Abstract
Altered cerebrovascular hemodynamics and low cerebral perfusion contribute to the development and progression of dementia. Dynamic cerebral autoregulation (dCA), a measure of the cerebral vasculature’s ability to buffer abrupt changes in mean arterial pressure and prevent hypoperfusion, such as during a supine-to-standing transition, have mixed results in people clinically diagnosed with mild cognitive impairment (MCI, people with objective cognitive impairment but maintained functional independence). Therefore, in 30 people with MCI, we tested the hypothesis that participants with a higher standing middle cerebral artery velocity (MCAv) at diastole (higher-velocity group) would have lower dCA values, to confer better cerebrovascular outcomes and enhanced cognitive function compared to participants with a lower MCAv at diastole (lower-velocity group). This study separated people with MCI into different diastolic MCAv groups. dCA was calculated as (MCAvnadir-MCAvsupine/MCAvsupine)/(MAPMCAnadir-MAPMCAsupine/MAPMCAsupine). This work led to the identification of a dysregulated dCA in the higher-velocity group (p = 0.009) compared to the lower-velocity group despite having greater cognitive scores (p = 0.008). Elevated levels of cerebral oxygen tissue saturation (p = 0.039) and lower end-tidal carbon dioxide (p = 0.042) suggest that a favourable dCA value may be a compensatory mechanism in the neurodegenerative disease processes. The unexpected results highlight the importance of uncovering hemodynamic pathways in clinical populations.
Keywords
Introduction
Dementia is the seventh leading cause of death worldwide, 1 and one of the most feared conditions among older adults. 2 Mild cognitive impairment (MCI) is a deviation from healthy aging with maintained functional independence, 3 and occurs in 14%–19% of community dwelling older adults.4–7 MCI progresses to dementia in 10%–24% of people,8,9 and otherwise remains stable 4 or reverts back to normal cognitive function.10,11 People with MCI represent a potential clinical cohort to target and improve factors impacting cognitive decline prior to the overt progression to dementia.
Reduced cerebral perfusion when lying down is a predictor for the development and progression of dementia.12–18 Middle cerebral artery velocity (MCAv) at diastole (MCAdv) is a hemodynamic index which profiles the lowest level of flow within a cardiac cycle and it is pronounced during reductions in MAP throughout an orthostatic stress test while MCAv at systole is maintained. 19 We recently found that lower standing MCAdv, and not supine MCAdv, is significantly associated with lower cognitive function on a Montreal Cognitive Assessment (MoCA) in people with MCI. 20 One potential pathway by which reduced MCAdv may be associated with lower cognitive performance is dynamic cerebral autoregulation (dCA).
When transitioning from lying to standing, mean arterial pressure and MCAv rapidly decrease before typically recovering within 30-seconds. During this transition, hydrostatic pressure in the arteries below the left ventricle increase, and above this level the pressure in the arteries decreases.21,22 dCA is the cerebral vasculature’s ability to modulate cerebrovascular resistance in order to maintain cerebral blood flow despite abrupt changes in atrial blood pressure, thus providing a rapid response to cerebral blood flow regulation.23–27 dCA is a conceptual framework to understand pressure-flow relationships experienced during activities of daily living and may be related to cognitive performance.
Several studies have demonstrated a vast range of conflicting evidence suggesting dCA is either implicated with neurocognitive dysfunction in people with MCI, 28 not different between Alzheimer’s disease, MCI or cognitively intact controls, 29 or improved with Alzheimer’s disease and MCI compared to controls. 30 Although these studies compared Alzheimer’s disease to MCI it is important to note that Alzheimer’s disease is a biological process which often, but not always, causes dementia, whereas MCI refers to someone with a measurable cognitive impairment but maintained functional independence. A systematic review investigating relationships between dCA in people with dementia and MCI only reported four studies which included participants with MCI. 31 A root source for the diverse study findings may be that only some people with MCI develop dementia, and some people revert to normal cognitive abilities. Therefore, only a portion of people with MCI may have dCA responses similar to people with dementia and others may respond similarly to cognitively intact controls.
As shown in our earlier report from this group, standing MCAdv was positively associated MoCA scores, 20 where two participants of the same age and sex can have similar supine MCAdv values but very different standing MCAdv profiles (Figure 1(a)). Knowing that not all people with MCI develop overt dementia, we propose to separate participants into cerebrovascular perfusion groups (higher and lower) to elucidate some of the conflicting reports regarding dCA in people with MCI. We tested the hypothesis that participants with MCI who had higher MCAdv during standing would have superior dCA and better performance on the MoCA compared to participants with MCI who had lower MCAdv during standing.
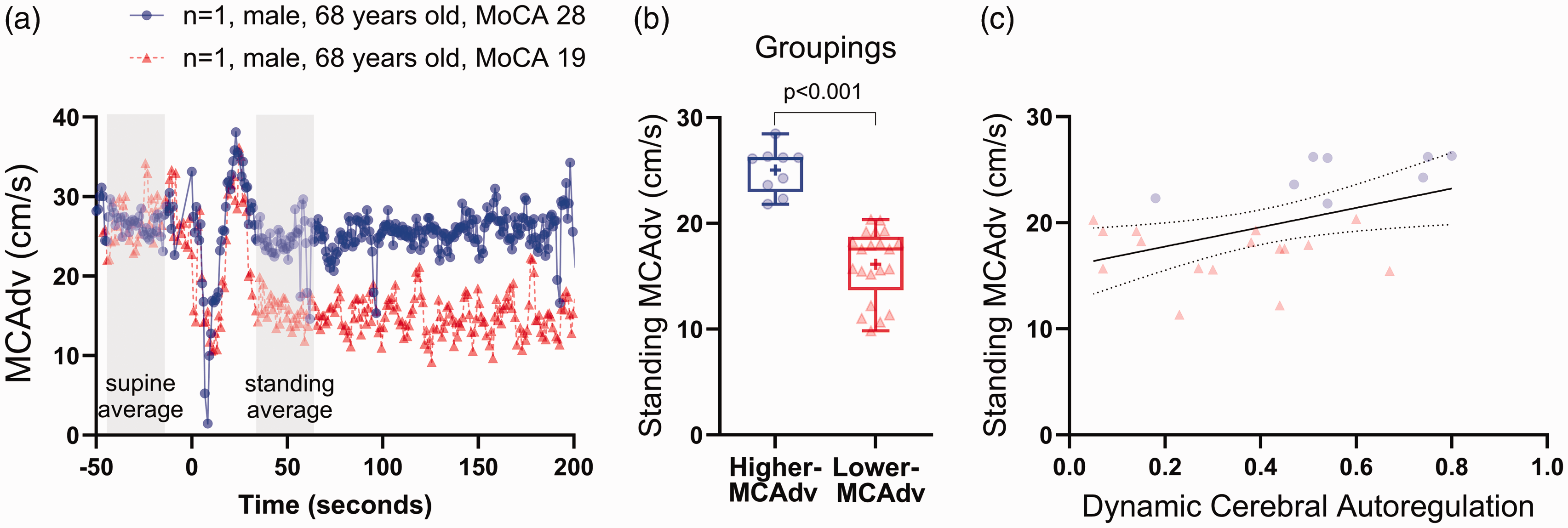
Cohorts based on standing middle cerebral artery velocity at diastole (MCAdv). (a) Beat-by-beat tracing of two age matched males, one with a higher MCAdv and one with a lower MCAdv during standing at 1-minute. (b) Groupings based off k-cluster variable, standing MCAdv. Positive symbol represents mean, box represents interquartile range, and whiskers represent maximum and minimum values and (c) plotted values for standing MCAdv and dynamic cerebral autoregulation in higher-velocity (red) and lower-velocity (Blue) groups.
Material and methods
Participant characteristics
The current study is part of the Comprehensive Assessment of Neurodegeneration and Dementia (COMPASS-ND) Study at the Parkwood Institute location in London, ON. The COMPASS-ND Study is a Canadian cohort research initiative, and it is a clinical study within the Canadian Consortium on Neurodegeneration in Aging (CCNA). All procedures were reviewed and approved by Clinical Trials Ontario (CTO750) at Western University and conformed with the Declaration of Helsinki. Informed written consent was obtained from all participants, after which nonidentifiable identifiers were generated to ensure the privacy of all participants. Investigators were unaware of the group allocation during the experimental and in assessing outcomes.
As part of the COMPASS-ND protocol, participants were asked to report years of education and prescription medications, including blood pressure lowering medications. General exclusion criteria included the presence of major psychiatric disorders due to confounding affects of cognitive capacity, 6 chronic brain disease (ex. leukoencephalopathy, or traumatic injury), multiple sclerosis, developmental disorders, malignant tumors, Huntington disease and other brain illnesses, alcohol or drug abuse, lack of a study partner, insufficient proficiency in English or French, and incapable of comprehending test instructions unassisted.6,32,33
Of the 88 participants who volunteered for this COMPASS-ND sub-study, participants were excluded from analysis due to inadequate MCAv signals (n = 35), a clinical diagnosis of dementia (n = 9), or classification criteria for cognitively intact controls (n = 14). 20 MCAv signal attrition rate was due to movement artifacts during the supine-to-stand transition which precluded the ability to calculate a true nadir value and calculate dCA. Aside from movement artifacts during a supine-to-stand transition, signal acquisition of MCAv is reduced with increasing age due to changes in bone morphology with age.34–37 Thirty individuals with classification criteria for MCI were included for analysis (10 female, 74 ± 6 years). Classification criteria for MCI included the National Institute on Aging, Alzheimer’s Association Clinical Criteria, 8 a self-reported concern of changes in cognitive function, preserved independence marked by a score of greater than 14 on the Lawton and Brody Scale, absence of dementia based on a Global Clinical Dementia Rating score less than 1, and the impairment in one or more of the following, Alzheimer’s Disease Neuroimaging initiative, Consortium to Establish a Registry for Alzheimer’s Disease, a MoCA score less than 24, or a Global Clinical Dementia Rating score of 0.5.8,32
Cardiorespiratory and cerebrovascular indexes
Continual measurements of hemodynamic variables were collected at 1KHz (PowerLab, LabChart, version 8.0; AD Instruments, Colorado Springs, CO). These recordings included finger plethysmography, reconstructed heart level brachial pressures, estimated stroke volume using the 3-element Modelflow algorithm 38 which is known to reliably track changes during orthostatic stress, 39 and calculated cardiac output by stroke volume and heart rate (Finapres Nova, Finapres Medical Systems, Enschede, NL). Offline calculations included total peripheral resistance by dividing mean arterial pressure (MAP) by cardiac output, heart rate by calculating 60 divided by the beat interval in seconds, and the normalization of stroke volume, cardiac output, and total peripheral resistance to body surface area. 40 MAP was height corrected to the vertical distance from the heart to the transcranial Doppler probe to produce MAP values for brain (MAPMCA). MCAv metrics were recorded by transcranial Doppler ultrasound (2 MHz probe, TCD-X; Atys medical, Soucieu en Jarrest, France). Offline, MCAv feature extraction included peak-systolic (MCAsv), MCAdv, and mean (MCAmv). Cerebrovascular resistance index (MAPMCA/MCAmv), PI (MCAsv-MCAdv/MCAmv),41,42 resistance index (MCAsv-MCAdv/MCAsv), and dynamic cerebral autoregulation (dCA) were later calculated. End-tidal carbon dioxide was collected using a nasal cannula and gas analyzer (Capnostream 35, Medtronics, USA and Ireland). Cerebral oxygen tissue saturation was collected by near-infrared spectroscopy (NIRS; Artinis Medical Systems BV, Netherlands).
MCAv is highly sensitive to changes in end-tidal carbon dioxide, where a 3–5% rise in MCAv results for a given increase of 1 mmHg of end-tidal carbon dioxide,23,43–45 therefore when assessing MCAv end-tidal carbon dioxide should always be considered. We Collected continuous end-tidal carbon dioxide (Capnostream35, Medtronic, Dublin, Ireland) with a nasal cannula throughout the posture transition.
Protocol
Following instrumentation, participants completed a practice supine-to-stand posture transition, with a subsequent 10 minutes of supine rest and a recorded supine-to-stand posture transition. To standardize the duration of the movement patterns from supine to standing, the same research technician assisted both the practice and the data recorded transitions for all participants. Three averages were calculated for analysis: supine, nadir, and standing. The 30 second supine average was calculated 15 seconds prior to the active posture transition to avoid anticipatory response (Figure 1(a)). Nadir was calculated as a 3-beat average at the time of the lowest single MCAdv beat and its neighboring beats. The 30 second standing average was calculated from 30-seconds to 60-seconds into the stand (Figure 1(a)). dCA was calculated as (MCAvt2-MCAvt1/MCAvt1)/(MAPMCAt2-MAPMCAt1/MAPMCAt1), where t1 was supine and t2 is nadir.46–48 A dCA value of 0.0 suggests the complete damping of the MAPMCA waveform into the cerebral vasculature, indicating perfect autoregulation. Conversely, a value of 1.0 implies lack of buffering of the MAPMCA changes to the cerebrovascular system, representing worse autoregulation. A dCA value represents the ability of the autoregulatory system of the brain to evoke changes on the vasculature which can then mitigate the effects of MAP on cerebral blood flow. This system is effective provided the changes of MAP are slow enough for the vasculature to act
Statistical testing
All analysis were performed using SPSS software (version 26, IBM Corp, Armonk, NY, 2019) and all tests were considered significant at p ≤ 0.05, trends were considered between p ≤ 0.1 and >0.05. Four participants were missing plethysmography data at nadir due to movement artifacts and placing pressure on the hand to get up from the supine position.
Participant grouping: We applied a K-means clustering algorithm to identify participant groups with similar responses. 49 Using the two-step cluster function in SPSS, two clusters (higher-velocity n = 9 and lower-velocity n = 21, Figure 1(b)) were optimally determined with a silhouette measure of cohesion and separation >0.5 (a good cluster quality).
A one-way analysis of variance was employed to determine if group differences existed for age, body mass index, MoCA scores, education, and dCA. A Chi square test and Phi were run to identify any group differences for sex and blood pressure lowering medications. When the assumptions for a Chi square test were violated a Fisher’s Exact test was utilized.
Effects of condition and group
A two-way repeated measures ANOVA identified group by position effects for all cardiorespiratory and cerebrovascular indices. Participant grouping included higher-velocity and lower-velocity, and positions included supine, nadir, and standing averages. Age was included in the statistical model as a covariate for all cardiovascular and cerebrovascular indices as these metrics change with age50,51 and age was significantly different between groups.
Results
Participant characteristics
No significant differences were observed between groups for the number of self identified females, body mass index, years of education, or presence of blood pressure lowering medication. However, participants in the higher-velocity group were significantly younger (70 ± 6 years vs. 75 ± 5 years, p = 0.027) and scored higher on the MoCA (27 ± 2 vs. 22 ± 5, p = 0.049, Table 1 and Figure 2).
Participant characteristics.
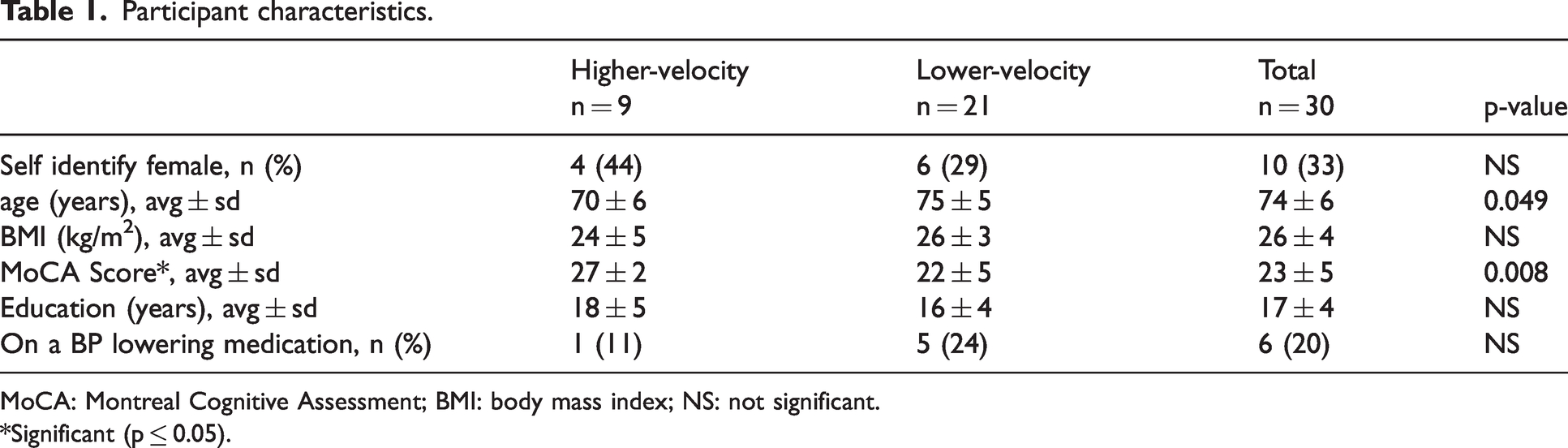
MoCA: Montreal Cognitive Assessment; BMI: body mass index; NS: not significant.
*Significant (p ≤ 0.05).
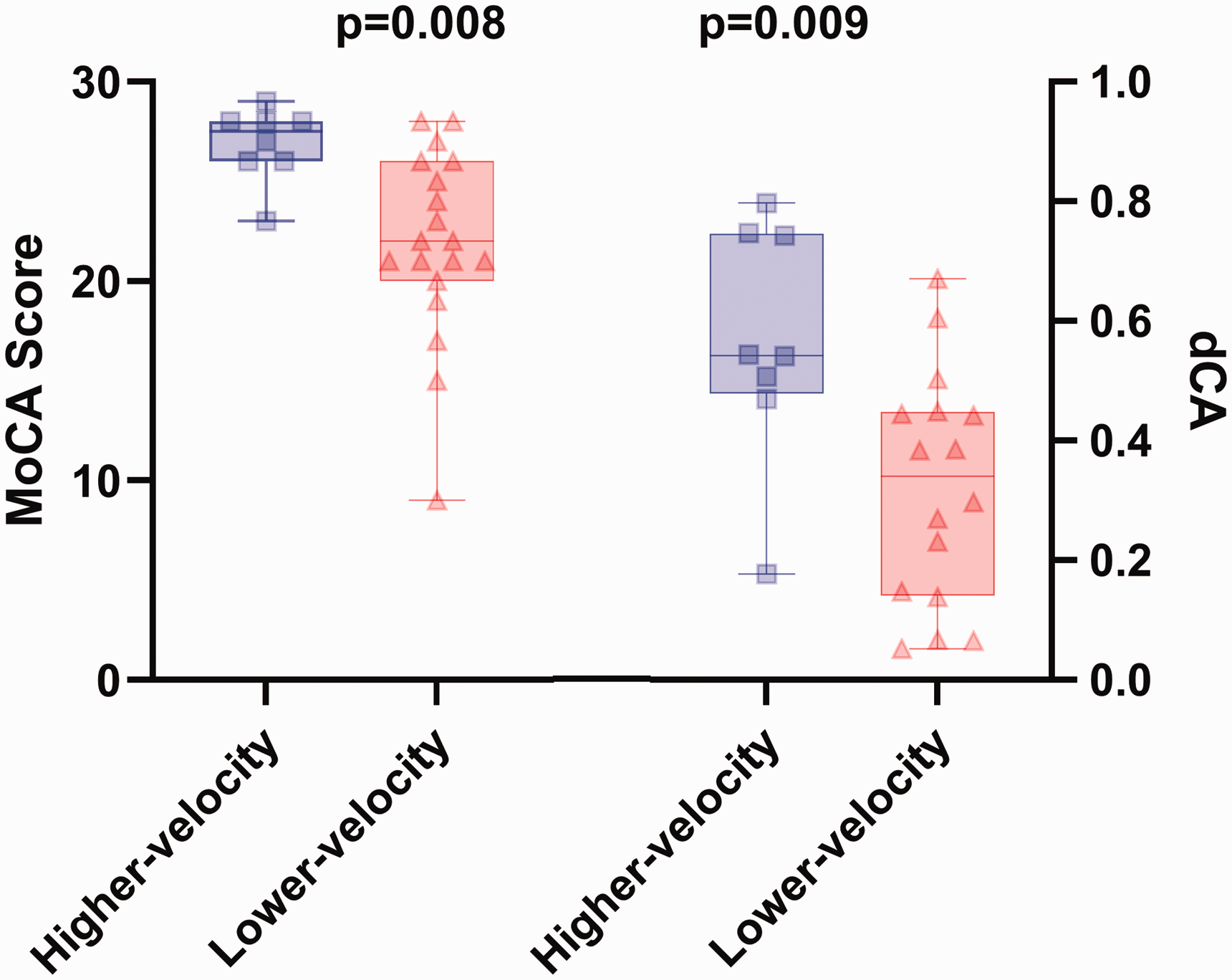
Montreal Cognitive Assessment (MoCA) score and dynamic cerebral autoregulation (dCA) are significantly greater in the higher-velocity group (solid blue bars) compared to the lower-velocity group (hashed red bars), even after controlling for age.
Dynamic cerebral autoregulation (dCA)
Compared to the lower-velocity group, dCA was significantly greater in the higher-velocity group (Figure 2). Figure 3 represents individual hemodynamic responses of MAPMCA and MCAv averages at supine rest and nadir which were used to calculate dCA. There were no significant differences of MAPMCA between groups. However, MCAv had a significant group by position interaction (p = 0.003), even after controlling for age (Figure 3). Pairwise comparisons identified differences between all positions for the higher-velocity group with the exception of supine versus standing MCAv, and for the lower-velocity group with the exception of nadir versus standing (Figure 3).
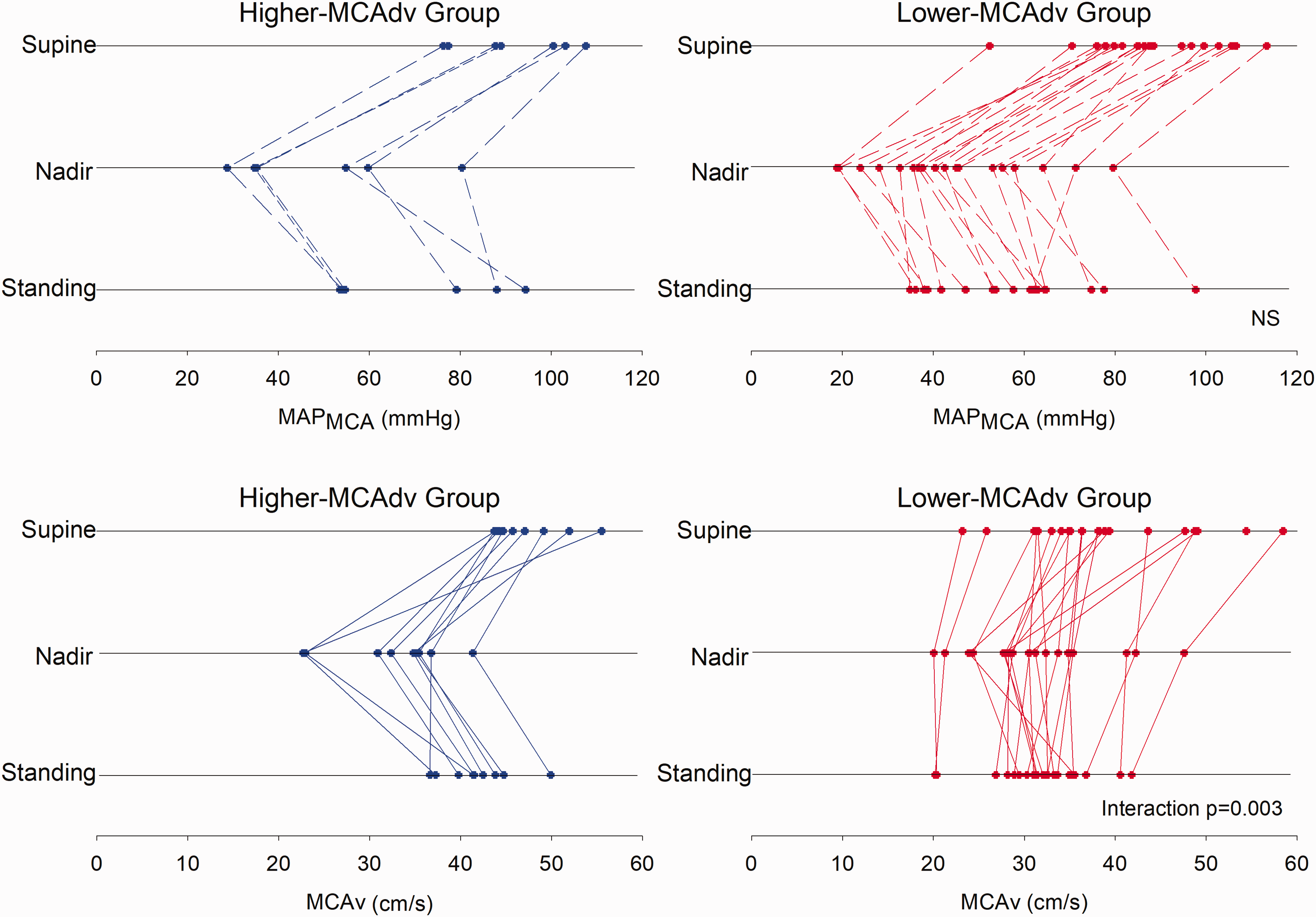
Individual hemodynamic responses from the supine-to-stand posture transition. Participants are separated by group (higher-velocity group left panel in blue), lower-velocity group right panel in red). Blood pressure at the level of the middle cerebral artery (MAPMCA, hashed lines) and middle cerebral artery mean velocity (MCAv, solid lines) at supine rest, 3-beat nadir, and standing. No significant relationships observed for MAPMCA (p > 0.05), a significant interaction effect (p = 0.003) was observed for the group by time responses for MCAv.
Hemodynamic responses
MCAdv was the clustering variable and the only hemodynamic variable to reach statistical significance (interaction p = 0.014, Table 2). Pairwise comparisons revealed similar relationships as observed for MCAv, whereby all group and position comparisons were significantly different, with the exception of the higher-velocity group for supine versus standing MCAdv (Table 2). Although not statistically significant, diastolic blood pressure, cerebrovascular resistance index, and resistance index all had trends for interaction effects (p = 0.063, p = 0.07, and p = 0.098 respectively, Table 2 and Figure 4). A significant group by position interaction was observed for cerebral oxygen tissue saturation, where the higher-velocity group did not demonstrate any differences in cerebral oxygen tissue saturation between positions, and the lower-velocity group had greater supine cerebral oxygen tissue saturation values compared to nadir and standing (Figure 4).
Hemodynamics 2-way repeated measures anova controlling for age.
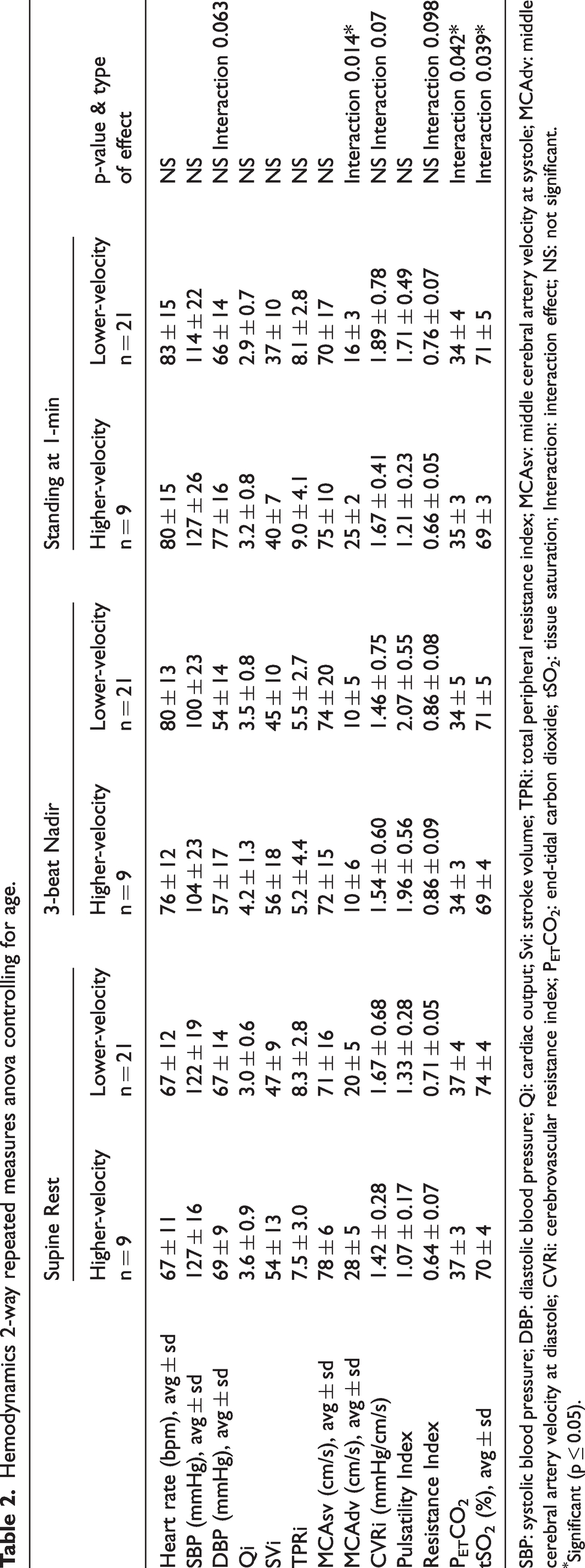
SBP: systolic blood pressure; DBP: diastolic blood pressure; Qi: cardiac output; Svi: stroke volume; TPRi: total peripheral resistance index; MCAsv: middle cerebral artery velocity at systole; MCAdv: middle cerebral artery velocity at diastole; CVRi: cerebrovascular resistance index; PETCO2: end-tidal carbon dioxide; tSO2: tissue saturation; Interaction: interaction effect; NS: not significant.
Significant (p ≤ 0.05).
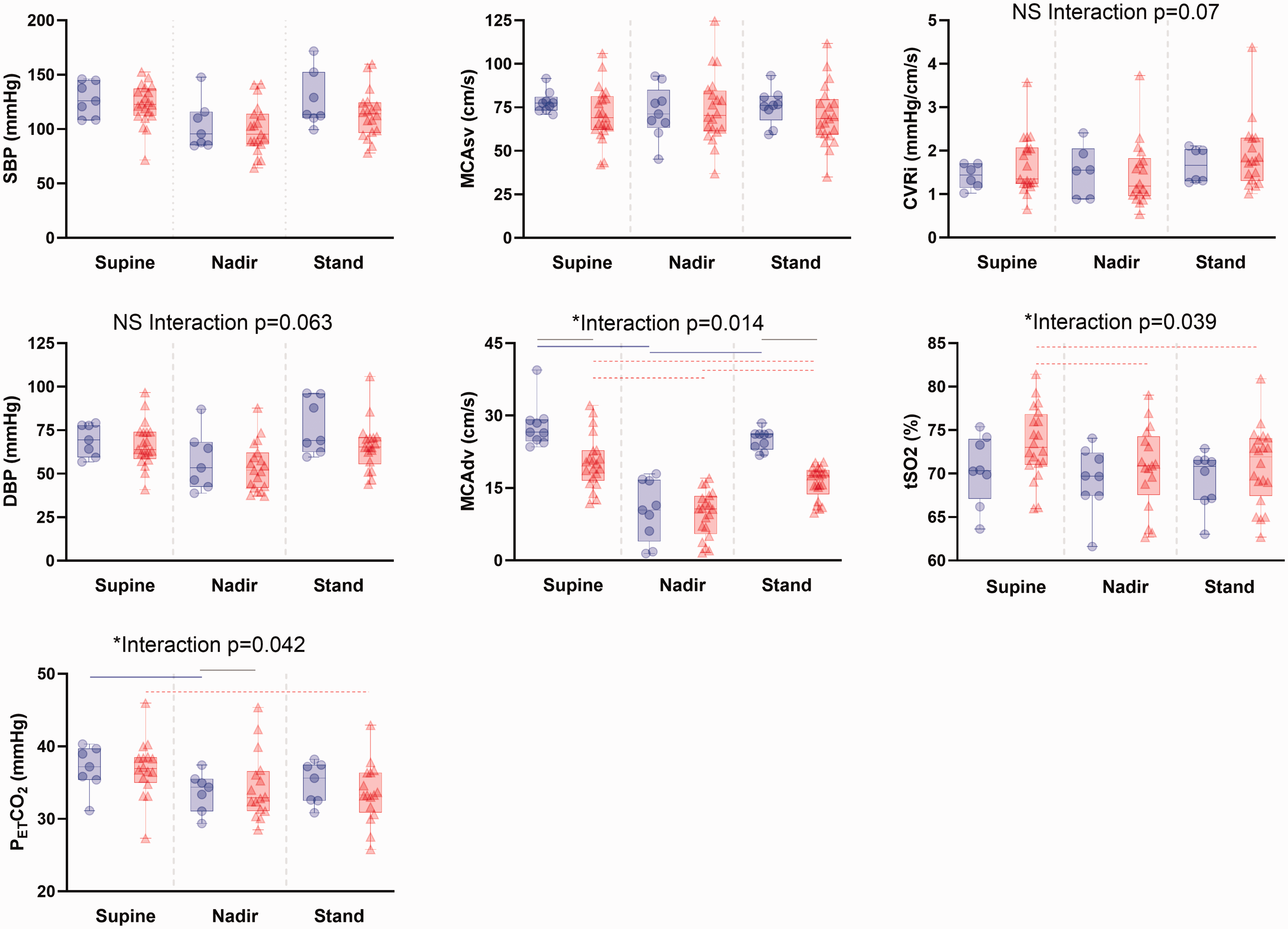
Group effects of a supine-to-stand transition on hemodynamic variables. Blood pressure at systole (SBP) and diastole (DBP), middle cerebral artery velocity at systole (MCAsv) and diastole (MCAdv), cerebrovascular resistance index (CVRi), and tSO2 cerebral oxygen tissue saturation, end-tidal carbon dioxide (PETCO2), for higher-velocity group (blue cicles) and lower-velocity group (red triangles bars). * For statistically significant differences (p ≤ 0.05), pairwise comparisons marked for higher-velocity group (solid blue lines), and lower-velocity group (red dashed lines) and between groups (solid grey line). The box represents interquartile range, and whiskers represent maximum and minimum values, with the mean as a bar in the middle of the box.
Heart rate, systolic blood pressure, cardiac output, stroke volume, total peripheral resistance, cerebrovascular resistance index, and pulsatility index, were not significantly different when comparing between groups and positions (supine, nadir, and standing), and controlling for age (Table 2). End-tidal carbon dioxide had a significant interaction effect (p = 0.042, Table 2) where the higher-velocity group had significantly lower end-tidal carbon dioxide value at nadir compared to supine values and the lower-velocity group had a higher end-tidal carbon dioxide value at supine rest compared to standing. When comparing nadir values of end-tidal carbon dioxide, the higher-velocity group had lower values than the lower-velocity group. Supine and nadir end-tidal carbon dioxide were not significantly correlated to dCA. There was a non-significant interaction effect for diastolic blood pressure (p = 0.062, Table 2) where post-hoc analysis identified a lower nadir value compared to supine and standing averages in the lower-velocity group, as well as differences between supine, nadir, and standing for participants in the higher-velocity group.
Discussion
The aim of the current study was to investigate if cognitive function and dCA are different between participant groups with higher and lower standing cerebral perfusion, but all of whom live with MCI. Aligning with our previous findings in a similar cohort of data, 20 the group with higher-velocity had significantly greater MoCA scores than the group with lower-velocity. However, contrary to our hypothesis, dCA was less efficient in the higher-velocity group (Figure 2). This finding is counterintuitive to what was expected in a cohort of older adults with higher MoCA scores and who are on average 5-years younger (Table 1).
Throughout an orthostatic challenge, the dCA values represent the correlation between the rate of reduction in MAPMCA and MCAv during the abrupt change in perfusion pressure. There were no differences between groups or among positions (supine, nadir, stand) for MAPMCA, suggesting any observed differences of the dCA calculation are driven by changes in the MCAv variable (Figure 3). MCAv had a significant group by position interaction effect (p = 0.003). Within the higher-velocity group, supine and standing MCAv did not differ, thus a full recovery of MCAv during a posture change was achieved. Within the lower-velocity group, nadir and standing values were not significantly different, indicating insufficient recovery of MCAv from supine to standing (Figure 3). The lower-velocity group also had a significantly lower end-tidal carbon dioxide from supine rest to standing (p = 0.04) which likely contributes to the lower standing MCAdv, as did the higher cerebrovascular resistance index, though this metric was not statistically significant (p = 0.07, Figure 3). Although dCA may be less effective in the higher-velocity group, previously this laboratory observed associations between steady state cerebral perfusion during standing and cognitive function 20 whereas the observed transient changes found in the current investigation did not support this earlier relationship.
Not all people with MCI progress to develop dementia, thus they represent a clinical cohort which may be influx with either reverting back to becoming cognitively intact, remaining in a stable MCI state, or progressing to dementia.4,8–11 Similarly, dCA values in people with MCI reportedly align with both cognitively intact controls and people with dementia. Beishon et al. 2021 demonstrated a reduced dCA in dementia and MCI relative to healthy controls 52 whereas Tarumi and colleagues did not identify group differences of dCA when comparing cognitively intact controls to amnestic MCI participants. 28 However when Tarumi and colleagues compared cerebral oxygen tissue saturation in response to dynamic changes in MCAv there appeared to be an association with memory performance. 28 Likewise, Gommer and colleagues did not find differences of spontaneously occurring supine dCA among cognitively intact controls, MCI or dementia groups. 29 In contrast, de Heuz et al. 2018, reported more efficient dCA indicators (higher autoregulatory index and lower transfer function gain) observed in people with dementia and MCI for a repeated sit-to-stand protocol when compared to cognitively intact controls. 30 In a systematic review on dCA in people with dementia and MCI it was concluded that autoregulatory function is preserved or potentially better in MCI and dementia participants. 31 It appears as though both approaches to assess dCA, spontaneous oscillations and an active transition to standing, are effective despite reports of spontaneously occurring supine dCA assessments producing insufficient blood pressure perturbations to effectively asses dCA. 53 The current study agrees with the reports by de Heuz et al., whereby the participants with a higher-velocity and higher MoCA scores had reduced dCA when compared to participants with lower-velocity and lower MoCA scores (Figure 2).
As outlined by Weijs et al. 2023, there are several explanations for the observed enhancement of dCA, i) elevated cerebrovascular resistance index, ii) reduced end-tidal carbon dioxide, iii) directional sensitivity of dCA, and iv) a compensatory mechanism. 54 As observed in the current study, elevated cerebrovascular resistance index has an inverse relationship with cerebral blood flow (Table 1, Figure 4) which is known to favourably augment dCA values,27,55,56 and has been observed in pre-clinical groups of people with subjective cognitive impairment. 41 Although cerebrovascular resistance index in the present study was not statistically different between groups, there was a trend for an interaction effect (p = 0.07, Table 1). Given that the participants in the lower- versus higher-velocity group had a greater cerebrovascular resistance index at supine rest, cerebrovascular resistance index may be a contributing factor for dCA values associated with better cerebrovascular outcomes. A calculation of MCA compliance index using a Windkessel model to assess the pulsatile components of cerebral blood flow would lend additional insight to the current findings and should be considered in future investigations when characterizing cerebrovascular hemodynamics.57,58 Hypercapnia and hypocapnia impairs and enhances dCA, respectively.27,59 We noted an interaction effect between groups and positions (supine, nadir, and standing, Figure 4) suggesting these end-tidal carbon dioxide metrics may have impacted both MCAv and dCA. Directional sensitivity of dCA during a repeated squat stand manoeuvre favours rises and not falls of mean arterial pressure, 60 and in-vivo research also demonstrates directional sensitivity for cerebrovascular adaptations in response to increases and not reductions of arterial pressure. 61 These studies indicate that vascular smooth muscle properties may respond faster to vasoconstriction stimuli than what is required for vasodilation during a large reduction in mean arterial pressure, 60 ultimately impacting the dCA responses. Weijs et al. 2023 also propose dCA values associated with better cerebrovascular outcomes as a compensatory mechanism to counter the effects of reduced cerebral blood flow, reduced ability to modulate flow to a change in carbon dioxide, and to protect against cognitive decline as it pertains to hypoperfsuion. 54 The current investigation noted increased cerebral oxygen tissue saturation (Table 1 and Figure 4) which also suggests a compensatory mechanism; whereby participants with lower-velocity have a greater proportion of oxygenated versus deoxygenated hemoglobin content. Once the effects of any compensatory mechanism begin to degrade, the true impact of reduced cerebral blood flow on cognitive function will ensue.
The current study applied a well-established calculation of dCA which utilizes MAPMCA and MCAv.27,56 However, to the best of our knowledge, pervious dCA calculations have not considered diastolic pressure or end diastolic velocity as determinants in the relationships between arterial pressure and brain blood flow velocity. Diastole represents the lowest point in the pressure waveform and the time at which cerebrovascular compliance would be most challenged. If insufficient cerebrovascular compliance is achieved, then reductions in flow would be evident. Investigating cerebrovascular compliance in the future may provide additional insight into the relationships between dCA and cognitive function. additional insight into the relationships between dCA and cognitive function.
Methodological considerations and limitations of the study
The dCA calculation performed in the current study does not consider the phase shift between MCAv and MAP, such that a time delay between the central and cerebral measures are not detected.59,60 The influence of sex on vascular measures was not quantified due to insufficient sample sizes for such comparisons, yet the impact sex has on the cardiovascular system62,63 and dementia64–66 does not go unacknowledged. Physical fitness and strength can influence the speed of the supine-to-stand transition and the posture-related reduction in blood pressure; however, the current study is part of a larger study (COMPASS-ND) where such metrics were not collected at intake.
MCAv is a surrogate marker of cerebral blood flow when the assumption of vessel diameter is constant. MCA diameter is altered during hypo- and hypercapnic conditions whereby a 0.4% change results by 1 mmHg of end-tidal carbon dioxide. 67 From supine-to-nadir and supine-to-standing, end-tidal carbon dioxide decreased by 2.7 ± 3.5 mmHg and 3.0 ± 2.7 mmHg respectively, resulting in a 1.1% and 1.2% change in MCA diameter. Based on previous assumptions the posture-related reductions in MCAv are under-estimated. Furthermore, MCA cross sectional area dilates during hypercapnic conditions with step changes in sustained end-tidal carbon dioxide, not during a slower ramp increase, and the time course of MCA cross sectional area would not impact the MCAv nadir values but potentially the standing MCAv values at 1 minute. 68
According to Fraenkel et al., a Pearson’s correlation test requires a sample size of n = 30 to ensure meaningful results and to reduce the risk of a type 1 (false positive) and type 2 (false negative) result.69,70 With a study sample size of n = 30 for MCAv parameters but n = 24 for dCA, the current study was underpowered to perform Pearson Correlations on these variables and concretely associate changes in MCAdv with dCA (Figure 1). As such we split our participants into groups (higher- and lower-velocity) to demonstrate the relationships which exist within each group.
The current investigation is part of a larger study which was powered to detect changes in orthostatic hypotension. The present study has a small sample size however work by Robertson 2013 48 was powered to detect similar changes in dCA with comparable sample sizes (3 groups of n = 12, n = 9, n = 10). Continued research to expand on this preliminary work could provide further insight to the relationships we have identified.
The MoCA was used in the current investigation to assess global cognitive ability. However, by using the total score we are unable to investigate specific domains. 71 The MoCA can also be skewed by metal health conditions such as depression which can result as a lower score without any cognitive impairment 72 MoCA score adjustments were not considered for the current investigation.
Conclusions
This study is unique in stratifying individuals with MCI into high and low MCAdv groups to further investigate dCA and cognitive ability. MCI is a transition stage between cognitively intact and progressive cognitive decline. Lower dCA values are expressed as large dampening effects from abrupt changes in mean arterial pressure into the cerebral vasculature which confers to better cerebrovascular control. Calculating dCA during an activity of daily living, such as a supine-to-stand transition, accurately reflects vascular hemodynamic responses experienced in everyday life. We found participants with lower standing MCAdv, had lower MoCA scores, a lower dCA value, which confers to better cerebrovascular outcomes, and elevated cerebral oxygen tissue saturation. Our findings point towards dCA and cerebral oxygen tissue saturation as a compensatory mechanism to buffer against reduced cerebral blood flow during acute changes in MAP. The steady state features of cerebral blood flow velocity appear to be more closely related than the dynamic changes in predicting cognitive function in people with MCI. 20 Emphasis on improving or maintaining vascular health should be prioritized in prospective studies to further investigate these relationships.
Footnotes
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article materials.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Academic Medical Organization of Southwestern Ontario (AMOSO) Opportunities Fund Project (Grant No. F20-004, JB), the Parkwood Institute Research Endowment Fund: In Care of Older Adults (LKFC), and the Vascular Training Platform (LKFC). Funding was also provided by the Canadian Institute of Health Research on Aging for the Canadian Collaboration on Neurodegeneration in Aging, NIH Alzheimer Disease Neuroimaging Initiative (ADNI3), Biogen, Alector, Eisai, Abbivie, Eli Lilly Medical Imaging Trials Network of Canada C6 Project – MITNEC – C6, Roche, Biogen, and Hoffman-La Roche Ltd (all MB).
Acknowledgements
We would like to thank Dr. Richard Hughson from the Schlegel-University of Waterloo Research Institute for Aging, Waterloo, ON, for supporting this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
All of the listed authors have made substantial contributions. Specifically, the authors contributed to the concept and design of the study (LKF-C, MB, SP, JKS, JB), the acquisition of data and analysis (LKF-C, JB), the interpretation of results (LKF-C, JKS, SP, JB), wirtting of the first draft (LKF-C, JB), and reviewing and editing the final manuscript versions (LKF-C, MB, SP, JKS, JB). All authors have critically revised the drafts to improve intellectual content and approve the version to be published.
