Abstract
Oxygen Extraction Fraction (OEF) is a critical measure of a tissue’s metabolic state post-ischemic stroke. This study investigated OEF changes in stroke-affected tissue compared to healthy tissue, post-reperfusion. OEF maps generated from gradient echo MRI images of 87 ischemic stroke patients at three time points after successful Endovascular Thrombectomy (EVT) were analysed in a prospective longitudinal multicentre study. Regions of interest (ROIs) delineating the infarct areas and corresponding mirror regions were drawn. The MR-derived OEF index values were obtained from the ROIs and compared using Wilcoxon signed rank tests. The cross-sectional comparison of OEF index values revealed lower values in the infarct areas than the corresponding contralateral areas at all three time points after successful EVT, presented as median (interquartile range) [24–72 hours: 20.84 (17.56–26.82)% vs 27.56 (23.22–31.87)%; 3 months: 27.37 (23.28–30.35)% vs 32.55 (28.00–35.81)%; 12 months: 24.38 (22.35–29.77)% vs 29.39 (25.86–34.04)%, p < 0.001 for all three time points]. Longitudinally, relative OEF index values increased gradually over time [24–72 hours: 0.81 (0.67–0.87); 3 months: 0.86 (0.79–0.95); 12 months: 0.88 (0.75–0.95)]. The findings revealed that following successful EVT, OEF in infarct tissue improves over time, indicating potential tissue recovery.
Trial registration name and URL: Post-Reperfusion Pathophysiology in Acute Ischemic Stroke https://trialsearch.who.int/Trial2.aspx?TrialID=ACTRN12624000629538
Keywords
Introduction
Cerebral Oxygen Extraction Fraction (OEF) is defined as the fraction of oxygen that is extracted by cerebral tissue from arterial blood. Cerebral OEF is a critical physiological parameter of brain tissue viability and energy metabolism.1,2
Ischemic stroke is caused by the sudden interruption of blood flow to a region of the brain. This sudden reduction in blood flow shuts the oxygen supply to the affected tissue, precipitating a cascade of events that lead to cell damage and cell death in the absence of prompt reperfusion. In the initial stages of the occlusion, the OEF in the endangered but salvageable ischemic tissue is elevated as a compensatory mechanism in response to reduced cerebral blood flow (CBF) to maintain the tissue’s energy consumption. However, when prolonged occlusion without reperfusion occurs, the ischemic stroke cascade progresses in the severely hypoperfused regions (CBF between 10 and 20 mL/100 g/min) leading to irreversible damage. The ability of the tissue to extract oxygen reduces in those regions, leading to a reduction in the OEF.3 –5 However, in those regions where the reduced CBF is above the threshold of infarction, OEF may remain elevated. The variation in tissue response in the affected area leads to heterogeneous tissue damage.6,7
Successful treatment of ischemic stroke involves restoring cerebral perfusion to the affected region by mechanically removing the thrombus with endovascular thrombectomy (EVT) or pharmacologically through intravenous thrombolysis, or a combination of both. EVT is the standard treatment for large-vessel occlusions. 8 EVT is performed to salvage tissue that is still viable, though electrically inactive.
Ischemic stroke imaging employs various MRI and CT techniques to determine the extent of damage and tissue that can still be salvaged. By complementing anatomical imaging with OEF imaging, clinicians can gain additional functional insight into underlying pathology in stroke-affected tissue that may not be apparent from anatomical imaging alone. This could provide an opportunity to design therapeutic strategies for individual patient profiles based on real-time assessment of metabolic status.
Positron Emission Tomography (PET) is the gold standard for OEF imaging; however, it is too complex to use in an acute care setting where time is a major factor, as in the case of ischemic stroke. Moreover, radiation exposure and logistic challenges, such as requiring a cyclotron on-site given the short half-life of 15O, limit its widespread adoption. Similarly, MRI is seldom performed acutely before thrombectomy but may be used post thrombectomy to assess tissue status. Several MRI-based methods have emerged to quantify OEF, based on the fact that when oxygen is extracted from the blood, oxyhemoglobin, which is diamagnetic, is converted to deoxyhemoglobin, which is paramagnetic. 2 Different MRI-based OEF quantification techniques have produced comparable results with PET.9,10
In the context of ischemic stroke research, OEF has been explored in a variety of settings, for example, penumbra detection,11,12 OEF changes in grey matter and white matter, 13 and its evolution through the different stroke phases.14,15 OEF within the infarct lesion was found to be lower in patients at the chronic stage of ischemic stroke, compared to the lesions of patients are earlier stages of ischemic stroke.14,15 However, these earlier retrospective studies included patients with variable chronicity, stroke sub-types and reperfusion treatment received. It is unclear whether the observed changes in OEF relates to persistent unresolved ischemia or the natural evolution of the OEF parameter in recovery cerebral tissue after ischemia. In addition, the heterogeneity of OEF values in infarct regions has been observed visually but not quantified in previous studies. 6
To investigate the nature of metabolic tissue changes after cerebral ischemia, the aim of this article was to prospectively explore the longitudinal changes in cerebral OEF in individual ischemic stroke patients with successful endovascular reperfusion at pre-specified and standardised timepoints.
We hypothesised that the infarct tissue has lower OEF values than the contralateral tissue owing to the existence of irreversibly damaged tissue within the infarct region. We also hypothesised that after successful EVT, there is a progressive increase and normalisation of OEF values within the ischemic tissue over time, reflecting the restoration of blood flow, tissue oxygenation and cellular metabolism.
By investigating the temporal changes in OEF values within ischemic tissue after patients have been successfully treated, we aim to contribute to the body of growing research on the underlying tissue changes in ischemic stroke-affected tissue, thereby providing insights that could guide future therapeutic strategies for these patients.
Materials and methods
Patient cohort
Eighty-seven patients with acute ischemic stroke with anterior circulation large vessel occlusion who received reperfusion treatment at two high volume comprehensive stroke centres were recruited to a prospective study carried out from 2018 to 2022 (post-reperfusion pathophysiology in ischemic stroke [PRAISE], Trial Registration Number - ACTRN12624000629538) in accordance with the Declaration of Helsinki. The Melbourne Health Human Research Ethics Committee approved the study on August 27th, 2018 (Melbourne Health Site Reference Number: 2018.247). Patients or caregivers provided informed and written consent before participating in this study. The patients or caregivers also consented to the results being published. All patient information was de-identified.
The PRAISE study performed longitudinal multimodal 3 T MRI scans at four pre-specified time points (under 12 hours, 24–72 hours, 3 months, and 12 months) after treatment. Our study focused on patient data at the three latter timepoints (24–72 hours, 3 months and 12 months) as the first time point (under 12 hours) imaging did not include the multi-echo gradient echo images required to generate the OEF images. Patients with successful EVT procedures determined by the modified Thrombolysis in Cerebral Infarction score (mTICI) of 2b, 2c and 3 were included in our study. Additionally, the patients’ presenting National Institutes of Health Stroke Scale (NIHSS) as well as the modified Rankin Scale (mRS) scores at 3 months post-reperfusion were obtained for our study.
MRI data acquisition and OEF maps generation
At each comprehensive stroke centre, patients were scanned on a 3 T Siemens Prismafit scanner; 80 patients at one centre, and 7 patients at the second. Diffusion-weighted imaging (DWI), fluid-attenuated inversion recovery (FLAIR), and multi-echo gradient echo (GRE) images using the following parameters: DWI-Resolve (b-value = 1000 s/mm2, Acquisition time =2 mins 19 secs, TR = 5100 ms, TE = 55 ms, Resolution =0.98 × 0.98 × 4 mm3), T2 FLAIR (Acquisition time =5 mins 4 sec, TR = 6700 ms, TE = 463 ms, TI = 2200 ms, FA = 120°, Resolution = 1 × 1 × 1 mm3), and multi-echo GRE (Acquisition time = 8 mins 4 sec, TR = 50 ms, 9 echoes TE = 5.84 ms–44.16 ms with a spacing of 4.79 ms, Pixel bandwidth = 310 Hz). The same protocol was used in both stroke centres.
OEF maps were obtained using a novel integrated Quantitative Susceptibility Mapping (QSM) and quantitative bold oxygenation level-dependent (qBOLD) method that utilises both phase and magnitude multi-echo GRE images to create OEF maps.
Briefly, the QSM term separates tissue susceptibility of any given ROI into non-blood tissue susceptibility and blood (mainly deoxyhemoglobin) susceptibility, with the latter being used in determining oxygen metabolism.16,17 The qBOLD term models the magnitude signal with the transverse relaxation rate (R2), signal intensity, signal decay due to the extravascular network, and the macroscopic field inhomogeneity.16,17 In the QSM term, the cerebral blood volume fraction is assumed to be linearly related to the cerebral blood flow (CBF) but is accurately measured in qBOLD; while in qBOLD, the non-tissue susceptibility is assumed to be constant throughout the brain, yet this is estimated in QSM. The two methods are combined to cater for the limitations in either method, hence the QQ term.
The QQ algorithm processes the images further using machine learning-based methods such as cluster analysis where voxels are classified according to tissue type (grey matter, white matter or cerebrospinal fluid) and their signal time evolution; and total variation regularization to reduce image noise. This OEF quantification method has been referred to as the QQ-CCTV method.16,18,19
Image processing
Regions of interest (ROIs) delineating the infarct region at each time point were manually drawn on DWI (24–72 hours) and FLAIR (3 and 12 months) by an expert neuroradiologist with over 30 years’ experience using ITK-SNAP software. 20 Mirror ROIs in the contralateral hemisphere were delineated automatically and manually verified. Figure 1 shows representative DWI images and OEF map of a patient, with the DWI containing the delineated infarct and contralateral ROIs.
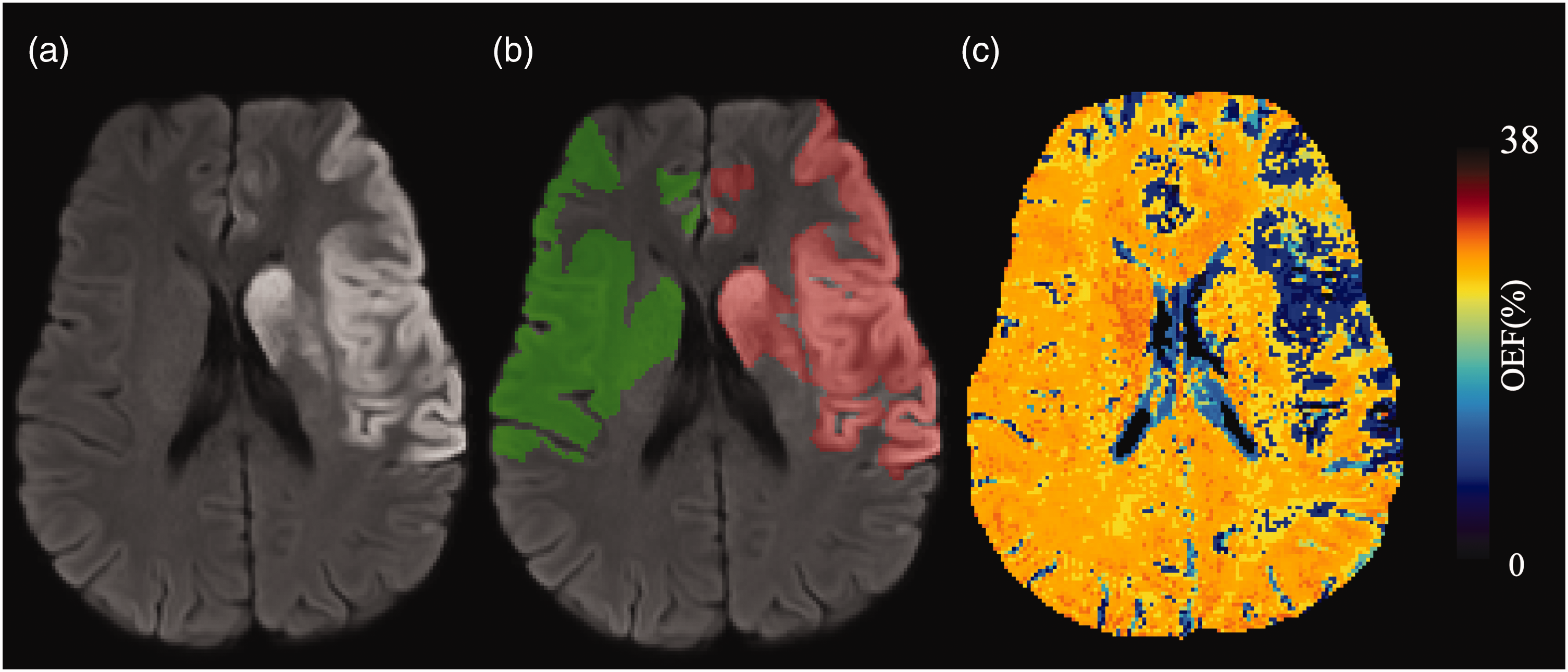
Representative DWI images without (a) and with ROI shading (b), and OEF (c) images of a 37-year-old female with a left MCA occlusion, presenting with NIHSS of 18, and complete reperfusion (mTICI 3). The red and green ROIs represent the infarct and contralateral tissue, respectively.
The regions of haemorrhage transformation and cerebrospinal fluid (CSF) were excluded from the ROIs at each time point using an FSL generated mask. The mask was created by identifying hypointense regions within the infarct region of the GRE magnitude image of the 6th echo that indicated regions of haemorrhage.
Once the haemorrhage was identified, they were manually delineated and subtracted from the infarct ROI at all time points using command line functions from the FMRIB software library (FSL).
The final ROIs with CSF and haemorrhages removed were co-registered to the OEF space using FSL’s FLIRT function.21,22 The ROIs were then overlaid onto the OEF maps at each time point, and the OEF index values were extracted using the FSL command line functions.
Heterogeneity analysis
We quantified the heterogeneity of the OEF index values in infarct and contralateral ROIs using first-order statistical analysis of grey-level histograms. Patient images were processed using the neurobase package in R to obtain histograms of the OEF index values in the infarct and contralateral ROIs. Heterogeneity of the OEF index values in the infarct and contralateral ROIs was then quantified by calculating the mean standard deviation and mean skewness using the moments package in R. Figure 2 is a display of histograms of the OEF index values of the infarct and contralateral ROIs at the three time points obtained from one patient.
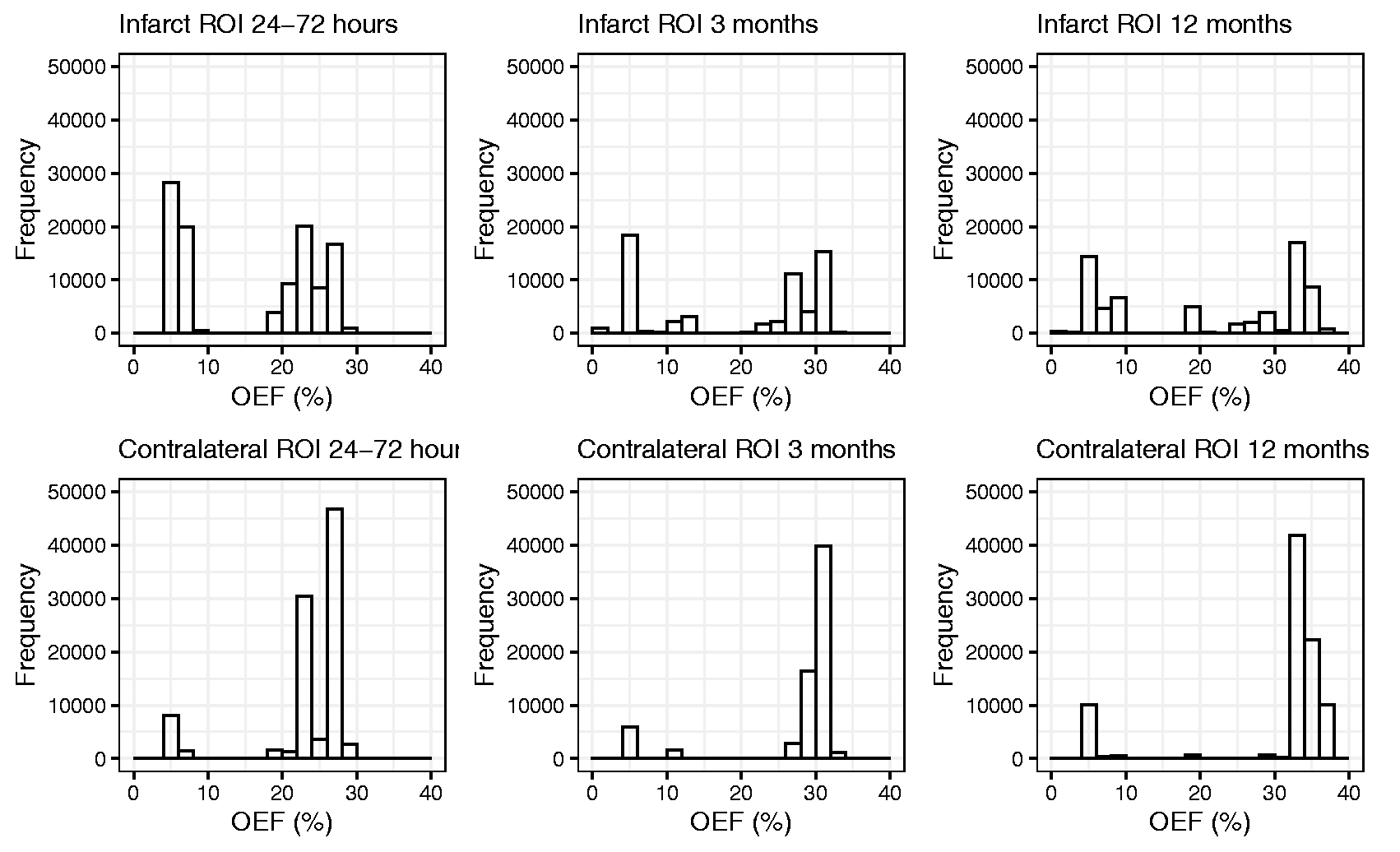
Distribution of OEF values within the different ROIs drawn on the OEF images of a 58-year-old male patient with a left MCA occlusion, presenting NIHSS 18, and an mTICI score 2 b.
Standard deviation quantifies the dispersion of values in relation to the mean. In our study, standard deviation quantified the dispersion of OEF index values within an ROI in relation to the mean OEF. Thus, a higher standard deviation indicated that there were more heterogeneous OEF index values within the ROI as the OEF index values were spread out over a wider range, varying significantly from one point to another, while a lower standard deviation indicated more homogeneous OEF index values within the ROI.
Skewness measures the symmetry of data distribution as observed on a histogram and indicates the direction of deviation from normal distribution. A skewness value less than 0 implies that the histogram is negatively skewed (the tail of the distribution is on the left). In this study, a more negative skewness measure indicated the tendency of OEF index values to cluster at the higher end of the OEF spectrum, closer to normal tissue ranges. If skewness greater is than 0, the histogram is positively skewed (the tail of the distribution is on the right). In this study, a more positive skewness measure indicated the tendency of OEF index values to cluster towards the lower end of the OEF spectrum. A skewness of 0 on the other hand implies a normal distribution. 23
Statistical analysis
Based on the normality of data, which was assessed by the Shapiro-Wilk test, continuous variables were expressed as mean ± SD or median (interquartile range). Statistical analysis was performed using IBM SPSS version 28.0.1.1 and R version 4.3.2.
The analysis was divided into two categories: analysis done on the whole dataset and analysis done on patients with complete imaging data at all three time points.
For the whole dataset, a cross-sectional analysis of the OEF index values in the infarct and contralateral ROIs was performed using the Wilcoxon signed-rank test. Standard deviation and skewness were also compared cross-sectionally across the infarct and contralateral ROIs using the Wilcoxon signed rank test.
The dataset of patients with complete imaging data at all three time points was analysed longitudinally using relative OEF index values, calculated as the ratio of the OEF index values in the infarct ROI to that in the contralateral ROI as well as absolute OEF index values. Absolute skewness and standard deviation values in this dataset were also compared longitudinally in the infarct ROIs, and in the contralateral ROIs. The Wilcoxon signed-rank test was used to perform both analyses.
An ordinal logistic regression analysis was performed to explore the association between infarct OEF index values at 24–72 hours and the mRS at 3 months using the polr function in R.
For all tests, statistical significance was set at P < 0.05.
The data used in this study is available from the corresponding author upon reasonable request.
Results
Patient characteristics
Among 87 patients in the PRAISE study, seven patients did not proceed to EVT after achieving significant clinical recovery after bridging intravenous thrombolysis, and four patients had unsuccessful reperfusion at EVT (mTICI <2 b). Fourteen patients were excluded due to suboptimal OEF image quality that could affect quantitative analysis or incomplete images required for analysis. In total, 62 patients with at least one time point of analysable OEF images were included in the current study. There were 53 patients who had analysable imaging data at 24–72 hours, 40 at 3 months, and 33 at 12 months. There were 23 patients with complete imaging data at all time points (Figure 3).
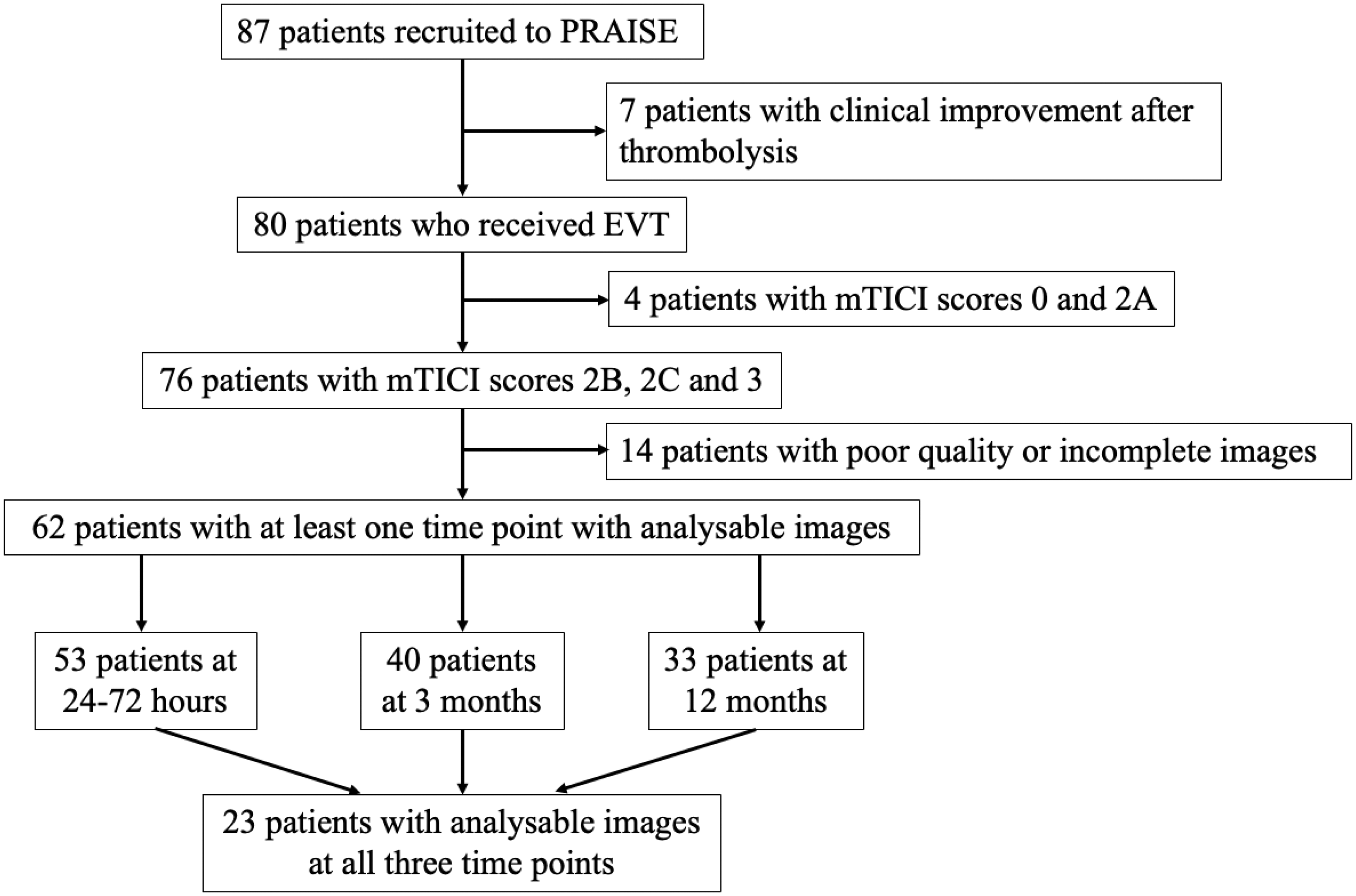
Patient selection flowchart.
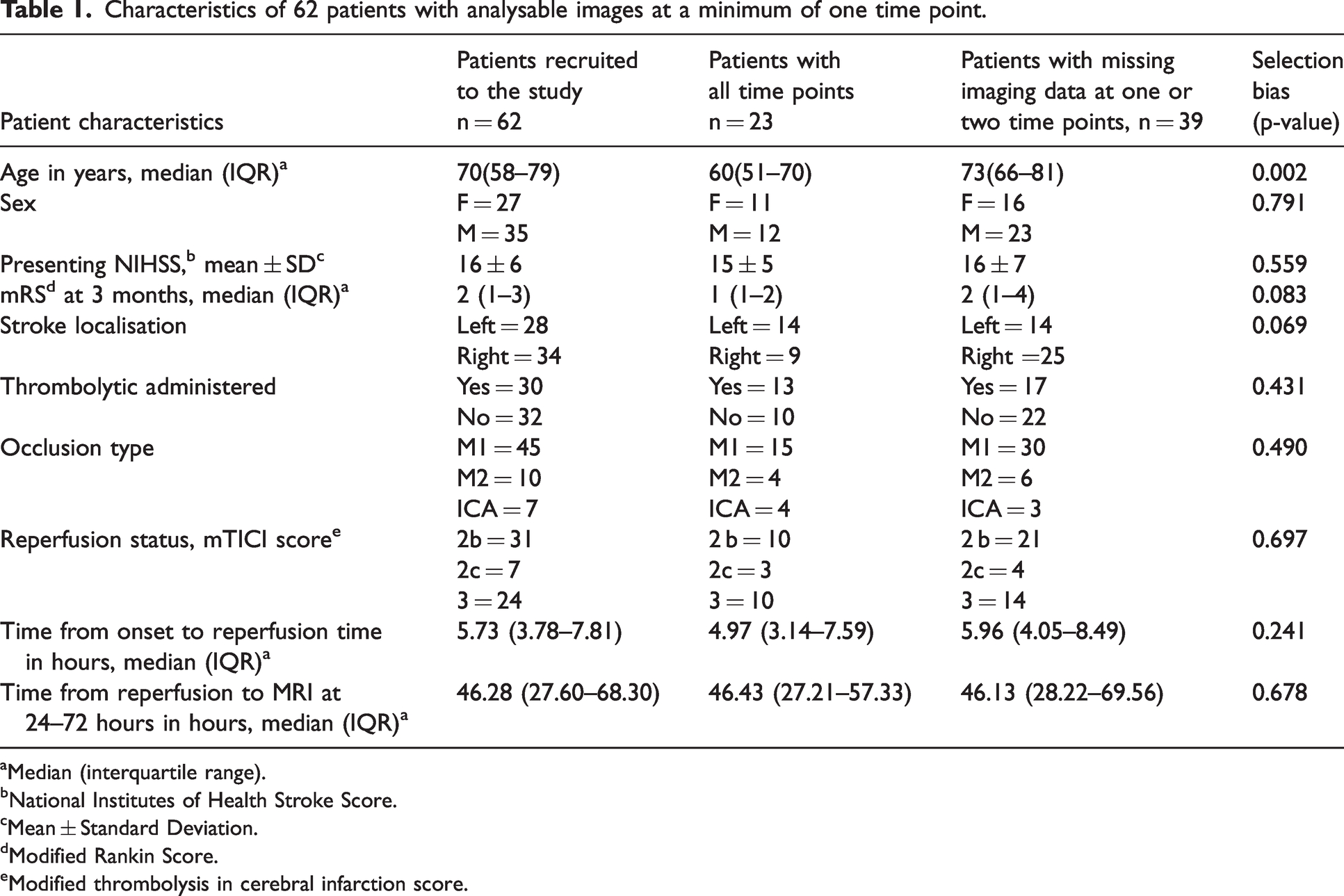
Table 1 shows the characteristics of the 62 patients who met the selection criteria and the 23 patients with complete imaging data at all three time points. Mann-Whitney U tests and Fisher’s exact tests were performed to assess for any difference in characteristics between the 23 patients with complete imaging data at all time points and the 39 patients with missing imaging data at one or two time points. The results show no statistical differences in all the patient characteristics except patient age, between the 23 patients with complete imaging data at all time points and the 39 patients with missing imaging data at one or two time points.
Characteristics of 62 patients with analysable images at a minimum of one time point.
Median (interquartile range).
National Institutes of Health Stroke Score.
Mean ± Standard Deviation.
Modified Rankin Score.
Modified thrombolysis in cerebral infarction score.
Cross sectional analysis of the whole dataset (62 patients)
The mean values of the OEF index, standard deviation and skewness are recorded in Table 2, and illustrated in Figure 4(a), (b) and (c) respectively. Cross-sectional comparison revealed that at each of the three time points, the OEF index values of the infarct ROIs were significantly lower than those of the contralateral ROIs (all p < 0.001)
Mean OEF, standard deviation and skewness values from infarct and contralateral ROIs for 53 patients at 24–72 hours, 40 patients at 3 months, and 33 patients at 12 months.
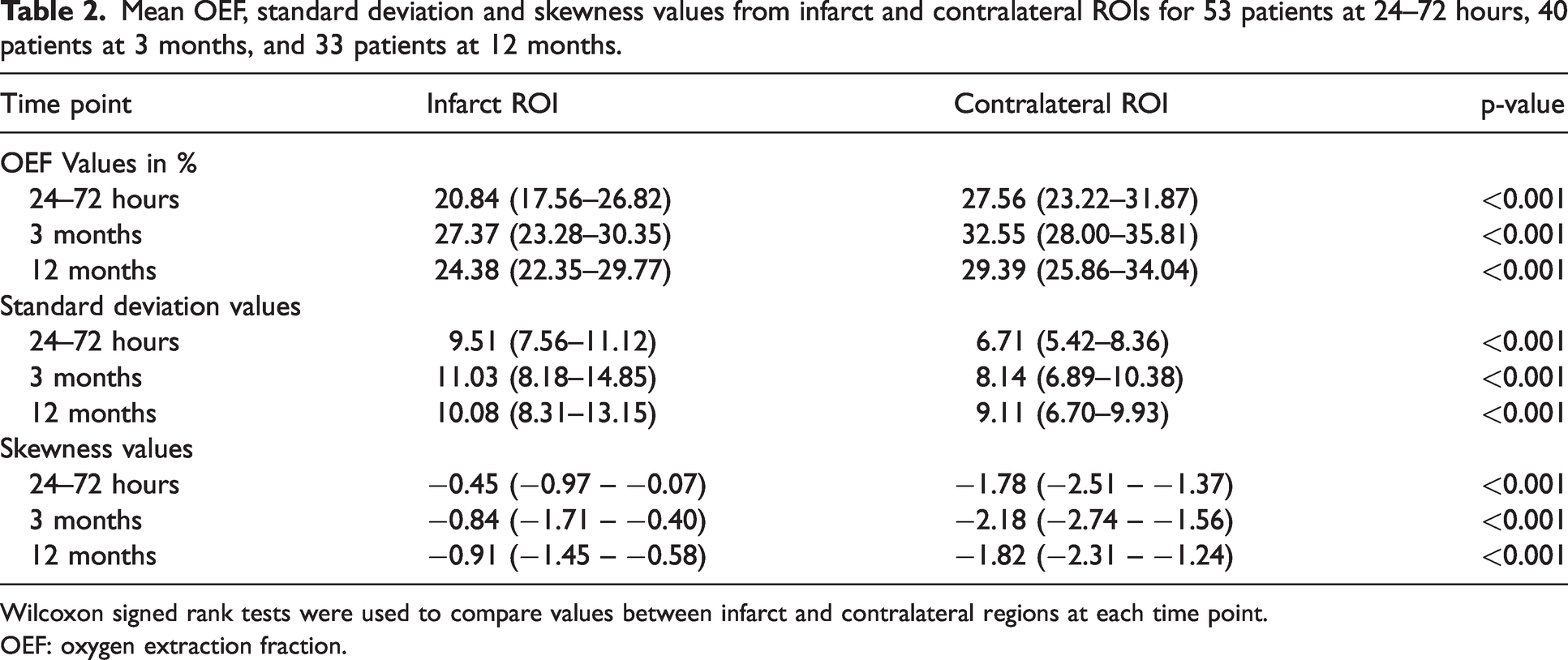
Wilcoxon signed rank tests were used to compare values between infarct and contralateral regions at each time point.OEF: oxygen extraction fraction.
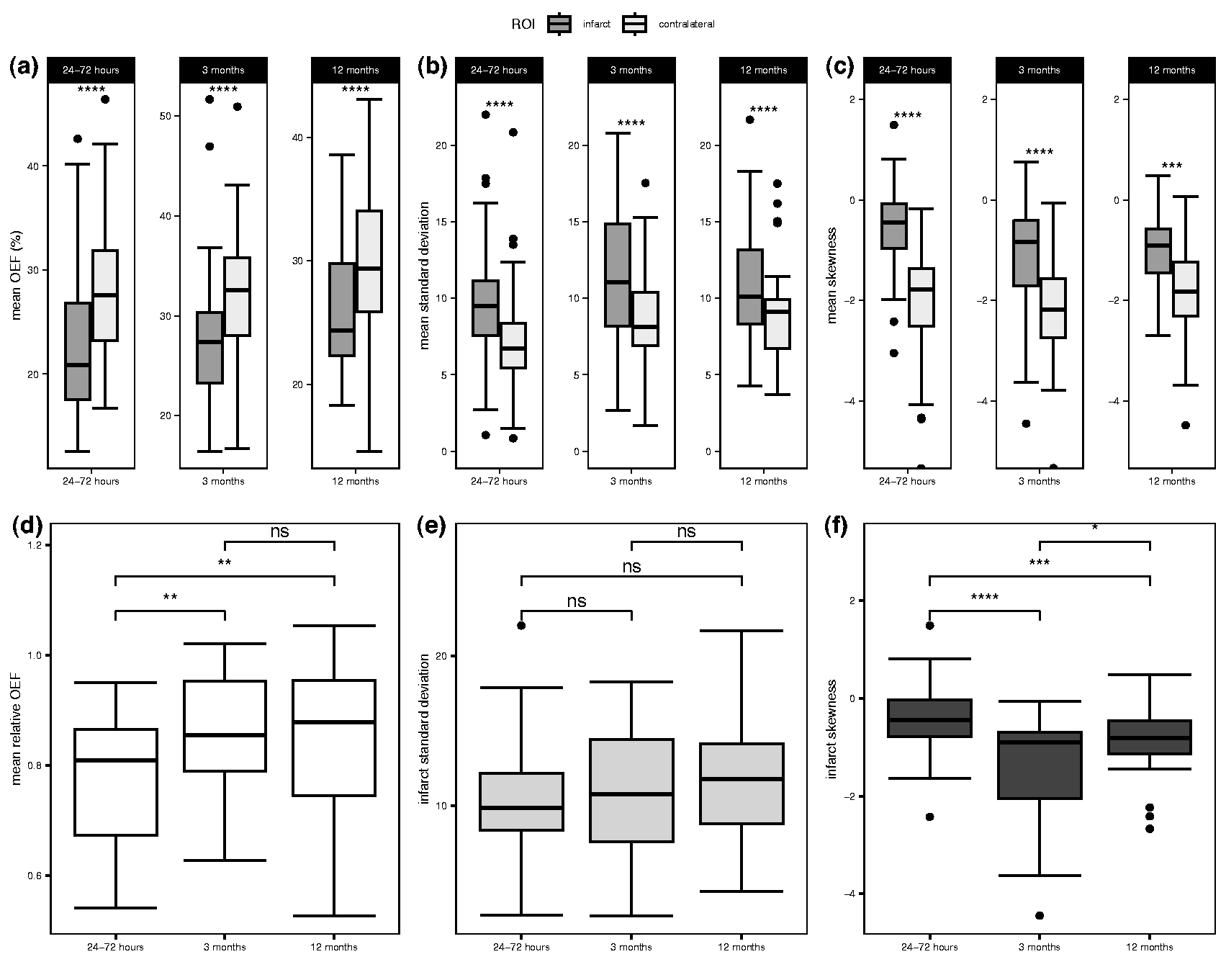
Cross-sectional and longitudinal analysis of OEF, mean standard deviation and mean skewness.
The standard deviation of OEF index values in the infarct ROIs was significantly higher than those in the contralateral ROIs at all three time points (p < 0.001 for all). The skewness values in the infarct ROIs on the other hand were less negatively skewed (closer to a normal distribution) than those in the contralateral ROIs (p < 0.001 for all).
Longitudinal analysis of the 23 patients with complete imaging data at all time points
The relative mean OEF index values generally increased from the 24–72 hours’ time point to the 12 months’ time point, with the Wilcoxon signed-rank test analysis revealing a statistically significant difference between 24–72 hours and 3 months (p = 0.009), 24–27 hours and 12 months (p = 0.006), but not between 3 months and 12 months (p = 0.760).
The absolute infarct OEF index values increased significantly between 24–72 hours versus 3 months (p = 0.004), as well as 24–72 hours versus 12 months (p = 0.023). There were no differences in absolute infarct OEF index values between 3 months versus 12 months (p = 0.484). The absolute contralateral OEF index values increased significantly from 24–72 hours versus 3 months (p = 0.033), but not between 24–72 hours versus 12 months (p = 0.223) and 3 months versus 12 months (p = 0.429).
The relative and absolute OEF index values for this analysis are tabulated in Table 3.
Relative OEF, Absolute OEF, standard deviation and skewness of OEF values from the infarct and contralateral ROIs for the 23 patients with complete imaging data at all time points.
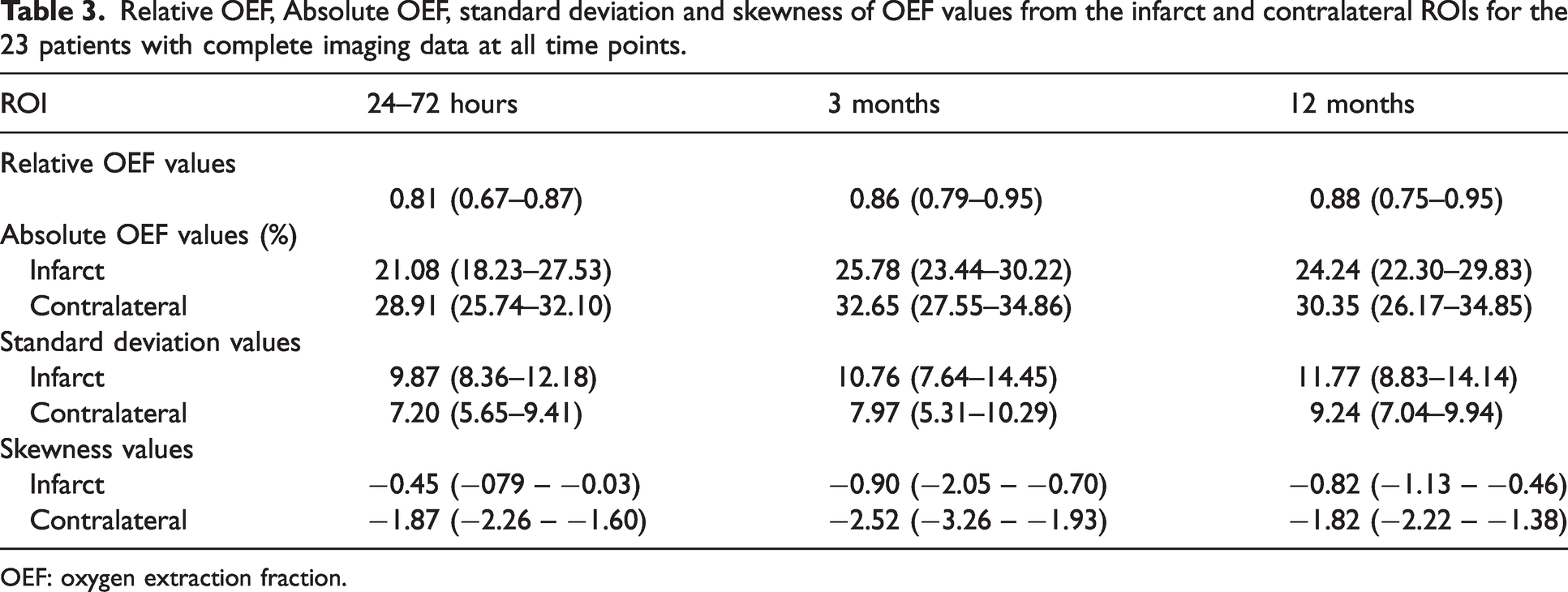
OEF: oxygen extraction fraction.
The standard deviation of the OEF index values in both the infarct and contralateral ROIs showed a slight but non-significant increase from the 24–72 hours’ time point to the 12 months’ time point, as shown in Table 3 and Figure 4(e) (contralateral results for standard deviation not illustrated).
The infarct OEF index values became more negatively skewed from 24–72 hours to 3 months (p < 0.001), and then less negatively skewed from 3 months to 12 months (p = 0.033). However, the skewness at 24–72 hours was significantly less negative (closer to the normal) than the skewness at 12 months (p = 0.002) as shown in Figure 4(f). For the contralateral ROIs, though the trend in skewness was similar to the infarct ROIs, there was no statistical difference in the skewness at 24–72 hours and 3 months (p = 0.128), and 24–72 hours and 12 months (p = 0.248). There was however a statistical significance between the skewness at 3 months and 12 months (p = 0.045) (contralateral results for skewness not illustrated).
Regression analysis
The results from the ordinal regression analysis revealed there was no significant association between mRS at 3 months and infarct OEF index values at 24–72 hours (unadjusted Odds Ratios = 1.000 [95% CI, 0.999–1.000], p = 0.89).
Discussion
The current study assessed the changes in the OEF index values in the infarct and contralateral regions in a cohort of successfully reperfused ischemic stroke patients using the recently developed QQ-CCTV OEF algorithm that combines both QSM and qBOLD OEF mapping techniques to produce a more accurate OEF map. The results showed a general and steady increase of both relative and absolute OEF index values from 24–72 hours to 12 months, with the OEF in the infarct ROIs being significantly lower than the OEF index values in the contralateral ROIs at all time points. The OEF index values in the infarct ROIs were less negatively skewed than OEF values in the contralateral ROIs cross-sectionally. Longitudinally, the OEF index values became more skewed negatively over time. Standard deviation of OEF index values on the other hand showed a non-significant increase over time. Cross-sectionally, the standard deviation of the OEF index values in the infarct ROIs was greater than that in the contralateral ROIs. The regression analysis of mRS at 3 months as a patient outcome measure and OEF index values at 24–72 hours yielded no significant results.
The results from our cross-sectional comparison of OEF index values agree with past comparison studies by Wu et al. 15 and Zhang et al. 14 who used a previous version of QQ pipeline that utilised the cluster analysis of time algorithm in its image processing. Both studies showed a decrease in OEF values in the infarct ROIs versus the contralateral ROIs, regardless of time point. In the current study, despite including patients with successful reperfusion, the infarct values were significantly less than those in the contralateral ROIs. This can be explained by the fact that the infarct tissue consists of both irreversibly damaged and reversibly damaged tissue, and that recovery will only occur in the regions of infarct tissue that are still viable at the time of treatment.
Our longitudinal results, however, contrast with some of the previous studies. Zhang et al. 14 found that the OEF values in the infarct core decreased from the acute to the late subacute stages of ischemic stroke. Cho et al. 6 who used the most recent version of the QQ pipeline employing a deep learning framework, and Wu et al. 15 observed a similar trend, reporting that patients with later stages of ischemic stroke had lower OEF values in the infarct regions compared to those in the contralateral region. It is important to note that the patients involved in these previous studies did not undergo EVT or intravenous thrombolysis before their MRI scans while in the current study, patients underwent successful reperfusion. Thus, the general increase in infarct OEF index values over time in the current study could be indicative of cell regeneration, as more cells recover and require oxygen to metabolize after successful reperfusion.
The statistically significant increase in relative OEF index values from 24–72 hours to 3 months and subsequent decrease in values at 12 months may reflect increased tissue recovery necessitating increased oxygen demand up to 3 months. Beyond 3 months, it may be that further tissue recovery is unlikely and thus the non-significant decrease in OEF values. These results possibly reveal the trend of OEF values beyond the patient’s hospitalization, thereby providing insights into long term patient outcome post-reperfusion.
Notably the absolute OEF index values in the contralateral hemisphere also increased from 24–72 hours to 3 months. Firstly, this may indicate that there are global metabolic changes causing reduction in OEF bilaterally in the acute (24–72 hours) phase of stroke which subsequently normalised in the chronic phase (3 months). However, we found that the OEF index was consistently lower in the infarct lesion at all three time points, which correlates with side-specific permanent tissue injury in the infarct in additional global metabolic changes. Secondly, previous studies have demonstrated that morphological,24,25 hemodynamic,26,27 metabolite 28 and gene expression 29 changes may occur in the contralateral hemisphere after ischemic stroke as a concurrent but independent process to the infarct. Analysis on a larger cohort with additional parameters such as cerebral blood flow inter-side comparison will provide further insight and validation to our data.
Most of the lesions displayed a heterogeneous distribution of OEF index values because of the heterogeneous nature of tissue damage.30,31 This heterogeneity was also seen in the multi-echo complex QQ study by Cho et al. 6 where they visually observed that the OEF maps were more heterogeneous in the infarct region compared to the contralateral hemisphere at the acute phase of ischemic stroke. They attributed the heterogeneity at the acute phase to rapid tissue changes. They postulated that the regions in the infarct ROIs with OEF values like those in the contralateral hemisphere could be reversibly damaged, while those regions in the infarct with significantly lower OEF values than those in the contralateral hemisphere could be irreversibly damaged tissue. In our study, we quantified these heterogeneity changes using standard deviation and skewness. The skewness of the OEF index values generally became more negative, that is, clustered around the higher OEF values over time. This aligns with the increase in relative OEF over time. The standard deviation however did not decrease as we expected but rather increased over time. There was also an unexpected significant decrease in the skewness values in the contralateral ROIs from 3 months to 12 months, the significance of which remains to be elucidated. An additional analysis exploring the association between the skewness of the OEF index values and the infarct volume at the three time points revealed significant results at all time points. However, when we explored the correlation between volume change and change in infarct skewness, we found no significant association (results in supplemental material).
Our contribution to the body of research lies in the quantification of OEF heterogeneity in ischemic stroke patients. Though the change in heterogeneity was visually observed in Cho et al.’s 6 study, this was quantified using standard deviation and skewness in this study. Our analysis shows a similar decrease in heterogeneity with the skewness values as observed by the previous study but not with the standard deviation. Repeating this analysis with a larger patient sample for longitudinal study and exploring the heterogeneity with advanced methods such as texture analysis using machine learning techniques could produce more robust results. Nevertheless, quantifying heterogeneity of OEF values in infarct regions could supplement current strategies like the patient mismatch profile used to identify patients who could benefit from EVT and thrombolysis procedures. 32 Furthermore, it could aid in understanding the underlying pathophysiology of patients who experience futile recanalization, which is experienced by up to 50% of patients with successful EVT procedures. 33
Correlating the OEF index values with mRS at 3 months did not yield significant results. Wu D. et al. 15 obtained NIHSS of the patients recruited at each stroke phase. Running a correlation analysis between NIHSS at each stroke phase and OEF at each stroke phase revealed a significant positive correlation only at the acute phase (less than 1 day) of stroke, which they attributed to the fact that in that phase, the brain is extracting more oxygen and patients are manifesting more severe symptoms. Our lack of significant results could be attributed to the small sample size of patients included and the intrinsic differences in NIHSS and mRS scores. 34
Our analysis of a prospectively recruited cohort of patients with successful angiographic reperfusion to study the longitudinal changes of OEF over time differs from previous longitudinal studies containing patients with variable reperfusion status analysed at variable time points and interval. In doing so, individual patient differences were eliminated. It makes for more accurate tracking of patient changes overtime as well as increasing statistical power. More to that, the present study examined patient data up to 3- and 12-months post-treatment, offering long term data on the patient's health beyond their hospitalization.
Quantification of brain oxygen consumption can provide information on brain tissue viability and function. This is especially important in the context of ischemic stroke, where the stroke lesion comprises irreversibly damaged tissue and a mixture of tissue that is damaged in varying proportions. 31 The use of the QQ-CCTV algorithm as an OEF evaluation method presents significant advantages as it can be easily integrated into routine patient imaging due to the use of routinely acquired multi-echo gradient echo images to obtain OEF maps. It eliminates the use of vascular challenges used in some MRI-based OEF images, which are not practical in the case of ischemic stroke patients in the acute stage. 14 More to that it enables quantification of OEF from tissue voxels, thus providing a more accurate measure of tissue status and tissue changes over time. 11
The longitudinal evaluation of the OEF parameter allows for a better understanding of the recovery or progression of metabolic activity within the heterogeneously viable regions in the infarct region following ischemic stroke treatment and may help to characterize brain oxygenation changes in patients with successful treatment but poor outcomes. Additionally, it would be beneficial to correlate these regions with perfusion measures of cerebral blood volume and cerebral blood flow. This will provide a comprehensive assessment of the OEF results obtained.
Limitations
In the current study, the generated OEF maps from 9 patient GRE images at different points were of poor quality due to incorrect processing and were excluded from analysis.
Cho et. al 6 noted that the current QQ processing methods available, including the QQ-CCTV algorithm employed in this study assume gaussian noise for both susceptibility and multi-echo gradient magnitude images. This assumption could however be unreliable in low signal-to-noise settings such as disease lesions where signal noise may not take a gaussian form. This could be responsible for the discontinuities observed in the histogram representations in our study in Figure 2. Cho et. al proposed a newer algorithm that models realistic noise and integrates it into the processing pipeline. 6
Cho et al. 18 also noted that due to the imperfect decoupling of the OEF and the venous blood fraction in the QQ CCTV model, a lower OEF value could result from a higher venous blood fraction. Thus, the OEF values tended to be lower than previous models of the QQ pipeline. In our study, we acknowledge that our OEF values were lower than some of the OEF values in published literature that used different OEF processing methods, for example Uchida et al. 11 and Kudo et al. 35 who used a QSM based method. We therefore referred to the absolute OEF values as index values. We suspect that the phenomenon explained by Cho et al. could be applicable to our cohort of patients who were successfully reperfused. Our OEF values were however comparable to literature that used similar QQ processing pipelines.14,36 The QQ method was previously validated against PET OEF method in 10 healthy volunteers in a comparison study by Cho et al. 9 They presented the OEF values they obtained using both methods in their publication. We therefore examined whether there were significant differences between the whole brain OEF values obtained in their study using the QQ and PET methods and our contralateral OEF index values obtained with another version of the QQ method. We found no significant differences between their QQ results or PET results and ours. Details of this analysis are presented in the supplemental materials.
We dealt with this limitation by focussing on the relative OEF measure for longitudinal measures, and showed the expected trend of increasing over time, which is a testament to ongoing tissue recovery. Thus, while the trends of OEF values in infarct and healthy tissue were as expected, the exact values should be interpreted with caution due to the above limitations. Performing another study with more recently developed QQ processing pipelines that are robust to the above limitations is recommended.
Using 9 echoes for patient imaging though advantageous in terms of image quality when it comes to processing, may not be ideal due to longer acquisitions. Faster acquisitions using fewer echoes can be explored, and the OEF images generated compared with the OEF images from the nine-echo GRE acquisitions.
The PRAISE study took place during the COVID-19 pandemic. Study participants retention and follow-up were impacted by changes in clinical and research practice to minimise non-essential physical contact. Four patients died during the study’s pre-specified 12-month follow-up period. This limitation was addressed by using only patient data with complete imaging at all three time points for the longitudinal analysis. Despite this measure, the small patient sample size underpowered the longitudinal analysis so interpretations of values should be taken with caution. Future studies with a larger cohort should be conducted to validate the findings obtained in this study.
Conclusion
In ischemic stroke patients with successful reperfusion, the OEF index values within the infarct lesion was reduced compared to the corresponding anatomical region in the normal hemisphere, but increased over time, owing to possible tissue recovery within the radiological infarct lesion. More to that the heterogeneity of the intralesional OEF index values decreased, and the values became more homogeneous in the direction of higher OEF values providing further evidence of possible tissue recovery.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251333940 - Supplemental material for Oxygen extraction fraction changes in ischemic tissue from 24–72 hours to 12 months after successful reperfusion
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251333940 for Oxygen extraction fraction changes in ischemic tissue from 24–72 hours to 12 months after successful reperfusion by Victoria Mercy Kataike, Patricia M Desmond, Christopher Steward, Bruce CV Campbell, Peter J Mitchell, Felix Ng and Vijay Venkatraman in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Ethics approval statement
The study was approved by the Melbourne Health Human Research Ethics Committee.
Consent to participate
The patient or caregiver provided informed consent prior to participating in the study.
Consent for publication
The patient or caregiver provided informed consent for the publication of the results obtained in the study.
Data availability
The data that supports the findings of this study can be made available upon reasonable request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Australian and New Zealand Association of Neurologists, Royal Melbourne Hospital Neuroscience Foundation, Australian Brain Foundation, Austin Medical Research Foundation, the Royal Australasian College of Physicians and the Melbourne Research Scholarship.
Acknowledgements
The authors acknowledge and extend their gratitude to Dr. Junghun Cho for providing the QQ-CCTV pipeline for the study. The authors would like to thank the MR radiographers and the neurointervention team at Royal Melbourne Hospital for their contributions to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceptualization: Felix Ng, Patricia M Desmond, Vijay Venkatraman, Bruce CV Campbell, Christopher Steward
Methodology: Victoria Mercy Kataike, Christopher Steward, Patricia M Desmond, Vijay Venkatraman, Felix Ng,
Data Curation: Felix Ng, Bruce CV Campbell, Christopher Steward, Patricia M Desmond, Vijay Venkatraman, Peter J Mitchell
Investigation: Victoria Mercy Kataike, Vijay Venkatraman, Patricia M Desmond, Felix Ng, Christopher Steward
Project Administration: Victoria Mercy Kataike, Vijay Venkatraman, Patricia M Desmond, Felix Ng, Christopher Steward, Bruce CV Campbell, Peter J Mitchell
Formal analysis: Victoria Mercy Kataike, Vijay Venkatraman, Patricia M Desmond, Felix Ng, Christopher Steward
Writing – original draft preparation: Victoria Mercy Kataike
Writing – review and editing: Victoria Mercy Kataike, Vijay Venkatraman, Patricia M Desmond, Felix Ng, Christopher Steward, Bruce CV Campbell, Peter J Mitchell
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
