Abstract
Cerebral blood flow (CBF) increases and dynamic cerebral autoregulation is impaired by acute hypoxia. We hypothesized that progressive hypocapnia with restoration of arterial oxygen content after altitude acclimatization would normalize CBF and dynamic cerebral autoregulation. To test this hypothesis, dynamic cerebral autoregulation was examined by spectral and transfer function analyses between arterial pressure and CBF velocity variabilities in 11 healthy members of the Danish High-Altitude Research Expedition during normoxia and acute hypoxia (10.5% O2) at sea level, and after acclimatization (for over 1 month at 5,260 m at Chacaltaya, Bolivia). Arterial pressure and CBF velocity in the middle cerebral artery (transcranial Doppler), were recorded on a beat-by-beat basis. Steady-state CBF velocity increased during acute hypoxia, but normalized after acclimatization with partial restoration of SaO2 (acute, 78%±2%; chronic, 89%±1%) and progression of hypocapnia (end-tidal carbon dioxide: acute, 34±2 mm Hg; chronic, 21±1 mm Hg). Coherence (0.40±0.05 Units at normoxia) and transfer function gain (0.77±0.13 cm/s per mm Hg at normoxia) increased, and phase (0.86±0.15 radians at normoxia) decreased significantly in the very-low-frequency range during acute hypoxia (gain, 141%±24%; coherence, 136%±29%; phase, −25%±22%), which persisted after acclimatization (gain, 136%±36%; coherence, 131%±50%; phase, −42%±13%), together indicating impaired dynamic cerebral autoregulation in this frequency range. The similarity between both acute and chronic conditions suggests that dynamic cerebral autoregulation is impaired by hypoxia even after successful acclimatization to an extreme high altitude.
Introduction
Exposure to high altitude radically alters factors that regulate the distribution of cerebral blood flow (CBF), such as hypoxia and hypocapnia. Hypoxia produces vasodilation in distal cerebral arterioles (Wadsworth, 1994), that is proportionate to the reduction in arterial oxygen content (Brown et al, 1985). Conversely, hypocapnia causes vasoconstriction in those vessels (Paulson et al, 1990). After prolonged acclimatization at high altitude, improvements in arterial oxygen saturation and increases in hemoglobin concentration restore arterial oxygen content, and progressive hypoxic hyperventilation induces further hypocapnia compared with the acute phase. The overall response of distal cerebral arterioles or steady-state CBF after acclimatization at high altitude depends on the relative magnitude of these two opposing effects of cerebral vasodilatation by reduction in arterial oxygen content and vasoconstriction by hypocapnia.
These factors may also alter dynamic regulation of CBF. Cerebral autoregulation adjusts tension in distal cerebral resistance vessels not only to keep steady-state CBF constant but also to minimize temporal changes or dynamic fluctuations of CBF (Aaslid et al, 1989; Giller, 1990; Zhang et al, 1998). Dynamic cerebral autoregulation is reported to be impaired immediately after the onset of exposure to either mild (Iwasaki et al, 2007a) or severe hypoxia (Subudhi et al, 2009). Conversely, impaired cerebral autoregulation in patients with intracranial diseases or circulatory dysfunction can be partly restored by hypocapnia (Newell et al, 1996; Strauss et al, 1998).
As there has been no report that examines steady-state CBF and dynamic cerebral autoregulation in subjects who are well acclimatized to very high altitude, it remains unclear whether the combination of hypoxia and hypocapnia results in significant changes in steady-state CBF and dynamic cerebral autoregulation after acclimatization. On the basis of previous work, we expected that acute exposure to severe hypoxia would induce profound vasodilation of distal cerebral arterioles and impairment of dynamic cerebral autoregulation; however, we hypothesized that acclimatization to an extreme high altitude would restore arterial oxygen content and induce progressive hypocapnia, normalizing dynamic cerebral autoregulation. To test this hypothesis, we examined the cerebral circulation in members of the Danish High-Altitude Medical Research Expedition to the Andes after successful acclimatization of 1-month exposure to a high altitude of 5,260 m.
Materials and methods
Subjects
The study was approved by the Scientific Ethics Committee for Copenhagen, Denmark, and the Institutional Review Boards of the University of Texas Southwestern Medical Center at Dallas and the Presbyterian Hospital of Dallas. Informed written consent was obtained from subjects before participation. In all, 11 healthy members of the Danish High-Altitude Medical Research Expedition to the Andes, 7 men and 4 women (aged 23 ± 4 years), were studied. Subjects refrained from heavy exercise and caffeinated or alcoholic beverages for ≥ 24 hours before testing. All experiments were performed ≥ 2 hours after a meal.
Protocols
Normoxia (Normobaric 21% FIO2):
All subjects had resided for over 6 months at 0 m (Copenhagen, Denmark), before the sea-level experiments. These experiments were conducted in a quiet, environmentally controlled laboratory at sea level (Copenhagen, Denmark) with an ambient temperature of 24°C.
Acute Hypoxia (Acute Exposure to Normobaric Hypoxia):
Following experiments for normoxia, humidified air with 10.5% FIO2 designed to match the FIO2 at 5,260 m was delivered to the subject from a large Douglas bag. The subject inhaled it for ∼30 minutes before the experiments for acute hypoxia, and the stability of SaO2 was confirmed by pulse oximetry. The same experiments as normoxia were repeated during inhalation of the hypoxic gas.
Chronic High Altitude (Chronic Exposure to Hypobaric Hypoxia):
All subjects had resided for over 1 month at 5,260 m (Chacaltaya, Bolivia), and had fully recovered from a successful summit of Huayna Potosi (6,088 m) the previous week. No subjects had any signs or symptom of acute mountain sickness (AMS). The same series of experiments as normoxia at sea level were performed at 5,260 m (Chacaltaya, Bolivia).
Instrumentation and Experiments
End-tidal carbon dioxide (ETCO2) was monitored at altitude using an infrared CO2 sensor (Sensormedics, Yorba Linda, CA, USA); at sea level, an infrared CO2 sensor acquired from a different manufacturer (MedGraphics, St Paul, MN, USA) was used, but with the same experimental set-up. Arterial oxygen saturation (SaO2) was monitored using a pulse oximeter (Criticare Systems, Waukesha, WI, USA). Continuous arterial blood pressure was obtained at the right radial artery by standardized applanation tonometry (CBM-7000, Colin, San Antonio, TX, USA) at heart level on a beat-by-beat basis. Intermittent blood pressure was also measured by oscillometric determination using cuff sphygmomanometry of the brachial artery to calibrate continuous arterial blood pressure. Cerebral blood flow velocity was measured continuously in the middle cerebral artery by transcranial Doppler (Multiflow, DWL, Sipplingen, Germany). A 2-MHz probe was placed over the subject's temporal window. To place it in the exact same position both at sea level and high altitude and to fix it at a constant angle, an individual probe holder was made with a polymer mold to fit each subject's facial bone structure and ear (Giller and Giller, 1997). The signal was obtained according to standard techniques with the Doppler sample volume adjusted to the proximal segment of the middle cerebral artery, optimizing the opportunity for obtaining true maximum velocities. All recordings were undertaken by the same experienced technician. The reproducibility of CBF velocity measured in the middle cerebral artery by transcranial Doppler especially when careful attention is paid to probe placement, has been confirmed to be good, indicated by the high intraclass correlation coefficient of ∼0.9 (Brodie et al, 2009) and the small coefficient of variation (10%) in repeated baseline measurements (Iwasaki et al, 2007b) for mean CBF velocity.
After establishment of quiet rest (usually ∼30 minutes), 6-minute data of arterial blood pressure and CBF velocity were recorded on the Multiflow during spontaneous respiration in the supine position. The analog arterial blood pressure and peak envelope of Doppler CBF velocity were sampled simultaneously at 100 Hz, and digitized at 12 bits. Real-time beat-to-beat systolic blood pressure, diastolic blood pressure, systolic CBF velocity, and diastolic CBF velocity were detected. Beat-to-beat values of mean blood pressure (MBP) and mean CBF velocity (MCBF) were calculated as a true average of each integrated waveform (Figure 1). Both ETCO2 and SaO2 were recorded every minute.
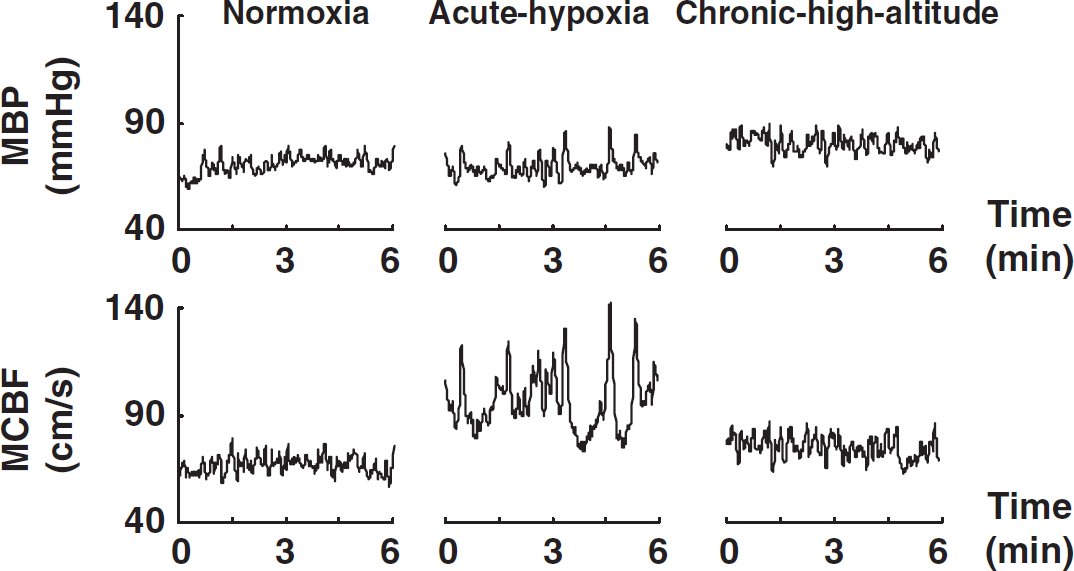
Representative data for mean blood pressure (MBP) and mean cerebral blood flow velocity (MCBF) in a subject for 6 minutes of spontaneous breathing. Normoxia, normobaric 21% FIO2 at sea level (0 m); acute hypoxia, acute exposure to normobaric hypoxia (10.5% FIO2) at sea level (0 m); chronic high altitude, chronic exposure to hypobaric hypoxia for 1 month at Chacaltaya in Bolivia (5,260 m).
To enhance the power of forced oscillations in arterial blood pressure as input signals to cerebral autoregulation at the primary autoregulatory frequency at which coherence may be too low during spontaneous breathing to reliably calculate the transfer function (0.05 Hz), controlled fixed-frequency breathing was used for all three protocols (namely normoxia, acute hypoxia, and chronic high altitude) to increase coherence. After spontaneous respiration, subjects were asked to control their respiratory frequency at a fixed rate of 3 per min by following a graph on a computer for 3 minutes; tidal volume was automatically adjusted on the graph to maintain constant alveolar ventilation, although ventilation was not strictly controlled.
Steady-State Indices Analysis
Steady-state arterial blood pressures (systolic blood pressure, diastolic blood pressure, and MBP), CBF velocities (systolic CBF velocity, diastolic CBF velocity, and MCBF), heart rate, SaO2, and ETCO2 were obtained from the average of the 6-minute data segments. A ‘cerebral vascular resistance index’ in the supine position was calculated by dividing steady-state MBP by steady-state MCBF to estimate relative changes in cerebrovascular resistance in the distribution of the middle cerebral artery (Aaslid et al, 1989).
Spectral Analysis
The 6-minute data of beat-to-beat MBP and MCBF were linearly interpolated and resampled at 1 Hz for spectral analysis. The spectrum by fast Fourier transform in MBP and MCBF variabilities and the transfer function by the cross-spectrum method between these two variabilities (Figures 2 and 3) were calculated to assess the dynamic relationship between arterial blood pressure and CBF velocity. This data analysis was performed using DADiSP software (DSP Development, Cambridge, MA, USA).
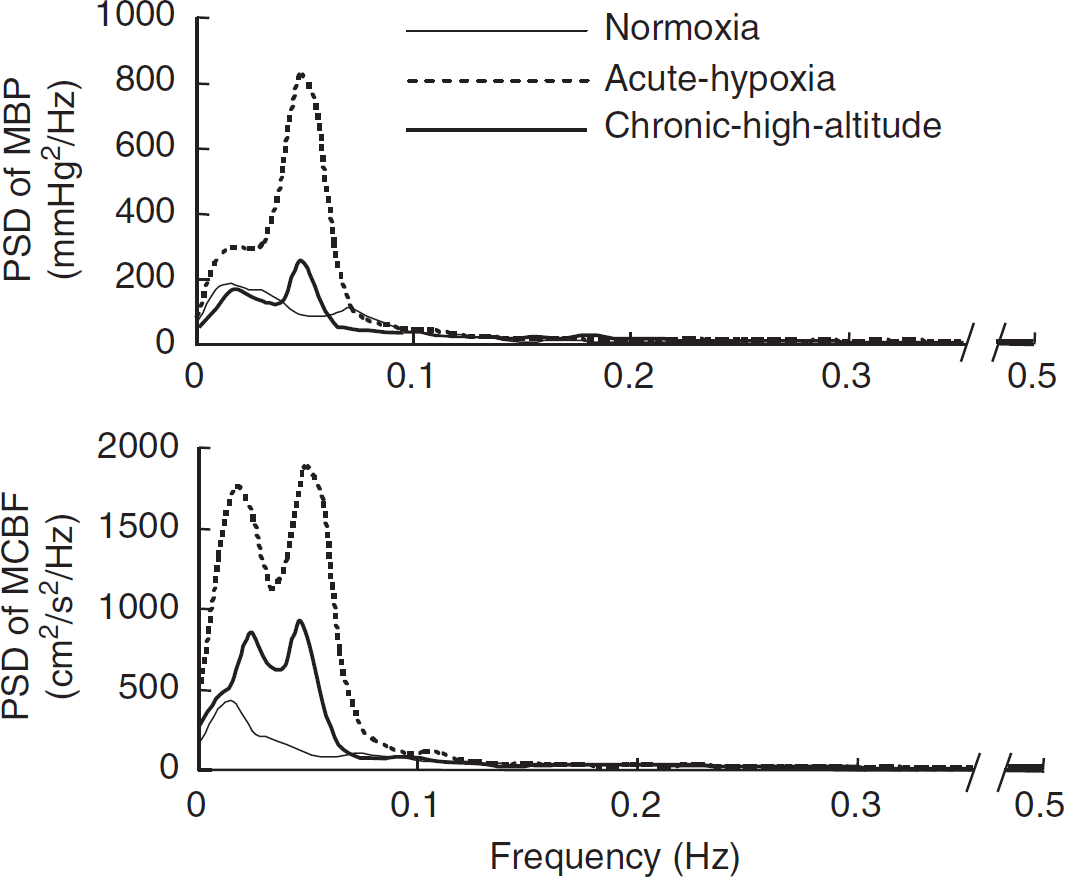
Group-averaged frequency-domain analysis of mean blood pressure (MBP) and mean cerebral blood flow velocity (MCBF) variabilities. The axis break on all the
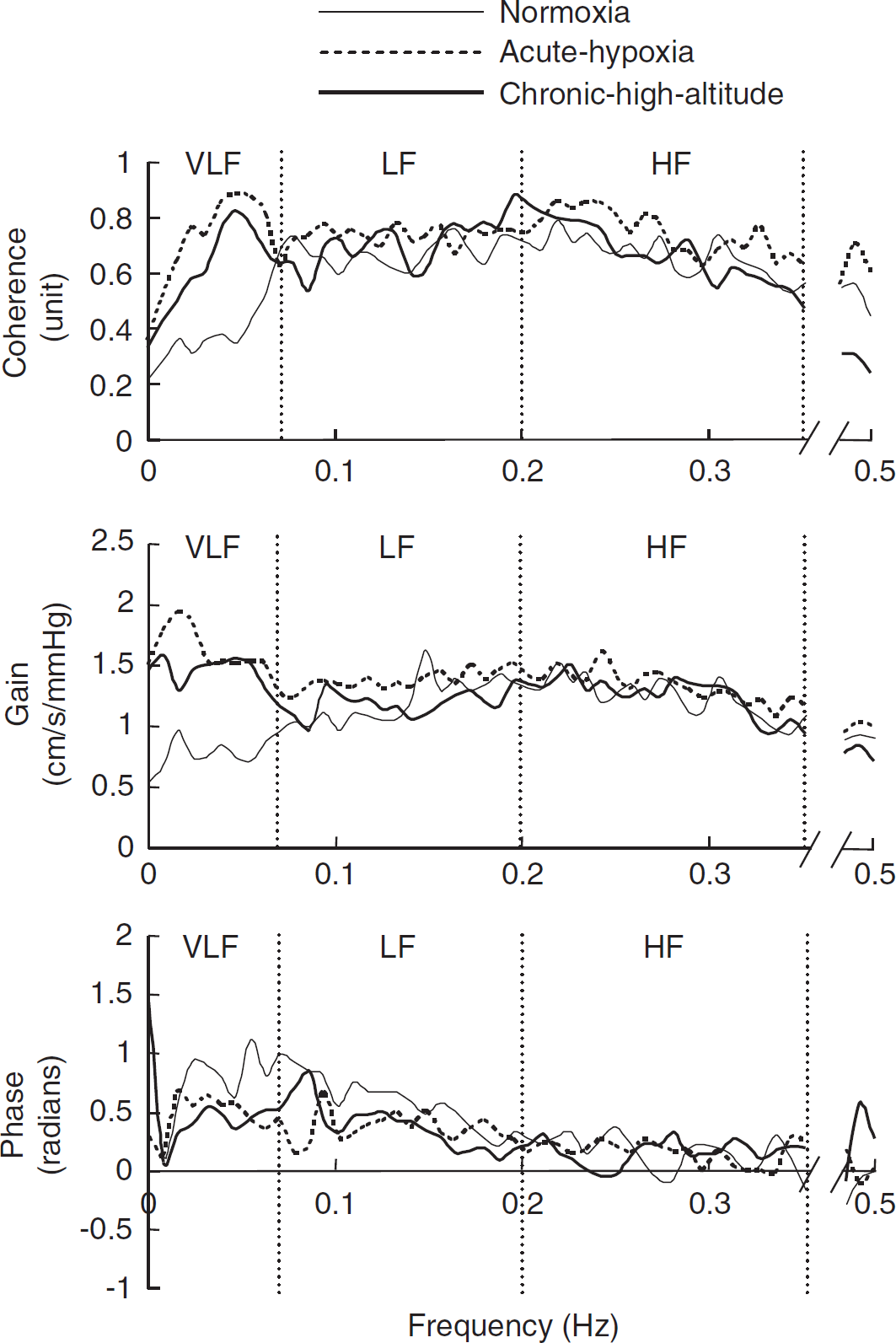
Group-averaged transfer function analysis between mean blood pressure (MBP) and mean cerebral blood flow velocity (MCBF) variabilities. Gain, transfer function gain between MBP and MCBF variabilities; VLF, very-low-frequency range (0.02 to 0.07 Hz); LF, low-frequency range (0.07 to 0.20 Hz); HF, high-frequency range (0.20 to 0.35 Hz). The axis break on all the
Transfer function H(f) between the two signals was defined as:
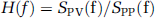
where SPP(f) is the Fourier-transformed autocorrelation function (autospectrum) of changes in MBP and SPV(f) is the Fourier-transformed cross-correlation function (cross-spectrum) between MBP and MCBF signals. Transfer function magnitude |H(f)| and phase spectrum Φ(f) were derived from the real part [HR(f)] and the imaginary part [HI(f)] of the complex function |H(f)| as:
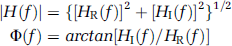
The mean squared coherence (MSC) function MSC(f) was defined as:
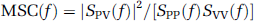
where Svv(f) is the autospectrum of MCBF.
The spectral power of MBP and MCBF variabilities, mean value of coherence function, transfer function gain, and phase were calculated in the very-low (0.02 to 0.07 Hz)-, low (0.07 to 0.20 Hz)-, and high (0.20 to 0.35 Hz)-frequency ranges, chosen to reflect frequency-dependent patterns of the dynamic pressure–flow relationship as reported previously (Zhang et al, 1998, 2002).
The coherence function (strength of association) was calculated to assess the linear relationship between these two variables and the reliability of the transfer function gain and phase. Estimates of the transfer function gain (magnitude of association) were used to quantify the amplitude of signal transmission from arterial blood pressure to CBF velocity. Phase was used to estimate the temporal relationship between these two variables. For example, when coherence is sufficiently high, pressure and velocity vary together very closely and both gain and phase can be used to evaluate the magnitude of the transfer and its temporal relationship. Under such circumstances, if gain is also large, changes in arterial blood pressure lead to larger changes in flow, implying impaired autoregulation. Moreover, it is reported that estimates of phase decrease under clinical conditions in which impairment of dynamic cerebral autoregulation is expected, such as occlusive cerebrovascular diseases (Diehl et al, 1995), carotid stenosis (Hu et al, 1999), or hypercapnia (Birch et al, 1995; Panerai et al, 1999).
Statistical Analysis
Data are represented as means ± s.e.m. Data across protocols were compared using a repeated-measures analysis of variance (ANOVA) with a Student–Newman–Keuls
Results
Steady-State Indices
Group-averaged steady-state respiratory conditions and hemodynamic data during 6 minutes of spontaneous breathing are presented in Table 1. As expected, SaO2 and ETCO2 decreased significantly during acute hypoxia (ANOVA,
Steady-state indices during spontaneous breathing
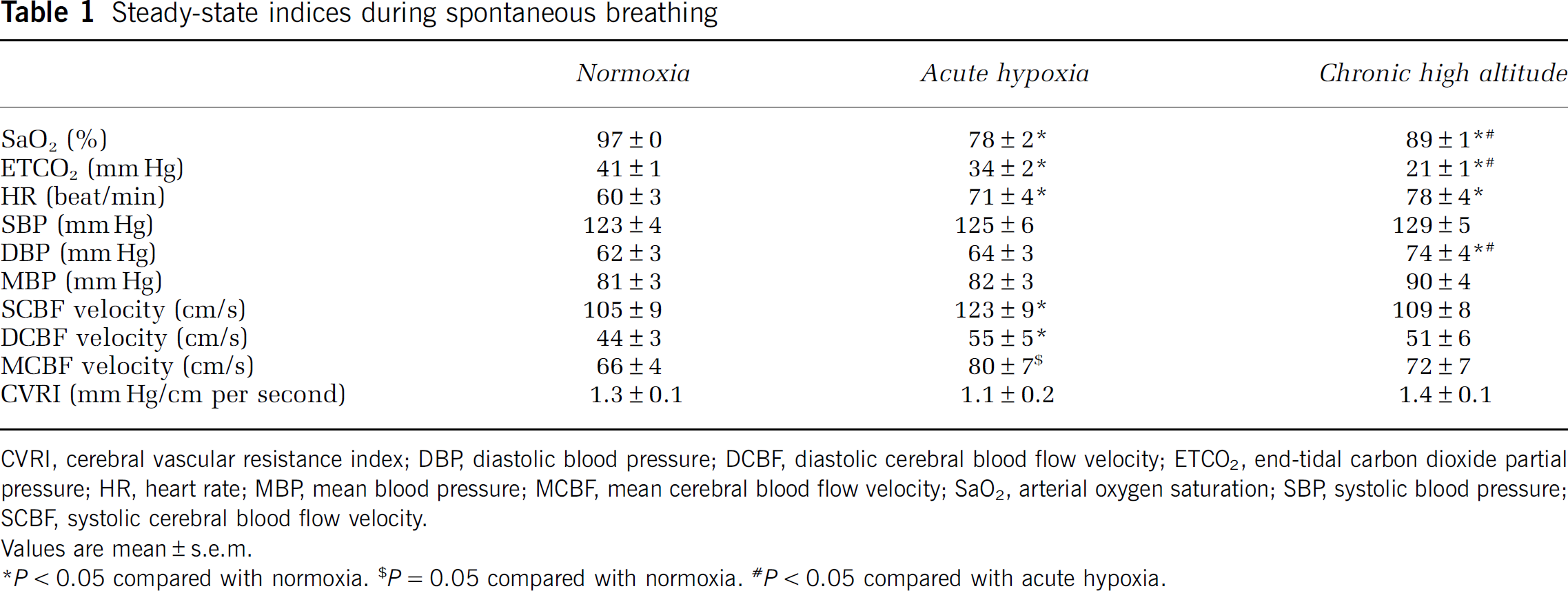
CVRI, cerebral vascular resistance index; DBP, diastolic blood pressure; DCBF, diastolic cerebral blood flow velocity; ETCO2, end-tidal carbon dioxide partial pressure; HR, heart rate; MBP, mean blood pressure; MCBF, mean cerebral blood flow velocity; SaO2, arterial oxygen saturation; SBP, systolic blood pressure; SCBF, systolic cerebral blood flow velocity.
Values are mean ± s.e.m.
Steady-state heart rate increased significantly during acute hypoxia and chronic high altitude (ANOVA,
Dynamic Cerebral Autoregulation
Group-averaged frequency analysis indices during spontaneous breathing are presented in Table 2. Very-low-frequency power of MBP variability increased significantly during acute hypoxia (468% ± 162%) (ANOVA,
Frequency analysis indices during spontaneous breathing
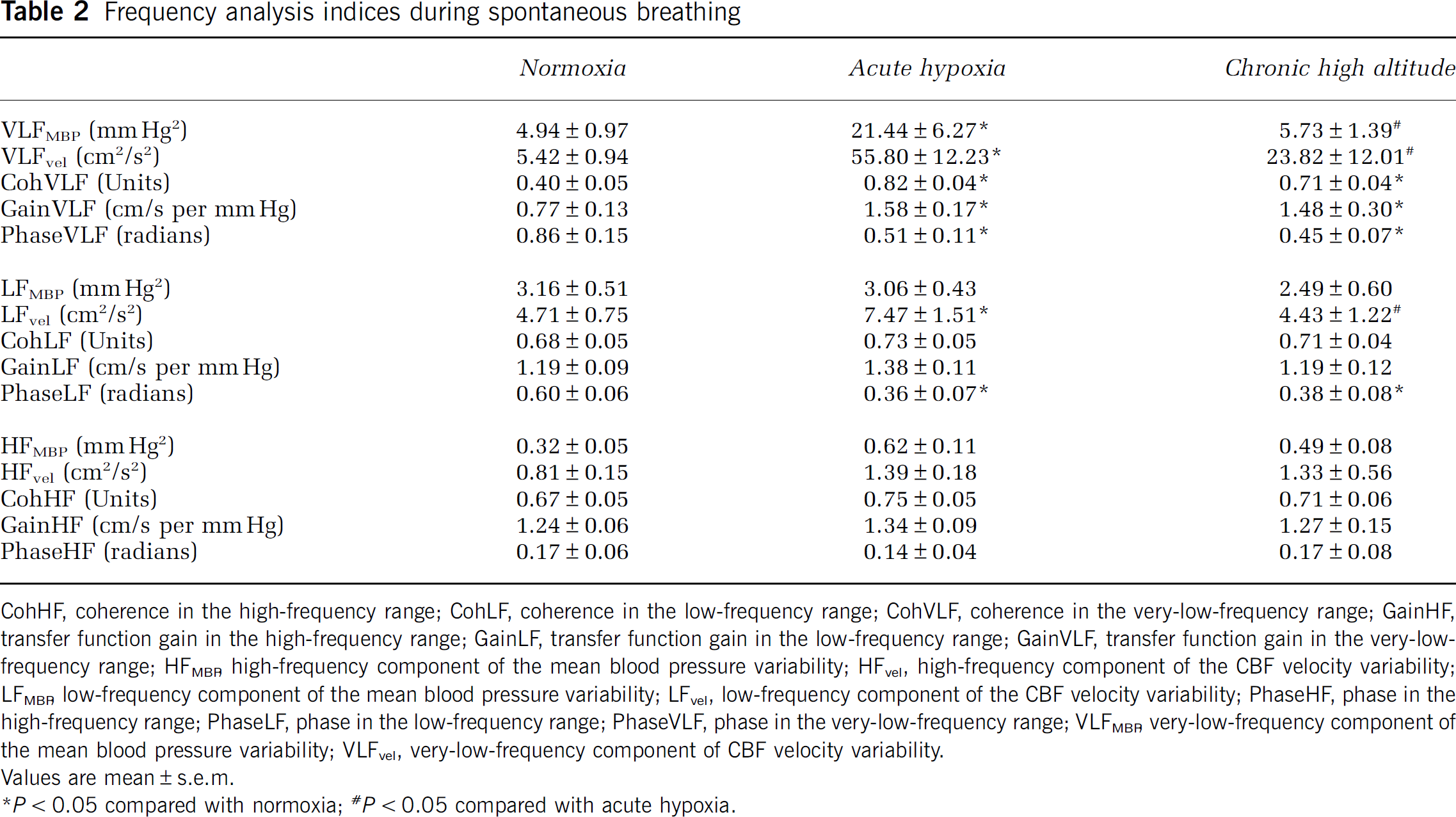
CohHF, coherence in the high-frequency range; CohLF, coherence in the low-frequency range; CohVLF, coherence in the very-low-frequency range; GainHF, transfer function gain in the high-frequency range; GainLF, transfer function gain in the low-frequency range; GainVLF, transfer function gain in the very-low-frequency range; HFMBR high-frequency component of the mean blood pressure variability; HFvel, high-frequency component of the CBF velocity variability; LFMBR, low-frequency component of the mean blood pressure variability; LFvel, low-frequency component of the CBF velocity variability; PhaseHF, phase in the high-frequency range; PhaseLF, phase in the low-frequency range; PhaseVLF, phase in the very-low-frequency range; VLFMBR very-low-frequency component of the mean blood pressure variability; VLFvel, very-low-frequency component of CBF velocity variability. Values are mean ± s.e.m.
Coherence in the very-low-frequency range was < 0.5 during normoxia; however, this index increased significantly to > 0.5 during acute hypoxia (136% ± 29%) and chronic high altitude (131% ± 50%) (ANOVA,
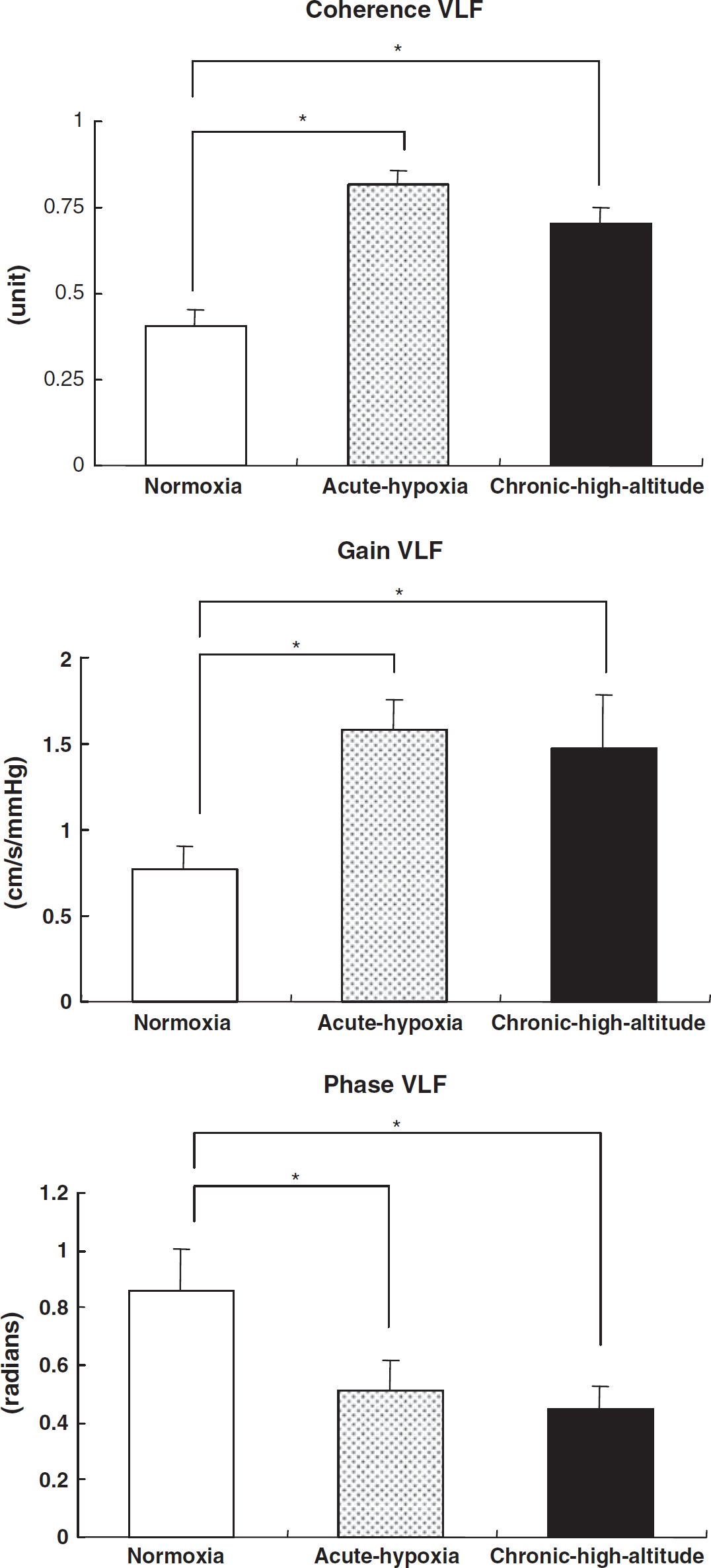
Mean changes (± s.e.m.) in coherence, transfer function gain (Gain), and phase between mean blood pressure (MBP) and mean cerebral blood flow velocity (MCBF) variabilities in the very-low-frequency range (VLF). The open bars identify values for normoxia (21% FIO2). The dotted bars identify values for acute exposure to normobaric hypoxia (10.5% FIO2). The solid bars identify values for chronic high altitude. *
There were no significant changes of any of the indices in the low- and high-frequency ranges, except for the low-frequency power of MCBF variability and phase in the low-frequency range.
The fixed-frequency breathing at 0.05 Hz induced sufficiently high coherence in the very-low-frequency range to ensure the reliability of the gain calculation under all three conditions (namely sea level, 0.62 ± 0.05; acute hypoxia, 0.81 ± 0.04; and chronic high altitude, 0.76 ± 0.04 unit), but did not change the pattern of increases in gain after either acute or chronic hypoxia (sea-level gain, 1.08 ± 0.13; acute hypoxia, 1.53 ± 0.18; chronic high altitude, 1.36 ± 0.16 cm/s per mm Hg). During this very slow, fixed-frequency breathing, there was a similar pattern of reduction in ETCO2 as was observed during spontaneous breathing: sea level, 43 ± 2; acute hypoxia, 36 ± 1; chronic high altitude,
Discussion
The major findings in this study were as follows: (1) Acute exposure to severe hypoxia led to increased steady-state CBF velocity and deterioration in dynamic cerebral autoregulation, manifested by increased coherence and gain of transfer function between MBP and MCBF within the optimal autoregulatory frequency range. Cerebral blood flow was more tightly coupled to arterial blood pressure than it was under normoxic conditions, with increased fluctuations in CBF induced by changes in arterial pressure. (2) After successful acclimatization to a high altitude of 5,260 m including near normalization of arterial oxygen content and despite normalization of steady-state CBF velocity, dynamic autoregulation remained remarkably impaired. Thus, we were forced to reject our hypothesis: acclimatization to an extreme high altitude did not normalize the dynamic pressure–flow relationship in the cerebral circulation altered by severe hypoxia.
Cerebral Blood Flow at Altitude
During the initial phase of exposure to high altitude, acute hypobaric hypoxia dilates cerebral arterioles, overriding the vasoconstrictive effect of hyperventilation-associated hypocapnia. During a prolonged stay at altitude, hypocapnia progresses and hypoxia is ameliorated by increased hemoglobin concentration and restoration in oxygen saturation (Brugniaux et al, 2007). These changes with acclimatization theoretically may reverse cerebral vasodilation. Previous studies have shown that CBF increases within the first few days after arrival at high altitude, followed by a gradual decrease within several days (Severinghaus et al, 1966; Huang et al, 1987). Furthermore, a previous report from the present research project in the same subjects showed that directly measured CBF using the Kety–Schmidt technique 3 weeks after arrival at 5,260 m was not different from sea level (Møller et al, 2002), confirming our findings in this study with transcranial Doppler. In addition, other factors have been proposed to alter cerebral arterioles in hypoxia, including sympathetic nerve activity or endothelium-derived factors at altitude (Brugniaux et al, 2007). Thus, the extent of change in cerebral arterioles at altitude is dependent on the balance between vasodilation and vasoconstriction. These vasomotor changes may also alter the dynamic regulation of CBF, which has not been extensively studied at altitude.
Transfer Function Analysis of Dynamic Autoregulation
In this study, we used transfer function analysis to quantify the dynamic relationship between arterial blood pressure and CBF. Previous studies using transfer function analysis between MBP and MCBF variabilities have shown frequency-dependent properties of the pressure–flow relationship in the cerebral circulation (Edwards et al, 2002; Giller, 1990; Kuo et al, 1998; Muller and Marziniak, 2005; Panerai et al, 1999; Zhang et al, 1998, 2002). The dynamic pressure–flow relationship has consistently shown a pattern like a high-pass filter. That is, coherence and gain are generally lower, and phase is higher at lower-frequency ranges, indicating that dynamic cerebral autoregulation is more effective with slower fluctuations in arterial pressure. Estimates of coherence function, which evaluate the linear relationships between these two variables, are generally < 0.5 in the very-low-frequency range (< 0.07 Hz) during supine rest (Edwards et al, 2002; Kuo et al, 1998; Muller and Marziniak, 2005; Ogawa et al, 2006; Zhang et al, 1998, 2002), signifying little linear correlation or dependence of CBF on arterial blood pressure (i.e., effective autoregulation).
The Effect of Hypoxia and High-Altitude Acclimatization
In this study, acute exposure to hypoxia increased steady-state CBF velocity, and also increased coherence significantly to > 0.5 in the very-low-frequency range, indicating augmented dependence of CBF on arterial blood pressure and impaired autoregulation. Moreover, transfer function gain increased significantly in this range, suggesting greater changes in CBF for any given change in arterial blood pressure. These results suggest that the buffering capacity of the cerebral circulation is reduced by severe hypoxia and are consistent with a previous report showing similar deterioration of cerebral autoregulation with acute exposure to even mild hypoxia (Iwasaki et al, 2007a). Somewhat more controversial, the change in phase also supports the interpretation of reduced buffering capacity of the cerebral autoregulation (Panerai, 2009). Although there are questions about the interpretation, reliability, and sensitivity of each transfer function index (Panerai, 2009; Subudhi et al, 2009), this is the first study that shows consistent, significant, and directionally appropriate alterations in all three indices of the transfer function analysis providing strong evidence of impaired dynamic cerebral autoregulation by acute severe hypoxia.
Despite significant changes in coherence, gain, and phase in the very-low-frequency range, these indices remained unchanged at higher frequencies at which autoregulation is typically less effective (Zhang et al, 1998). Thus, the frequency-dependent ‘high-pass filter’ nature of the dynamic pressure–flow relationship in the brain was abolished by acute hypoxia (Figure 3), suggesting that oscillations in arterial blood pressure at all frequencies were readily transmitted as fluctuations in CBF velocity.
Unexpectedly, and contrary to our hypothesis, the pressure–flow relationship observed during chronic exposure to high altitude was very similar to that during acute hypoxia (Figure 3). The similarity observed in all three indices between both conditions, suggests that dynamic cerebral autoregulation may be impaired by hypoxia even after successful acclimatization to a high altitude that restores arterial oxygen content and normalizes CBF and resistance. Thus, factors induced by hypoxia other than resistance vessel vasodilation might alter dynamic cerebral autoregulation after acclimatization. For example, dynamic cerebral autoregulation in the very-low-frequency range may be partly modulated by the autonomic nervous system (Zhang et al, 2002), although there has been much debate about the effects of sympathetic activity on CBF (Strandgaard and Sigurdsson, 2008; Levine and Zhang, 2008). Sympathetic neural activity to the skeletal muscle increases to extraordinarily high levels, even in well-acclimatized subjects, as shown by Hansen and Sander (2003) in these same subjects, although it is less certain about regional autonomic activity in the brain itself. In the face of such intense sympathetic hyperactivity, the autonomic nervous system may not function appropriately to regulate cerebral vascular resistance, although moderate sympathetic hyperactivity may have protective effects against impairment of cerebral autoregulation. Alternatively, both hypoxia and sympathetic activity may alter endothelial and/or neural nitric oxide synthase activity, thus altering vascular responsiveness (Brugniaux et al, 2007). A myriad of other hypoxia-induced changes (such as various autacoids and cytokines, endothelin, adenosine, angiogenesis, blood viscosity) that influence CBF may alter dynamic cerebral autoregulation as recently elegantly reviewed by Ainslie and Ogoh (2010). It is also possible that reduced stroke volume which is known to occur with chronic altitude acclimatization could alter cerebral vascular pulsatility and thereby alter the dynamic pressure–flow relationship (Zhang and Levine, 2007).
The cerebral circulation is exquisitely sensitive to arterial partial pressure of CO2 (PaCO2), and hypocapnia by itself is reported to decrease coherence and transfer function gain (Muller et al, 2003), as well as increase phase in the lower-frequency ranges (Birch et al, 1995; Edwards et al, 2002). Thus, hypocapnia could in theory counteract the changes in coherence, gain, and phase observed in this study. However, even greater and progressive hypocapnia after acclimatization did not normalize these three indices compared with the acute phase. Thus, the protective effect of hypocapnia is likely to be less important than the inhibitory effect of hypoxia on the dynamic relationship between arterial blood pressure and CBF at altitude. This possibility is supported by the findings in mild hypoxia (Nishimura et al, 2010).
There have been a few studies that investigated dynamic cerebral autoregulation at high altitude, although these studies mostly reported the changes before the acclimatization process was complete. For example, Ainslie et al (2008) investigated dynamic cerebral autoregulation 1 to 2 days after arrival at high altitude (∼5,400 m) by transfer function analysis. Their findings of increased transfer function gain (but not coherence) in the low- and very-low-frequency ranges at high altitude are roughly consistent with our results. Subudhi et al (2010) also reported impairment of dynamic cerebral autoregulation after 4 to 9 hours of hypobaric hypoxia (≈4,875 m) by transfer function analysis. Conversely, Van Osta et al (2005) investigated the effects of altitude on dynamic cerebral autoregulation by a thigh cuff method. They found no difference between sea level and altitude of 4,559 m on dynamic cerebral autoregulation, which is inconsistent with the present findings. However, these discrepancies may be explained by differences in analysis techniques and particularly the frequency range in question. In particular, Van Osta et al (2005) estimated dynamic autoregulation using an index calculated from quick restoration of CBF to baseline (normal time at rest, 5 seconds) after a transient decrease in blood pressure. The time scale of their estimates for dynamic autoregulation was similar to that of the transfer function indices in the low- and high-frequency ranges in this study, which also did not change. Thus, on careful inspection, their results are actually quite similar to those reported herein.
Clinical Implications
The impairment in cerebral autoregulation could be a significant risk factor for the development of AMS or high-altitude cerebral edema. However, a direct relationship between impaired dynamic cerebral autoregulation and AMS remains speculative and unproven. Previous reports have been controversial regarding the relationship between AMS and cerebral autoregulation: related in some studies (Bailey et al, 2009; Van Osta et al, 2005) and unrelated in others (Subudhi et al, 2010). Given the vigorous health of our subjects despite impaired dynamic autoregulation, it could even be hypothesized that this impairment might be adaptive to the hypoxic environment or completely independent of altitude pathology. Conversely, impaired cerebral autoregulation could result in overperfusion of capillaries and vasogenic cerebral edema (Hackett and Roach, 2001). For example, when large and rapid increases in blood pressure occur such as during exercise, impaired cerebral autoregulation may leave the brain relatively unprotected from increases in arterial blood pressure. A similar pathophysiology has been shown by Heistad and Marcus (1979) who increased blood pressure in cats by peripheral infusion of norepinephrine, and protected only one side of the brain with electrically induced sympathetic vasoconstriction; the unprotected side developed severe cerebral edema which may represent the consequence of a ‘functional sympatholysis’ induced by hypoxia leading to impaired autoregulation. Although acclimatization is generally considered protective against AMS, it is also clear that even acclimatized individuals remain at risk if they ascend too high, too fast—perhaps because cerebral autoregulation never returns to normal at very high altitudes.
Limitations
Transcranial Doppler ultrasonography was used for noninvasive and beat-to-beat estimation of CBF in this study. This approach is based on the assumption that the diameter of the insonated artery changes minimally and CBF velocity is directly proportional to CBF. Therefore, the results obtained by this technique should be interpreted with caution. However, a constant diameter in the proximal segment of the middle cerebral artery has been confirmed during hypoxia (Poulin and Robbins, 1996), hypocapnia with hyperventilation (Serrador et al, 2000; Valdueza et al, 1997), hypotension and hypertension (Larsen et al, 1994), or various stimuli (Aaslid et al, 1991; Giller et al, 1993; Serrador et al, 2000). Moreover, the reliability of CBF velocity measured in the middle cerebral artery by transcranial Doppler has been confirmed to be excellent (Brodie et al, 2009). Perhaps most importantly, absolute CBF was actually directly measured in these same subjects using the Kety–Schmidt technique confirming no change in CBF (or cerebral oxidative metabolism) between baseline and chronic acclimatization to 5,260 m, although no such quantitative data are available for acute hypoxia (Møller et al, 2002).
In this study, tonometry was used for noninvasive and beat-to-beat estimation of arterial blood pressure. Although the reliability of this blood pressure measurement has not been examined at high altitudes, the accuracy of this device has been confirmed by comparisons with tonometric and intraarterial pressure monitors under a wide range of arterial blood pressures (Kemmotsu et al, 1991).
Another limitation of this study is using ETCO2 that is not perfectly equivalent to PaCO2. It is possible that we overestimated the decreases in ETCO2. A previous report from the present research project showed that PaCO2 was 25 mm Hg 3 weeks after arrival at 5,260 m (Møller et al, 2002). In addition, a previous report showed that PaCO2 was 24.3 mm Hg 1 to 2 days after arrival at 5,400 m (Ainslie et al, 2008), with another study by the same investigators showing that PaCO2 was 23.7 mm Hg at 3,840 m after a 9-day ascent to altitudes > 5,000 m (Ainslie et al, 2007). Thus, shorter or lower altitude exposure than that reported in this study induced a PaCO2 of at least 25 mm Hg. These observations provide evidence that a PaCO2 in the range of the 21 mm Hg reported in this study is reasonable after prolonged acclimatization to an extreme high altitude such as in this study.
Conclusions
During acute hypoxia, steady-state CBF velocity increased, whereas indices of dynamic regulation of cerebral perfusion deteriorated suggestive of impaired cerebral autoregulation. As expected, steady-state CBF velocity was normalized associated with intense hypocapnia and partial restoration of SaO2 during chronic exposure to an extreme high altitude. However, contrary to our hypothesis, dynamic cerebral autoregulation remained grossly abnormal. The similarity between acute and chronic exposure to hypoxia suggests that dynamic cerebral autoregulation remains impaired with chronic hypoxia even after successful acclimatization to an extreme high altitude.
Footnotes
Acknowledgements
We thank Rob Roach from the University of Colorado, and Bengt Saltin from the Copenhagen Muscle Research Center for all their support.
The authors declare no conflict of interest.
