Abstract
Adenosine A2 receptors (A2ARs) have shown promising therapeutic properties despite their controversial role in modulating stroke outcome. However, the temporal evolution of cerebral A2ARs density after cerebral ischemia and its subsequent neuroinflammatory response have been scarcely explored. In this study, the expression of A2ARs after transient middle cerebral artery occlusion (MCAO) was evaluated in rats by positron emission tomography (PET) with [11C]SCH442416 and immunohistochemistry (IHC). In addition, the role of A2ARs in stroke inflammation with pharmacological modulation was assessed with magnetic resonance imaging (MRI), PET imaging with [18F]DPA-714 (TSPO), IHC, western-blot, and autoradiography. After cerebral ischemia, [11C]SCH442416 and IHC revealed neural expression of A2ARs in the striatum in healthy brains, followed by a binding decrease at day 1 and a subsequent significant increase at day 3 after ischemia in microglia and infiltrated leukocytes. Furthermore, activation of A2ARs with the agonist CGS-21680 resulted in a reduction in stroke volume, along with an increase in TSPO expression in immune cells in the striatum. Our results provide novel evidence on A2ARs density dynamics after cerebral ischemia that might guide the therapeutic management of stroke by modulating adenosine receptors.
Introduction
Adenosine purinergic A2A receptors (A2ARs) are highly expressed in striatal neurons, with much lower presence in other parts of the brain, such as cortex and hippocampus. 1 The activation of A2ARs is critical for the control of a variety of cognitive behaviors including working memory, motor sequence learning and decision making, among others.2 –4 In addition, adenosine exerts a variety of effects on hearth muscle, coronary arteries, platelets, and cells involved in innate (microglia, macrophages, mast cells, and neutrophils) but also in adaptive immunity (lymphocytes) cells, supporting its control of the neuroinflammatory response.5,6
Ischemic stroke triggers a dramatic increase in adenosine extracellular concentration, which can stimulate A2ARs expressed by neurons and both innate and adaptive immune cells, suggesting their control of both primary and secondary ischemic injury, respectively.7 –9 A2ARs might play a detrimental or protective role depending on the time window following cerebral ischemia. 9 A2AR activation triggers excitotoxicity in a very short time window (minutes to few hours), followed by a protective role in secondary inflammatory damage during days after stroke onset. 10 Therefore, a potential therapeutic strategy could involve an early treatment with A2ARs antagonists to reduce excitotoxicity followed by treatment with agonists for the later control of secondary ischemic injury. However, other studies have shown neuroprotective effects in ischemic rats treated with only the A2ARs agonist CGS-21680 from a few hours until several days after ischemia. 11 Despite this controversy, these receptors have attracted increased interest in recent years because of their promising therapeutic properties after cerebral ischemia. 12 However, the temporal evolution of cerebral density after cerebral ischemia remains unknown hence, the use of non-invasive positron emission tomography (PET) would be extremely helpful to gain knowledge of their expression and the protective role that they might play in ischemic stroke.
In recent decades, promising radiotracers such as [11C]SCH442416, which bind selectively and reversibly to striatal A2ARs with nanomolar affinity, have been developed.13,14 [11C]SCH442416 is a selective antagonist of A2ARs with high affinity and signal-to-noise ratio that has shown low radioactive metabolite levels in the brains of rats, primates, and humans.13,15,16 Nevertheless, only a limited number of PET imaging studies using [11C]SCH442416 have reported alterations in A2AR density in brain pathologies such as Parkinson’s disease and schizophrenia.15,16 Further evaluation of A2ARs expression in brain ischemia may aid in identifying novel diagnostic and therapeutic options for stroke damage.
Our study aimed to investigate the density of A2ARs during subacute and chronic phases of ischemic stroke by combining PET and magnetic resonance imaging (MRI). In particular, the role of these receptors in the neuroinflammatory response after ischemic stroke was studied using in vivo imaging, ex vivo studies, and neurofunctional evaluation. Ischemic rats treated with the A2ARs agonist CGS-21680 or vehicle were subjected to T2W-MRI and PET studies with [18F]DPA-714 for imaging the translocator protein 18 kDa (TSPO) as a surrogate imaging biomarker for the inflammatory reaction.17,18 Finally, immunohistochemistry (IHC) was used to evaluate the modulatory effect of A2ARs on purinergic and TSPO expression in both microglia and infiltrated macrophages following stroke onset.
The results reported here shed light on the role of A2ARs in brain ischemia and the potential of these receptors as modulators of acute injury and neuroinflammation. Hence, these findings could pave the way for the development of novel treatments that ultimately improve the clinical management of ischemic stroke.
Materials and methods
Animal model and experimental setup
Adult male Sprague‒Dawley rats (n = 74; 308 ± 6.2 g body weight; Janvier, France) were used for in vivo imaging studies, ex vivo immunohistochemistry, and western blot analysis (Table S1).
Animal experimental protocols and relevant details regarding welfare were approved by the Ethical Committee at CICbiomaGUNE, UPV/EHU, and local authorities and were conducted in accordance with the Spanish legislation (Real Decreto 53/2013) and the Directives of the European Union on animal ethics and welfare. Results are reported following the ARRIVE guidelines. All the studies were conducted under same experimental conditions in AAALAC certified animal facilities.
Transient focal ischemia was produced under anesthesia by a 90-min intraluminal occlusion of the middle cerebral artery (MCAO) followed by reperfusion as described previously. 19 In brief, rats were anesthetized with 2.5% isoflurane in 100% O2, and a 2.6-cm length of 4–0 monofilament nylon suture was introduced into the right external carotid artery up to the level where the MCA branches out. Animals were then sutured and placed in their cages with free access to water and food. Ten minutes before the end of the 90-min occlusion, the animals were reanesthetized, and the filament was removed to allow reperfusion. In the case of sham rats, animals were subjected to the same surgical procedure without the occlusion of the MCA. Following surgery, rats were kept in their cages with free access to food and water.
Perioperative and postoperative pain management was conducted using subcutaneous injections of buprenorphine at a dose of 0.05 mg/kg. The first dose was administered 15 min before MCAO, and subsequent doses were given every 12 h for the next 72 h.
Ischemic rats (n = 6) were subjected to T2-weighted (T2W) MRI scans at 24 h after reperfusion (i) to include animals showing cortico-striatal infarction in PET studies and (ii) to measure the volume of infarction. Rats were repeatedly scanned by PET with [11C]SCH442416 at day 0 (before MCAO) and at days 1, 3, 7, 14, 21, and 28 after cerebral ischemia to determine the distribution of A2ARs in the rat brain. Additionally, a sham group of rats (n = 4) were subjected to [11C]SCH442416 PET studies to evaluate the effect of surgery on A2AR binding.
A2AR protein expression was analyzed in a group of 49 rats using immunohistochemistry, western blotting and autoradiography before (day 0) and at days 1 and 3 after stroke. (For 6 consecutive days, starting on day 1 following MCAO, the first group of five rats was treated daily with an intraperitoneal (i.p.) injection of CGS-21680 (0.1 mg/kg), an agonist of A2ARs, and a control ischemic group of five rats received the same daily volume of vehicle (physiologic saline solution) in a randomized and blinded fashion. At day 7, all the rats from these two groups were subjected to PET and neurological assessment to determine the effects of CGS-21680 on TSPO expression and neurological outcomes. Ex vivo ICH studies for TSPO and A2ARs were also performed on those animals to validate the PET imaging findings during the first 72 h after ischemia onset. Finally, five rats were used for the validation of [18F]DPA-714 PET acquisitions (Figure S1).
MRI scans and analysis
T2W-MRI scans were performed in rats anesthetized with isoflurane (2%–2.5%) in a 30%/70% mixture of O2/N2. Animals (n = 10) were placed into an MRI rat compatible holder and maintained under normothermic conditions (37 ± 0.2 °C) via a hot air flow-based system, interfaced and controlled by a SAII M1030 system (SA Instruments, NY, USA), which was also used to ensure animal welfare through continuous monitoring of the temperature and respiration rate while animals remained in the MRI scanner. MRI in vivo studies were performed on a 7T horizontal bore Bruker Biospec USR 70/30 MRI system (Bruker Biospin GmbH, Ettlingen, Germany) interfaced with an AVANCE III console running ParaVision 7.0 and with a BGA12-S imaging gradient insert (maximal gradient strength 400 mT/m, switchable within 80 ms). Measurements were performed with a 72 mm volumetric quadrature coil for excitation and a 20 mm rat brain surface coil for reception. The imaging session started with the acquisition of a scout scan, which was used to plan the whole study, with a focus on the region of interest. T2W images were acquired with a Bruker RARE (Rapid Acquisition with Relaxation Enhancement) sequence (Effective TE = 40 ms, TR = 4400 ms, NA = 2; Matrix = 256 × 256 points; FOV = 25.6 × 25.6 mm; spatial resolution = 100 × 100 mm; 24 contiguous slices of 1 mm thickness covering the whole brain), which was used to quantify the volume of the lesion.
For image analysis, regions of interest (ROIs) were manually defined using ImageJ (version 1.53k, NIH) software. The calculation of the lesion volume using MRI was carried out by summing the areas of infarcted regions showing hyperintense signals of all slices affected by the lesion. The evaluation of midline displacement in the entire brain was performed using the following formula: ((volume of ipsilateral hemisphere/volume of contralateral hemisphere) − 1) × 100.
Radiochemistry
The synthesis of [11C]SCH442416 was carried out as previously described, 20 via the direct alkylation of the des-methylated precursor with [11C]CH3I in the presence of sodium hydroxide as the base, using the captive solvent method. 21 In brief, [11C]CH4 was directly generated in an IBA Cyclone 18/9 cyclotron and transferred to a TRACERlab FXCPro synthesis module (GE Healthcare), where [11C]CH3I was generated. At the end of the process, [11C]CH3I was distilled under continuous helium flow (20 mL/min) and introduced in a 2 mL stainless steel reaction loop, pre-charged with a solution of the precursor (1 mg) in dimethylformamide (100 µL) and 5 M aqueous sodium hydroxide solution (10 µL). After reaction (5 min, room temperature) purification was performed by high-performance liquid chromatography on a Mediterranea Sea18 column (250 × 10 mm, 5 µm particle size) using 0.05 M aqueous NaH2PO4 (pH = 3)/acetonitrile (50/50) as the mobile phase. The collected fraction (retention time = 14–15 min) was formulated by retention on a C-18 cartridge (Sep-Pak® Light; Waters) and elution with ethanol (0.7 mL) and saline (6.3 mL). Filtration through 0.22 µm sterile filters yielded the final tracer solution. Average decay corrected radiochemical yield and molar activity were 25% ± 7.2% and 50.2 ± 26.32 GBq/µmol, respectively. Radiochemical purity was higher than 98% at injection. [18F]DPA-714 was produced exactly as previously described. 17
PET scans and analysis
PET images for different studies were acquired using an Explore Vista PET-CT camera (GE Healthcare, Waukesha, WI, USA) except for PET studies in sham rats which were performed using β- and X-cube microsystems (Molecubes, Belgium). Rats were anesthetized with 2%–2.5% of isoflurane in 100% O2 and were placed in a PET acquisition system rat compatible holder. Rats were maintained in normothermia and their respiration rate and temperature were continuously monitored while they remained in the PET camera, using a SAII M1030 system (SA Instruments, NY, USA). The tail vein was catheterized with a 24-gauge catheter for intravenous administration of the radiotracers. For longitudinal assessment of A2ARs, rats (n = 10) were scanned at sham control and during the following month after ischemia with [11C]SCH442416. The radiotracer was injected concomitantly with the start of the PET acquisition and dynamic images of the brain were acquired for 60 min using 36 frames (2 × 5, 4 × 10, 5 × 20, 5 × 30, 5 × 60, 5 × 120, 5 × 180, 5 × 300 s) in the 400–700 keV energetic window. For evaluation of treatment on TSPO binding after ischemia (n = 10), [18F]DPA-714 was injected at the start of the PET acquisition and dynamic brain images were acquired for 60 or 30 min using 31 frames (3 × 5, 3 × 10, 3 × 15, 3 × 30, 4 × 60, 4 × 120, 5 × 180, 6 × 300 s) or 23 frames (3 × 5, 3 × 15, 4 × 30, 4 × 60, 4 × 120, 5 × 180 s), respectively. [18F]DPA-714-PET studies were conducted to evaluate the effect of CGS after ischemia using 30-min PET scans once it was showed that tracer equilibrium is reached within 20–30 min time frame for adequate %ID/mL calculation using 60 min PET scans (Figure S1). After each PET scan, CT acquisitions were also performed (140 mA intensity, 40 kV voltage), to provide anatomical information of each animal as well as the attenuation map for the later PET image reconstruction. Dynamic and static acquisitions were reconstructed (decay and CT-based attenuation corrected) with filtered back projection (FBP) using a Ramp filter with a cut off frequency of 0.5/mm.
PET images were analyzed using PMOD image analysis software (Version 3.5; PMOD Technologies Ltd., Zurich, Switzerland). For the analysis of PET signal, both PET images and a MRI (T2W) rat brain template from PMOD were separately manually co-registered to the CT of the same animal to generate a spatial normalization. Subsequently, MRI brain template was automatically co-registered to PET images. Two different types of volumes of interest (VOIs) were defined: (1) A first set of VOIs was established to assess the whole brain [11C]SCH442416 PET signal. Those VOIs were manually drawn in both the ipsilateral and contralateral hemispheres on slices of MRI-T2W rat brain template from PMOD software. (2) Another set of VOIs was automatically defined in the striatum, hypothalamus, thalamus, cortex, hippocampus, and cerebellum by using the regions proposed by PMOD rat brain template, to assess the uptake of [11C]SCH442416 and [18F]DPA-714 PET signal in brain regions after stroke. The last four frames were used to quantify [11C]SCH442416 and [18F]DPA-714 uptake during the last 15 and 10 min, respectively. Average values were calculated as percentage of injected dose per cubic centimeter (%ID/mL).
PET data modeling
The simplified reference-tissue model (SRTM) 22 from the PMOD software package was used to assess the non-displaceable binding potential (BPnd) of [11C]SCH442416 in the striatum, hypothalamus, thalamus, cortex, and hippocampus. This model relies on a two-tissue reversible compartment for the target region (brain regions) and a single-tissue compartment for the reference region without specific binding of the radiotracer (cerebellum). Three parameters were estimated for each kinetic: R1, which represents the ratio of tracer delivery; k2a, which is the clearance from the target tissue back to the vascular compartment; and BPnd, which is the non-displaceable binding potential of the tracer to the tissue.
Autoradiography
Rat brains (n = 13) were removed, frozen, cut in 20-μm-sections using a cryostat and stored at −80 °C. First, sections were dried at RT for 2 h and were pre-incubated for 15 min in Tris buffer and BSA 1% adjusted at pH 7.4. Sections were then incubated with the radioligand [11C]SCH442416 for 20 min (185 MBq) and then rinsed for 2 min with cold buffer, followed by a quick wash in cold distilled water. Brain sections were then placed in a cassette with the phosphor screen for 10 min and scanned with the Amersham Typhoon Biomolecular Imager (Cytiva, Marlborough, UK). Regions of interest were manually defined in the ischemic striatum and cerebellum using PMOD. The concentration of radioactivity in the regions analyzed were obtained as cpm/mm2. Values were expressed as the ratio between the concentration of radioactivity in the ipsilateral striatum versus in the cerebellum.
Immunofluorescence and cell counting
Immunofluorescence studies were performed to validate the results obtained by PET images. For that purpose, rat brains (n = 22) were removed, frozen and cut in 6-μm-sections using a cryostat. Samples were fixed in 4% paraformaldehyde, washed with phosphate-buffered saline (PBS) and blocked using NH4Cl, followed by another wash with PBS. For the permeabilization, sections were introduced in methanol:acetone (1:1) at −20 °C and in triton (0.1%), followed by a wash with PBS. Sections were then saturated using a solution of bovine serum albumin (BSA) 5% and Tween (0.5%) in PBS and were incubated for 1 h at RT with primary antibodies in BSA (5%)/Tween (0.5%) in PBS. For the first set of sections (n = 12), the primary antibodies used were: for A2ARs staining, rabbit anti-rat A2AR (1:50; Alomone Labs, Israel); for NeuN staining, guinea pig anti-rat NeuN (1:1000; Synaptic Systems, Goettingen, Germany); and for CD11b, mouse anti-rat CD11b (1:300; Serotec, Raleigh, NC, USA). Additionally, a second set of sections was performed (n = 10) for TSPO expression with rabbit anti-rat (NP155, 1:1000) and CD11b, and a third set of sections with A2ARs and CD11b. Samples were washed with PBS incubated for 30 min at RT with secondary antibodies Alexa Fluor 594 goat anti-rabbit IgG, Alexa Fluor 488 goat anti-chicken IgG and Alexa Fluor 647 goat anti-mouse IgG (1:1000; Molecular Probes, Life Technologies, Madrid, Spain) in BSA 5%/Tween 0.5% in PBS. Samples were mounted with a Prolong Antifade Kit with DAPI (Molecular Probes Life Technologies, Madrid). Axio Observer Z6 (Zeiss) was used to the acquisition of images at 63× magnifications to cell-counting, which was manually performed with Image J (Version 2.0.0-rc-69/11.52p; National Institute of Health, Maryland, USA).
Immunohistochemistry
Immunohistochemical studies were performed to demonstrate the neurodegeneration after ischemic stroke. For that purpose, rat brains (n = 6) were cut in 6-μm sections using a cryostat. For the permeabilization, sections were introduced in H202 in phosphate-buffered saline (PBS) for 30 min, followed by three washes in PBS. Sections were then saturated using a solution of bovine serum albumin (BSA) 0.5% and Triton (0.1%) in PBS for 30 min and incubated overnight at 4 °C with primary antibodies in BSA/PBS. The primary antibody used for neuronal cells was rabbit anti-rat NeuN (1:200; Abcam, Cambridge, United Kingdom), and for microglial/macrophage staining, mouse anti-rat CD11b (1:300; Serotec, Raleigh, NC, USA). Samples were washed 3× with PBS and then were incubated at RT for 1 h with secondary antibodies conjugated with biotin. The secondary antibodies used were goat anti-rabbit (1:1000; Abcam, Cambridge, UK) and horse anti-mouse (1:100; Vector Laboratories, Newark, USA). After wash in PBS, biotin-peroxidase complex (Vector Laboratories, Newark, USA) was added for incubation of 30 min, followed by 3× washes in PBS. Detection was performed with 3,3′-diaminobenzidine (DAB) revelation (Vector Laboratories, Newark, USA) and samples were mounted with Mowiol. Images were acquired with the Pannoramic MIDI II automated digital slide scanner (3DHistech Ltd., Hungary).
Protein extraction and western blotting of A2ARs
Cerebral striatum samples were extracted before (day 0) and at days 1 and 3 after stroke to semi-quantify the expression of A2ARs (n = 18). All samples were homogenized separately adding lysis buffer and protein concentration was determined by BCA protein assay (ThermoFisher, USA). Samples (10 µg of protein) were run in a denaturing 10% SDS–PAGE and proteins transferred to an activated polyvinylidene difluoride (PVDF) membrane (Bio-Rad, USA), which was incubated at RT for 2 h with the primary antibody, rabbit anti-rat A2AR (1:5000; Alomone Labs, Israel), and washed three times with TBS and incubated at RT for 1 h with the secondary antibody, goat anti-rabbit (1:5000; Bio-Rad, USA). Immunoreactive bands were visualized by chemiluminescent HRP substrate (Bio-Rad, USA) using ChemiDoc (Bio-Rad, USA) and ImageLab (Bio-Rad, USA) was used for the quantification. Subsequently, A2ARs antibody was removed by stripping buffer during 30 min at 55 °C, followed by the incubation with the primary antibody, rabbit anti-rat GAPDH (1:5000, Abcam, Cambridge, UK) for 1 h and washed three times with TBS and incubated at RT for 1 with the secondary antibody, goat anti-rabbit (1:10000; Bio-Rad, USA). Immunoreactive bands were visualized by chemiluminescent HRP substrate (Bio-Rad, USA) using ChemiDoc (Bio-Rad, USA) and ImageLab (Bio-Rad, USA) was used for the quantification.
Neurological assessment
The assessment of neurological outcome induced by cerebral ischemia was based on a previously reported 9-neuroscore test. 19 This neurological evaluation (composed of four consecutive tests) is a global neurological assessment that was developed to measure neurological impairments following stroke and assesses a variety of motor, sensory and reflex responses. Before imaging evaluation, neurological evaluation was assessed at days 1 and 7 after ischemia as described previously.19,23
Statistical analysis
The data are expressed as the means ± SDs (standard deviations). Normality tests were performed to check the data distribution. Two-tailed Student’s unpaired t tests or Mann–Whitney tests were performed, as appropriate, for comparisons between two different groups. Similarly, to define significant differences between more than two groups, one-way ANOVA followed by Bonferroni’s multiple-comparison test for post hoc analysis or the Kruskal–Wallis test followed by Dunn’s test or two-way ANOVA was performed. The level of significance was set at p < 0.05, and statistical analyses were performed with GraphPad Prism version 8 software.
Results
PET imaging evaluation of A2ARs in the healthy brain
The expression of A2ARs in healthy rat brains (control) was evaluated by PET. With that aim, time–activity curves (TACs) were obtained in different brain regions (striatum, hypothalamus, thalamus, cortex, hippocampus and cerebellum) to assess [11C]SCH442416-PET signal uptake (%ID/mL). The radiotracer showed fast uptake in the different brain regions studied, followed by wash-out until a steady state was reached in the hypothalamus, thalamus, cortex, hippocampus and cerebellum over the last 15 min of the scan. However, such a steady state was not reached in the healthy striatum, which is the region with higher uptake (Figure 1(a)–(c)), even though a tracer equilibrium was observed at some days after ischemia (Figure S2).
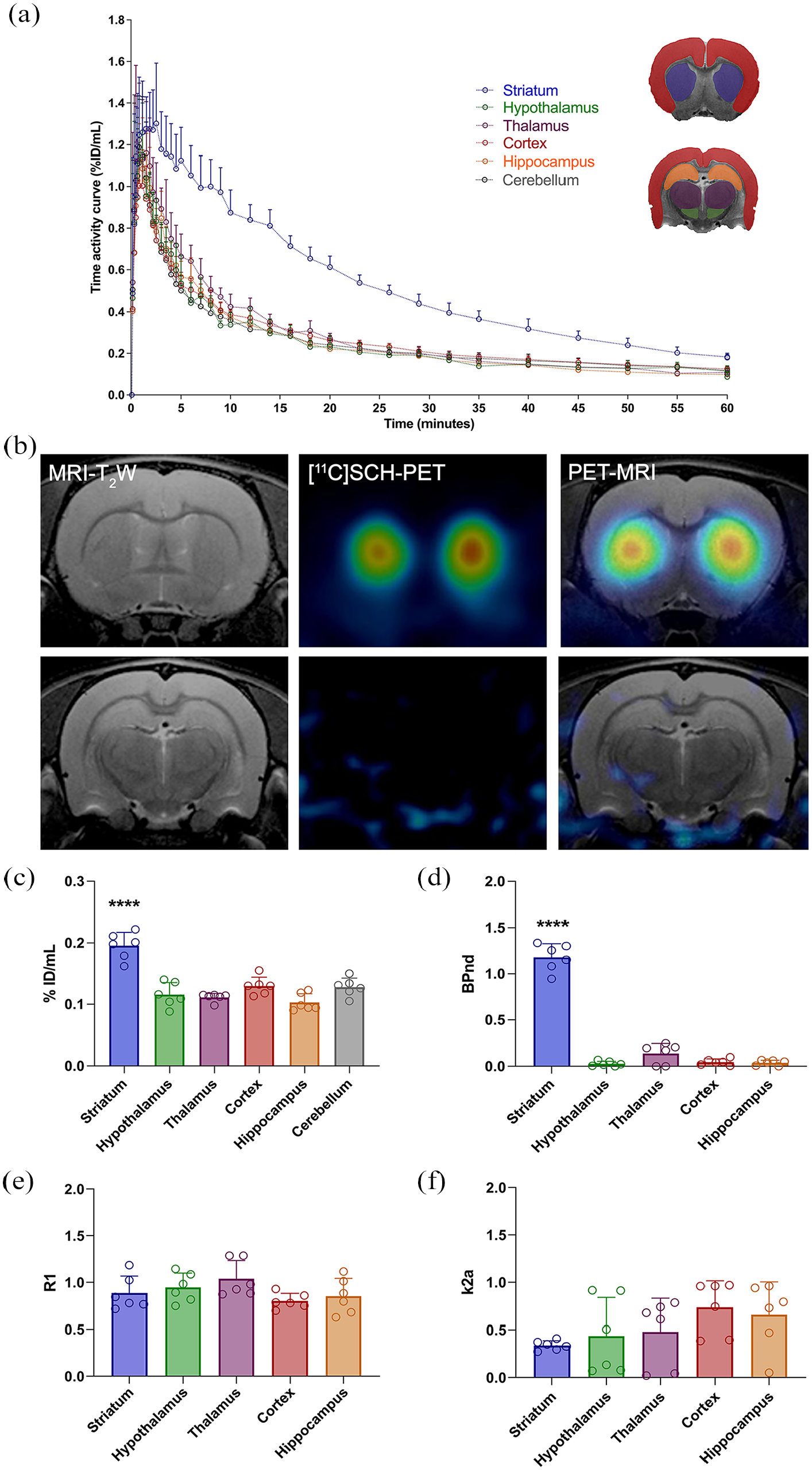
Expression of A2ARs in healthy rat brain. Time–activity curves (%ID/mL) of [11C]SCH442416 in different rat brain regions (a). MRI-T2W, [11C]SCH442416-PET and coregistered MRI–PET coronal axial images show A2AR distribution in the healthy rat brain (b). %ID/mL and kinetic parameters (BPnd, R1, and k2a) in different brain regions determined via SRTM (n = 6; c–f). ****p < 0.0001 compared with the other brain regions. The values are presented as scatter dot plots (means ± SD).
In this scenario, the determination of uptake as %ID/mL (Figure 1(c)) was not considered fully appropriate, and a more accurate quantification model was applied. As no blood input function was available, SRTM was employed to verify the specific binding in the different brain regions (Figure 1(d)-(f)). From the analysis, kinetic parameters, including the BPnd (ratio of bound to free radiotracer in a specific region in the brain), R1 (influx of the radiotracer to a specific region of the brain), and k2a (efflux of the radiotracer from a specific region to the blood compartment), were obtained using the cerebellum as the reference tissue region. The results revealed a significant increase in BPnd in the striatum compared with the hypothalamus, thalamus, cortex, and hippocampus (p < 0.001; Figure 1(d)), supporting the existence of specific [11C]SCH442416 binding in the striatum and the lack of specific binding in other regions. In contrast, no significant differences in R1 or k2a were observed among the different regions (Figure 1(e) and (f)).
Assessment of brain infarction with MRI
MRI-T2W images of the coronal planes corresponding to an ischemic rat brain at day 1 after stroke are shown in Figure 2(a). The hyperintense regions shown in the images demonstrated vasogenic edema formation due to the consolidation of the cerebral infarction.
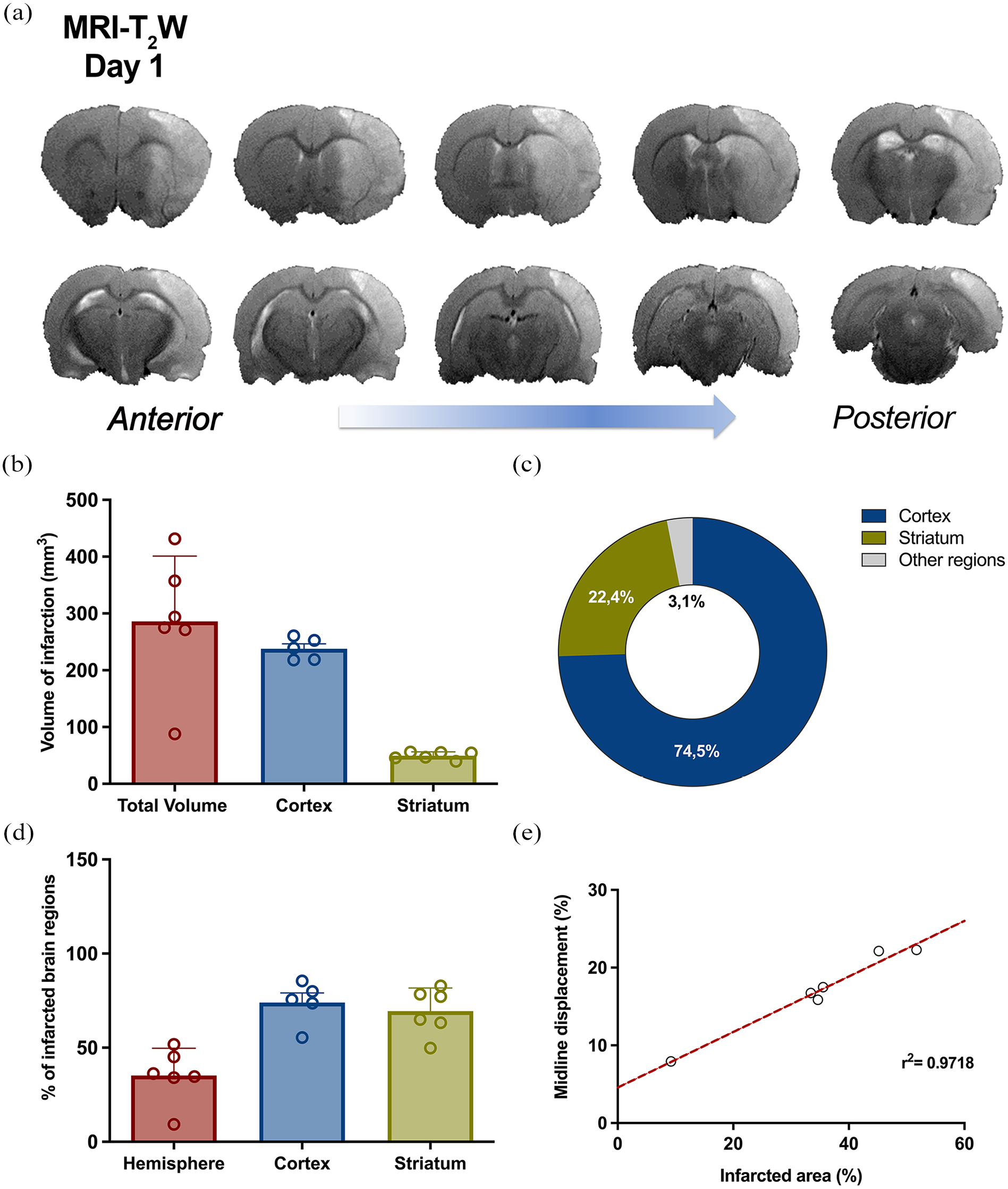
Characterization of brain infarction. MRI-T2W images on day 1 after cerebral ischemia (a). Total volume of infarction and volumes of infarction per brain region (cortex and striatum; b). Regional distribution of cerebral infarction in the rat brain (c). Percentages of the cerebral hemisphere, cortex and striatum affected by infarction (d). Linear regression between total brain infarction and midline displacement (e). r2 = 0.9718, p < 0.0001. The values are presented as scatter bar plots (means ± SD; b, d) or scatter dot plots (e).
The extent of the brain lesions was assessed at 24 h, and the total volume of infarction was 282.78 ± 121.82 mm3 (mean ± SD; Figure 2(a) and (b)). The areas most affected by ischemic stroke were the cortex and striatum, since almost all the animals experienced cortico-striatal infarctions. The volume of infarction in the cortex was 238.19 ± 19.95 mm3, whereas the striatum volume was 48.65 ± 9.58 mm3. As a result, more than 90% of the infarctions were distributed in the cortex and striatum, and the rest were distributed in other brain regions, such as the hippocampus (data not shown; Figure 2(b) and (c)).
The percentage of infarct volume within each region (including the ipsilateral hemisphere, cerebral cortex and striatum) was also analyzed. These results revealed that 35% of the ischemic hemisphere developed infarction at 24 h after ischemia onset and that most of the area of both the cerebral cortex (74%) and the striatum (69%) was infarcted (Figure 2(d)). Moreover, linear regression was performed to study the relationship between the volume of the infarcted ipsilateral hemisphere (%) and midline displacement (%). These results revealed a significant correlation between the volume of infarction and midline displacement at 24 h after cerebral ischemia (r2 = 0.9718, p < 0.001; Figure 2(e)).
In vivo PET imaging of A2ARs after cerebral ischemia in rats
The longitudinal expression of A2ARs was evaluated with in vivo PET imaging using the radiotracer [11C]SCH442416 administration at sham and ischemic rats during the following 28 days after ischemia onset (molar activity was 66.1 ± 14.3 GBq/µmol; Table S2). Visual inspection of the images reveals that the intensity of the PET signal decreased in the ipsilateral striatum at day 1 after cerebral ischemia when compared to sham levels, followed by an increase at day 3 after ischemia. Subsequently, PET signal decreased from days 7 to 28 in relation to day 3 (Figure 3). In contrast, the contralateral striatum did not differ across days after cerebral ischemia. The standardized uptake value (SUV) and kinetic parameters in the striatum and cortex were analyzed in sham rat brains and during the month following ischemic stroke (Figures 4 and 5). In the ischemic striatum, SUV showed higher values at day 3 in relation to sham animals and at the following time points after ischemia (Figure 4(a)). In contrast, the peak value of BPnd was observed at sham, followed by a decrease at day 1 after stroke onset. Subsequently, BPnd increased at day 3 and declined progressively from days 7 to 28 (p < 0.05, p < 0.01, p < 0.001 vs day 3 and p < 0.01, p < 0.001 and p < 0.0001 vs sham; Figure 4(c)). In the contralateral striatum, SUV values showed a non-significant increase from days 3 to 28 compared to control conditions and day 1 after stroke (Figure 4(b)). However, these results were not supported by the BPnd values, in which similar values were observed at different days after ischemia in relation to sham rats (Figure 4(d)). Finally, R1 and k2a values in the striatum revealed greater [11C]SCH442416 entrance into the brain than returning from the brain to the blood. Despite this, non-significant differences were observed for R1 and k2a values in both the ipsilateral and contralateral striatum (Figure 4(e)–(h)).
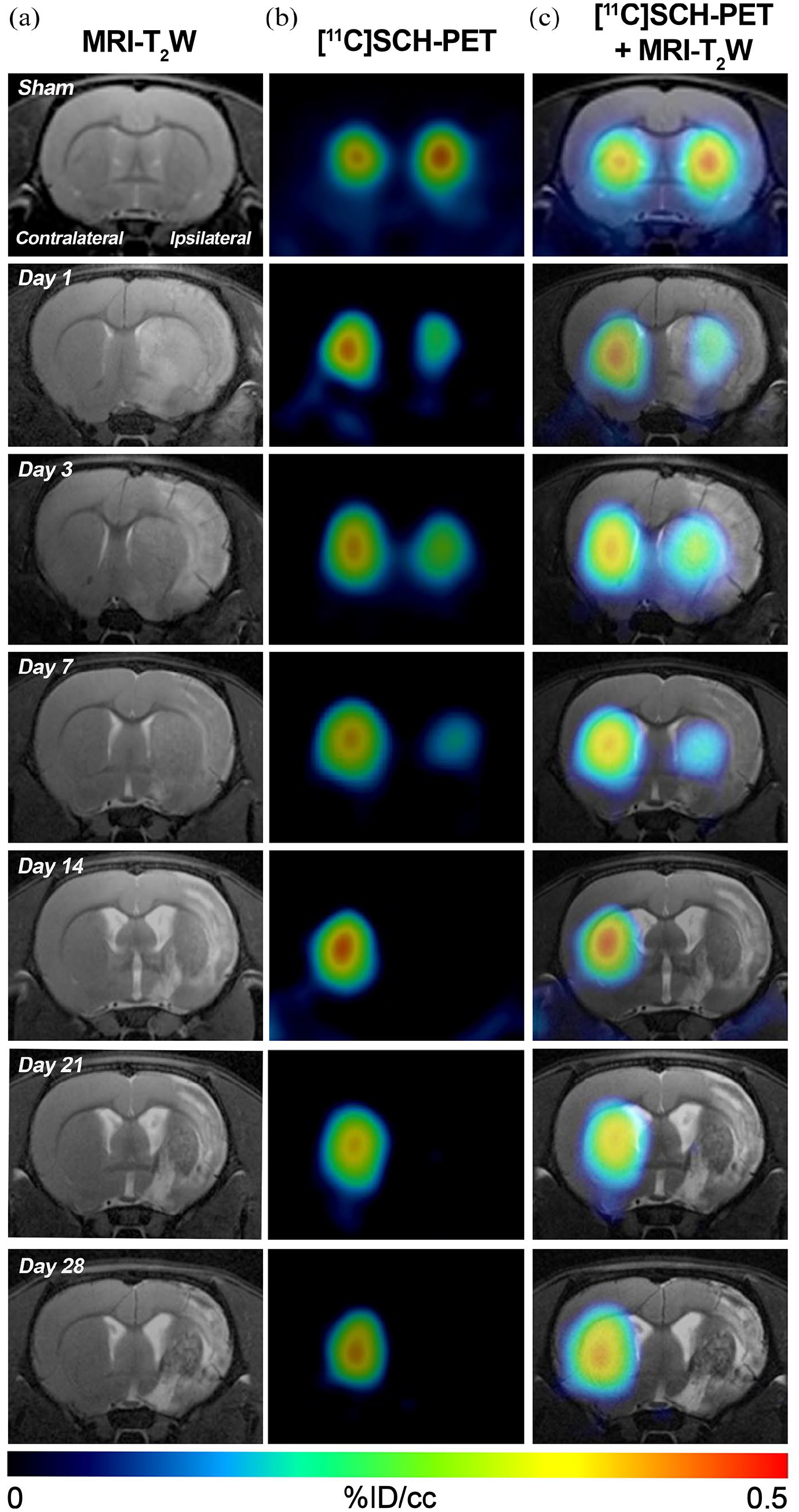
MRI and [11C]SCH442416 PET brain images after ischemia. (a) MRI-T2W, (b) [11C]SCH442416-PET and (c) coregistered axial PET–MRI images of a representative rat brains from sham- and ischemic rats at 1, 3, 7, 14, 21, and 28 days after ischemic stroke show the evolution of ischemic lesion and A2AR expression over the month following stroke onset.
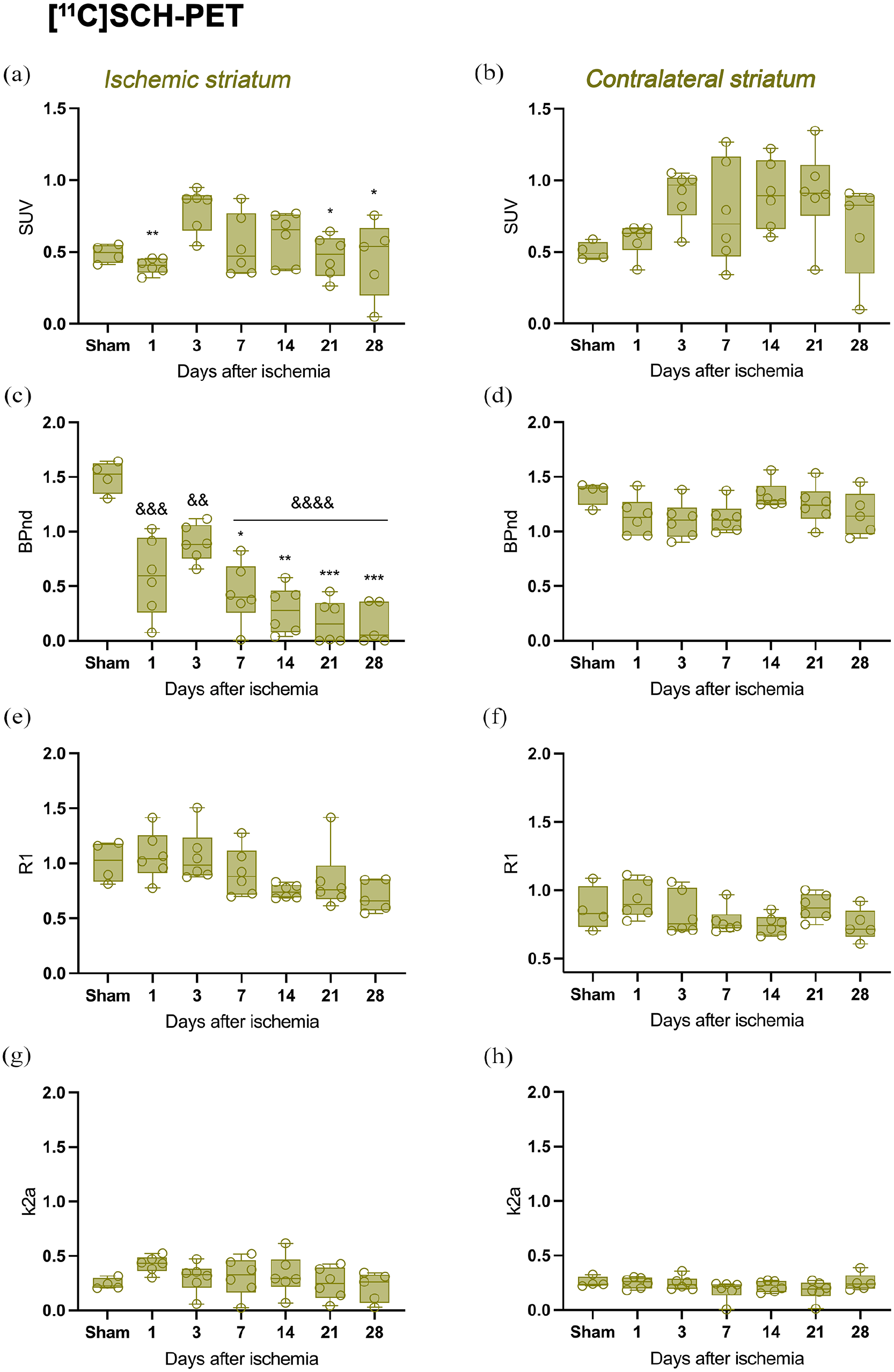
Time course of the progression of the [11C]SCH442416 PET signal at different days after ischemia. The PET signal at sham (n = 4) and ischemic rats (n = 6) was quantified by the SUV and kinetic parameters (BPnd, R1, and k2a) in the ischemic (a, c, e, g) and contralateral striatum (b, d, f, h). *p < 0.05, **p < 0.01, and ***p < 0.001 compared with day 3; &&p < 0.01, &&&p < 0.01, and &&&&p < 0.0001 compared with sham. The values are presented as boxes and whiskers and represent all points (means ± SD).
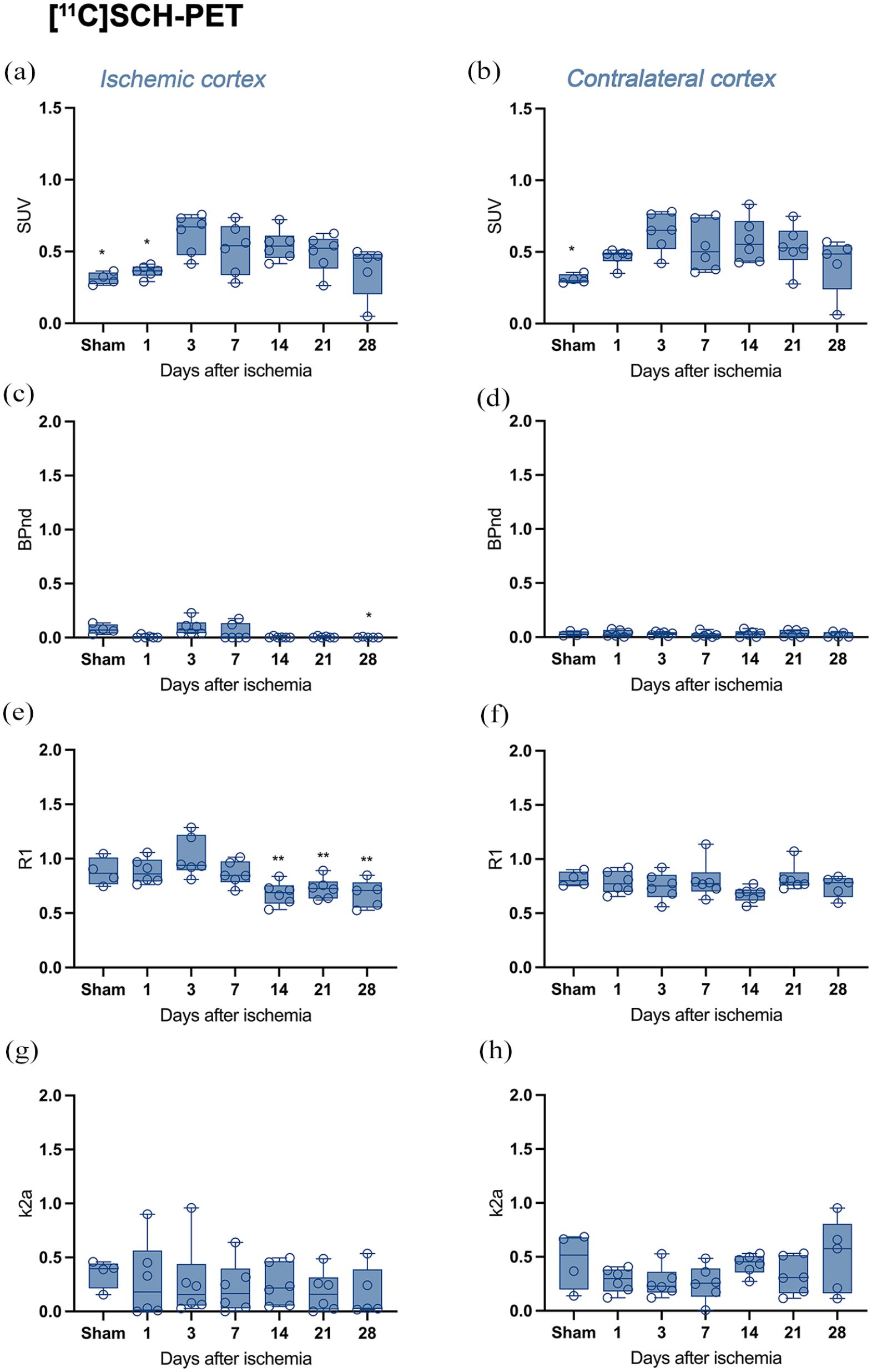
Time course of the progression of [11C]SCH442416 PET signals in the contralateral brain hemisphere. The PET signal at sham (n = 4) and ischemic rats (n = 6) was quantified by the SUV and kinetic parameters (BPnd, R1, and k2a) in the ischemic (a, c, e, g) and contralateral cerebral cortex (b, d, f, h). *p < 0.05 and **p < 0.01 compared with day 3. The values are presented as boxes and whiskers and represent all points (means ± SD).
Similarly, the cerebral cortex presented the highest SUV values at day 3 in relation to the sham control and at different time points after ischemia in both brain hemispheres (p < 0.05; Figure 5(a) and (b)). However, this brain region presented negligible BPnd values before and after ischemia in both hemispheres (Figure 5(c) and (d)). In addition, our results revealed similar radiotracer influx (R1) values and impaired radiotracer efflux to the blood (k2a) after ischemia in both the ipsilateral and contralateral brain cortex (Figure 5(e)–(h)). Hence, these results suggest that SUV values observed in both the ipsilateral and contralateral cerebral cortex were the result of non-specific binding.
Ex vivo expression of A2ARs before and after ischemia
The cellular expression of A2ARs in healthy and ischemic conditions was evaluated using immunofluorescence studies before (day 0) and at days 1 and 3 after stroke in the penumbra and core of infarction affecting the ischemic striatum (Figure 6(a)–(e)). In healthy conditions, A2ARs showed mainly neuronal expression (A2ARs+/NeuN+ cells) and negligible expression in GFAP+ astrocytes (data not shown). Following stroke, the ischemic penumbra showed a significant decrease of A2ARs+/NeuN+ cells at days 1 and 3 in comparison to control conditions (p < 0.001; Figure 6(d)). In contrast, the core of the infarction showed a sharp significant decrease in A2ARs+/ NeuN+ cell number from days 1 to 3 in relation to day 0 (p < 0.001; Figure 6(e)). Hence, ischemic lesion was replaced by a progressive increase of microglia, macrophages and infiltrated leukocytes expressing A2ARs (A2ARs+/CD11b+ cells) at day 3 after ischemia onset in both striatal penumbra and infarct core (p < 0.01 and p < 0.001 at day 3 vs 0, p < 0.05 and p < 0.001 at day 3 vs 1; Figure 6(d) and (e)). Overall, the increase of A2ARs in immune cells (A2ARs+/CD11b+ cells) observed at day 3 confirmed the increase of the value of BPnd obtained by [11C]SCH442416-PET at same time point in relation to day 1 in the ipsilateral striatum (Figure 4). Progressive neurodegeneration followed by increase of immune response was confirmed by immunohistochemistry staining with NeuN and CD11b before and after ischemic stroke (Figure 6(f) and (g)). To confirm these findings, the expression of A2ARs in the ipsilateral striatum was also evaluated by western blot analysis and autoradiography before (day 0) and at days 1 and 3 after brain ischemia (Figures 6(h)–(k) and S3). Results showed an immunoreactive band of 43 kDa recognized by A2ARs antibody, corresponding to A2ARs. Particularly, brain ischemia triggered a decrease in the protein expression level of A2AR at day 1 in relation to day 0 and followed by a pseuro-recovery at day 3 (Figures 6(f) and (g) and S2), which was consistent with PET findings (Figure 4(c)) and ex vivo autoradiography with [11C]SCH442416 (Figure 6(j) and (k)).
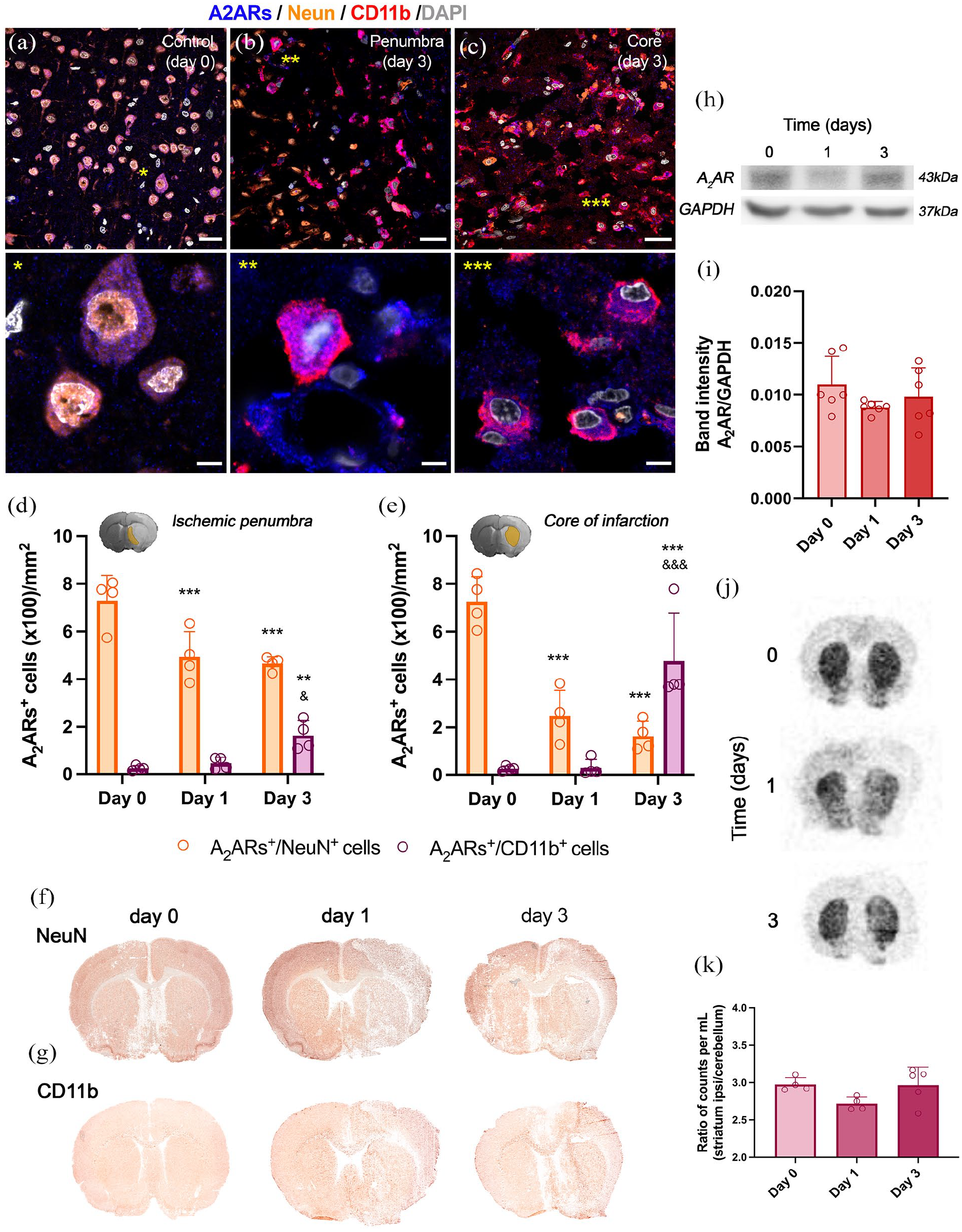
Temporal ex vivo evaluation of A2AR expression after cerebral ischemia. Immunofluorescent labeling of A2ARs (blue), NeuN (orange), CD11b (red), and DAPI (gray) in the penumbra and core of the infarction in the control and day 3 groups is shown in four channels (a–c). The data show A2AR expression in NeuN+ and CD11b+ cells from the control group (day 0) and at days 1 and 3 after cerebral ischemia in both the ischemic penumbra and the core of infarction (d, e). Immunohistochemistry labeling of NeuN and CD11b in the whole brain section before (day 0) and at days 1 and 3 after ischemia (f, g). The protein expression of A2ARs during the first 3 days after ischemia was evaluated by immunoblotting (h, i) and autoradiography (j, k). **p < 0.01 and ***p < 0.001 compared with day 0; &p < 0.05 and &&&p < 0.001 compared with day 1. The values are presented as scatter dot bar plots (means ± SD). Scale bars, 30 μm (top panels) and 10 μm (bottom panels).
Evaluation of inflammatory reactions after the modulation of A2ARs
The modulatory effect of the A2ARs agonist CGS-21680 (0.1 mg/kg) on the ischemic response was studied with in vivo MRI and [18F]DPA-714-PET techniques, as well as with behavioral evaluation (Figure 7). MRI-T2W images were obtained at day 1 after stroke to demonstrate the presence of similar volumes of infarction between the treated and control ischemic groups before the start of the treatment (Figure 7(a) and (c)). Likewise, neurofunctional impairment was assessed with the 9-neuroscore test at days 1 and 7 after stroke. Ischemic animals showed the major neurologic impairment at day 1 after cerebral ischemia (before the start of the treatment) in relation to day 7 in both groups (p < 0.001; Figure 7(d)). However, ischemic rats treated with CGS-21680 did not show neurological improvement at day 7 in relation to control animals. Additionally, the activation of microglia/macrophages after the pharmacological modulation of A2ARs with CGS-21680 after stroke was analyzed by [18F]DPA-714 PET imaging at day 7 after cerebral ischemia (Figure 7(b)). These results showed higher striatal [18F]DPA-714 uptake values (%ID/mL) in the treated group in relation to ischemic control group (p < 0.05; Figure 7(e)). However, the cerebral cortex showed no differences in PET signal between the two groups (Figure 7(f)). Furthermore, IHC studies show increased expression of TSPO+/CD11b+ cells among the treated animals in the area of the striatum, and this trend was also observed in the cerebral cortex (Figure 7(g)–(j)), thus supporting the results obtained in PET studies. Furthermore, the treatment induced a non-significant increase in striatal A2A+/CD11b+ cells after ischemic in relation to vehicle (Figure S4).
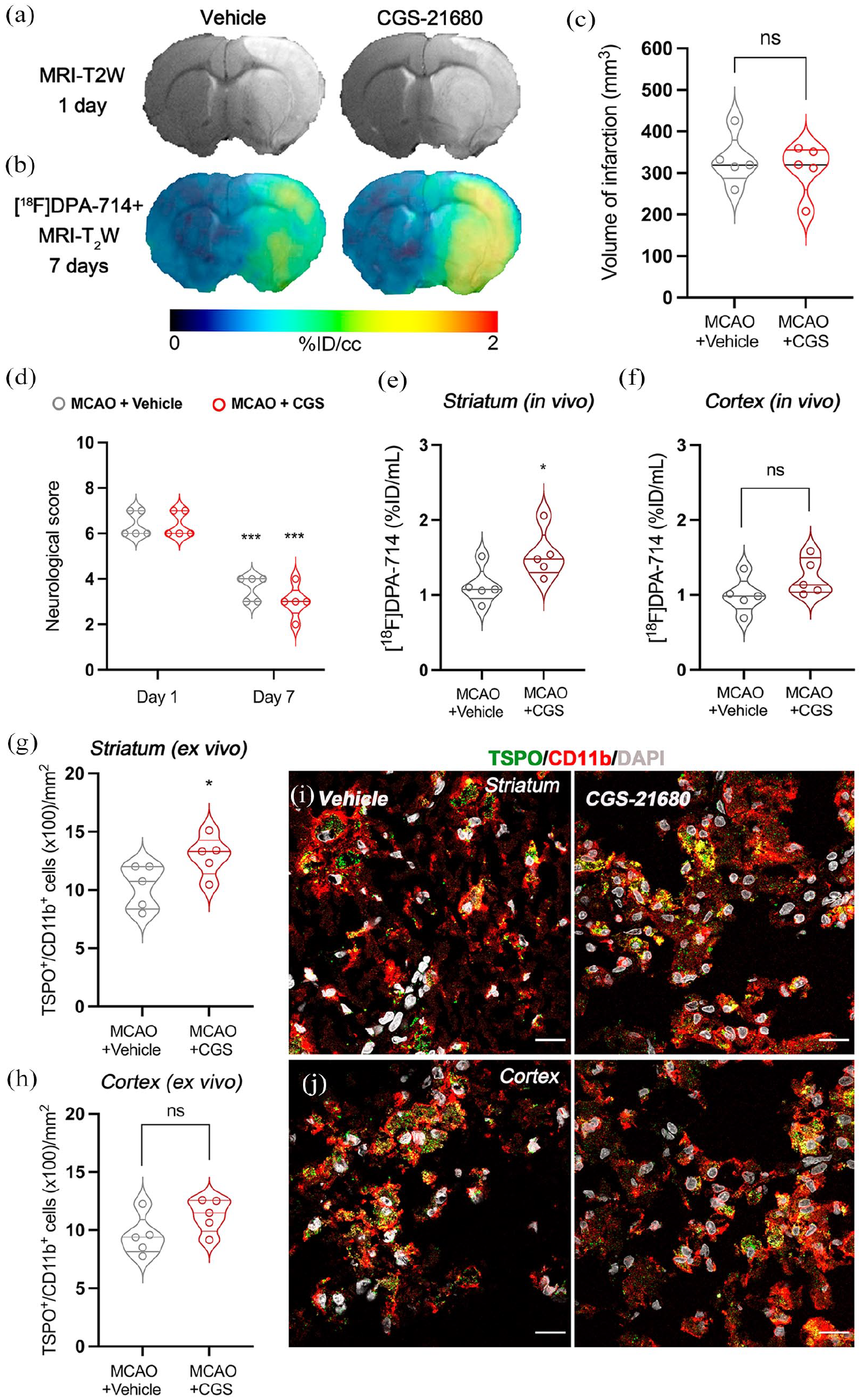
Effect of A2ARs on neuroinflammation after cerebral ischemia. MRI (T2W; a) and PET axial images of [18F]DPA-714 (b) in vehicle- and CGS-treated ischemic rats at the level of the ischemic lesion. The infarct volume was evaluated via MRI (T2W) at day 1 after ischemia (c), and the neurological score (d) was determined at day 1 (before the start of treatment) and day 7 after MCAO in vehicle (n = 5) and CGS-treated (n = 5) rats. In vivo [18F]DPA-714 PET signals and ex vivo immunohistochemical evaluation of TSPO expression in CD11b-positive cells were quantified on day 7 after ischemia in both the striatum and cerebral cortex (e–j). ***p < 0.001 compared with day 1, *p < 0.05 compared with the vehicle. The values are presented as violin plots showing all points (means ± SD). Scale bars, 20 μm.
Discussion
In recent years, adenosine and A2ARs have attracted increased interest as therapeutic targets in the field of ischemic stroke because of their key role in hypoxic conditions.7,12,24,25 However, the identification of effective treatments based on the pharmacological modulation of A2ARs will require a better understanding of the changes in these receptors during ischemic stroke. For the first time, we have performed MRI and PET imaging studies to evaluate changes in A2AR expression during the first month after experimental stroke and the effects of their modulation on brain damage, neurological function and the neuroinflammatory response.
Expression of A2ARs in healthy and ischemic rat brains
The in vivo distribution of PET radiotracers for A2ARs, such as [11C]SCH442416, [11C]preladenant, or [18F]FLUDA, has been characterized by the distribution pattern of these receptors in the striatum of rodent, primate and human brains, among others.13,15,16,26,27 Similarly, previous assays with in situ hybridization revealed that the tissue distribution of A2AR mRNAs was also restricted to the striatum. 28 Therefore, the findings reported above are consistent with the in vivo BPnd distribution of [11C]SCH442416 in healthy rat brains observed in our study (Figure 1).
Following the first 24 h after transient MCAO, rat brains showed vasogenic edema formation and consolidation of the brain infarction in the cerebral cortex and striatum, which are regions of the brain that are irrigated by the middle cerebral artery (Figure 2). In fact, the uptake of [11C]SCH442416, measured as SUV in sham rats and after cerebral ischemia, was similar in both the ischemic cortex and the striatum (Figures 3, 4(a), and 5(a)), despite the absence of PET signal detected in the brain cortex via kinetic modeling analysis (Figure 1). Hence, to avoid false-positive values from unspecific binding in the cerebral cortex, among other regions, the reference tissue model (SRTM) was applied for the quantification of receptor kinetics without the need to measure arterial input function. 22 As a result, we observed a decrease in the BPnd at day 1 in the ischemic striatum followed by recovery at day 3 and a subsequent decline later (Figures 3 and 4(c)). In contrast, no substantial changes in BPnd were observed in the cerebral cortex over time after cerebral ischemia (Figure 5(c)). Our [11C]SCH442416-PET findings are in agreement with results obtained by Trincavelli et al., who did not observe any alterations in A2AR binding in the cerebral cortex at 24 h after MCAO but detected a significant decrease in A2AR ligand affinity in the ischemic striatum. 29
Cellular expression of A2ARs before and after cerebral ischemia
Immunofluorescence studies revealed that the neural expression of A2ARs under healthy conditions was replaced by the expression of A2ARs in microglia and infiltrated macrophages (A2ARs+/CD11b+ cells) during subacute ischemic stroke (Figure 6). Thus, the expression of these receptors in different cell types over time supports the dual role that these receptors might play in primary and secondary ischemic damage led by neurons and immune cells, respectively. 9 Furthermore, the reduction of A2ARs expression at day 1 in relation to healthy conditions followed by a pseudo-recovery at day 3 observed with immunohistochemistry, western blot, and autoradiography analysis is in agreement with the [11C]SCH442416-PET binding pattern observed during the first 72 h after cerebral ischemia (Figure 4). Similarly, these results revealed that the increase in BPnd observed at day 3 after ischemia was due to the overexpression of A2ARs in microglia and infiltrated macrophages (CD11b+ cells) in both the penumbra and the core of infarction in the striatum (Figures 4 and 6).
Effect of A2ARs modulation after experimental stroke
Previous findings have shown that microglial activation under pathological conditions involves elevated levels of A2Ars.30 –32 In this sense, Orrú et al. reported that A2ARs are upregulated during the retraction of microglial processes before becoming amoeboid shaped, defining A2AR as a key purinergic receptor in the cytoskeletal rearrangement of microglia. 33 The activation of microglia triggers the secretion of bioactive molecules, many of which are related to neuroinflammation. 34 Therefore, owing to the involvement of A2ARs in microglial dynamics,33,35 we investigated the effects of the selective A2AR agonist CGS-21680 on the neurological and inflammatory outcomes of ischemic rats. Unlike Melani et al., who reported that rats treated with either 0.01 or 0.1 mg/kg CGS-21680 improved neurological outcomes after stroke compared with control rats, 11 our findings revealed no differences in lesion volume and neurofunctional evolution between vehicle-treated and treated ischemic rats. In fact, these differences could be explained by the drug administration protocol used in both studies, while we used a daily single dose of 0.1 mg/kg CGS-21680 from day 1 and during the following week after MCAO, Melani et al. used twice-daily therapy that began 4 h after ischemia and lasted for the next 7 days. 11 Additionally, the expression of TSPO by [18F]DPA-714-PET was also studied following the pharmacological activation of A2ARs. In fact, cellular TSPO expression is related to inflammation and specifically to the role of proinflammatory myeloid cells in rodents. 36 We observed an increase in the striatal [18F]DPA-714-PET signal along with an increase in the number of immune cells expressing TSPO at day 7 after daily treatment with CGS-21680 (Figure 7). In this sense, our work suggests that the use of A2AR agonists induces the activation of the microglial proinflammatory phenotype after ischemic stroke, which limits their protective role in stroke recovery. In agreement with our results, subchronic administration of the selective A2AR antagonist SCH-58261 after focal ischemia decreased the activation of p38MAPK, protecting against neurological deficits and brain damage in cerebral ischemia.37,38 Similarly, KO mice lacking A2ARs showed an attenuation on infarction volume, neurological impairment, leukocyte infiltration, blood brain barrier leakage, and edema in experimental rodent models of ischemic stroke.39,40 However, these studies contrast with the protective role of adenosine receptors during secondary inflammatory damage after ischemic stroke, which reduces microgliosis and astrogliosis and improves myelin organization.10,11 Hence, these findings demonstrate the controversial role of A2ARs in the modulation of ischemic damage evolution.
Conclusion
To our knowledge, this is the first study using MRI and PET imaging to evaluate the temporal expression of A2ARs during the subacute and chronic phases of ischemic stroke in rats. We observed a decrease in the PET signal at day 1 after ischemia followed by recovery during the following 72 h due to the overexpression of A2ARs by immune cells in the ischemic striatum. In addition, the pharmacological activation of A2ARs induced an increase in activated microglia expressing TSPO as a surrogate proinflammatory markers after ischemic stroke. Overall, our work provides novel insights into the involvement of A2ARS in ischemic damage and the neuroinflammatory response, contributing to the future development of novel therapeutic strategies for stroke involving purinergic receptors.
Limitations
Future studies should consider including animals of both sexes, as it is well known that the progression of stroke differs between males and females. Finally, the comorbidities associated with the disease should be studied in parallel with ischemic stroke. Ischemic stroke often occurs simultaneously with other medical conditions that could worsen or improve the stroke prognosis. Given their relevance, comorbidities must be incorporated into studies on ischemic stroke.
Supplemental Material
sj-docx-1-jcb-10.1177_0271678X251370835 – Supplemental material for Longitudinal imaging evaluation of the inflammatory role of purinergic A2A receptors during subacute and chronic ischemic stroke
Supplemental material, sj-docx-1-jcb-10.1177_0271678X251370835 for Longitudinal imaging evaluation of the inflammatory role of purinergic A2A receptors during subacute and chronic ischemic stroke by Maider Garbizu, Naroa Mocha-Muñoz, Esther Rubio-López, Laura Palacios, Laura Aguado, María Ardaya, Ana Joya, Unai Alduntzin, Sandra Plaza-García, Daniel Padro, Vanessa Gómez-Vallejo, Unai Cossío, Makoto Higuchi, Pedro Ramos-Cabrer, José Luis Zugaza, Jordi Llop and Abraham Martín in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Acknowledgements
The authors would like to thank A Lekuona and V Salinas for their technical support in radiosynthesis and L Iglesias for their technical support in histology. The authors are also thankful for their technical and human support with analytical and high-resolution microscopy at the Biomedicine Service of Bizkaia provided by SGIker (UPV/EHU).
Correction (September 2025):
The article has been updated to interchange Figures 2 and 3, along with their corresponding citations.
Ethical considerations
Animal experimental protocols and relevant details regarding welfare were approved by the Ethical Committee at CICbiomaGUNE, UPV/EHU, and local authorities and were conducted in accordance with Spanish legislation (Real Decreto 53/2013) and the Directives of the European Union on animal ethics and welfare. The results are reported following the ARRIVE guidelines. All the studies were conducted under the same experimental conditions in AAALAC-certified animal facilities.
Data availability statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the Spanish Ministry of Education and Science/FEDER RYC-2017-22412, PID2019-107989RB-I00, PID2022-138022OB-I00, and PCI2022-134986-2 funded by MICIU/AEI/10.13039/501100011033 and European Union NextGenerationEU/PRTR). Jordi Llop also acknowledges the Spanish Research Agency for financial support (MCIN/AEI/10.13039/501100011033; PID2020-117656RB-I00). Part of the work was performed under the Maria de Maeztu Units of Excellence Programme—grant MDM-2017-0720 funded by MCIN/AEI/10.13039/501100011033, Fundació TV3 (248/C/2020) and PIBA_2023_1_0013 program from the Basque Government.
Declaration of conflicting interests
The author(s) declare that there are no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
MG, NM-M, ER-L, LP, LA, MA, AJ, UA, SP-G, VG-V performed experiments and acquired data. DP, MH, PR-C, JLZ, JL, AM designed experiments. MG, MA, UC, PR-C, JL, AM analyzed data, prepared the manuscript, and approved the final version of the manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
