Abstract
Evidence increasingly suggests that dyslipidemia diminishes dilatory function of resistance arteries by altering ion channel activity. Focusing on the cerebral vasculature, this study investigated whether inwardly rectifying K+ 2.1 (KIR2.1) channels are targeted early in dyslipidemia. Experiments began at the cellular level (patch-clamp electrophysiology), then progressed to isolated arteries (pressure myography) and whole animals (arterial spin-labeling magnetic resonance imaging). Lipid analysis confirmed dyslipidemia in Ldlr−/− (normal chow) and C57BL/6 mice fed a high-fat high-cholesterol (HFHC) diet for 8 weeks; no aortic plaques were observed. Patch-clamp electrophysiology revealed a marked reduction in endothelial but not smooth muscle KIR activity in both dyslipidemic models; this KIR activity was recoverable by plasma membrane cholesterol depletion. These cellular changes notably diminished flow-induced vasodilation in cerebral arteries isolated from both dyslipidemic models; such deficits were observed in endothelial Kir2.1−/− arteries. A blood pressure challenge induced a perfusion phenotype in HFHC-C57BL/6 but not genetic deletion (Ldlr−/−) mice, consistent with reduced KIR activity and flow-mediated dilation. Our findings highlight that endothelial KIR2.1 channels are targeted early in dyslipidemia, which was associated with attenuated flow-mediated dilation in our acute HFHC model. This change likely moderates the range of blood flow control and substrate delivery to active brain tissue.
Introduction
The optimization of neuronal function necessitates tight regulation of cerebral perfusion by a network of resistance arteries. This is accomplished by sensing hemodynamic stimuli such as flow and pressure, which together set base arterial tone, the foundation upon which local neurovascular stimuli elicit dilation (neurovascular coupling). Mechanistically, arterial tone is driven by the balance of depolarizing and hyperpolarizing currents in smooth muscle cells (SMCs) and endothelial cells (ECs), which set membrane potential (VM) and Ca2+ influx through voltage-gated Ca2+ channels.1–4 One of these key currents is encoded by inwardly rectifying K+ (KIR) channels, notable for their strong inward rectification, potentiation by extracellular K+, and blockade by micromolar Ba2+.5,6
KIR channels exhibit a distinct cell-specific expression pattern in cerebral arteries, with KIR2.1 dominating in ECs and KIR2.2 in SMCs.7,8 Both channel pools display mechanoregulation, with flow increasing EC KIR activity and pressure inducing the opposite in the SMC pool—effects conferred by specific lipid-channel interactions. 9 This lipid regulatory effect is intriguing when considered in the context of pathobiology and the functional consequences of dyslipidemia. Consider, for example, EC KIR channels and their interaction with phosphatidylinositol 4,5-bisphosphate (PIP2), which typically places the channel in a preferred open state. If replaced with cholesterol—plausible during the dyslipidemic state—the channel will be pushed to a preferred closed state, and lose its flow sensitivity, a key endothelial feedback response. This line of thinking began to mature as work examined KIR2.1 activity in mesenteric arteries from apolipoprotein E-deficient (Apoe−/−) mice. 10 As expected, flow-mediated responses, albeit at the channel or vessel level, were markedly diminished. 10 While a valuable first step, this work is delimited by the severity of its animal model, its focus on peripheral arteries, and the absence of macrohemodynamic measurements.10,11 Extending their investigation, the authors also confirmed that shear-stress activation of KIR is blunted in mesenteric arteries in a milder mouse model of lipidemia with glycocalyx degradation as the underlying mechanism. 12 Intriguingly, EC KIR2.1 dysfunction was also observed in adipose arteries harvested from morbidly obese patients with elevated low density lipoprotein (LDL) levels. 12 However, questions remain as to whether EC KIR2.1 channel dysfunction arises early in the dyslipidemia response in vital organs, such as the brain, prior to the advent of structural manifestations.
The objective of this study was to determine the impact of early, mild dyslipidemia on endothelial KIR channels in cerebral arteries. We focused on two mouse models, C57BL/6 mice fed a high-fat high-cholesterol (HFHC) diet and the low-density lipoprotein receptor knockout (Ldlr−/−) on normal chow, both of which display dyslipidemia without plaque formation. Whole-cell patch-clamp electrophysiology first showed that KIR activity was diminished in ECs isolated from dyslipidemic mice and that activity could be restored by depleting membrane cholesterol. We next demonstrated that flow-induced endothelial KIR activity was decreased in each dyslipidemic model, changes that paralleled reduced flow-mediated dilatory responses in isolated cerebral vessels. Diminished flow-induced EC KIR activity and vasodilation were confirmed in cerebral arteries of EC Kir2.1−/− mice. In vivo, when cerebral blood flow responses to a blood pressure challenge that increased shearing force were monitored, we observed a perfusion phenotype in HFHC-C57BL/6 but not genetic deletion Ldlr−/− mice, consistent with diminished KIR activity and flow-mediated dilation. We conclude that KIR2.1 channels, through their interaction with cholesterol, are an early target during mild dyslipidemia, leading to impaired endothelial feedback and diminished cerebral blood flow responses needed to maintain neuronal function.
Material and methods
Animal model
Animal procedures followed the ARRIVE guidelines 13 and were done in accordance with the Canadian Council on Animal Care regulations; specific protocols were approved by the University of Western Ontario Animal Care Committee (Protocol #2017-144 and #2016-057). C57BL/6J male mice were obtained from The Jackson Laboratory; colonies of endothelial cell Kir2.1−/− mice (EC Kir2.1−/−; B6.Cg-Kcnj2tm1Swz tg(Tek-cre)1Ywa), floxed Kir2.1 control mice (B6.Cg-Kcnj2tm1Swz)14,15 and Ldlr−/− mice (D2.129S7(B6)-Ldlrtm1Her/J) were maintained at the University of Western Ontario. Control C57BL/6 and Ldlr−/− mice were fed normal chow. At 8 weeks of age, a second randomized group of C57BL/6 mice was placed on a high-fat high-cholesterol diet (42% calories from fat, plus 0.2% cholesterol by weight; TD.09268-HFHC; Harlan Teklad, Envigo) for 8 weeks. The mice were housed under constant temperature and humidity with a 12-h light/dark cycle and given ad libitum access to food and water. At endpoint, mice were 16 weeks old and weighed 25 – 40 g.
Blood and tissue collection, and plasma lipid analysis
Mice were fasted 6 h before euthanasia to standardize blood lipid levels. Under ketamine (100 μg g−1; Bioniche Animal Health) and xylazine (10 μg g−1; Bayer Healthcare, Animal Health Division) anesthesia, blood was collected via cardiac puncture in syringes primed with 80 µL of 7% Na2-EDTA. Plasma was separated by centrifugation; cholesterol and triglycerides were measured on a Cobas Mira S autoanalyzer (Roche Diagnostics). Calibrators, controls and enzymatic reagents for cholesterol (cholesterol CHOD-PAP, #11491458-216) and triglycerides (triglycerides/glycerol blanked, #11877771 216) were from Roche Diagnostics. As previously described, 16 plasma lipoproteins were separated by fast protein liquid chromatography using an AKTA purifier and Superose 6 column. An aliquot of each fraction was used to measure cholesterol enzymatically in samples and standards on a microtiter plate with added enzymatic reagents (WAKO Diagnostics, Cholesterol E, CHOD-DAOS method, #439-17501).
The thoracic cavity was accessed via midline incision; hearts were flushed with heparinized saline (10 units mL−1) via left ventricle inlet and right atrium outlet. The heart and aortic sinus were extracted. The top half of the heart was placed in OCT medium, frozen on dry ice, and stored at −80°C. The aortic sinus was serially sectioned (10 μm) using a Leica CM 3050S cryostat. Adjacent cross-sections of the aortic sinus were stained with hematoxylin and eosin (H&E) and Oil Red O (ORO, Sigma-Aldrich), and visualized with brightfield microscopy (Olympus BX50 microscope).
Isolation of cerebral arteries
Mice were euthanized via CO2 asphyxiation. The brain was removed and placed in chilled phosphate-buffered solution (PBS; pH 7.4) containing (in mM): 137 NaCl, 2.7 KCl, 10 Na2HPO4, 1.8 KH2PO4, and 5 glucose. Segments of middle and posterior cerebral arteries were isolated and cleaned for electrophysiology and myography experiments.
Isolation of SMCs and ECs
For SMC extraction, arterial segments were placed in isolation medium containing (in mM): 60 NaCl, 80 Na-glutamate, 5 KCl, 2 MgCl2, 10 glucose, and 10 HEPES with 1 mg mL−1 bovine serum albumin (BSA; pH 7.4) on ice for 10 min. The vessels were warmed at 37°C for 10 min, followed by a two-step digestion process: (1) a 6-min incubation in 0.9 mg mL−1 papain and 1 mg mL−1 dithiothreitol and (2) a 7-min incubation in 0.3 mg mL−1 H-type collagenase, and 0.7 mg mL−1 F-type collagenase. The digested vessels were then washed with ice-cold isolation medium and placed on ice for 15 min before trituration with a fire-polished pipette. SMCs were identified by their spindle-like shape and contractile behavior. Cells were stored in ice-cold isolation medium before use within 5 h.
For EC extraction, arterial segments were placed in isolation medium containing (in mM): 140 NaCl, 5.5 KCl, 1 MgCl2, 1.2 NaH2PO4, 5 glucose, 2 Na+ pyruvate, 0.02 EDTA, and 10 HEPES with 0.1 mg mL−1 BSA (pH 7.4) on ice for 10 min. This was followed by incubation in isolation medium with addition of 0.1 mM CaCl2 at 37 °C for 10 min. Vessels were then exposed to a two-step digestion process at 37 °C: (1) a 30-min incubation in 1 mg mL−1 BSA, 100 µmol L−1 Ca2+, 1 mg mL−1 papain, and 1 mg mL−1 dithioerythritol and (2) a 9-min incubation in 1 mg mL−1 BSA, 100 µmol L−1 Ca2+, 0.9 mg mL−1 type-F collagenase, 0.6 mg mL−1 type-H collagenase, 5 mg mL−1 elastase, and 1 mg mL−1 trypsin inhibitor. The digested vessels were then washed with ice-cold isolation medium and placed on ice for 15 min before trituration with a fire-polished pipette. ECs were identified by their rough shape and lack of voltage-dependent K+ conductances. Cells were stored in ice-cold isolation medium before use within 4 h.
Electrophysiology
Conventional whole-cell patch-clamp electrophysiology was used to measure Ba2+-sensitive K+ currents (an index of KIR activity) in isolated SMCs and ECs. Recording electrodes (pipette resistance, 5–8 MΩ) were pulled from borosilicate glass microcapillary tubes (Sutter Instruments), fire-polished (Narishige MF-830), covered in dental wax to reduce capacitance, and backfilled with pipette solution containing (in mM): 5 NaCl, 35 KCl, 100 K-gluconate, 1 CaCl2, 0.5 MgCl2, 10 HEPES, 10 EGTA, 2.5 Na2-ATP, and 0.2 GTP (pH 7.2). A pipette was gently placed onto a cell and negative pressure was applied to rupture the membrane and attain whole-cell access. Cells were voltage-clamped at −60 mV and equilibrated for 15 min in bath solution containing (in mM): 135 NaCl, 5 KCl, 0.1 MgCl2, 10 HEPES, 10 glucose, and 0.1 CaCl2 (pH 7.4). Whole-cell currents were recorded on an Axopatch 200B amplifier (Axon Instruments), filtered at 1 kHz, digitized at 5 kHz, and analyzed with Clampfit 10.3 software (Molecular Devices). Cell capacitance ranged between 14 and 18 pF in SMCs and 4–8 pF in ECs and was measured with the cancellation circuity in the voltage-clamp amplifier. Cells that displayed a noticeable shift in capacitance (>0.3 pF) during experiments met exclusion criteria. A 1 M NaCl-agar salt bridge between the Ag-AgCl reference electrode and bath solution was used to minimize offset potentials (<2 mV). All experiments were performed at room temperature. KIR currents were quantified by elevating extracellular [K+] from 5 to 60 mM K+ via equimolar replacement of NaCl by KCl. Voltage was stepped to −100 mV for 100 ms and then ramped to +20 mV at a rate of 0.04 mV ms−1. Currents from three trials were averaged. To differentiate between KIR currents and whole-cell K+ currents, KIR activity was blocked by addition of Ba2+ (100 µM) to the bath solution. Maximal peak current was divided by cell capacitance to obtain a peak current density (pA/pF) for each cell.
To assess the effects of shear stress on ECs, extracellular [K+] was maintained at 20 mM and the magnitude of the inward K+ current was measured before and after application of laminar shear stress to cells using a custom patch-clamp chamber. This chamber measured 1.0 cm × 2.5 cm × 0.2 cm (height). Laminar flow across the chamber was achieved by directing solution through a diffuser with gravity flow set to 2 mL min−1 (sufficient to keep isolated cells in place). The solution was continuously removed by wicking from a dam at the chamber’s outlet. Shear stress was calculated to be approximately 10 mPa—a value that sustains patch seal integrity.
To determine the effects of cholesterol depletion on EC inward K+ current, extracellular [K+] was maintained at 60 mM and methyl-β-cyclodextrin (MβCD; 5 mM) was added to the pipette solution. Current recordings were taken at baseline and 10 min after the addition of MβCD.
Myography
Flow-induced responses were studied in endothelium-intact arteries cannulated in a custom chamber filled with physiological salt solution (PSS; 37°C) containing (in mM): 119 NaCl, 4.7 KCl, 20 NaHCO3, 1.7 KH2PO4, 1.2 MgSO4, 1.6 CaCl2, and 10 glucose. The inflow and outflow pressures were controlled by a pressure Servo-controller (Living Systems Instrumentation). The bores of a pair of glass pipettes were matched to achieve equal resistance. Following equilibration at 15 mmHg for 30 min, 60 mM K+ was briefly superfused to assess vessel viability; vessels with no response were excluded. Intravascular pressure was raised from 15 to 80 mmHg and exposure to bradykinin (15 µM) was used to confirm the presence of intact endothelium. Vessels were pressured to 60 mmHg and diameter was measured before and after stepwise intraluminal flow increments (2, 4, and 6 µL min−1; 5 min steps) under control conditions or in the presence of external/intraluminal 100-µM BaCl2. Tone was assessed in EC Kir2.1−/− and floxed Kir2.1 control vessels by raising the pressure from 15 mmHg to 80 mmHg before and after addition of Ba2+. Passive vessel diameter was determined by bathing vessels in 2 mM EGTA in Ca2+-free PSS. Arterial diameter was measured with an automated edge detection system (IonOptix, Inc) under a 10× objective on a Zeiss Axiovert 200 microscope (Carl Zeiss).
Arterial Spin-labeling MRI
Under 2% isoflurane anesthesia, a small incision was made in the skin and underlying linea alba. A polyethylene catheter (PE10; Instech Laboratories) was inserted through the incision and secured with a purse-string stich (5-0 silk suture). The catheter was connected to a syringe containing 1 mM phenylephrine. The animal was placed in a custom-built insert in the prone position and inserted in an Agilent Animal magnetic resonance imaging (MRI) scanner with a 9.4-Tesla, 31-cm horizontal bore magnet (Magnex Scientific), 60-mm gradient coil set of 1000mT/m strength (Agilent), and Bruker Avance MRI III console with Paravision-6 software (Bruker BioSpin Corp). A 40-mm millipede volume coil (Agilent) was used for data acquisition.
An anatomical reference scan was acquired using a 2D fast spin echo (Turbo-Rapid Acquisition with Relaxation Enhancement (RARE)) sequence with the following parameters: field of view (FOV) = 19.2 × 19.2 mm2, matrix size = 128 × 128, 11 slices with slice thickness of 1 mm, repetition time = 5000 ms, echo time (TE) = 10 ms, effective echo time = 40 ms, RARE factor = 8, number of averages = 1.
A flow-sensitive alternating inversion-recovery spin echo planar imaging sequence with a 180° hyperbolic secant radiofrequency inversion pulse was used (imaging parameters: TE = 17 ms; five slices with imaging slice thickness of 2 mm; image matrix = 64 × 50; FOV = 19.2 × 15 mm2; inversion parameters: inversion slab thickness = 13 mm; pulse length = 3 ms) for perfusion images. Eleven images with increasing inversion times (100 ms +
During acquisition, the animal was maintained at 1.7% isoflurane and breath rate was monitored (PC-SAM model #1025; SA Instruments, Inc.) with a pneumatic pillow. Body temperature was measured with a rectal probe and maintained at approximately 37°C with a homeothermic warm air blower. Mean arterial pressure was measured with a tail cuff (CODA™ Monitor, Kent Scientific) during MRI acquisition. After a baseline scan of the brain was acquired, phenylephrine hydrochloride (0.816 mg kg−1; Sigma-Aldrich) was injected intraperitoneally and the scan was repeated. Following completion of MRI, mice were euthanized via cervical dislocation under deep anesthesia. Exclusion criteria included presence of brain anatomic abnormality (one HFHC mouse; enlarged ventricles), heating equipment malfunction during data acquisition (one control mouse), and inability to get a tail cuff blood pressure reading (two HFHC mice). Cerebral blood flow (mL 100 g−1 min−1) was quantified in the cortex, cerebral nuclei, hippocampus, thalamus, and hypothalamus using custom software written in MATLAB (MathWorks, Inc.).
Statistical analysis
Data are expressed as mean ± SD. Analysis was conducted using GraphPad Prism 8 (GraphPad Software). Based on our previous electrophysiology and myography work, power calculations revealed n = 6 is sufficient. p < 0.05 was considered statistically significant. Data distribution was determined using Kolmogorov-Smirnov test. Differences in lipid levels were compared using two-way ANOVA with Bonferroni correction. K+ currents before and after addition of 100 μM Ba2+, or under static versus flow conditions, or before and after addition of MβCD, were compared using paired t test while subtracted currents between different groups of mice were made using unpaired t test. Vessel diameter changes before and after exposure to Ba2+ were compared using paired t test while floxed control versus knockout responses were compared using unpaired t test. For cerebral blood flow measurements, two-way ANOVA with Šídák’s multiple comparisons test was performed for control versus dyslipidemic group comparisons; paired t test was performed for pre- versus post-phenylephrine treatment.
Results
Documenting mild dyslipidemia
Atherosclerosis is a pathophysiological state intimately tied to prolonged dyslipidemia, with microvascular dysfunction presumptively arising at an earlier phase. To study microvascular dysfunction, two models of dyslipidemia were used: 1) C57BL/6 mice fed a HFHC diet for 8 weeks, and 2) Ldlr−/− mice displaying impaired lipoprotein removal; their dyslipidemic status was confirmed by blood lipid levels. Plasma cholesterol was significantly higher in both models compared to C57BL/6 controls while triglyceride and very low-density lipoprotein (VLDL) cholesterol remained low (Figure 1(a)). Low-density lipoprotein (LDL) cholesterol was elevated in Ldlr−/− mice while high-density lipoprotein (HDL) cholesterol rose in HFHC-fed C57BL/6 mice. Our dyslipidemic mice did not develop atherosclerosis in their aortic roots, a key site of lesion development (Figure 1(b)) 17 ; this contrasts Ldlr−/− mice fed a HFHC diet (positive control). The mean weight was 28.5 ± 1.1 g in the control group, 35.2 ± 3.9 g in the HFHC group (p < 0.0001 compared to control) and 28.0 ± 1.7 g in the Ldlr−/− group. Of note, the floxed Kir2.1 group weighed 28.6 ± 1.0 g and EC Kir2.1−/− group weighed 29.5 ± 2.9 g.
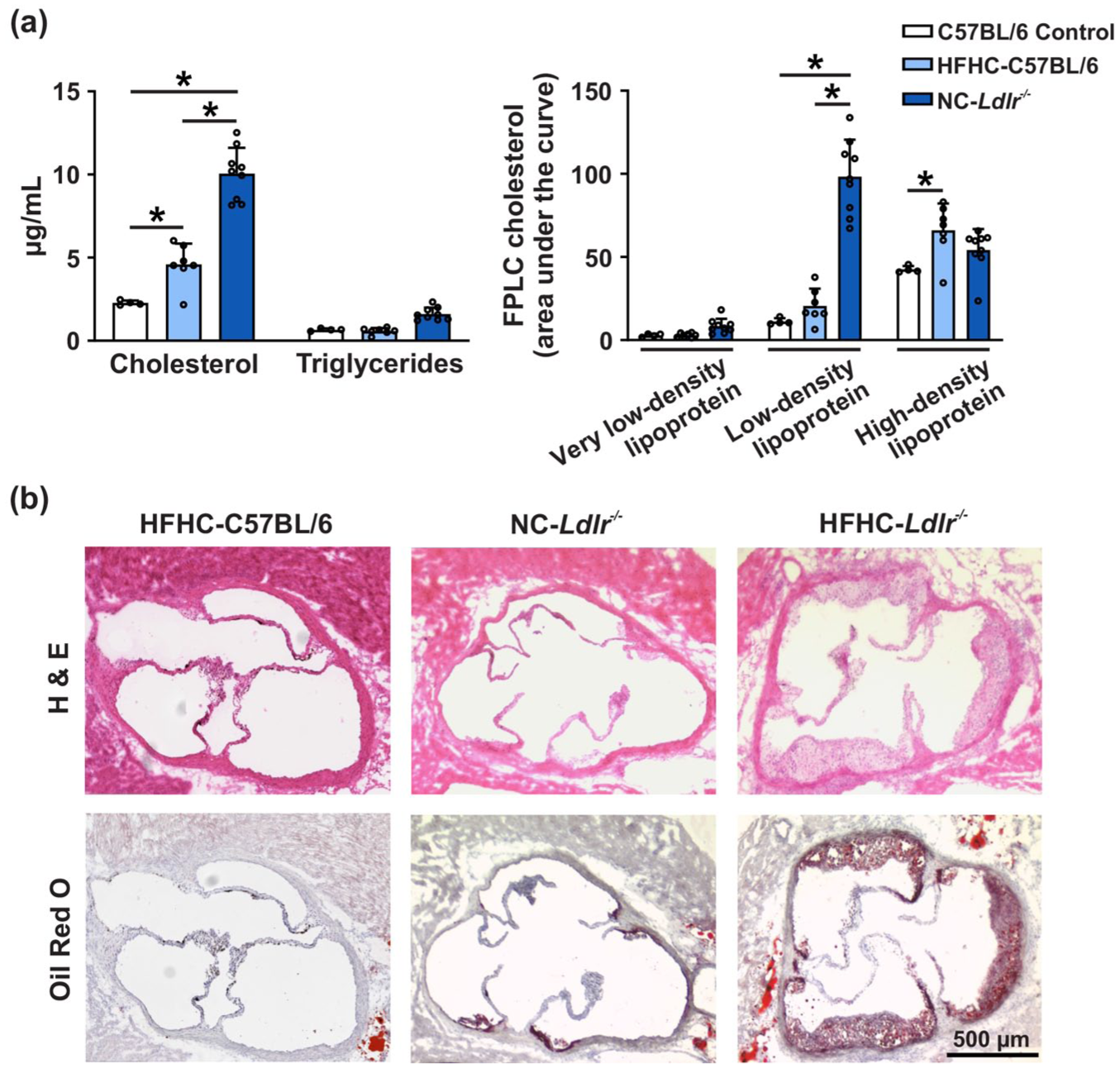
C57BL/6 mice on high-fat high-cholesterol (HFHC) diet and Ldlr−/− mice on normal chow (NC) both exhibit mild dyslipidemia without plaque formation in the aortic root. (a) C57BL/6 mice fed a HFHC diet for 8 weeks have significantly increased cholesterol and high-density lipoprotein cholesterol levels in their plasma while Ldlr−/− mice on normal chow have increased cholesterol and low-density lipoprotein cholesterol levels. Data presented as mean ± SD, differences were assessed using two-way ANOVA with Bonferroni’s correction. p < 0.05 was considered significant. (b) Plaques did not form in the aortic roots of the HFHC-fed C57BL/6 and NC-fed Ldlr−/− mice. HFHC-fed Ldlr−/− mice were used as a positive control for plaque detection. Note, Oil Red-O stains neutral lipids red (primarily triglyceride and cholesteryl ester). FPLC; fast protein liquid chromatography.
Mild dyslipidemia reduces endothelial but not smooth muscle KIR activity
KIR channels set base arterial tone, with cell-specific pools differentially regulated by membrane lipids. 9 To determine whether these channels are dysregulated in dyslipidemia, we monitored the Ba2+-sensitive KIR current (peak inward current density at −100 mV before and after application of Ba2+) in cerebral arterial ECs and SMCs using conventional patch-clamp electrophysiology. Negative control experiments first confirmed that the Ba2+-sensitive current was absent in ECs isolated from EC Kir2.1−/− mice, with no impact on SMC KIR current (Figures S1 and S2). We secondarily documented a marked reduction in the Ba2+-sensitive KIR current in ECs harvested from HFHC-fed C57BL/6 and Ldlr−/− cerebral arteries (Figure 2). Specifically, the Ba2+-subtracted current (pA/pF) was −19.2 ± 3.1 in the control (normal chow/wild type) group, −8.7 ± 1.7 in the HFHC group (p < 0.0001 compared to control), and −7.9 ± 1.9 in the Ldlr−/− group (p < 0.0001 compared to control). In contrast, the Ba2+-sensitive KIR current in cerebral arterial SMCs was not impacted in any of the models tested (Figure 3). Here, the Ba2+-subtracted current (pA/pF) was −1.8 ± 0.1 in the control group, −1.5 ± 0.3 in the HFHC diet group, and −1.8 ± 0.3 in the Ldlr−/− group, not statistically significant compared to control. Thus, early dyslipidemia has a notable impact on KIR in cerebral arterial endothelium as plasma lipid composition changes with disease.
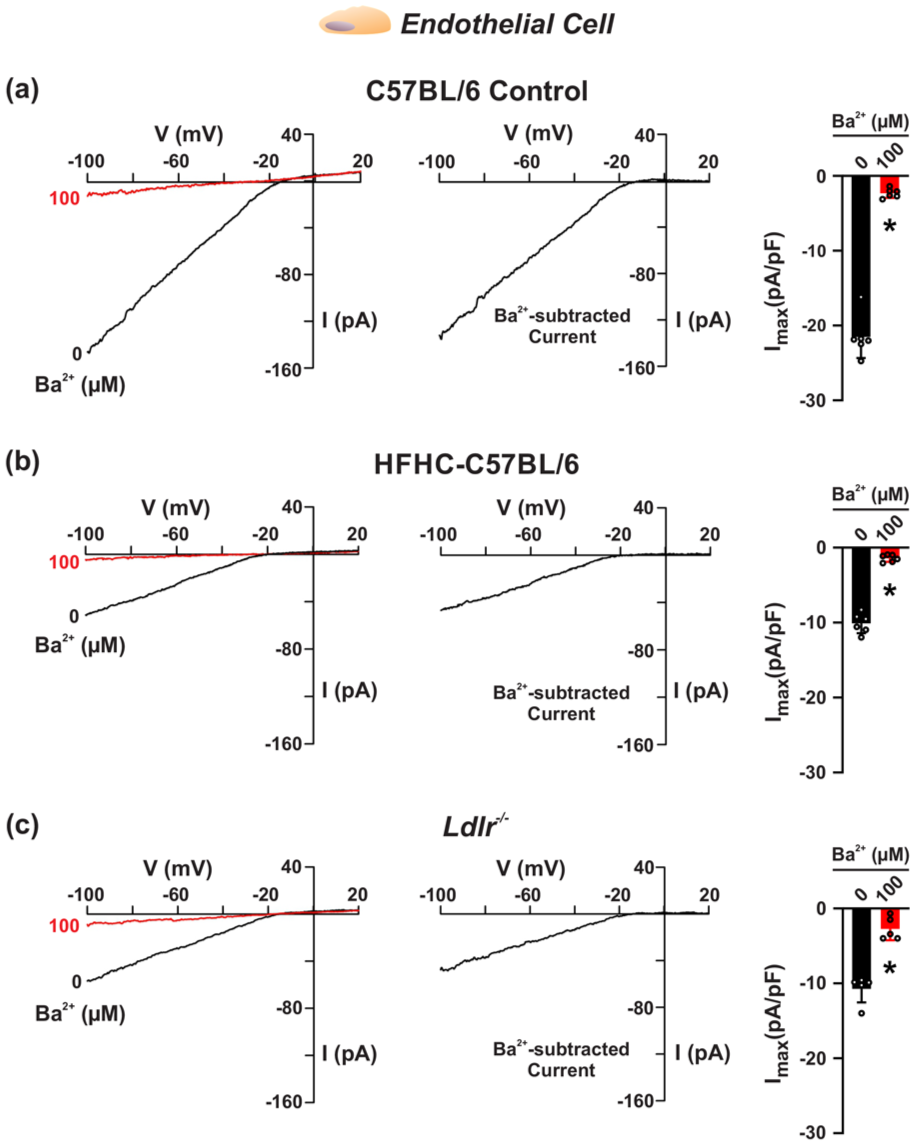
KIR current in cerebral artery endothelium is diminished in mild dyslipidemia. C57BL/6 mice on an 8-week high-fat high-cholesterol (HFHC) diet and Ldlr−/− mice on normal chow were used in parallel with C57BL/6 mice on normal chow. Whole-cell K+ currents were recorded in freshly isolated endothelial cells bathed in 60 mM K+ using voltage ramps from −100 to +20 mV before and after blockage of KIR with Ba2+ (100 μM). Representative whole-cell K+ current traces in endothelial cells of (a) control and (b and c) dyslipidemic animals with Ba2+-subtracted traces are shown. Summary data compare peak inward current density at −100 mV before and after application of Ba2+. Data presented as mean ± SD. *p < 0.05, paired t test.
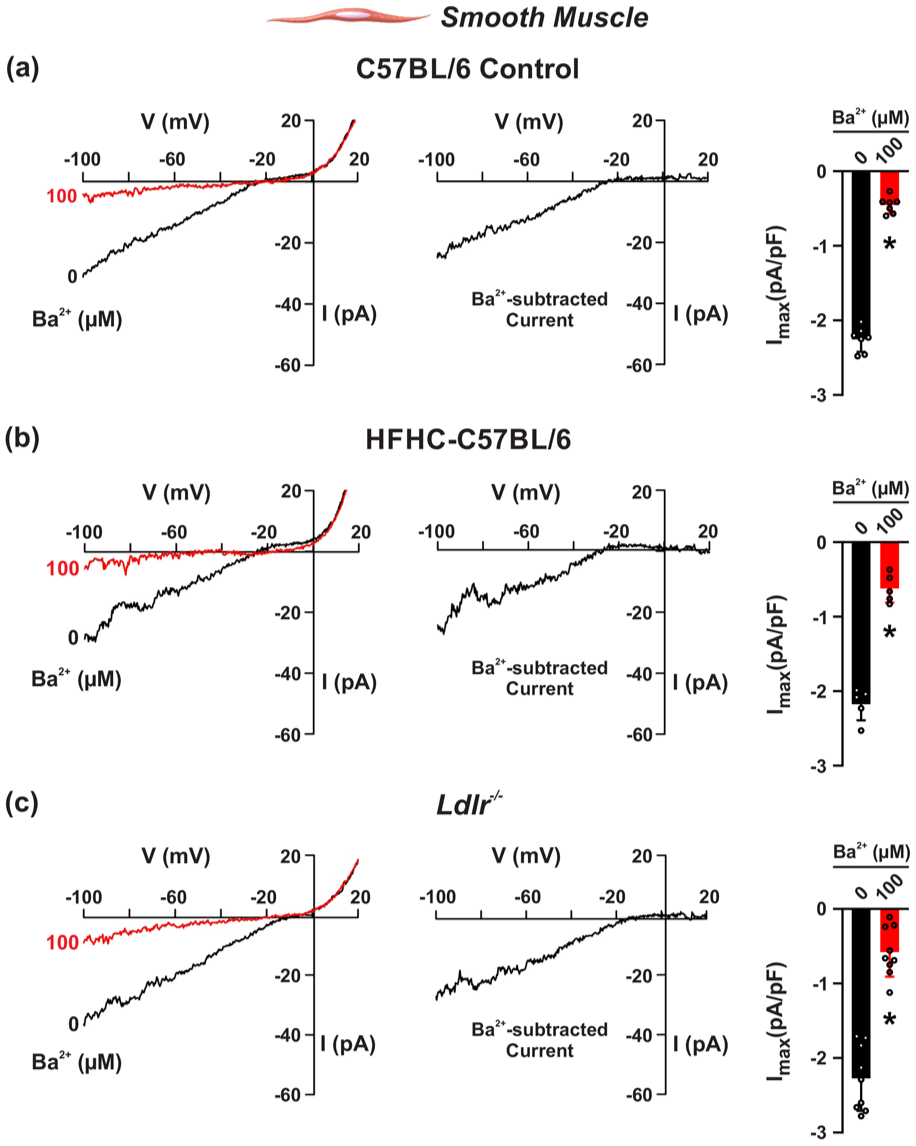
Mild dyslipidemia does not impact smooth muscle cell KIR current in cerebral arteries. C57BL/6 mice on an 8-week high-fat high-cholesterol (HFHC) diet and Ldlr−/− mice on normal chow were compared to C57BL/6 mice on normal chow. Whole-cell patch-clamp electrophysiology was used to measure KIR current with voltage ramps from −100 to +20 mV in the absence and presence of Ba2+ (100 μM) in 60 mM K+. Representative whole-cell K+ current traces in smooth muscle cells of (a) control and (b and c) dyslipidemic animals with Ba2+-subtracted traces are shown. Summary data compare peak inward current density at −100 mV before and after application of Ba2+. Data presented as mean ± SD. *p < 0.05, paired t test.
Flow-induced endothelial KIR activity is attenuated by mild dyslipidemia
As dyslipidemia supresses EC KIR currents, we next tested whether flow-induced modulation was also diminished. ECs isolated from cerebral arteries were placed in a custom chamber where a shearing force of 10 mPa was applied. In control mice, we noted increased whole cell KIR current (Figure 4(a)) following the induction of flow, a rise that was absent in ECs isolated from EC Kir2.1−/− mice (Figure S3; negative control). In contrast, flow failed to augment the EC KIR current in both dyslipidemic models, presumptively due to the rise in membrane cholesterol and its association with KIR2.1 (Figure 4(b) and (c)). If membrane cholesterol is elevated, limiting KIR activity, then its depletion by MβCD should restore EC KIR activity in these disease models. Figure 5(a) shows that cholesterol sequestration by MβCD had little effect on the EC KIR current from control mice, consistent with preferential PIP2 binding to KIR2.1. In sharp contrast, the same treatment in cerebral arterial ECs from dyslipidemic mice had a notable restoring effect on KIR activity, in agreement with cholesterol being replaced by PIP2 as the key regulatory lipid (Figure 5(b) and (c)). This rapid recovery also aligns with the idea that surface expression of EC KIR channels does not change as a result of dyslipidemia.
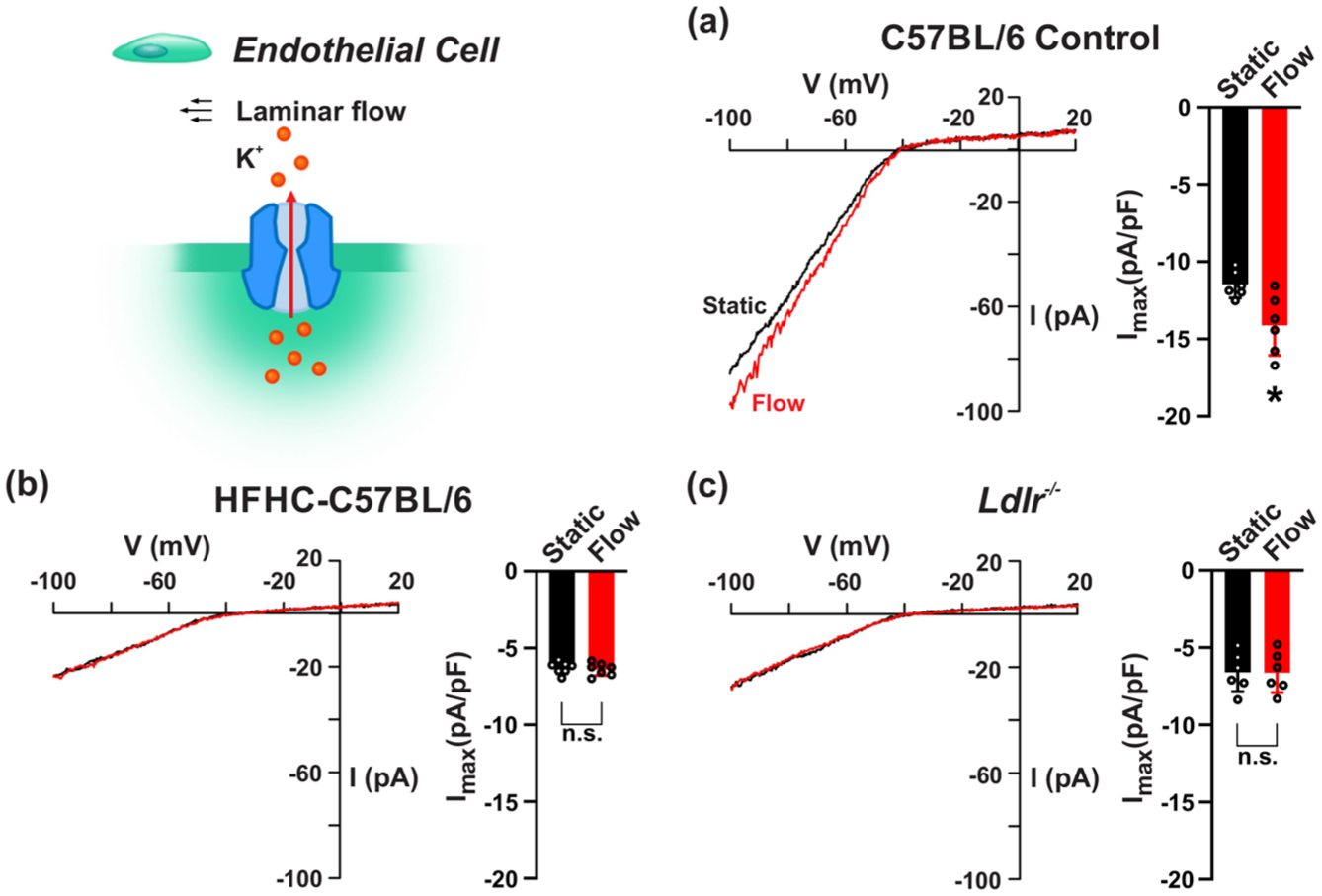
Flow-induced endothelial KIR activity is diminished in mild dyslipidemia. Extracellular [K+] was maintained at 20 mM (voltage ramp, −100 to +20 mV) and the magnitude of the inward K+ current was measured before and after application of laminar shear stress to cerebral ECs in a patch-clamp chamber. Representative traces and summary data of the endothelial KIR current from (a) control and (b and c) dyslipidemic animals are shown. Data presented as mean ± SD. *p < 0.05, paired t test.
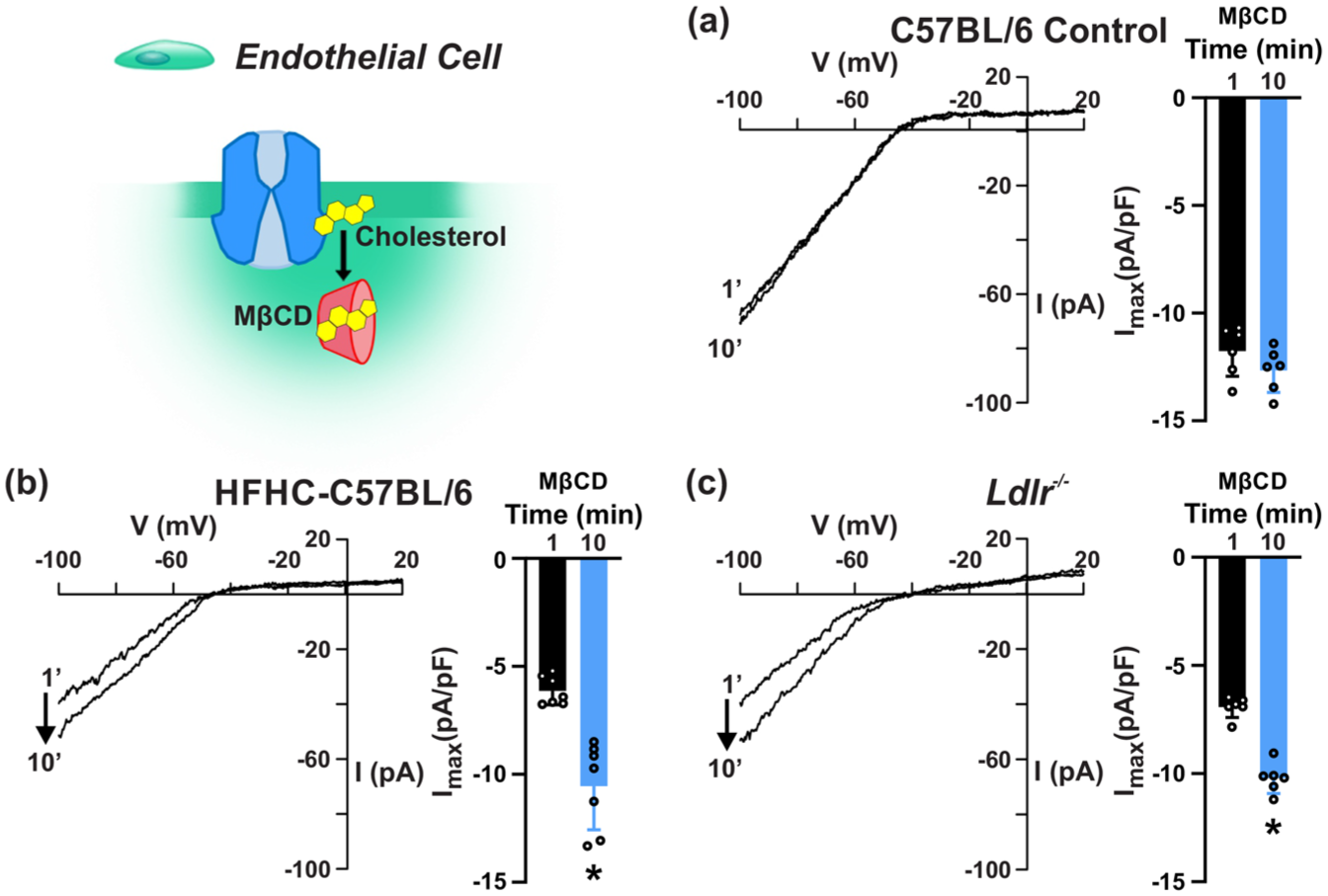
Blunted endothelial KIR current in dyslipidemia is restored by cholesterol depletion. Cerebral endothelial cells were bathed in 20 mM K+ and methyl-β-cyclodextrin (MβCD; 5 mM) was added to the pipette solution to sequester cellular cholesterol. Inward K+ current recordings were taken at baseline and 10 min after addition of MβCD. Representative current traces and summary data for (a) control and (b and c) dyslipidemic mice are shown. Data presented as mean ± SD. *p < 0.05, paired t test.
Dyslipidemia abrogates flow-induced vasodilation in cerebral arteries
The endothelium is responsible for flow-induced dilation. 9 To confirm a role for KIR2.1, we monitored this hemodynamic response in cerebral arteries isolated from floxed Kir2.1 and EC Kir2.1−/− mice. Cannulated arterial segments were positioned in a custom chamber and flow was increased stepwise from 2 to 4 to 6 μL min−1. We observed a progressive vasodilation in floxed Kir2.1 mice (one abrogated by intraluminal Ba2+), which was absent in cerebral arteries harvested from EC Kir2.1−/− mice (Figure S4(a)). Of note, the baseline vessel diameters did not significantly differ between the floxed control and knockout, 131 ± 13 µm versus 136 ± 8 µm, respectively. We further confirmed that the myogenic response was not altered by EC Kir2.1 knockout, as Ba2+-sensitive tone was comparable in EC Kir2.1−/− and floxed Kir2.1 vessels at either 15 mmHg or 80 mmHg (Figure S4(b)). In line with these observations, flow-induced dilation was markedly diminished in endothelium-intact cerebral arteries from HFHC-fed C57BL/6 and Ldlr−/− mice compared with control C57BL/6 mice (Figure 6); mean baseline vessel diameter in the control group was 137 ± 5 µm, 147 ± 7 µm in the HFHC-diet group (p = 0.0271 compared with control), and 133 ± 6 µm in the Ldlr−/− group. These findings are consistent with vessel-level changes in arterial contractile function early in mild dyslipidemia.
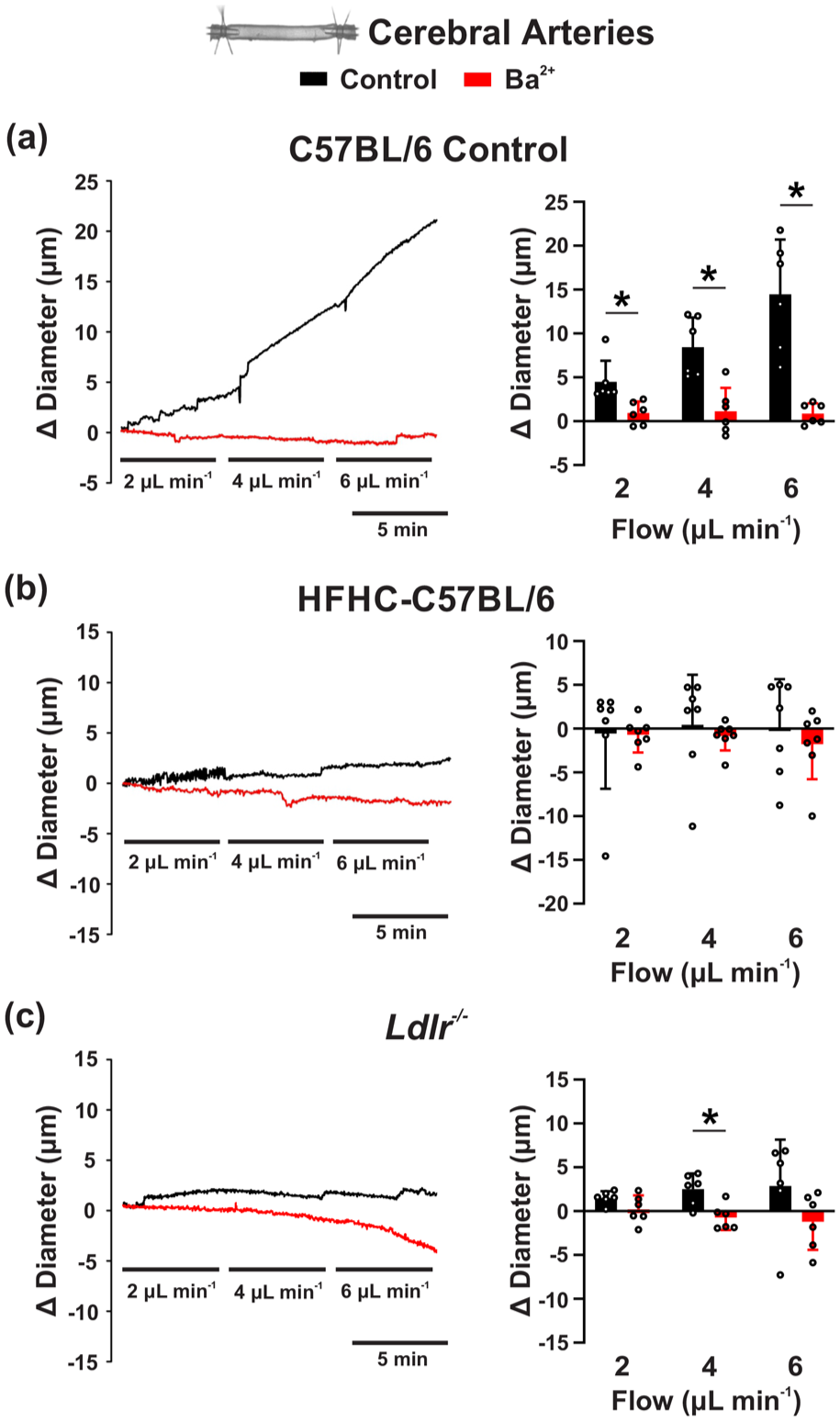
Flow-induced vasodilation is diminished in dyslipidemia. Shear stress-induced responses were studied in endothelium-intact arteries mounted in a flow chamber and vascular diameter changes were assessed while flow was increased by 2 μL min−1 increments before and after application of 100-µM Ba2+. Representative traces and summary data for (a) control C57BL/6 and (b and c) dyslipidemic animals are shown. Data presented as mean ± SD. *p < 0.05, paired t test.
Brain blood flow regulation is altered by mild dyslipidemia
To test whether vessel-level changes in arterial tone translate to brain perfusion, arterial spin-labeling MRI was used to assess blood flow prior to and following a blood pressure challenge with intraperitoneal phenylephrine. We reasoned that as elevated blood pressure should foster feedback dilation, the control blood flow response in key brain structures should be distinct from dyslipidemic mice. In control mice, brain blood flow varied between 150 and 200 mL 100 g−1 min−1 dependent on the brain structure (Figure 7), a rate that modestly but significantly increased after a phenylephrine infusion, which elevated systemic blood pressure by ~18 mmHg (C57BL/6, 103 ± 16 to 120 ± 9; HFHC-C57BL/6, 97 ± 16 to 116 ± 11; Ldlr−/−, 109 ± 7 to 128 ± 14 mmHg). The pressure-induced rise in steady state cerebral blood flow was lower in HFHC-C57BL/6 mice, consistent with a reduction in flow-mediated feedback. A similar reduction was not observed in the Ldlr−/− mice, a chronic (genetic model) of dyslipidemia. Note, resting cerebral blood flow was higher in C57BL/6 mice compared to controls, with the cortex being an exception, while Ldlr−/− mice exhibited similar perfusion levels as controls (Figure 7(b)).
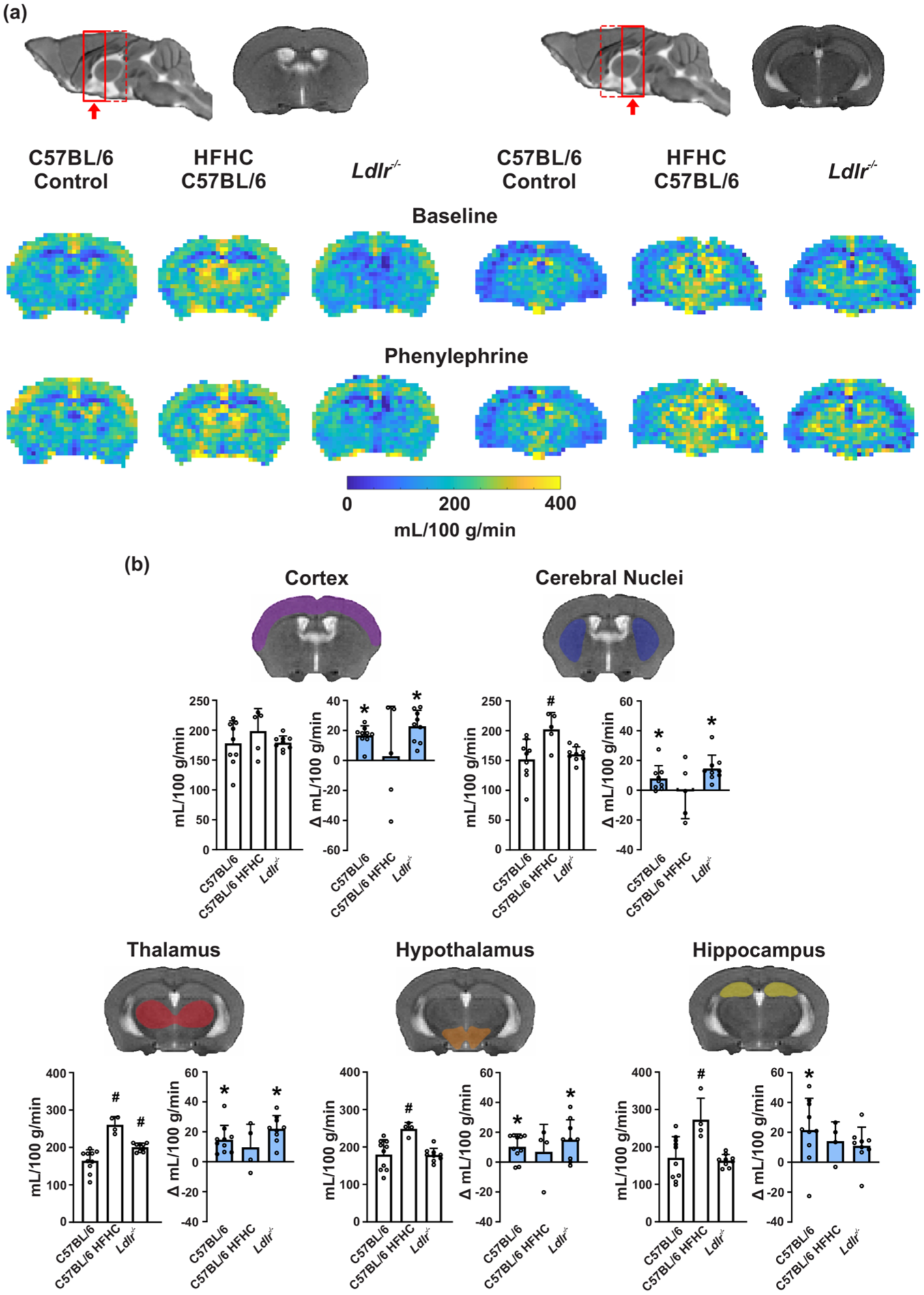
Region-specific brain perfusion is altered in dyslipidemic C57BL/6 mice on a high-fat high-cholesterol (HFHC) diet. (a) Arterial spin-labeling MR brain perfusion maps: scans were done in a posterior-to-anterior direction and the volume of brain scanned was divided into coronal slices. Coronal slices from 2 regions of the brain (red boxes) are shown, spanning cerebral nuclei, hypothalamus, thalamus and hippocampus. Perfusion was measured at baseline and after intraperitoneal phenylephrine injection. (b) Cerebral blood flow is shown before and after the blood pressure challenge; white bars show baseline flow and blue bars show change in flow after phenylephrine infusion. Data presented as mean ± SD. *p < 0.05, two-way ANOVA with Šídák’s multiple comparisons test was performed for control versus dyslipidemic group comparisons; paired t test was performed for pre- versus post-phenylephrine treatment.
Discussion
Cerebral arterial KIR channels aid in blood flow control by responding to hemodynamic forces via a signaling mechanism tied to the lipid microenvironment. Of particular value to this investigation is flow regulation of EC KIR2.x, mediated in part through its binding to PIP2. This unique form of regulation raises an interesting query: would a mild dyslipidemic state, one that conceivably alters membrane composition, impact hemodynamic sensing and consequently blood flow control in the brain? Herein, we determine whether elevated plasma lipids in two mildly dyslipidemic models (HFHC-C57BL/6 and Ldlr−/− mice) impact the cerebrovascular KIR2.x channels as directly measured in cells and functionally assayed in isolated vessels and in live brain tissue, using EC Kir2.1−/− mice as negative controls. In brief, we found that EC but not SMC KIR channel activity decreased with mild dyslipidemia, prior to the development of atherosclerosis. This cell-specific alteration impaired vasodilatory responses to flow, a change we attributed to cholesterol replacing PIP2 as the primary lipid binding to and regulating this channel. Consistent with the ex vivo diminishment of EC KIR activity, we noted the cerebral perfusion response to a blood pressure challenge, one that causes a flow-induced feedback response, was attenuated in one (HFHC-C57BL/6) but not the other (Ldlr−/−) dyslipidemia model. Taken together, mild dyslipidemia induces endothelial dysfunction in key resistance arteries well in advance of macrovascular changes; this dysfunction is evident in flow-mediated responses ex vivo and at times in vivo.
Background and initial observations
Dyslipidemia is characterized by a rise in cholesterol and an imbalance amongst low- and high-density lipoproteins, the long-term consequence being atherosclerotic lesions in conduit arteries that impede blood flow and which are a source of clots. 18 While this aspect of vascular disease has been well characterized, the effects of dyslipidemia on small resistance arteries is less certain, with literature largely focused on the impact of diminished endothelial nitric oxide availability. Recent work has expanded perspective toward ion channels, particularly those whose activity is intimately tied to the surrounding lipid microenvironment. Case-in-point are EC KIR2.x channels which typically bind PIP2, a phospholipid that places the channel in a preferred open state and confers flow sensitivity. In an animal model of severe hypercholesterolemia, basal and flow-mediated KIR2.1 activity markedly decreased in ECs of arteries of the mesenteric arcade—a change tied to elevated membrane cholesterol and its binding to the channel, pushing it to a preferred closed state. 10 Similar reductions in EC KIR activity have also been noted in arteries isolated from human adipose tissue. 12 Interestingly, mild dyslipidemia also blunts endothelial KIR2.1 activity and flow responses in first-order mesenteric arteries but in a glycocalyx-dependent manner. 12 While such work has provided valuable insight, it is limited by the choice of test vasculature, as cells from critical vascular beds, such as those in the brain, have escaped experimental attention. Notably, blood flow regulation in mesenteric arteries is dominated by sympathetic control while cerebral blood flow largely depends on local metabolic control and pressure responses to critically maintain a stable oxygen supply and protect the brain from injury. 19 In this regard, we began investigating cerebral arterial KIR2.x channel function in two mild models of dyslipidemia, one being C57BL/6 mice fed a high-fat high-cholesterol diet and the second, an Ldlr−/− mice on normal chow. Increases in plasma cholesterol were evident in both models, as were modest changes in LDL/HDL profile without atherosclerotic plaque formation (Figure 1). Whole-cell electrophysiology on cerebral arterial vascular cells revealed that the EC KIR current was indeed diminished by mild dyslipidemia (Figure 2), presumptively due to increased binding of membrane cholesterol to EC KIR. A similar reduction in SMC KIR activity failed to emerge, an expected result as these cells are not in direct contact with plasma and are preferentially known to bind cholesterol under control conditions (Figure 3). Supplemental experiments in EC Kir2.1−/− mice confirmed that this subunit encodes the EC KIR and that its deletion has no compensatory impact on SMC KIR current (Figures S1 and S2).
Flow regulation of KIR channels
Hemodynamic stimuli such as pressure and flow set arterial tone and tissue perfusion by altering arterial VM and consequently cytosolic Ca2+ through the gating of voltage-operated Ca2+ channels. 20 While a dominant hyperpolarizing current in both endothelium and smooth muscle, 21 KIR channels are often viewed as a background conductance with limited regulatory potential.5,22 This view has shifted as recent work has noted that membrane lipids impact channel gating 23 and help confer hemodynamic sensitivity. First, consider SMC KIR current encoded by KIR2.2 subunits, where binding to cholesterol confers pressure sensitivity, and consequent inhibition leads to myogenic tone development. Next ponder the effects of PIP2 when bound to EC KIR2.x channels, helping confer flow sensitivity while its replacement with cholesterol, as in severe dyslipidemia, impairs this hemodynamic regulation. 9 The latter finding spurred the monitoring of KIR in ECs from cerebral arteries exposed to laminar shear stress (~10 mPa; Figure 4). It is clear that shear stress increases EC KIR activity from C57BL/6 control mice, a change that was notably absent in ECs from our mildly dyslipidemic models. Consistent with this loss reflecting increased binding of membrane cholesterol over PIP2, we subsequently noted that depletion of the former with MβCD restored basal KIR activity (Figure 5). The logical extension of this cell-level work, and this regulatory switch from PIP2 to cholesterol, would be a blunting of Ba2+-sensitive flow-induced dilation in cerebral arteries from HFHC-C57BL/6 and Ldlr−/− mice (Figure 6). This blunting of flow-induced vasodilation was indeed observed in our mild dyslipidemia models, just as in arteries in which endothelial Kir2.1 was deleted (Figure S4). In theory, two mechanisms can explain how diminished EC KIR activity impairs cerebral arterial dilation. First, without robust EC hyperpolarization, the bioavailability of nitric oxide decreases, as the electrochemical force needed to drive extracellular Ca2+ influx in support of endothelial nitric oxide synthase decreases. Second, there will also be less hyperpolarizing charge to spread across myoendothelial gap junctions to decrease smooth muscle VM and consequently the activity of voltage-operated Ca2+ channels.
In presenting this work, one should recognize interpretational limitations, the most important being that the laminar shear stress applied in the cell-level experiments was only 10%–15% of the shear stress in vivo. While higher shear stress would have been ideal, that would be problematic as patch seals destabilize, limiting both the validity and reliability of whole-cell recordings. Similarly, myography experiments were conducted at the low end of intraluminal flow range, a pragmatic decision needed to limit the pressure gradient across the vessel which could theoretically introduce a strong myogenic response, confounding measures of flow-mediated vasodilation. Nonetheless, KIR-dependent endothelial dysfunction was notable during mild dyslipidemia well in advance of atherosclerosis, a finding aligned with human flow-mediated vasodilation tests, supporting their use as an early indicator of cardiovascular disease. 24
Cerebral blood flow control in vivo
Base tone development in the cerebral circulation is set by blood pressure, its elevation driving a myogenic constriction that secondarily increases shear stress in vivo. The latter is sensed by the endothelium and, through its activation of KIR, initiates feedback dilation. The dynamic balance created by these two hemodynamic forces is referred to as cerebral autoregulation and it ensures brain blood flow remains relatively constant across a range of blood pressures. It follows that if mild dyslipidemia impairs flow-mediated feedback, a misbalance in arterial tone will ensue and this will translate into altered brain blood flow dynamics. To test this idea, we used arterial spin-labeling MRI to monitor steady state blood flow in control and dyslipidemic mice, prior to and following phenylephrine infusion to elevate blood pressure and secondarily shear stress (Figure 7). Resting cerebral blood flow was higher in HFHC-fed mice compared to controls; an observation congruent with slightly elevated baseline diameter of cerebral arteries extracted from this group. Cerebral autoregulation was evident in control C57BL/6 mice across all five major brain structures when blood pressure was elevated (by ~18 mmHg), as the small rise (10–15 mL 100 g−1 min−1) in blood flow represented only 5%–7% of baseline. However, this small but significant rise was blunted in C57BL/6 mice fed a HFHC diet, a finding aligned with mild dyslipidemia impairing flow-mediated dilation. While an intriguing observation and one aligned with our cell and tissue observations, it is somewhat delimited by the higher baseline perfusion observed in this mild dyslipidemia model. Contrary to these findings, no attenuation of the response was observed in Ldlr−/− mice; indeed, a trend toward elevated brain perfusion was observed following the phenylephrine infusion. We explain these findings by compensatory changes that often arise in chronic deletion models as biology attempts to re-establish normal blood flow dynamics. While findings are somewhat mixed, this study is alone in translating cell/tissue level observations of EC KIR dysregulation in the in vivo setting.
Considering the subtle distinctions in blood lipid profiles 25 and hormonal regulation between sexes, it is plausible that cerebral vascular function may vary accordingly. Therefore, a limitation of our study is that, for logistical purposes, it included only male mice. Furthermore, it is evident that there are inherent difficulties in working with live animals and developing experiments to selectively probe an aspect of ion channel regulation. For example, while the blunted blood flow responses in HFHC-C57BL/6 mice are consistent with KIR dysregulation, changes in membrane cholesterol impact other endothelial ion channels setting cerebral arterial tone and blood flow control.26,27 Membrane cholesterol suppresses transient receptor potential vanilloid cation channels, like TRPV 28 and TRPV4, 29 the latter providing the Ca2+ to activate small/intermediate conductance Ca2+ activated K+ channels involved in arterial dilation. 30 Cholesterol can also potentiate others, such as Piezo1, a flow-sensitive channel in brain endothelium, whose activity is tied to both dilatory and constrictor responses.31,23 As well, disruption of endothelium-dependent vasodilation with increased cholesterol involves endothelin-1, with receptor antagonism showing therapeutic potential. 32 Considering the various dilatory mechanisms, it is important to highlight that KIR2.1 amplifies the effects of other vasodilators through its characteristic negative slope conductance, and, thus, impairment of KIR2.1 function by cholesterol would diminish hyperpolarization along the arterial wall, reducing the effects of a range of vasodilatory pathways.14,21 To garner greater interpretational precision, one could perhaps incorporate the use of transgenic mice, one of note being where endothelial KIR channels have been rendered insensitive to cholesterol binding with a mutation in the Kir2.1 sensitivity belt (L222I) 33 to study the cerebral vasculature.
Summary
Our cerebral arterial work revealed that endothelial dysfunction arises early with mild dyslipidemia, as manifested by cholesterol-induced changes in EC KIR2.1 activity (Figure S5). Projecting forward, it is intriguing to consider whether endothelial KIR dysfunction is observable in other vascular structures such brain capillaries. If so, dyslipidemia would be expected to impair the K+-induced conducted response, a key element of neurovascular coupling. In closing, this work extends our mechanistic understanding of endothelial dysfunction in critical organs, and suggests that lipidemic control is likely to preserve brain blood flow control in service of neurological function.24,12
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251399019 – Supplemental material for From cells to organism: impact of dyslipidemia on inwardly rectifying K+ channels and cerebral vascular function
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251399019 for From cells to organism: impact of dyslipidemia on inwardly rectifying K+ channels and cerebral vascular function by Paulina M Kowalewska, Maria Sancho, Sergio Fabris, Murray W Huff, Robert Gros and Donald G Welsh in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
We thank: Suzanne Brett and Michelle Kim (Western University) for excellent animal care support; Miranda Bellyou and Alex Li (Western University) for assistance with live animal imaging experiments and operating the MRI system; Dawn Telford and Brian Sutherland for assistance with the dyslipidemic animal models (Western University).
Author contributions
PMK, MS, and SF performed experiments and analysed data. MWH and RG provided expertise and resources. PMK and DGW wrote the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Canadian Institutes of Health Research (Project # RN354019) and the Heart and Stroke Foundation of Canada. DGW is the Rorabeck Chair of Molecular Neuroscience and Vascular Biology at Western University.
Ethical considerations
Animal research protocols were approved by the University of Western Ontario Animal Care Committee (Protocol #2017-144 and #2016-057), in compliance with the Canadian Council on Animal Care regulations for care and use of laboratory animals.
Data availability
The data that support the findings of the study are included in this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
