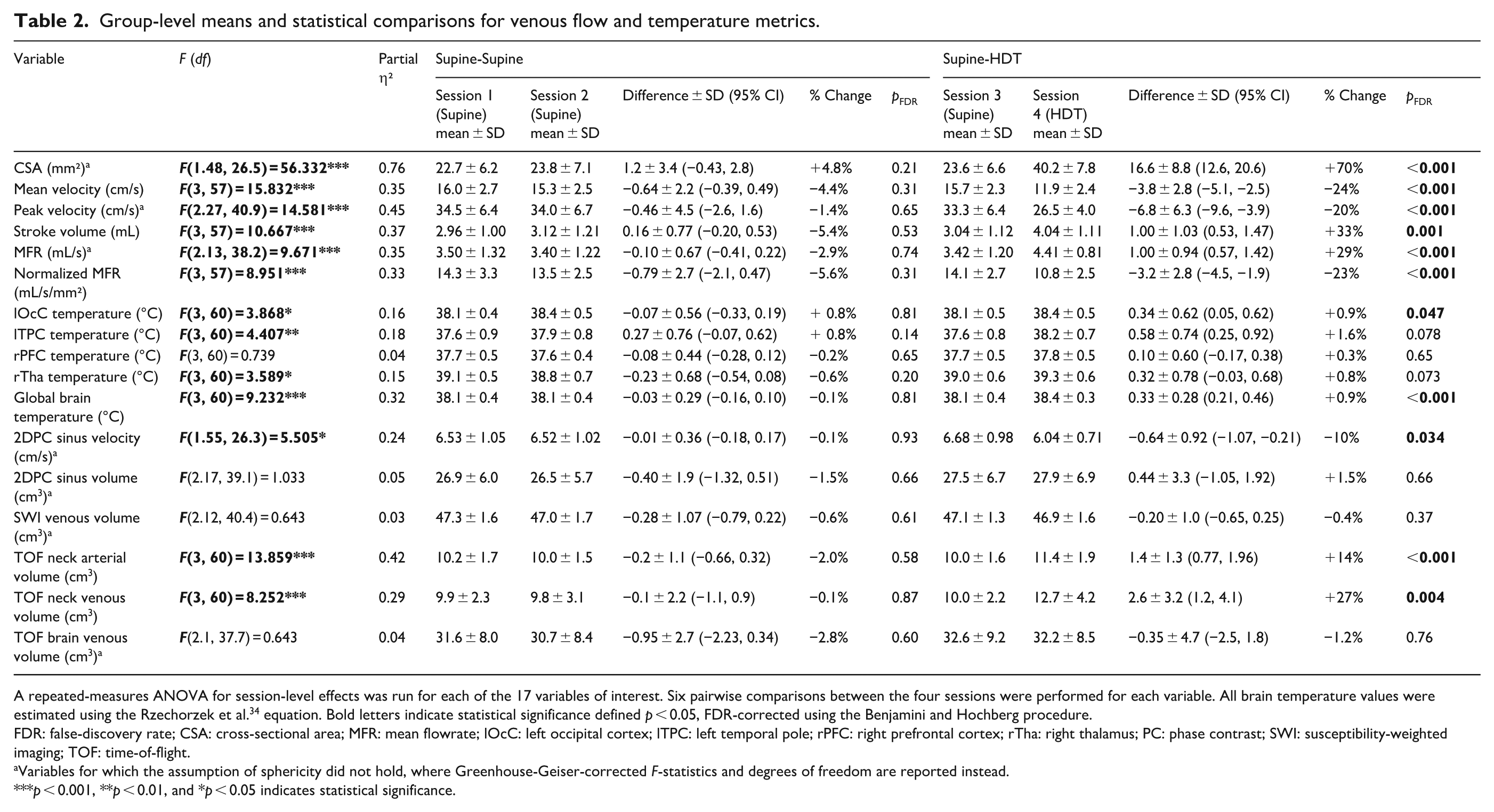
A repeated-measures ANOVA for session-level effects was run for each of the 17 variables of interest. Six pairwise comparisons between the four sessions were performed for each variable. All brain temperature values were estimated using the Rzechorzek et al. 34 equation. Bold letters indicate statistical significance defined p < 0.05, FDR-corrected using the Benjamini and Hochberg procedure.
FDR: false-discovery rate; CSA: cross-sectional area; MFR: mean flowrate; lOcC: left occipital cortex; lTPC: left temporal pole; rPFC: right prefrontal cortex; rTha: right thalamus; PC: phase contrast; SWI: susceptibility-weighted imaging; TOF: time-of-flight.
Variables for which the assumption of sphericity did not hold, where Greenhouse-Geiser-corrected F-statistics and degrees of freedom are reported instead.
p < 0.001, **p < 0.01, and *p < 0.05 indicates statistical significance.
Flow dynamics of the internal jugular veins
The 4D-flow MRI data for session 2 was motion corrupted in two subjects and thus excluded from the respective within-subject analyses. Streamline visualizations of IJV flow revealed extensive inter-individual variability in jugular hemodynamics, but largely confirmed the general trends seen in the quantitative analysis (Figure 2(b) and (c)). No significant main effects of vessel (i.e., lIJV vs rIJV) were observed for any flow variables (all p > 0.05), and session by vessel interactions were also statistically insignificant for CSA (p = 0.846), SV (p = 0.811), MFR (p = 0.824), nMFR (p = 0.380), vmean (p = 0.576), and vpeak (p = 0.513). Accordingly, rmANOVA results are reported as an average across both IJVs unless otherwise specified.
We observed a significant cross-sectional expansion of the IJVs following head-down tilt (HDT) compared to all supine sessions (Table 2 and Supplement; p < 0.001). When averaged across both IJVs, CSA increased by approximately 70% between sessions 3 and 4 (Table 2; pFDR < 0.001). There was a larger percent increase in the lIJV compared to the rIJV (80.7% vs 61.2%); however, this difference was not statistically significant (t₂₀ = 0.425, p = 0.675). Overall, rIJV-CSA was ~20% larger on average than lIJV-CSA across all sessions (31.3 ± 13.0 mm2 ± vs 24.5 ± 12.2 mm2; Z = 3.779, p < 0.001). Interestingly, the degree of change in CSA between sessions 3 (supine) and 4 (HDT) correlated with BMI (ρ = 0.444, p = 0.044).
There was a significant slowdown in jugular vein flow velocity during HDT, with vpeak and vmean decreasing by ~20% and ~24%, respectively (Table 2; p < 0.001). Compared to session 3, the vpeak in the lIJV was 9.21 ± 7.95 cm/s slower in session 4 (pFDR = 0.001), with a similar, but insignificant, trend seen in the rIJV (−4.66 ± 9.89 cm/s; pFDR = 0.09). There was a slightly smaller asymmetry when looking at the vmean, with HDT leading to a similar slowdown in both vessels (lIJV: −4.43 ± 4.53 cm/s, rIJV: −3.19 ± 2.82 cm/s; both pFDR < 0.001). SV was on average ~33% higher during the Supine-HDT condition (lIJV: 0.80 ± 1.34 mL, t20 = 2.714, pFDR = 0.027 and rIJV: 1.14 ± 1.55 mL; t20 = 3.350, pFDR = 0.019). We observed a similar 29% average increase in the MFR between sessions 3 and 4 (Table 2; pFDR < 0.001) with a 0.70 ± 1.35 mL/s increase in the lIJV (pFDR = 0.047) and 1.22 ± 1.57 mL/s increase in the rIJV (pFDR = 0.01). Across all sessions, the SV and MFR were consistently higher in the rIJV at the group level (SV: 0.75 ± 2.72 mL/beat; Z = 2.124, p = 0.034; MFR: 0.92 ± 3.05 mL/s; p = 0.008).
On the other hand, we observed a significant reduction in the per second flow of blood during session 4 when controlling for the IJV-CSA. Namely, nMFR was on average 23% lower during HDT compared to the same-day supine baseline (Table 2, pFDR < 0.001). While there were no contralateral differences in nMFR during session 4 (lIJV: 0.106 ± 0.04 mL/s/mm² vs rIJV: 0.112 ± 0.03 mL/s/mm², p = 0.48), a more significant drop in nMFR was seen on the left side (lIJV: −0.044 mL/s/mm² vs rIJV: −0.018 mL/s/mm²; t20 = 2.651, p = 0.015).
Structural changes in the other cephalic vessels
2D-PC data was missing for three subjects, and they were excluded from the group-level analysis. Across all subjects, mean vSSS decreased by 10% between sessions 3 and 4 (Table 2; pFDR = 0.034). Correlation analysis with 4D-flow metrics revealed that vSSS was directly related with nMFR (r = 0.28, p = 0.014), vpeak (r = 0.29, p = 0.011), and vmean (r = 0.31, p = 0.007). While we found no significant differences between sessions in the group-level volumetric analysis of the 2D-PC or SWI masks (Table 2), voxel-wise analysis suggested a significant expansion of the cortical venules after HDT (Figure 3). Furthermore, correlations with IJV flow parameters suggest that the volume of the major venous sinuses covaried with some 4D-flow variables, including CSA (ρ = 0.26, p = 0.02), SV (ρ = 0.30, p = 0.008), and MFR (ρ = 0.36, p = 0.001).
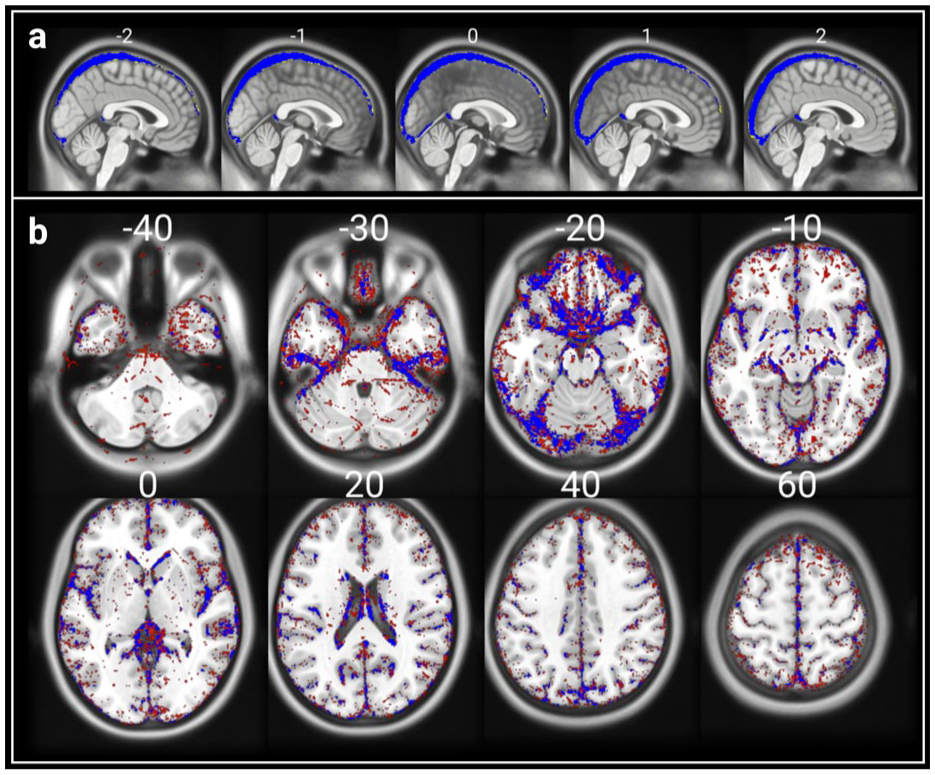
Cranial venous vascular segmentation findings across subjects. (a) Mean superior sagittal and rectal sinus volume across supine sessions (blue) showed only minimal increase for the HDT session (increase in yellow). (b) Cortical venules segmented from SWI on the other hand showed a substantial increase (red) in caliber and visible number compared to the general venous vasculature (blue) found during the supine sessions across all subjects.
Quantitative analysis of the vascular masks derived from TOF images revealed a volumetric expansion of both the arterial and venous cervical vasculature during HDT (Figure 4). Specifically, we saw an increase in arterial vessel volume by 14% between sessions 3 and 4 (Table 2; pFDR < 0.001), while venous volume increased by 27% (pFDR = 0.004). However, no significant changes were seen across sessions in the volume of the cranial veins obtained from the bTOF mask (Table 2, p = 0.76). Cervical arterial volume was strongly related to the CSA (r = 0.417, p = 0.002), SV (r = 0.393, p < 0.001), and MFR (r = 0.453, p < 0.001). Similarly, the volume of the vTOF mask was positively correlated with the CSA across all sessions (r = 0.339, p = 0.002), with the degree of change during the Supine-HDT condition closely related to the increase in IJV-CSA as obtained from the 4D-flow measurements (r = 0.563, p = 0.008).
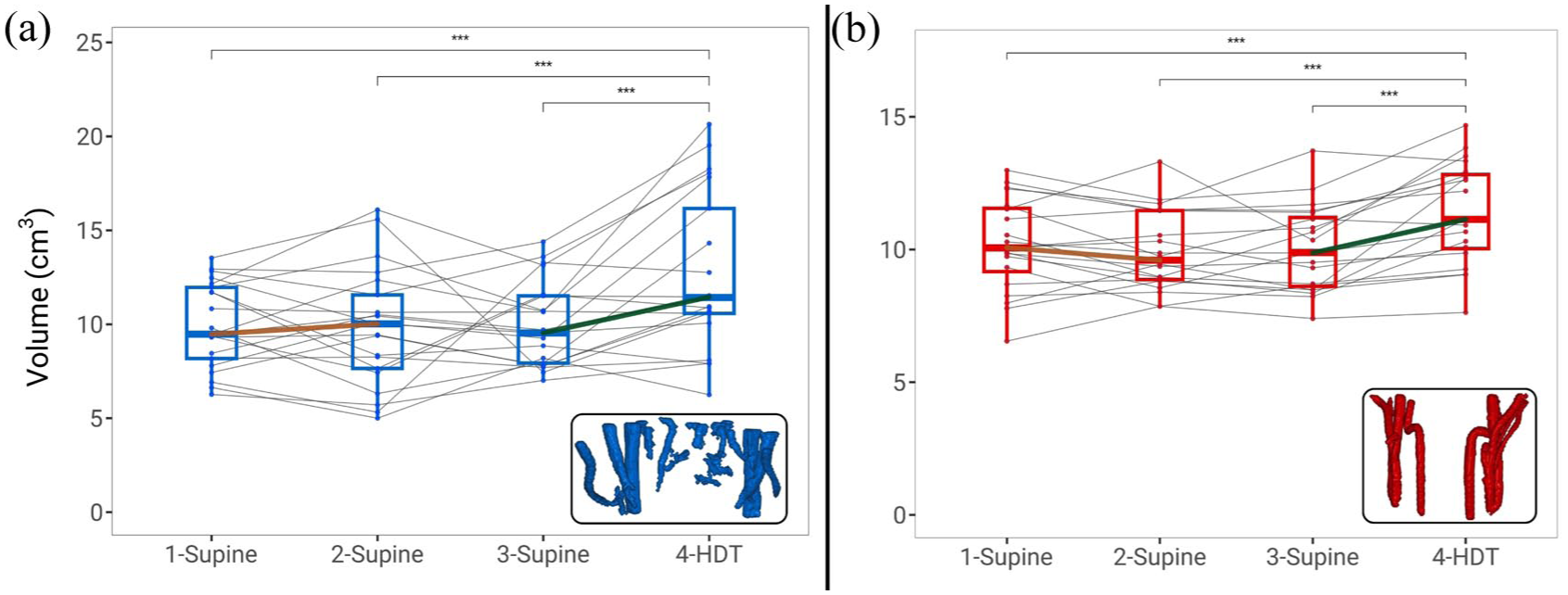
Volumetric changes in the cervical vasculature across sessions. (a) Volume of the venous time-of-flight (vTOF) mask across all sessions, centered on the C2/C3 vertebral boundary. Individual subject values are shown with overlaid boxplots and insets display a representative vTOF segmentation used for quantification. A significant expansion in volume was seen during HDT, mirroring 4D-flow results. (b) Volume of the arterial time-of-flight (aTOF) mask across sessions, where a post-HDT increase in arterial volume was also observed. Inset shows a representative aTOF mask.
Elevation of global brain temperature
A two-way rmANOVA on TBr revealed a significant main effect of session, regardless of the calibration equation used (TRz: F2.12, 42.5 = 9.232; TCa: F3, 60 = 9.211; TZh: F3, 60 = 9.356; all pFDR < 0.001). Nonetheless, the absolute temperature estimates produced by each calibration equation were significantly different across all sessions (TZh > TRz > TCa, 38.4 ± 0.4°C vs 38.2 ± 0.4°C vs 37.5 ± 0.4°C; p < 0.001). As TRz values generally fell between TCa and TZh, we report findings here according to this equation 34 ; results for the other two equations can be found in the Supplement. Overall, our data suggests a mean TBr of 38.1 ± 0.4°C in the supine position (averaged across sessions 1, 2, and 3 from all subjects). MRS-derived TBr across all sessions was significantly higher relative to TBo and TSu and displayed much less variability (Figure 5(c); 38.2 ± 0.3°C vs 36.7 ± 0.3°C and 33.2 ± 0.5°C, respectively; t19 = 14.4, p < 0.001; t19 = 46.2, p < 0.001).
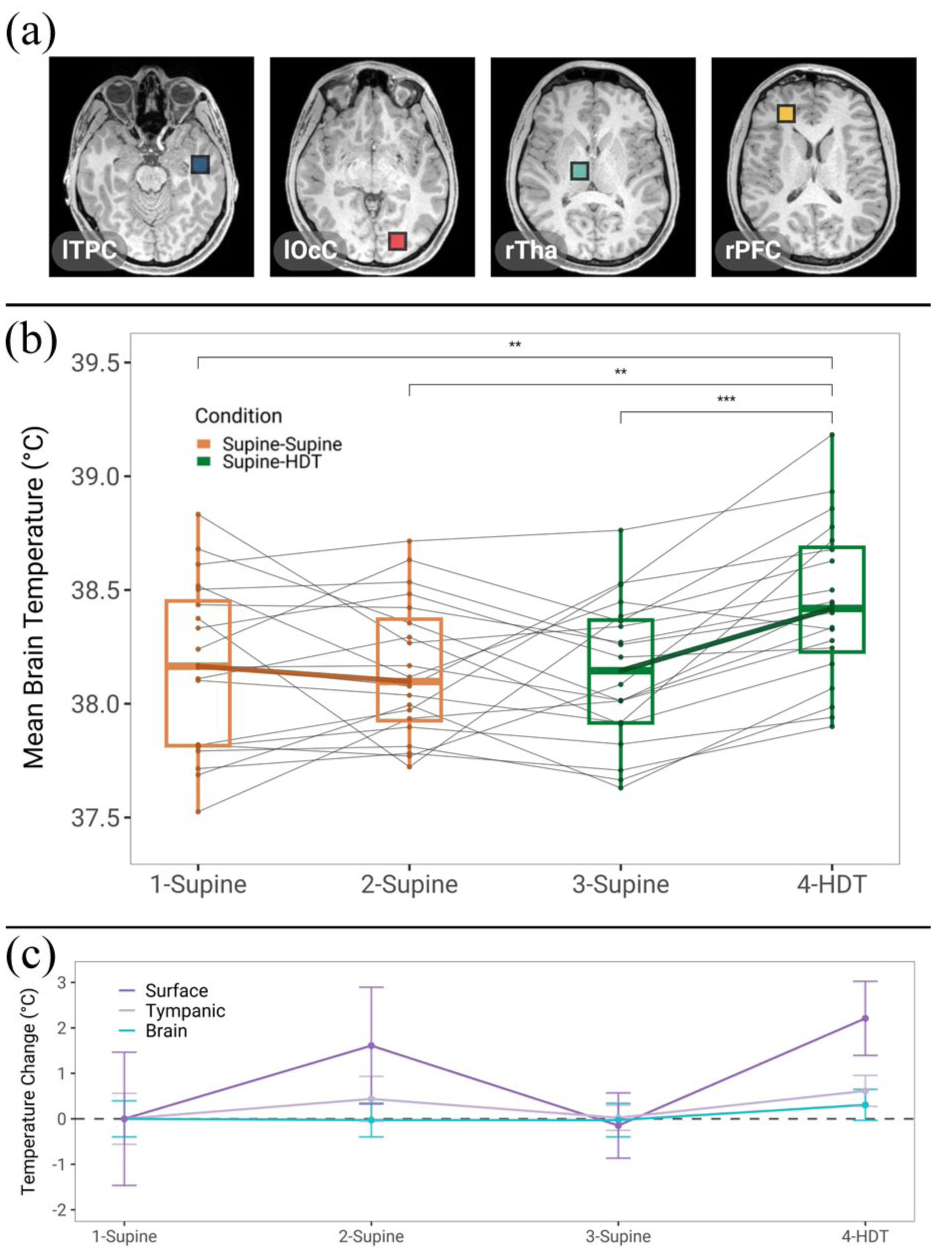
Temperature changes across sessions. (a) MRS voxel placements for four regions of interest (ROIs): left temporal pole (lTPC), left occipital cortex (lOcC), right thalamus (rTha), and right prefrontal cortex (rPFC). (b) There was a significant elevation in mean global brain temperature (TBr) following HDT (session 4), with generally no differences between supine sessions. Individual subject trajectories are overlaid. (c) Change in surface, core body (tympanic), and brain temperature from baseline. While surface temperature increased significantly across both conditions, the change in Supine-HDT was greater than Supine-Supine. Overall, there was much greater variation in surface and tympanic temperatures than in the brain.
The global TBr in session 4 was ~0.3°C higher than all supine sessions (Figure 5; all pFDR < 0.01). Further analysis revealed that the elevation in global TBr between sessions 3 and 4 was closely tied to the change in the TlOcC (r = 0.61, p = 0.003) and TrTha (r = 0.77, p < 0.001). However, only the increase in TlOcC was significant after correction for multiple comparisons (Table 2; t20 = 2.58, pFDR = 0.047). While trends were occasionally seen for the other ROIs, all pairwise comparisons between sessions were statistically insignificant (pFDR > 0.05). Notably, regression analysis showed that the CSA, MFR, vpeak, and vmean all had a significant effect on global TBr (F4, 78 = 3.41, R2 = 0.149, p = 0.013).
Discussion
To our knowledge, this is the first study to investigate the effects of HDT on the hemodynamic and thermal regulation of the brain. By combining phase-contrast, spectroscopy, and angiography techniques, we demonstrate that even ~2 h of −10° HDT can lead to noticeable changes in the dynamic and structural properties of the IJVs, cervical veins and arteries, intracranial veins, and major venous sinuses. We additionally report a global elevation in TBr without consistent regional changes. These effects were largely independent of the time spent in the supine position and varied considerably between individuals. The findings support the role of impeded cranial venous outflow as a driver for CMR.
As a principal finding, we observed significant changes in all the hemodynamic parameters of interest following HDT. Most notably, the left and right IJV-CSA increased by 81% and 61%, respectively, replicating previous findings in space and using ground-based analogues.9,42,45,67–70 We saw a comparative volumetric expansion in the TOF-derived venous masks highly correlated with the change in CSA, serving as an anatomical proof of concept.4,71–73 Since the IJVs are thin-walled vessels, they are prone to changes in central venous pressure (CVP) and will expand in situations where venous flow is inhibited.74,75 Thus, this vasodilatory response also implies an elevation of CVP (or intracranial pressure (ICP) more generally) due to a cephalad fluid shift-mediated stagnation of venous outflow.70,71,76,77 Moreover, the obtained values for SV and MFR suggest an increased total blood volume in both IJVs as predicted by mathematical modelling 78 in similar acute HDT settings.44,45,79 At first glance, greater venous outflow from the head without a commensurate increase in arterial inflow appears to violate the Monro–Kellie doctrine.80,81 Although dynamic flow information for the cervical arteries was not acquired, the TOF-derived arterial mask provides strong evidence that arterial inflow did in fact increase during HDT, mirroring results from earlier studies.38,79,82–84 Since the cranium naturally has a lower vascular resistance than most other peripheral areas, this result likely indicates both an exacerbated hydrostatic pressure gradient from heart to head in HDT as well as central vasodilatory adjustments to maintain cerebral perfusion. 85 Lastly, the increased MFR could also reflect a further transfer of venous outflow toward the IJVs similar to the shift of primary venous drainage when going from an upright to supine posture; as the other cervical veins were not included in the 4D-flow analysis, we cannot rule out this possibility.44,86
Given that the increase in IJV-CSA was nearly twice the drop in vmean, we would expect a much larger volume flowrate than observed from a fluid dynamics standpoint (Q = A × v). This mismatch indicates less efficient venous blood offloading and is supported by our analysis with nMFR, which reflects changes in flowrate independent of vessel CSA. Specifically, we saw a 23% drop in nMFR during HDT, consistent with the notion of cranial blood pooling and observations of facial edema (“puffy faces”) in astronauts during early spaceflight. 18 Both v and v also decreased by ~20% post-HDT, with patches of near-static flow near vessel edges in many subjects, mirroring earlier results.10,44,45,87,88
Considering that slow venous flow is a known risk factor for thrombosis in the lower extremities, 89 these results provide additional evidence for a link between IJV flow stasis in microgravity and elevated blood clot risk.8–11 Furthermore, our results show that this slowdown in the IJVs is closely tied to a reduction in vSSS. As the brain’s major site of venous and CSF drainage, a HDT-mediated congestion of SSS outflow would naturally slow return downstream; this is corroborated by both mathematical modelling and empirical observations.72,90–94 Although correlation results suggested a direct relationship between IJV-CSA and SSS volume, there were no group-level volumetric changes of the 2DPC- and TOF-derived sinus masks. Since the major dural sinuses sit in rigid connective tissue that limits their distensibility, 16 a measurable enlargement in an acute setting is quite unlikely. 70 In fact, Rosenberg et al. 5 showed that the SSS only expands after weeks of microgravity exposure, and moreover, exclusively in astronauts who developed SANS. Lastly, the voxel-wise expansion of the SWI-derived masks during HDT suggests venous congestion in major cranial veins may also be having an upstream effect on drainage from the cortical venules. Since smaller cerebral veins are generally more vulnerable to changes in venous pressure, this may lead to an elevated risk of microbleeds and associated cerebrovascular complications during LDSF. 6
Previous work suggests the lIJV is anatomically more vulnerable to flow disruption in microgravity due to its relatively perpendicular angle of entry into the left subclavian vein.78,95,96 Our results further support this claim, with both vpeak and nMFR showing more significant decreases in the lIJV compared to the rIJV during HDT. Even the consistently greater SV, MFR, and CSA of the rIJV across sessions can be attributed to more efficient outflow resulting from differences in lower IJV resistance,92,97 resembling findings from other microgravity experiments.10,45,69,98 However, we did not find any vessel-based effect at the level of vertebra C2, suggesting the response to the rather short HDT manipulation eclipsed any bilateral differences so close to the skull.92,97
These individual responses were likely modulated by factors such as BMI, since higher BMI was linked to greater CSA expansion during HDT. In fact, greater intrathoracic pressures in heavier individuals could lead to increased CVP and IJV expansion, likely intensifying the effects of HDT compared to lower BMI subjects.99,100 Additionally, we observed that older participants tended to have lower stroke volumes, possibly reflecting age-related declines in vessel compliance.75,101 Lastly, the strong correlation between HR and SV demonstrates that 4D-flow-derived volumetric flow metrics are often confounded by HR variability.102–104 Since astronauts are typically younger, fitter, and male compared to the civilian population,16,105,106 future studies must account for such individual heterogeneity in the next generation of spacefarers.
To our knowledge, this protocol is the first to implement 4D-flow MRI with MRS to jointly examine venous blood flow and brain temperature. Our major finding is a discrete but consistent ~0.3°C rise in global TBr following HDT, as suggested by the data of Stahn et al. 29 upon arrival in microgravity. This unprecedented result suggests that thermoregulatory impairment in microgravity extends beyond the body to include the brain as well.28–30,107,108 There is compelling evidence that compromised CBF leads to an increase in TBr,22,24,26,109,110 and that the venous system plays a crucial role in cerebral autoregulation.111–113 Hence, stagnant venous flow due to a microgravity-mediated cephalad fluid shift would likely prevent an adequate offloading of metabolic heat through the cerebral veins and sinuses, contributing to an elevation in TBr as we observe here and supported by the regression results. This increase may have been driven by the temperature changes in the lOcC and rTha as they correlated most strongly with global TBr during HDT. The higher CSF and GM fractions in these regions could have rendered them more vulnerable to venous stasis-induced impairment of glymphatic heat clearance, particularly given the elevated metabolic activity of GM.22,114,115
By demonstrating that HDT had no effect on TBo and that TBo remained significantly lower than TBr across all sessions, we illustrate how surface-level temperature readings cannot be used as a reliable proxy for brain tissue temperature changes.29,30,34,116,117 Furthermore, while the significant increase in TSu during both conditions can be partially explained by the confined scanner environment, the additional 0.7°C difference after HDT points to vascular adjustments (e.g., facial vasodilation) related to the cephalad fluid shift and impaired venous heat offloading.29,107,108,111
Given that central nervous tissue is particularly sensitive to heat, 118 these findings raise significant concerns about overall brain health during space travel. Prolonged increases in brain temperature are linked to neuronal injury, parenchymal edema, and blood-brain barrier leakage. 119 These effects would likely contribute to an accumulation of fluid in the brain, increased ICP, and an exacerbation of symptoms associated with obstructed venous outflow. It also remains unclear whether brain temperature continues to rise in space or stabilizes, similar to CBT, 29 or if the observed changes are simply artifacts of immediate exposure to microgravity, highlighting the need for further research in this area.
The absence of significant changes across all measured parameters during the Supine-Supine condition provides strong evidence that the neurovasculature is well adapted to uniform gravitational gradients. 45 In contrast, HDT introduces a sudden gravitational challenge in the form of a headward fluid shift that resembles the primary physiological mechanisms contributing to the CMR. The strongest evidence for this comes from longer-duration HDT bed-rest studies, which reliably reproduce key neurovascular changes associated with prolonged exposure to microgravity such as cranial venous congestion, IJV distension, PVS expansion, and elevated ICP.39,42–46,120–123 Generally, the longer the duration of bed-rest, the more robust the replication of these effects. However, even SANS-related ophthalmic findings such as optic nerve sheath distension and increased choroidal thickness have been observed after only 1 h of −6° HDT,124,125 further supporting the ability of short-duration protocols like ours to elicit CMR-like responses.
Study limitations and future directions
This study has a few limitations worth considering. First, the short HDT exposure duration (~2 h) and small sample size, while sufficient to detect acute group-level effects, may have limited our ability to observe slower-developing physiological changes.126–128 Second, our 4D-flow analysis did not examine collateral venous outflow (e.g., vertebral venous plexus) and arterial inflow, preventing a proper inflow-outflow comparison, and resolution of Monro–Kellie discrepancies. Additionally, the selected venc range may have been suboptimal for capturing very slow or stagnant flow, potentially leading to underestimation of IJV-CSA or false instances of aliasing.75,129 While temperature changes were largely independent of equation used, the accuracy of absolute temperature estimates with MRS remains limited by the assumptions of each calibration. 60 Spectral quality in deeper ROIs, especially the lTPC, was occasionally reduced, likely due to its high vascularization and corresponding motion artifacts. 33
As mission durations increase and spaceflight becomes more accessible to the general population, the development of tailored countermeasures to enhance astronaut health and safety will become increasingly important. 130 Lower body negative pressure (LBNP) is a leading candidate, proven to improve blood flow, reduce ICP, and decrease venous pulsatility in both HDT and spaceflight settings.73,93,122,131,132 However, the effects of LBNP often dissipate rapidly,67,133 necessitating repeated or prolonged use to sustain benefit and heightening the risk of cardiovascular events like syncope and orthostatic intolerance.134,135 Thus, research into other viable alternatives is urgently needed. Given that the present setup induced observable structural and functional changes within just a few hours, it presents a rapid-turnaround model for the evaluation of future countermeasures against CMR. This paradigm may therefore serve as a practical platform to screen, compare, and optimize interventions, identifying important biomarkers in the process.
Conclusion
As humans have only spent extended month-long periods in space for the past 15 years, data on long-term neurological health remains scarce. 16 Additionally, prevention and management strategies for the adverse effects of spaceflight can only be developed in combination with active astronaut surveillance and analogue-based experiments on Earth.136,137 Such data will be essential in evaluating the long-term impact of weightlessness on the brain, particularly in light of the present findings suggesting impeded cranial venous outflow and elevated TBr in the early phases of microgravity exposure. In conclusion, this work contributes to the growing body of literature on the CMR and introduces a novel experimental model for countermeasure development. 15 Ongoing efforts to safeguard astronaut health will be vital as we set our sights towards the Moon, Mars and beyond.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251400470 – Supplemental material for The immediate functional and structural consequences of head-down tilt on the cranial venous vasculature and brain temperature
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251400470 for The immediate functional and structural consequences of head-down tilt on the cranial venous vasculature and brain temperature by Mehul Nimpal, Patrick Winter, Susanne Schnell and Peter zu Eulenburg in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-docx-2-jcb-10.1177_0271678X251400470 – Supplemental material for The immediate functional and structural consequences of head-down tilt on the cranial venous vasculature and brain temperature
Supplemental material, sj-docx-2-jcb-10.1177_0271678X251400470 for The immediate functional and structural consequences of head-down tilt on the cranial venous vasculature and brain temperature by Mehul Nimpal, Patrick Winter, Susanne Schnell and Peter zu Eulenburg in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
We thank all volunteers for their participation.
Author contributions
Mehul Nimpal: Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Validation, Visualization, Writing – Original Draft, Writing – Review & Editing. Patrick Winter: Methodology, Resources, Software, Writing – Review & Editing. Susanne Schnell: Software, Methodology, Resources, Writing – Review & Editing. Peter zu Eulenburg: Conceptualization, Methodology, Supervision, Funding Acquisition, Validation, Visualization, Writing – Review & Editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Space Agency (DLR) on behalf of the Federal Ministry of Economics and Technology/Energy (50WB2027 to PzE).
Ethical considerations
This experiment was carried out in accordance with the Declaration of Helsinki and approved by the ethics committee of the Ludwig-Maximilians-University Hospital.
Consent to participate
All human participants provided written and verbal informed consent to the experiment before participation, and could withdraw at any time before, during, and after the study without consequence.
Consent for publication
Informed consent for publication of images and data was provided from all participants. Identifying details are omitted wherever possible, and all data was anonymized prior to analysis.
Data availability statement
Processed data and code supporting the findings of this experiment can be made available upon reasonable request to the corresponding author (MN).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.