Abstract
Ceramide is a major constituent of membrane microdomains and controls cell signaling. Previous studies showed that ceramide exacerbates cerebral ischemia/reperfusion injury and that inhibition of acid sphingomyelinase (ASM), which converts sphingomyelin to ceramide, promotes microvascular remodeling via induction of extracellular vesicles (EVs) release. ASM inhibitors have so far not translated into clinical stroke therapies. Of note, ceramide levels can also be lowered by inhibition of serine palmitoyltransferase (SPT), which is the rate-limiting enzyme of ceramide de novo synthesis. To evaluate this pathway as a potential therapeutic target for stroke, we exposed cerebral microvascular endothelial cells to the SPT inhibitor myriocin or induced SPT knockdown by siRNA. Deactivation of de novo ceramide synthesis increased endothelial tube formation, migration, VEGF secretion, and EV release. These EVs did not induce angiogenesis in tube formation assays. Using light sheet microscopy, we demonstrate that myriocin-mediated SPT inhibition increases the length and branch density of thin microvessels (<4 µm diameter), which are drivers of angiogenesis, in the previously ischemic brain tissue of mice exposed to middle cerebral artery occlusion. These data highlight the importance of ceramide de novo synthesis for post-ischemic microvascular remodeling and angiogenesis, while its mode of action differs from ASM inhibitors.
Keywords
Introduction
The sphingolipid ceramide is critically involved in regulating biological properties of cell membranes. 1 As part of membrane microdomains, ceramide is critically involved in many cell signaling pathways, regulating cellular proliferation, differentiation, and survival.2–4 Ceramide responses to injuries are triggered within seconds by sphingomyelin hydrolysis through pH-dependent sphingomyelinases located on the outer and inner plasma membrane leaflets, respectively.1,5 Following cerebral ischemia/reperfusion (I/R) injury, ceramide rapidly accumulates in cerebral microvessels, orchestrating signaling processes that exacerbate cell damage.6–9 In ischemic stroke patients, increased plasma ceramide levels were associated with poor functional neurological recovery. 10 Pre-clinical studies demonstrated that deactivation of acid sphingomyelinase (ASM), which is located in lysosomes and the outer membrane leaflet, 11 by antidepressants enhanced neuronal survival and angiogenesis and mitigated brain inflammatory responses in global and focal cerebral ischemia models of rats and mice.8,12 An important mechanism how ASM inhibitors stimulate brain remodeling post-I/R, is the release of small extracellular vesicles (sEVs), which, when taken up by cerebral endothelial cells, augment endothelial migration, formation of capillary-like tube structures, and release of vascular endothelial growth factor (VEGF) of target cells. 8 ASM knockdown abolished these actions, providing the proof that ASM mediates the restorative actions of antidepressants in cerebral I/R injury. 8 Despite a promising double-blind, randomized controlled phase II clinical study demonstrating that the ASM inhibitor fluoxetine improved stroke outcome, 13 these findings could not be validated by large phase III multicenter trials.14,15
Sphingolipids crucially influence membrane fluidity and fusogenicity,16,17 and ceramide in particular induces membrane curvatures when enriched in one of the two membrane leaflets, which is due to the cone-shaped physicochemical configuration of ceramide. 18 Both mechanisms likely explain why ASM inhibitors facilitate sEV release. 8 Another target that allows to modulate cellular ceramide levels is the de novo ceramide synthesis pathway. The de novo synthesis pathway begins with the condensation of serine and palmitoyl-CoA by serine palmitoyltransferase (SPT). 19 Subsequently, 3-ketosphinganine is reduced to sphinganine by 3-ketosphinganine reductase. 20 Sphinganine is then acylated by ceramide synthases to dihydroceramide. 20 Introduction of a trans-4,5 double bond by dihydroceramide desaturase yields ceramide. 21 As SPT catalyzes the first and rate-limiting step, manipulation of the de novo pathway is usually conducted by targeting this enzyme, for example, by administration of the small molecule myriocin, which is a potent SPT inhibitor. An in vitro study previously revealed that myriocin increased cell viability and inhibits oxidative damage of neurons exposed to glutamate. 22 These findings were further corroborated by in vivo observations showing that delivery of myriocin at a dose of 0.3 mg/kg reduced ceramide accumulation in the cerebral cortex and induced acute neuroprotection in rats exposed to middle cerebral artery occlusion (MCAO). 23 In myocardial I/R injury, the intraventricular injection of nanocarriers loaded with myriocin at the onset of reperfusion counteracted ceramide synthesis, reduced infarct size, and reduced pro-inflammatory cytokine formation. 24 SPT inhibition also stimulated cardiac remodeling, as shown by an experimental study in which mice received myriocin-containing chow 2 weeks after myocardial infarction. 25 This dietary intervention prevented left ventricular dilation, reduced fibrosis, and improved systolic function, indicative for successful tissue regeneration. 25 Studies on the effects of de novo ceramide synthesis inhibitors on microvascular reposes post-I/R were so far lacking.
In contrast to ASM inhibitors, which selectively reduce ceramide concentration in the outer cell plasma membrane leaflet, 11 thus influencing cell membrane curvatures as a consequence of ceramide’s cone-shaped structure, 18 de novo ceramide synthesis deactivation is supposed to reduce ceramide levels in both membrane leaflets throughout the cell.
In the present study, we examined how SPT deactivation achieved by myriocin or siRNA-mediated knockdown influences angiogenic features of human cerebral microvascular endothelial cells (hCMEC/D3) in vitro and microvascular remodeling of mice exposed to transient middle cerebral artery occlusion (MCAO). We herein demonstrate that pharmacological deactivation and genetic downregulation of SPT reduced endothelial ceramide content, increased angiogenesis evaluated by tube formation and migration assays, increased VEGF secretion, and sEV release by cerebral microvascular endothelial cells. In line with these data, 3D light sheet microscopy studies showed that pharmacological SPT deactivation enhanced microvascular network remodeling in the brains of MCAO mice. Unlike in earlier studies, in which sEVs released by endothelial cells in response to ASM inhibitors increased angiogenesis, the sEVs released by endothelial cells upon myriocin lacked angiogenic properties, indicating that SPT deactivation increased angiogenesis via a paracrine, presumably VEGF-related mode of action.
Materials and methods
Legal issues, animal housing, randomization, and blinding
All experiments were performed with permission from the Landesamt fuer Natur, Umwelt, und Verbraucherschutz (LANUV) which is part of the Ministry for Environment, Agriculture, Conservation, and Consumer Protection (MULNV) of the State of North Rhine–Westphalia and in accordance with EU guidelines (Directive 2010/63/EU) for the care and use of laboratory animals, STAIR, STEPS, and ARRIVE guidelines. Animal experiments were strictly randomized and the experimenter performing the animal experiments was fully blinded at all stages of the study by another researcher preparing the vehicle and myriocin solutions. Mice were kept in a regular inverse 12 h:12 h light/dark cycle in groups of five animals/cage and had free access to food and drinking water. As exclusion criteria, mice were removed from the study if suffering from respiratory abnormalities or from severe nurturing handicaps resulting in a weight loss >20%.
Cell culture
Immortalized brain microvascular endothelial cells (hCMEC/D3) were cultured in endothelial basal medium (EBM-2; Lonza, Basel, Switzerland) supplemented with 5% fetal bovine serum (FBS; Life Technologies, Carlsbad, CA, USA), 100 U/ml penicillin/streptomycin (Life Technologies), 1.4 µM hydrocortisone (Sigma–Aldrich, St Louis, MO, USA), 5 µg/ml ascorbic acid (Sigma–Aldrich), 1% chemically defined lipid concentrate (Life Technologies), and 10 mM HEPES (Life Technologies).
Small interfering RNA (siRNA) knockdown
SPT knockdown in vitro was achieved by small interfering RNA (siRNA). Cells were transfected with ON-TARGETplus SMARTpool siRNA (Dharmacon, Lafayette, CO, USA, catalog number L-006673-00-0005) targeting human SPTLC1 (Entrez Gene ID: 10558). This reagent consists of a pool of four distinct siRNAs. Transfections were performed according to the manufacturer’s instructions using Dharmafect transfection reagents. Scrambled siRNA was used as a negative control.
Tube formation assay
To evaluate the formation of capillary-like tubular structures, 60 µl matrigel (Corning, NY, USA) were pipetted into 96-well plates. The gel solidified at 37 °C for 30 min. Cells 3 × 104 were seeded and treated with vehicle solution (DMSO) or 1 µM myriocin (Sigma–Aldrich). After 20 h, photomicrographs were taken with a 2× objective using the EVOS digital inverted microscope (Advanced Microscopy Group, Bothell, WA, USA). Closed tubes were counted using Image J (NIH) in each well. Microvascular length and branching point numbers were determined. Experiments were done in triplicates for which mean values were formed.
Transwell migration assay
Cell migration was assessed using a modified Boyden chamber. Cells 3 × 104 were seeded in the upper compartment of polycarbonate membrane inserts (8.0 μm pores) that contained serum-reduced (1.25% FBS) medium. Vehicle solution or 1 µM myriocin were administered into the lower compartment that contained 5% FBS. Cells that did not migrate were removed after 24 h. Migrated cells were fixed with 4% PFA and stained with Hoechst 33342 (Thermo Fisher Scientific, Waltham, MA, USA). Photomicrographs were taken with a 20× objective using the EVOS digital inverted microscope (Advanced Microscopy Group). Experiments were done in duplicates, for which mean values were formed.
Cell viability assay
Cells 2 × 104 were seeded into 96-well plates and treated with vehicle solution, 1 µM myriocin, scrambled siRNA or SPT siRNA. After 24 h, cells were incubated with 0.5 mg/ml 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT; Biomol, Hamburg, Germany) for 2 h. Formazon formation was measured at 570 nm on a microplate reader (iMark Detection; Bio-Rad Laboratories, Hercules, CA, USA). Samples were analyzed in triplicates, of which mean values were formed.
Middle cerebral artery occlusion
Focal cerebral ischemia was induced in male C57BL/6j mice (10–12 weeks; Envigo, Horst, The Netherlands). Mice were anesthetized with 1.0%–1.5% isoflurane (30% O2, remainder N2O). Rectal temperature was maintained between 36.5 °C and 37.0 °C using a feedback-controlled heating system. Cerebral blood flow was recorded by laser Doppler flow (LDF) measurement using a flexible probe with a diameter of 0.5 mm attached to the animals’ skull above the core of the middle cerebral artery territory. For MCAO, a midline neck incision was made and the left common and external carotid arteries were isolated and ligated. The internal carotid artery (ICA) was temporally clipped. A silicon-coated monofilament was introduced via a small incision into the common carotid artery (CCA) and advanced to the carotid bifurcation for MCAO. Reperfusion was initiated after 30 min by monofilament removal. Wounds were carefully sutured. Myriocin (1 mg/kg, i.p.) or vehicle solution were intraperitoneally administered for 14 days starting immediately after reperfusion. The opioid buprenorphine (0.1 mg/kg; Reckitt Benckiser, Slough, UK) was subcutaneously administered before surgery, and the antiphlogistic carprofen (5 mg/kg; b.i.d.; Bayer Vital, Leverkusen, Germany) was subcutaneously administered for 3 days after surgery.
FITC–albumin perfusion and light sheet fluorescence microscopy
Mice were deeply anesthetized and transcardially perfused with 4% paraformaldehyde, followed by infusion of 10 ml FITC-albumin hydrogel (Sigma–Aldrich). Brains were subsequently removed, post-fixed overnight at 4 °C in 4% PFA. After brain dehydration and clearing using a tetrahydrofuran gradient (Sigma–Aldrich) and ethyl cinnamate (Sigma–Aldrich), brains were scanned at 2 μm steps using an UltraMicroscope Blaze (LaVision BioTec, Bielefeld, Germany) light sheet microscope. Regions of interest measuring 508 × 508 × 1000 µm were chosen in the dorsolateral striatum, as previously reported, 8 which is the core of the middle cerebral artery territory.
Analysis of light sheet microscopy data
The microvascular network analysis pipeline VesselExpress was applied to analyze brain vasculature in FITC-albumin perfused brains as described before. 26 The pipeline utilizes a dual Frangi filter strategy for vessel segmentation, which allows analyzing diameters of small, intermediate, and large microvessels with high accuracy and unprecedented speed. After segmentation of raw images, vessel centerlines are extracted from the binary images using a parallel thinning algorithm from the scikit-image Python package. These centerlines are then converted into undirected graphs via the 3scan Python toolkit. A depth-first search (DFS) traces the vessels and vascular parameters such as vessel length density, number of branches, and mean branch density are extracted.
Western blot analysis
Cells were lysed with NP-40 buffer containing protease and phosphatase inhibitors (Sigma–Aldrich). Lysates were centrifuged at 13,400 rpm at 4 °C and the supernatant was collected. Equal amounts of cell lysates or sEV preparations were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and subsequently transferred to nitro-cellulose membranes (GE Healthcare Life Sciences, Chicago, IL, USA). Non-specific binding was blocked with 5% non-fat milk powder (Sigma–Aldrich) dissolved in 0.1% Tween in 0.1 M Tris-buffered saline (TBS-T). Membranes were incubated with anti-SPT long chain base subunit-1 (SPTLC1; ABS1642; Merck Millipore, Burlington, MA, USA), rabbit anti-syntenin (clone EPR8102; Abcam), rabbit anti-calnexin (ab10286; Abcam), rabbit anti-CD81 (GTX101766; GeneTex, Irvine, CA, USA), rabbit anti-TSG101 (E6V1X; Cell Signaling Technology, Danvers, MA, USA), mouse anti-Alix (3A9; Cell Signaling Technology), or rabbit anti-β-actin (4967; Cell Signaling Technology) antibodies overnight at 4 °C, rinsed and incubated in peroxidase-conjugated secondary antibodies (Santa Cruz, Heidelberg, Germany) for 1 h at room temperature. Signals were detected by enhanced chemiluminescence using prime Western blotting detection reagent (GE Healthcare Life Sciences). SPTLC1 expression was normalized to β-actin abundance.
Serine palmitoyltransferase activity assay
SPT activity was measured by tracking de novo sphingolipid synthesis using isotope-labeled amino acids as previously described. 27 These labeled substrates were incorporated into long-chain bases (LCBs), which were subsequently isolated by chemical hydrolysis of lipids to remove head groups and acyl chains. The resultant LCBs were then analyzed and quantified using liquid chromatography–mass spectrometry (LC–MS) with a targeted multiple reaction monitoring (MRM) approach.
Acid sphingomyelinase activity assay
ASM activity was measured using BODIPY-labeled sphingomyelin as substrate. Samples were lysed in 250 mM sodium acetate buffer (pH 5.0) containing 1% NP-40 detergent (Fluka BioChemika, Morristown, NJ, USA). The cellular membrane integrity was disrupted with a sonicator. After centrifugation for 5 min at 300g at 4 °C, supernatants were collected. Lysates were adjusted to a specific protein concentration and incubated with 100 pmol BODIPY-labeled sphingomyelin (Thermo Fisher Scientific) in 250 mM sodium acetate (pH 5.0) and 0.1% NP-40 for 1 h at 37 °C. Chloroform:methanol (2:1, v/v) was added, samples were vortexed and centrifuged for 5 min at 15,000g to achieve a phase separation. The lower phase was collected and concentrated in a vacuum centrifuge (SPC111V; Thermo Fisher Scientific) for 45 min at 37 °C. Lipids were dissolved in 20 µl chloroform:methanol (2:1, v/v) and spotted onto thin layer chromatography (TLC) plates (Macherey Nagel, Düren, Germany). The TLC run was performed with chloroform:methanol (80:20, v/v). TLC plates were analyzed with a Typhoon FLA 9500 scanner (GE Healthcare Life Sciences) and lipid spots were quantified with Image Quant (GE Healthcare Life Sciences).
Liquid chromatography tandem–mass spectrometry of ceramides
Following lipid extraction with a mixture of methanol:chloroform (2:1, v/v) that contained C17:0 ceramide and d7-sphinganine (both from Avanti Polar Lipids, Alabaster, USA) as internal standard, ceramide species (C16:0, C18:0, C20:0, C22:0, C24:0, and C24:1) and sphinganine were quantified by liquid chromatography tandem–mass spectrometry (LC–MS/MS) using a 6495C triple–quadrupole mass spectrometer (Agilent Technologies, Waldbronn, Germany) operating in the positive electrospray ionization mode (ESI +). 28 Quantification was performed with MassHunter Software (Agilent Technologies).
Isolation and analysis of small extracellular vesicles (sEVs)
To isolate EVs, conditioned cell culture media were collected and centrifuged at 2000g for 15 min at 4 °C, followed by centrifugation at 10,000g for 45 min at 4 °C (5810R centrifuge; Eppendorf, Hamburg, Germany). Supernatants were filtered through a 0.22 µm filter (Sartorius, Göttingen, Germany) and supplemented with 75 mM NaCl and 10% polyethylene glycol-6000 (PEG; Sigma–Aldrich). sEVs were concentrated at 1500g for 30 min at 4 °C (Avanti J-26 XP centrifuge; Beckmann Coulter, Brea, CA, USA). Pellets were then dissolved in 0.9% NaCl (Sigma–Aldrich), transferred to ultra-clear centrifuge bottles (Beckmann Coulter) and precipitated by ultracentrifugation at 110,000g for 130 min at 4 °C (Optima L7-65, k factor: 133; Beckmann Coulter). sEV pellets were resuspended in 0.9% NaCl supplemented with 10 mM HEPES (Life Technologies) and stored in low retention microcentrifuge tubes (Kisker Biotech, Steinfurt, Germany). EV size was analyzed by nanoparticle tracking analysis (NTA; Particle Metrix, Meerbusch, Germany), as described previously. 29 For quantitative measurements, sEVs were labeled with CD9–FITC (MEM-61; Exbio, Vestec, Czech Republic) and CD63–APC (MEM-259; Exbio) antibodies and analyzed by ImageStreamX MkII imaging (Merck Millipore). 30 For tube formation assays, hCMEC/D3 were treated with sEVs at a concentration of 25 µg/ml.
Transmission electron microscopy of sEV preparations
Two hundred-mesh copper grids (Plano, Wetzlar, Germany) were physically charged by a glow discharge procedure to allow strong adherence of particles to the electron microscopy grid during further processing. Four point five microliter of sEV preparations were added to the pre-treated grid surface and dried at room temperature. For subsequent removal of salts, grids were successively transferred on three droplets of deionized water. Samples were incubated with 1.5% phosphotungstic acid (Electron Microscopy Science, Hatfield, PA, USA) and dried. Images were acquired using a JEM 1400Plus electron microscope (JEOL, Tokyo, Japan) operating at 120 kV that was equipped with a 4096 × 4096 pixel CMOS camera (TemCam-F416; TVIPS, Gauting, Germany). Image acquisition software EMMENU (Version 4.09.83) was used for taking 16 bit images. Image post-processing was carried out using ImageJ software (version 1.52b; NIH).
Enzyme-linked immunosorbent assay for VEGF
VEGF was measured by enzyme-linked immunosorbent assay (ELISA; R&D Systems) in supernatants of hCMEC/D3 cells according to the manufacturer’s instruction.
Statistical analysis
Data are expressed as mean ± standard deviation (SD). In case of multiple group comparisons, two-way ANOVA followed by least significant difference (LSD) posthoc tests was used and for comparisons between two groups, unpaired or paired two-tailed t tests were applied, as appropriate. All values were normally distributed. p < 0.05 were defined to indicate statistical significance. The statistical details (n numbers and tests) are given in the figure legends. Statistical analyses were performed using GraphPad Prism version 9.5.1 software.
Results
Serine palmitoyltransferase deactivation reduces cerebral microvascular endothelial ceramide levels
To evaluate the role of the de novo ceramide synthesis pathway in regulating angiogenesis, we administered the SPT inhibitor myriocin (1 µM) to human microvascular cerebral endothelial cells (hCMEC/D3). Enzyme activity measurements revealed a near-complete reduction of SPT activity by 98.8% ± 1.4% after myriocin treatment (Figure 1(a)). In order to test that myriocin did not modulate other ceramide formation pathways, specifically sphingomyelin hydrolysis, ASM activity was measured using BODIPY-labeled sphingomyelin as substrate. ASM activity was not altered by myriocin (Figure 1(b)).
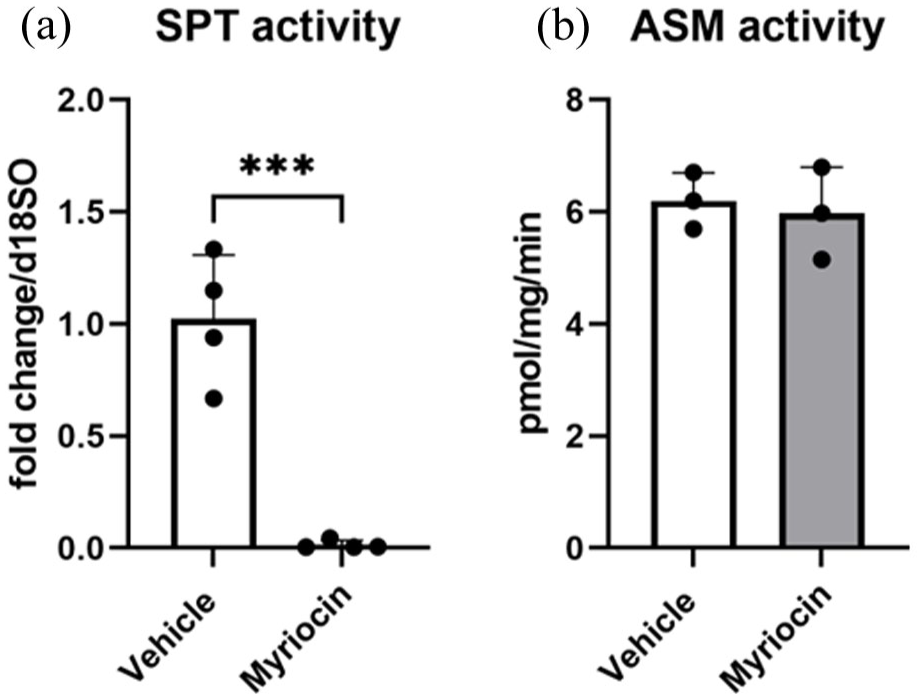
Myriocin inhibits SPT activity and does not affect the sphingomyelin hydrolysis pathway. (a) SPT activity was measured by LC–MS and (b) ASM activity was assessed using BODIPY-labeled sphingomyelin in hCMEC/D3 after treatment with vehicle or myriocin (1 µM). Data are means + SD values.
To rule out that observations made are attributed to off-target effects, we knocked down SPT by small interfering RNA (siRNA) in hCMEC/D3 cells in a second approach. The efficacy of transfection was examined by Western blots for serine palmitoyltransferase long chain base subunit 1 (SPTLC1), which is an essential subunit of the heterodimeric SPT complex and is indispensable for enzymatic action. 31 SPTLC1 protein abundance after 72 h was efficiently reduced by 43.2% ± 5.4% (Figure 2(a) and (b)), demonstrating the successful transfection with SPT siRNA.
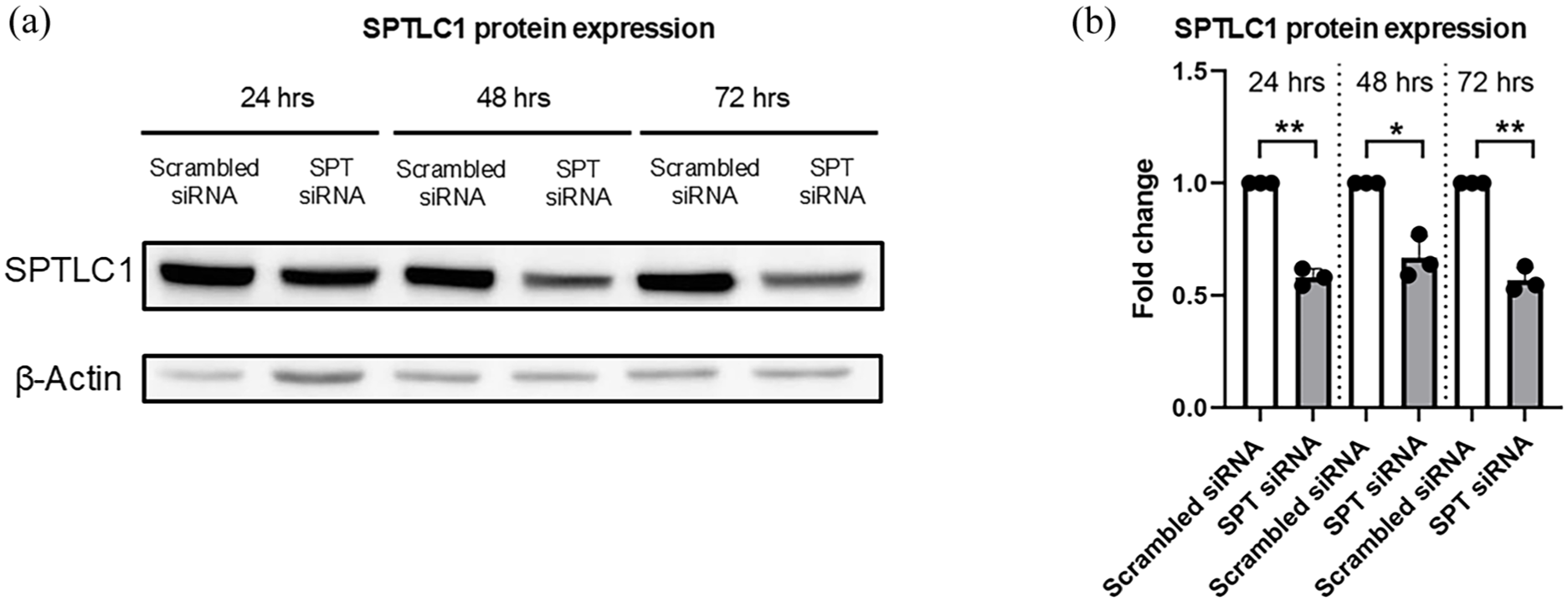
SPTLC1 abundance is effectively downregulated by siRNA in vitro. (a, b) Successful downregulation of SPTLC1 protein expression was confirmed by Western blot in hCMEC/D3 transfected with scrambled siRNA and SPT siRNA for 24–72 h. Data are means + SD values.
Both pharmacological SPT inhibition and genetic SPT knockdown significantly reduced ceramide levels in hCMEC/D3, as assessed by LC-MS/MS (Figure 3(a) and (c)). Ceramide species can be distinguished according to their acyl chain length, which have distinct cellular functions.32–34 Of note, both C16:0 and C24:0 ceramides were reduced by myriocin (Figure 3(a)). Additionally, sphinganine, a ceramide precursor in the de novo pathway, was measured (Figure 3(b) and (d)). Myriocin treatment significantly reduced sphinganine levels (Figure 3(b)).
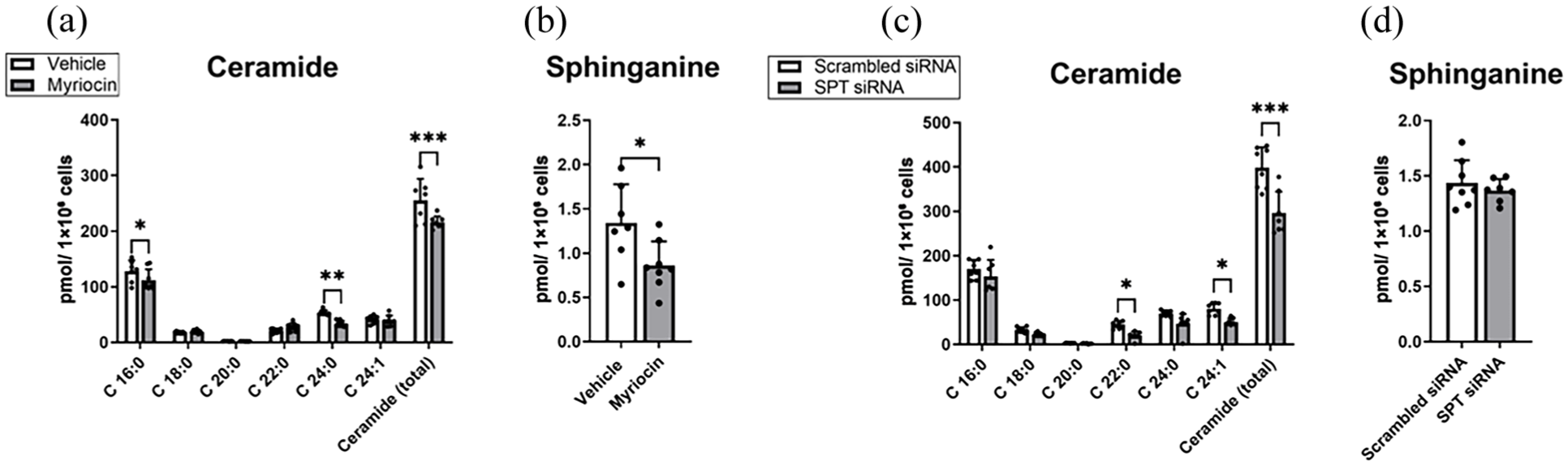
Pharmacological inhibition and genetic downregulation of SPT reduces ceramide abundance. In order to assess if ceramide reduction is achieved by pharmacological and genetic downregulation of SPT activity, ceramide, and sphinganine content was measured by LC–MS/MS in hCMEC/D3 (a, b) after treatment with vehicle or myriocin (1 µM) or (c, d) after transfection with scrambled siRNA or SPT siRNA. Both strategies reduced total ceramide levels. Data are means + SD values.
Deactivation of ceramide de novo synthesis stimulates cerebral angiogenesis in vitro
Next, we analyzed the effect of SPT deactivation on angiogenesis in vitro. Matrigel-based tube formation and migration assays showed that SPT inhibition by myriocin increased the tube formation and migration of hCMEC/D3 cells (Figure 4(a)–(d), (m), and (n)). Besides the number of closed tubes, the number of branching points was increased by myriocin (Figure 4(a), (b), and (m)), while mean branch length was non-significantly reduced (Figure 4(c) and (m)). Cell viability was slightly, but not significantly increased by pharmacological SPT inhibition (Figure 4(e)). ELISA revealed that VEGF released in supernatants was significantly increased by myriocin (Figure 4(f)).
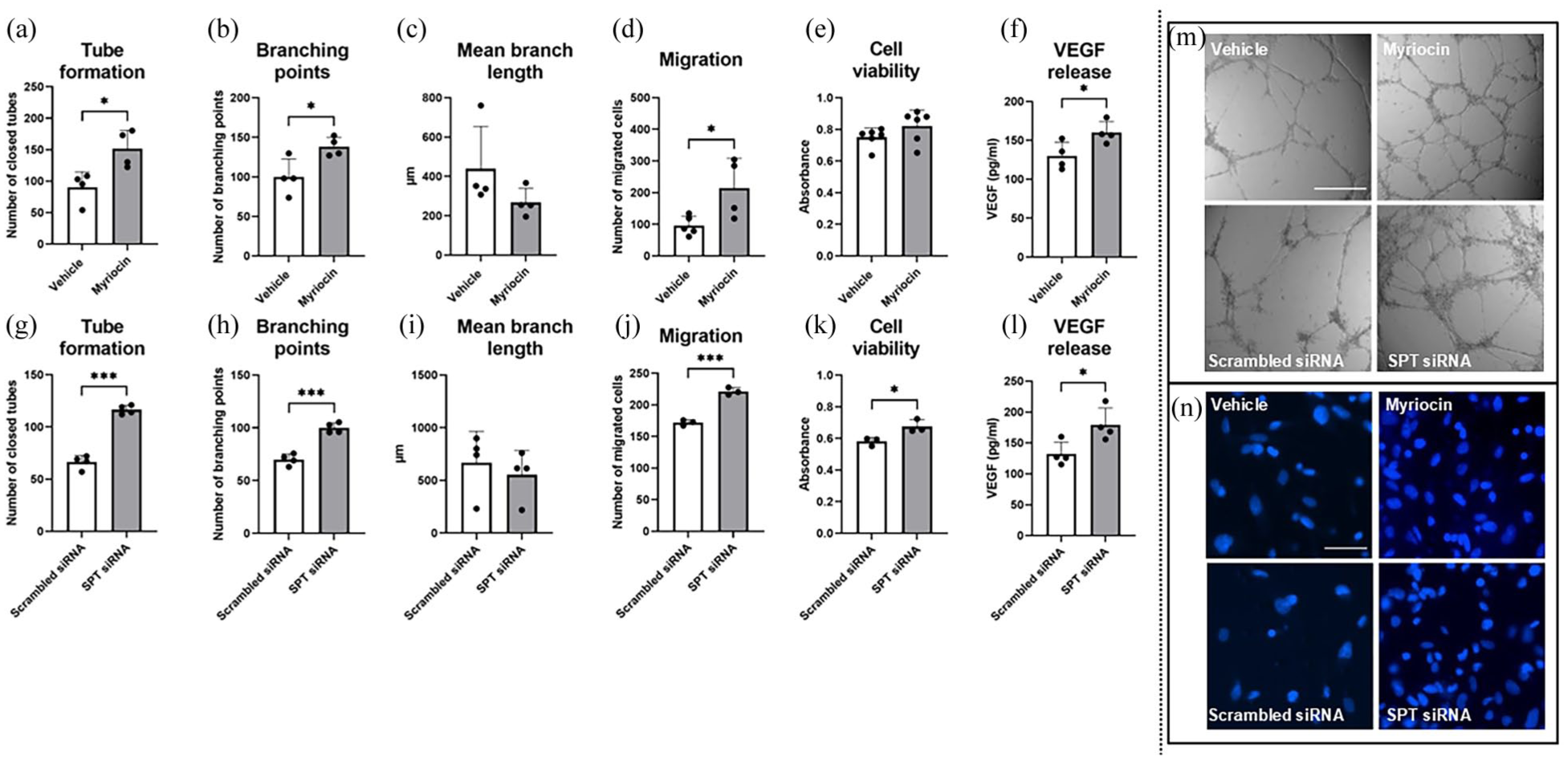
Pharmacological inhibition and genetic downregulation of SPT induces angiogenesis in vitro. (a, g) Number of closed tubes, (b, h) branching point number and (c, i) mean branch length assessed by matrigel-based tube formation assay, (d, j) endothelial migration assessed by transwell migration assay, (e, k) endothelial viability, and (f, l) VEGF release of hCMEC/D3 after treatment with vehicle or myriocin (1 µM; a–f) or transfection with scrambled siRNA or SPT siRNA (g–l). Representative microphotographs of (m) matrigel-based tube formation and (n) migration assay images that depict nuclei of cells that migrated through the transwell-filter are also shown. Scale bar in (m), 500 µm and in (n), 50 µm. Data are means + SD values.
In hCMEC/D3 exhibiting SPT siRNA knockdown, tube formation, cell migration, and cell viability were likewise increased compared to cells exposed to scrambled (control) siRNA (Figure 4(g)–(k), (m), and (n)), as was VEGF release assessed by ELISA (Figure 4(l)). These findings confirm that the inhibition of ceramide de novo synthesis indeed promotes angiogenesis.
Inhibition of ceramide de novo synthesis stimulates release of small extracellular vesicles which lack angiogenic properties
sEVs are important vehicles of paracrine cell communication. 35 Since it was previously shown that ceramide lowering by ASM inhibitors increases the release of sEVs which enhanced angiogenesis, 8 we aimed to evaluate if similar changes are induced by SPT deactivation. The pharmacological inhibition by myriocin increased the release of CD9+ and CD63+ sEVs (Figure 5(a) and (b)), which are tetraspanin proteins highly enriched on the surface of sEVs (Figure 5(a) and (b)). Genetic knockdown of SPT by siRNA enhanced the secretion of CD9+ sEVs but had no significant effect on CD63+ sEVs (Figure 5(c) and (d)). To assess the functional role of these sEVs, sEVs were isolated from cell supernatant by ultracentrifugation and PEG precipitation, as previously described. 36 The particle size of EVs was assessed by NTA. The mean size of EVs was 125.97 ± 2.29 and 117.84 ± 6.45 nm after vehicle and myriocin treatment, respectively (Figure 5(f)), which is in the range of exosomes. Myriocin increased the proportion of sEVs (<150 nm diameter), measured by NTA, by 30%–35% (Figure 5(e)). The total EV particle number was 6.03 × 1011 ± 6.66 × 1010 and 5.46 × 1011 ± 1.45 × 1011/ml, respectively. Of note, a main limitation of NTA is its reduced sensitivity for EVs <50 nm, 37 which may lead to underestimation of the total EV particle count for small EV populations. Transmission electron microscopy confirmed that the sEVs exhibited the characteristic size and cup-shaped morphology of exosomes (Figure 5(g)). Western blot analysis demonstrated the presence of exosomal markers syntenin, CD81, TSG101, and Alix while the cellular contamination marker calnexin was absent in the sEV preparations (Figure 5(h)). The tube formation experiments revealed that angiogenesis was not stimulated by EVs derived from vehicle-treated cells or myriocin-treated cells (Figure 5(i)), suggesting that, in contrast to ASM inhibition, SPT deactivation does not modify sEVs in a way that confers angiogenic properties.
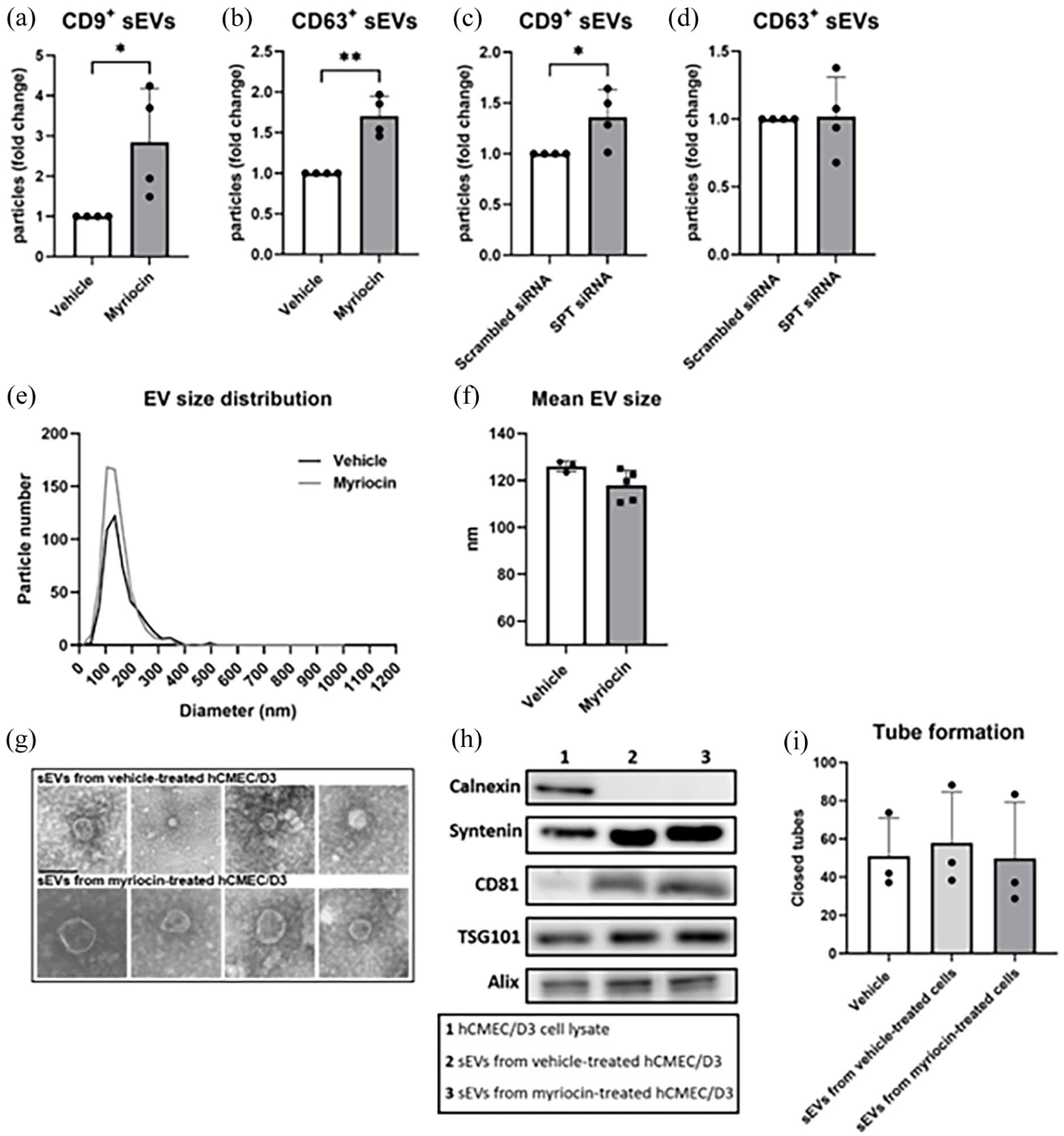
SPT inhibition or downregulation amplifies release of sEVs, which do not have angiogenic properties. To evaluate if SPT influences the release of sEVs, (a, c) CD9+ sEVs and (b, d) CD63+ sEVs were quantified by AMNIS in the supernatant of hCMEC/D3 after treatment with vehicle or myriocin (1 µM; a, b) or following transfection with scrambled siRNA or SPT siRNA (c, d). Both approaches robustly increased the release of sEVs. (e, f) The EV size distribution and mean EV size was assessed by NTA. (g) Transmission electron microscopy showing representative sEVs obtained from supernatants of hCMEC/D3 exposed to vehicle or myriocin (1 µM). Scale bar, 100 nm. (h) Western blots for the cellular contamination marker calnexin, and the exosomal markers syntenin, CD81, TSG101, and Alix using protein samples obtained from hCMEC/D3 lysates, sEVs from vehicle-treated hCMEC/D3 and sEVs from myriocin-treated hCMEC/D3. (i) Tube formation assay of hCMEC/D3 after treatment with vehicle, 25 µg/ml sEVs obtained from vehicle treated hCMEC/D3 or 25 µg/ml sEVs obtained from myriocin (1 µM) treated hCMEC/D3, showing that sEVs released upon SPT deactivation did not exhibit any angiogenic properties. Data are means + SD values.
Inhibition of ceramide de novo synthesis increases microvascular remodeling after ischemic stroke in vivo
We next explored if SPT inhibition stimulates vascular remodeling in vivo by analyzing the network structure of cerebral microvessels using 3D light sheet fluorescence microscopy (LSFM) in mice exposed to MCAO which were treated with vehicle or myriocin (1 mg/kg) for 14 days. Vessels were labeled by the microvascular tracer FITC–albumin. To obtain a comprehensive understanding of the vascular system, microvessels were discriminated according to their diameter size and subsequently classified as small (<4 µm), intermediate (4–5.4 µm), and large microvessels (>5.4 µm) using the VesselExpress pipeline (Figure 6(a)–(i)), given that we previously showed that the formation of new microvessels post-MCAO is predominated by the appearance of small microvessels (<4 µm). 38 In vehicle treated mice, ischemic stroke markedly reduced the length density of intermediate microvessels (4–5.4 µm diameter) in the reperfused striatum by 38.88% ± 21.59% (Figure 6(b)). These intermediate microvessels make up ~80% of all microvessels in our study, as evidenced by length densities in mm/mm3. The length density of small microvessels (<4 µm) significantly increased in the previously ischemic striatum and the contralateral non-ischemic striatum of myriocin-treated compared with vehicle-treated mice by 181.35% ± 197.51% and 161.68% ± 193.7% (Figure 6(a)). The number of branches of small microvessels was also increased by myriocin in the previously ischemic striatum (Figure 6(d)), whereas branch length was reduced (Figure 6(g)). Further analysis showed that the vessel length density and branch density of intermediate-sized microvessels was not altered by myriocin (Figure 6(b) and (e)). The branch length of intermediate-sized microvessels in the ischemic striatum was reduced by myriocin (Figure 6(h)). SPT inhibition by myriocin increased the microvascular length density and number of branches of large microvessels in the ischemic striatum (Figure 6(c) and (f)). The branch length of large microvessels was neither influenced by MCAO nor myriocin (Figure 6(i)).
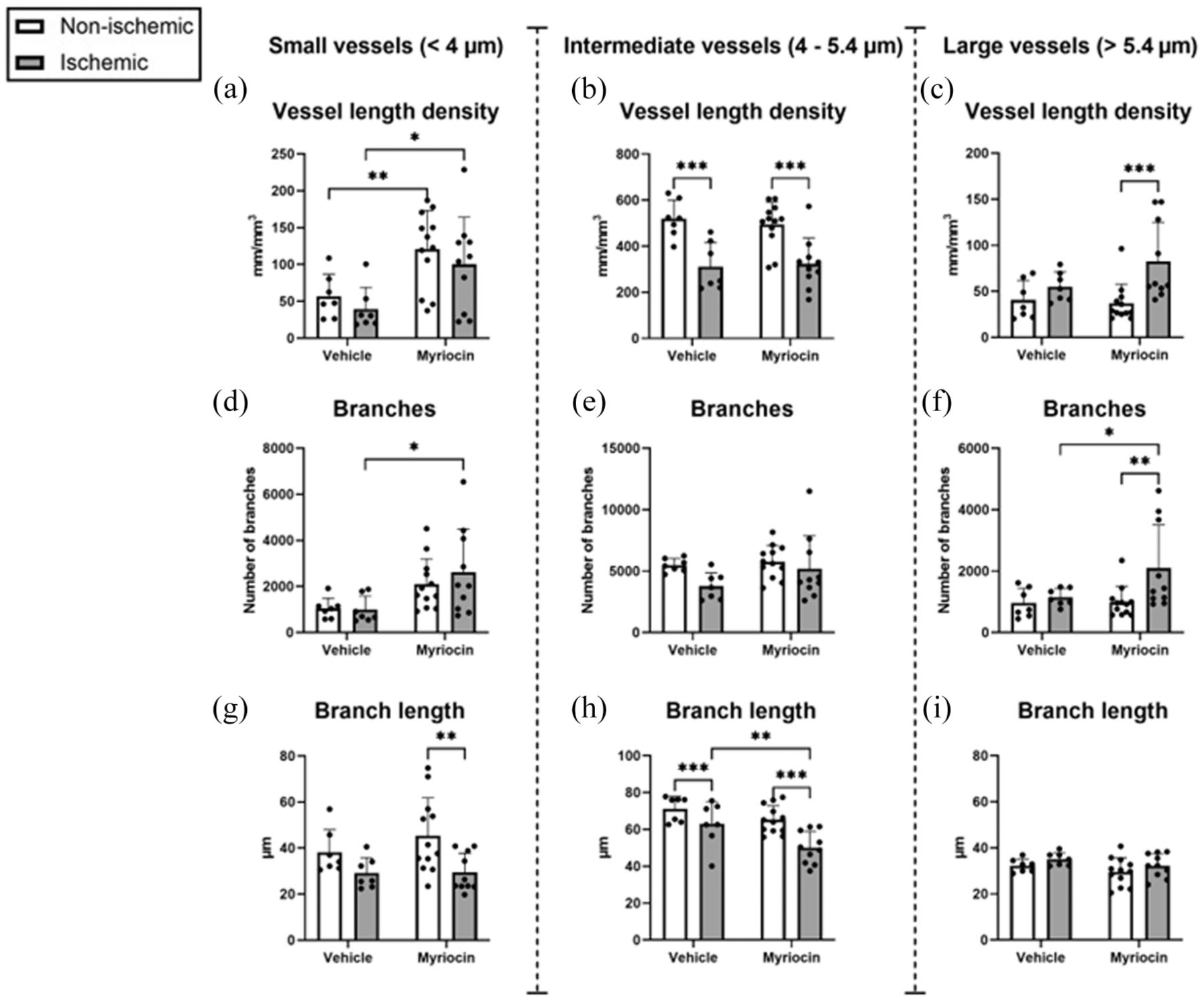
SPT deactivation increases the density of small diameter microvessels after ischemic stroke in vivo. Microvessels were discriminated according to their diameter and analyzed for vessel length density, number of branches, and mean branch length. Total microvascular length of (a) small diameter (<4 µm), (b) intermediate diameter (4–5.4 µm), and (c) large diameter (>5.4 µm) microvessels, number of branches with (d) small diameter (<4 µm), (e) intermediate diameter (4–5.4 µm), and (f) large diameter (>5.4 µm), and branch length of (g) small diameter (<4 µm), (h) intermediate diameter (4–5.4 µm), and (i) large diameter (>5.4 µm) microvessels were evaluated by LSFM in the ischemic and contralesional non-ischemic striatum of MCAO mice treated with vehicle or myriocin (1 mg/kg, s.c.) for 14 days. Note that SPT deactivation by myriocin increased the length density and branch density of small microvessels (<4 µm) in both hemispheres. Data are means + SD values.
Discussion
We herein show that deactivating the ceramide de novo synthesis pathway by pharmacological SPT inhibition or genetic SPT knockdown promotes cerebral angiogenesis of hCMEC/D3 in vitro and increases vascular remodeling and angiogenesis in mice exposed to intraluminal MCAO. Although sEV release from endothelial cells was increased by SPT deactivation, tube formation was not enhanced by treatment with sEVs from hCMEC/D3 treated with the SPT inhibitor myriocin, suggesting that other mechanisms, independent of sEVs, mediated the pro-angiogenic responses. Indeed, VEGF release of hCMEC/D3 was increased both by pharmacological SPT inhibition and genetic SPT knockdown, pointing towards a role of this growth factor in the angiogenic responses noted.
The present study complements previous studies of our group, in which we demonstrated that pharmacological ASM deactivation by the antidepressants amitriptyline and fluoxetine increased angiogenesis, mitigated brain inflammatory responses and increased neuronal survival in the same MCAO model in mice.8,39 In our earlier studies, we found that sEV release from cerebral endothelial cells (again hCMEC/D3) was increased by amitriptyline, which, when taken up by hCMEC/D3 cells, increased angiogenesis revealed by endothelial tube formation and migration assays, furthermore increasing the release of VEGF of target cells. 8 ASM knockdown abolished these actions in vitro and in vivo, providing the proof that ASM mediates the restorative actions of antidepressants in cerebral I/R injury. 8 Despite a promising double-blind, randomized controlled phase II study demonstrating that the ASM inhibitor fluoxetine administered starting 5–10 days post-stroke improved stroke outcome after 90 days in ischemic stroke patients, 13 these findings could not be validated by large phase III multicenter trials, in which fluoxetine was applied starting between 2 and 15 days post-stroke.14,15 In view of the broad window of treatment onset, it is conceivable that the critical time window for induction of angiogenesis, which in our experience is within the first 7–maximum 14 days, 40 was missed in the latter phase III trials. It is also conceivable that the dose administered was too low to achieve a clinically relevant ceramide reduction in cerebral microvascular cells.
Besides the formation of membrane microdomains, ceramide is critically involved in the endosomal sorting complex required for transport (ESCRT)-independent processes that govern the budding of intraluminal vesicles (ILVs) into multivesicular bodies (MVBs). 18 These MVBs, in turn, release their contents as exosomes, a key type of small extracellular vesicle involved in intercellular communication. 18 Concentration gradients of ceramide in the two endosomal membrane leaflets are critical for initiating membrane budding and the formation of ILVs. Ceramide gradients are rapidly controlled by sphingomyelin hydrolysis through ASM, which is localized on the luminal endosomal membrane leaflet, and neutral sphingomyelinase (NSM), which is found on the cytosolic membrane leaflet. 11 An elevated ceramide levels on the luminal membrane leaflet induces a negative membrane curvature, presumably due to ceramide’s cone-shaped physicochemical structure, which leads to membrane repulsion towards the cytosolic side. 18 Conversely, ceramide lowering on the luminal membrane leaflet by administering ASM inhibitors increases membrane budding and sEV formation. 8 In contrast, ceramide formation on the cytosolic membrane leaflet promotes membrane budding and sEV formation.18,41 Since NSM expression in endothelial cells is low, 8 ASM decisively controls sEV release in endothelial cells.
Unlike ASM inhibition, SPT deactivation does not lead to leaflet-specific ceramide reduction and therefore likely does not influence EV budding the same way. This difference in the subcellular site of action might explain why sEVs released upon SPT deactivation unlike those released by ASM inhibitors did not reveal angiogenic actions in hCMEC/D3. That both sEV isolates had diverse biological activity highlights the complexity of the underlying EV release mechanisms. EVs are highly heterogeneous. They vary in size, composition, and biological function, and can be classified into three main types: exosomes (60–150 nm), microvesicles (100 nm–1 µm), and apoptotic bodies (⩾1 µm). 42 Their heterogeneity is influenced by the cellular origin, and microenvironment, which determine the specific cargo (proteins, lipids, RNAs), as well as surface markers and biological effects.43,44 In contrast to exosomes, which are derived from ILVs as MVBs fuse with the plasma membrane, microvesicles are released directly from the plasma membrane through outward budding. 44 Besides exosomes, a number of other EV classes are released from the inside of cells, for example, from the nucleus and endoplasmic reticulum. 45 The method of EV isolation used in this study does not allow us to discriminate between different EV classes, and there are currently no markers specific for one of these categories. Given the different angiogenic properties, it is likely that the inhibition of the de novo ceramide synthesis pathway and ASM inhibition affected different sEV sets, which differed in their cargo and biological activity. In our angiogenesis studies, EV concentration was normalized based on protein content, which is a widely used approach for the quantification of EV samples.46,47 Of note, cell number did not differ between groups in hCMEC/D3 cultures, in which EVs were harvested. Thus, EV normalization based on cell number would not have led to different results. We used state-of-the-art strategies of EV isolation and characterization throughout this study in line with ISEV criteria. 48
In view of the absence of angiogenic effects of sEVs released by SPT deactivation, the angiogenic effects of the inhibition of the de novo ceramide synthesis pathway are likely attributed to VEGF, which was released by pharmacological and genetic SPT deactivation. VEGF receptors are expressed on endothelial cells, inducing downstream signaling through the phosphatidylinositol 3-kinase (PI3K)/Akt (protein kinase-B) pathway, the Raf–mitogen-activated or extracellular signal-regulated protein kinase kinase (MEK)–mitogen-activated protein kinase (MAPK) pathway and the Src–focal adhesion kinase (FAK) pathway, which control cell survival, proliferation, migration, and differentiation.49–53 VEGF has been shown to reduce infarct volume, mitigate neurological deficits and enhance angiogenesis and neuroplasticity after ischemic stroke.50,54,55 To evaluate the effect of systemic myriocin delivery after stroke on the cerebral vascular system, we conducted a detailed analysis of the cerebral microvascular network using 3D light sheet microscopy using the VesselExpress pipeline. 26 Our data show that SPT inhibition significantly promoted vascular remodeling. As previously demonstrated, stroke-induced microvascular injury mainly affects small-to-intermediate microvessels. 38 In our study, ischemic stroke markedly reduced the length density of intermediate microvessels (4–5.4 µm diameter) in the reperfused ischemic striatum, which according to our data make up ~80% of all microvessels. Strikingly, SPT inhibition specifically increased the length density and branch density of small (<4 µm diameter) and large (>5.4 µm diameter) microvessels in the reperfused ischemic striatum. Of note, post-stroke angiogenesis is largely driven by the reappearance of small microvessels. 38 Hence, the increased formation of small microvessels indicates that SPT inhibition amplifies the brain’s restorative response. Besides providing oxygen and nutrients to the previously ischemic tissue, new-born microvessels are a major source of growth factors in the injured brain. 54 New-born microvessels furthermore act as scaffolds for the migration of neuroblasts in direction to injured tissue, which support brain parenchymal remodeling processes. 56
In summary, we herein demonstrated that ceramide lowering by the deactivation of the de novo ceramide synthesis pathway increases cerebral angiogenesis in vitro and microvascular remodeling in mice exposed to MCAO. Given the overriding importance and complexity of signaling processes induced by ceramide modulation, further studies evaluating ceramide’s role in brain restorative processes are warranted. In view of the failure of the fluoxetine phase III multicenter trials,14,15 such studies may provide us necessary insights in order to develop targeted therapies allowing us to enhance stroke recovery in human patients.
Footnotes
Acknowledgements
We thank the Imaging Center Essen (IMCES) at the Faculty of Medicine of the University of Duisburg-Essen, Germany for providing access to its electron microscopy services including sample preparation and transmission electron microscopy.
Author contributions
AMY, TH, AL, NH, TH, TT, FS, BK contributed to the data acquisition and analysis. AMY, TH, and DMH developed the study concept and design, interpreted data, and drafted the manuscript. UB, BG, MG, EG, critically revised the manuscript together with all other coauthors. All authors approved the final version of the manuscript.
Funding
Supported by German Research Foundation (DFG) grants 259317790 (GRK2098), 389030878, 405358801/428817542 (within FOR2879), 449437943 (within TRR332, project C06), and 514990328.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
