Abstract
Acute ischemic stroke results from blockage of a cerebral artery or impaired cerebral blood flow due to cervical or intracranial arterial stenosis. Ischemic stroke prevention seeks to minimize the risk of developing impaired cerebral perfusion by controlling vascular and cardiac disease risk factors. Similarly, ischemic stroke treatment aims to restore cerebral blood flow through recanalization of an occluded artery or dilation of a severely narrowed artery that supplies cerebral tissue. Stroke prevention and treatment are increasingly informed by imaging studies, and neurovascular and cerebral perfusion imaging has become essential in in guiding ischemic stroke prevention and treatment. Here we review the latest advances in ischemic stroke prevention and treatment with an emphasis on the neuroimaging principles emphasized in recent randomized trials. Future research directions that should be explored in ischemic stroke prevention and treatment are also discussed.
Introduction
Ischemic stroke is the leading cause of disability and the fifth leading cause of death in the United States. 1 Ischemic stroke is caused by embolic or thromboembolic occlusion of a cerebral artery, which results in reduced cerebral blood flow (CBF) to the brain. This impaired CBF results in brain ischemia, which is separated into two distinct components: (1) the irreversibly damaged “core” infarction and (2) the ischemic, but viable, surrounding tissue that is called the “penumbra.”2,3 The goal of all ischemic stroke treatments is restoration of CBF to the penumbra, as penumbra reperfusion minimizes the final size of core infarction and results in improved patient outcomes.2,4,5
The past several years have seen significant advancements in both ischemic stroke prevention and treatment. There have been significant changes in medical stroke prevention due to cardiac arrhythmias and intracranial atherosclerosis in recent years, and more stepwise advancements in stroke prevention due to cervical atherosclerotic disease. Furthermore, the recent publication of five randomized trials4–8 that demonstrate an overwhelming benefit of endovascular mechanical thrombectomy for the treatment of ischemic stroke compared to medical management is the most significant advance in ischemic stroke therapy since the approval of intravenous tissue plasminogen-activator (tPA) in 1995. 9
Here we review the latest advances in ischemic stroke prevention and treatment with an emphasis on recent randomized trials and neuroimaging principles. We aim to provide an update on the treatment of ischemic stroke to the imaging community and to emphasize the important advances in imaging as they pertain to stroke prevention and treatment. First, recent advances in clinical ischemic stroke prevention are discussed with an emphasis on cardioembolic, cervical carotid artery atherosclerosis, and intracranial atherosclerosis. Next, cervical and intracranial atherosclerotic plaque and vessel wall imaging advances that may identify patients at increased risk of an ischemic stroke risk are discussed. Third, recent advances in ischemic stroke treatment are discussed with an emphasis on endovascular therapy. Fourth, we consider neuroimaging techniques for endovascular stroke therapy with an emphasis on the variety of approaches in practice and on neuroimaging biomarkers that predict patient outcome. Lastly, the success of the recent ischemic stroke trials has raised many questions with respect to the neuroimaging evaluation of ischemic stroke patients, and we also discuss future research directions that should be explored to define the optimal imaging evaluation of these patients.
Ischemic stroke prevention clinical and imaging advances
Ischemic stroke is most commonly caused by atherosclerotic disease or cardiac arrhythmias, such as atrial fibrillation, and stroke risk reduction focuses on the medical control of these diseases. 10 The most common medical comorbidities that increase the risk of ischemic stroke include hypertension, diabetes mellitus, hyperlipidemia, and smoking.10–17 Effective ischemic stroke prevention requires aggressive blood pressure control (target systolic blood pressure less than 140 mm Hg and diastolic blood pressure less than 90 mm Hg), hyperlipidemia reduction with exercise, diet, and statin medications (target low-density lipoprotein levels < 100 mg/dl), and smoking cessation. 10 To date, aggressive glycemic control in patients with diabetes mellitus has not been shown to result in a reduced risk of ischemic stroke. 18 However, most stroke neurologists suggest a hemoglobin A1C of < 6.5% as an appropriate therapeutic target. 10
In the past several years, there have been advancements in the prevention of ischemic stroke due to cardioembolic, cervical carotid atherosclerotic disease, and intracranial atherosclerotic disease (ICAD). These clinical advances and emerging neuroimaging techniques to predict the risk of stroke due to cervical or ICAD are briefly reviewed below.
Cardioembolic stroke risk reduction
Patients with non-valvular atrial fibrillation are at an increased risk of ischemic stroke secondary to cardiac thromboembolism, 19 and the majority of these patients should be anticoagulated to minimize their stroke risk.10,20–25 For many years, most patients were anticoagulated with warfarin. Warfarin is an effective anticoagulant that targets vitamin K-dependent enzymes in the coagulation cascade, but it interacts with various foods and many medications and requires frequent international normalized ratio blood tests to ensure adequate anticoagulation.
More recently, non-vitamin K antagonist oral anticoagulant (NOAC) medications have become the preferred anticoagulant in the prevention of ischemic stroke secondary to atrial fibrillation. Dabigatran, rivaroxaban, apixaban, and edoxaban are all NOACs that result in non-inferior (rivaroxaban and edoxaban) or reduced (edoxaban and dabigatran) rates of ischemic stroke compared to warfarin.26–30 These medications also have a similar (rivaroxaban) or reduced (dabigatran, rivaroxaban, edoxaban) frequency of bleeding complications compared to warfarin.26–30 NOACs also have the advantage of not requiring routine blood tests to ensure adequate anticoagulation. Thus, NOACs represent an important iterative advancement in the non-invasive prevention of ischemic stroke in patients with atrial fibrillation. Future studies and patient registries will be important to determine the long-term safety of the NOAC medications.
Cervical carotid artery atherosclerosis risk reduction
Atherosclerotic disease affecting the common carotid artery and cervical internal carotid artery is responsible for 25% of ischemic strokes in the United States. 31 Atherosclerotic plaque may reduce CBF as the arterial lumen is reduced in caliber by the plaque, and this impaired CBF may manifest as a transient ischemic attack (TIA) or ischemic stroke. Alternatively, atherosclerotic plaque rupture exposures the sub-endothelial cholesterol-rich plaque to the bloodstream, which may cause thrombotic or thromboembolic occlusion of the cervical internal carotid artery or intracranial circulation. 31
All patients with a TIA or ischemic stroke undergo imaging evaluation of the cervical common and internal carotid arteries to evaluate for a significant stenosis due to atherosclerotic plaque. Carotid ultrasound is the most common imaging modality used to evaluate the cervical arteries, and computed tomographic angiography (CTA) or magnetic resonance angiography (MRA) are also frequently performed (Figure 1).
Acute ischemic stroke secondary to a critical cervical internal carotid artery stenosis. A DWI image demonstrates a small area of cerebral infarction in the subcortical white matter of the right temporal lobe (a, arrow). Cervical artery vascular imaging using carotid color Doppler ultrasound (b) and CTA (c) after presentation identified a critical cervical right internal carotid artery stenosis (b and c, arrows). The patient underwent treatment by placement of a carotid artery stent. A pre-stent digital subtraction angiogram in the lateral projection shows a severe cervical right internal carotid artery stenosis that measures 1 mm in diameter (d, arrow). This lesion was treated by uneventful placement of a carotid artery stent (e, arrow), and a post-stent digital subtraction angiogram in the lateral projection demonstrates a significant improvement in the caliber of internal carotid artery (f, arrow).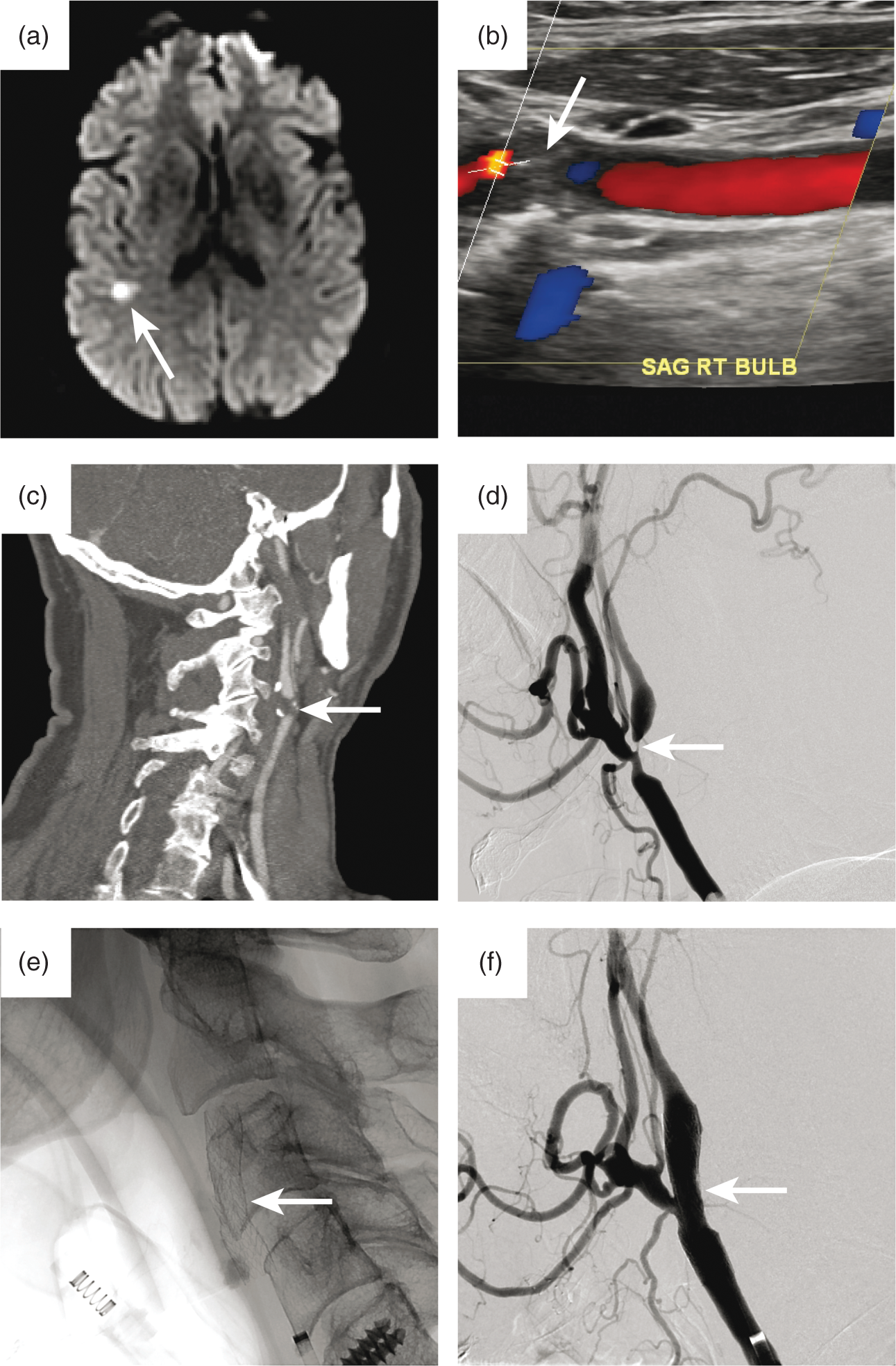
Patients with ischemic symptoms referable to a common or internal carotid artery stenosis that measures 50–99% compared to the normal adjacent vessel caliber benefit from treatment by carotid endarterectomy (CEA)32–34 or carotid artery stenting (CAS) (Figure 1).35,36 Both CEA and CAS improve the caliber of the treated cervical carotid artery and lead to a reduced risk of subsequent stroke. Importantly, the long-term results of the Carotid Revascularization Endarterectomy versus Stenting Trial (CREST) were published in 2016. The CREST study randomized 2502 patients with symptomatic carotid artery stenosis to CEA or CAS, and this study demonstrated no significant difference between CEA and CAS with respect to periprocedural stroke, myocardial infarction, death, post-procedure ipsilateral stroke, or long-term ipsilateral stroke at up to 10 years of follow up. 36 However, a meta-analysis of randomized trials comparing CEA and CAS showed found a higher rate of any stroke or death in patients over 70 years who were treated with CAS compared to CEA. 37 Although CEA and CAS may be considered equivalent therapies in the treatment of symptomatic cervical carotid artery stenosis, CEA is likely still superior in older patients.
Although cervical common or internal carotid artery narrowing of 50% or greater is associated with an increased risk of stroke, 88–90% of asymptomatic patients with this degree of stenosis do not develop an ischemic stroke.34,38,39 Therefore, the degree of luminal stenosis is only one component of the atherosclerotic plaque that determines the risk of a future ischemic stroke, and additional studies have identified atherosclerotic plaque composition to be important for risk stratification.40–43 Imaging advances in cervical atherosclerotic plaque characterization are discussed in the “Vessel Wall Imaging in Stroke Prevention” Section below.
Intracranial atherosclerosis risk reduction
ICAD is an important cause of ischemic stroke, and ICAD accounts for up to 10% of all ischemic strokes in the USA and up to 50% of all ischemic strokes outside of North America.44–46 Similar to cervical atherosclerotic disease, ICAD may reduce CBF through artery luminal narrowing or acute thrombotic occlusion in the setting of a ruptured atherosclerotic plaque (Figure 2)47–49 Similar to cervical atherosclerotic disease, reduction of stroke risk in the setting of ICAD focuses on minimizing the risk of further arterial lumen narrowing and preventing plaque rupture.
Transient ischemic attacks due to a severe middle cerebral artery stenosis. A 66-year-old patient developed transient ischemic attacks localizing the right middle cerebral artery circulation. Brain MRI demonstrated no evidence of acute or chronic cerebral infarction (a), and MRA demonstrated a focal severe stenosis in the proximal M1 segment of the right MCA (b, c, arrows). This stenosis was confirmed on digital subtraction angiography (d, arrow), and the appearance was consistent with ICAD. The patient was initially treated medically, including dual anti-platelet medication initiation, but her symptoms were persistent. She underwent endovascular treatment with cerebral artery angioplasty (e, f) given that she had failed medical management. An angioplasty balloon was inflated across the right M1 stenosis (e, arrow). Following angioplasty, there was a marked improvement in the caliber of the right M1 segment (f, arrow). She has had recurrent symptoms after treatment.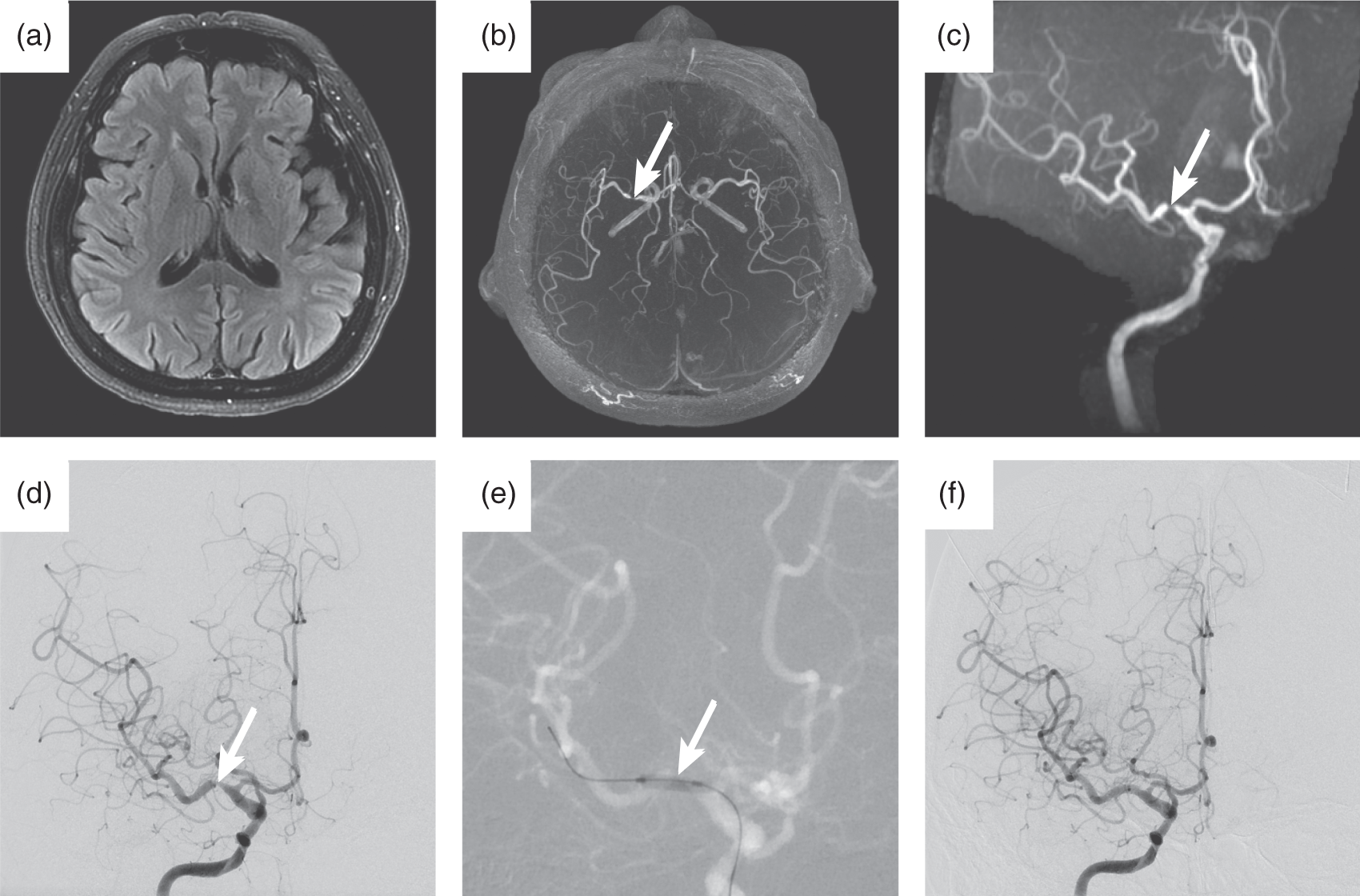
The medical management of ICAD requires aggressive lipid lowering with atorvastatin 80 mg daily or rosuvastatin 40 mg daily, regardless of LDL level, and aggressive antiplatelet medical therapy to minimize the risk of in situ atherosclerotic plaque thrombosis. 50 The randomized trials “A Comparison of Warfarin and Aspirin for the Prevention of Recurrent Ischemic Stroke” (WARSS) and “Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis” (WASID) showed aspirin to be equivalent to warfarin for prevention of ischemic stroke, cerebral hemorrhage, or death.51,52 Patients treated with warfarin also had a significantly higher adverse event rate. 52 The WARSS and WASID trials became the basis for treatment of ICAD with single antiplatelet agents, most commonly either aspirin or clopidogrel.
Subsequent studies comparing dual antiplatelet medical therapy using both aspirin and clopidogrel to either aspirin alone or clopidogrel alone did not find a benefit of dual antiplatelet medication therapy in the prevention of ischemic stroke.53,54 However, these studies were not restricted to patients with ICAD, and the ICAD specific “Stenting versus Aggressive Medical Therapy for Intracranial Arterial Stenosis” (SAMMPRIS) trial found a lower rate of recurrent ischemic stroke and death among ICAD patients treated with aspirin and clopidogrel compared to historical controls. 55 Based upon the SAMMPRIS trial results, most stroke neurologists and neurointerventionalists consider dual antiplatelet medical therapy to be the standard of care in the treatment of ICAD. However, additional studies are required to define better the most optimal medical therapy. 56
More aggressive endovascular treatments for ICAD include angioplasty and intracranial stenting. Initial studies showed promise for these techniques in preventing ischemic strokes in patients with symptomatic ICAD.57–62 However, the SAMMPRIS trial, which randomized patients to either cerebral artery angioplasty and stenting or maximal medical therapy, was terminated after an interim analysis demonstrated a significantly higher rate of stroke or death in the angioplasty and stenting group (14.7%) compared to the medical management group (5.8%) within 30 days of treatment. Based upon the SAMMPRIS results, cerebral artery angioplasty and stenting are not recommended for the treatment of ICAD. 10 Future studies are required to determine if subsets of patients with ICAD, such as those who develop ischemic stroke or TIAs while on maximum medical management, benefit from cerebral artery angioplasty or stenting.
Atherosclerotic plaque and vessel wall imaging in stroke prevention
The advances in medical and interventional therapy for stroke prevention in patients with cervical carotid atherosclerotic disease or ICAD have been coupled with the more recent identification of imaging biomarkers that may more accurately predict the risk of ischemic stroke in these populations. The degree of luminal narrowing has been the mainstay of vessel imaging for over 20 years, but emerging techniques that image the composition, shape, and inflammatory characteristics of atherosclerotic plaques may provide superior information regarding the risk of a future thrombotic or thromboembolic event due to plaque rupture.40–43,63–65 It is not our intention to provide a complete overview of the advances in carotid and intracranial atherosclerotic plaque imaging, which are well reviewed elsewhere.66,67 However, we will briefly highlight the principal advances in atherosclerotic plaque and vessel wall imaging.
The most important imaging biomarkers of atherosclerotic plaque instability are: (1) intraplaque hemorrhage (IPH), (2) vessel wall enhancement, and (3) plaque ulceration.68–71 MRI is the preferred imaging modality for the assessment of carotid and intracranial atherosclerotic plaque, as CT/CTA and CT/positron emission tomography are less reliable in the detection of IPH and plaque enhancement.72–74
Intraplaque hemorrhage imaging
IPH is present in one-third of atherosclerotic plaques with a luminal stenosis of at least 50%.64,75 Histologic analysis of cervical carotid artery atherosclerotic plaque demonstrates regions of hemorrhage within the plaque, which occur secondary to transient endothelial breakdown overlying the plaque or hemorrhage due to vasa vasorum breakdown adjacent to the plaque. 76 These hemorrhagic foci are hypothesized to perpetuate ongoing plaque growth and further instability.64,76,77 Consistent with this idea, the presence of IPH strongly correlates with an increased risk of plaque progression and stroke in patients without symptoms irrespective of the degree of associated arterial stenosis.41,64,78 Furthermore, retrospective studies demonstrate that IPH absence confers a 90–100% negative predictive value of a subsequent ischemic stroke.40,41,64,78 Larger prospective studies are needed to validate IPH as a biomarker of stroke risk, but this biomarker holds promise in the evaluation of patients at risk of ischemic stroke.
MRI identifies IPH by imaging methemoglobin within the plaque as a region of T1 shortening (T1 hyperintense signal abnormality). Most protocols use T1-weighted two-dimensional spin echo sequences or three-dimensional magnetization prepared gradient echo sequences to image IPH.41,64,68,78,79 Volumetric 3D acquisitions and the use of fat suppression techniques may further increase the conspicuity of these lesions in the vessel wall. 66
Vessel wall and plaque enhancement
Atherosclerotic plaque inflammation results in plaque instability and a higher risk of atherosclerotic plaque rupture and ischemic stroke.80,81 The mechanism by which plaque inflammation results in plaque instability remains incompletely understood, but it is thought to result in cytokine signaling that promotes macrophage recruitment, endothelial and fibrous cap breakdown, and plaque rupture. 82 Carotid plaque enhancement reflects this atherosclerotic plaque inflammation, and this enhancement has been used as a marker of increased ischemic stroke risk.83–87 Similar to IPH, plaque enhancement is associated with an increased risk of ischemic stroke regardless of the degree of associated vessel stenosis.85,86
Plaque enhancement is measured by dynamic contrast enhanced MRI following intravenous gadolinium-based contrast agent injection.69,83–85 Post-contrast T1-weighted images are typically acquired in the axial plane so as to be perpendicular to the course of the cervical common and internal carotid arteries. Fat-saturation techniques can help to increase the conspicuity of the plaque enhancement. 69
Plaque ulceration imaging
Rupture of an atherosclerotic plaque occurs when there is a sudden exposure of the highly thrombogenic lipid core to the blood traversing the arterial lumen. Plaque rupture then results in platelet adhesion and formation of a thrombus at the lipid–blood interface, and this thrombus may result in an ischemic stroke. Therefore, high-risk atherosclerotic plaques are those with a high likelihood of exposure of the lipid-rich core to arterial blood flow.
The lipid-rich core of an atherosclerotic plaque is separated from the endothelial lumen by a fibrous cap. Discontinuity in this fibrous cap represents plaque ulceration, which has been correlated with an increased risk of ischemic stroke.71,88 On MRI, this fibrous cap appears as a discrete structure between the vessel lumen and the underlying plaque, and this cap is hypointense on conventional MRA images. Plaque ulceration is best demonstrated on contrast enhanced MRA as a focal disruption in the hypointense cap with T1 shortening. 89 Future prospective studies are required to validate fibrous cap ulceration as a marker of increased stroke risk.
Intracranial artery wall and atherosclerotic plaque imaging
Similar to cervical artery atherosclerotic plaques, atherosclerotic plaques in the intracranial circulation and are at risk of in situ thrombosis and thromboembolism that may result in ischemic stroke. Cerebral artery IPH and wall enhancement have been linked to a higher risk of ischemic stroke in the vascular territory of the affected vessel,90–92 but the smaller size of the cerebral arteries introduces challenges in the imaging of these high-risk plaques. To our knowledge, cerebral artery plaque ulceration imaging has not yet been described.
Atherosclerotic plaques commonly involve the middle cerebral artery (MCA), the intracranial vertebral arteries, and basilar artery. Interestingly, MCA atherosclerotic plaques typically localize asymmetrically to one wall of the cerebral artery, 93 and symptomatic plaques are more commonly located on the superior and posterior wall of the MCA. These findings suggest that symptomatic plaques are more closely associated with the origins of the lenticulostriate perforating arteries, which may become occluded. 94 Most ischemic strokes affect the MCA vascular bed, and MCA IPH is present in up to 20% of patients with ischemic stroke due to ICAD.95,96 These data suggest that cerebral vessel wall imaging to identify IPH may be valuable in the evaluation of ischemic stroke patients, especially those who otherwise have cryptogenic ischemic strokes.
Plaque enhancement is seen in 70% of patients with symptoms referable to the enhancing plaque, but whereas only 8% of asymptomatic plaques demonstrate enhancement (Figure 3).
97
In support of these findings, a recent meta-analysis found enhancement of cerebral artery plaque to be highly associated with ischemic infarction within the territory of the enhancing vessel with an odds ratio of 10.8.
92
Future prospective studies are needed to validate these findings and determine the relative risk of cerebral vessel wall enhancement in predicting ischemic stroke risk.
Vessel wall enhancement in a patient with intracranial atherosclerotic disease. A middle aged patient presented with recurrent transient ischemic attacks localizing to the right MCA circulation. An MRI FLAIR image demonstrated evidence of prior cerebral infarction (a, arrow), and MRA identified a severe stenosis in the M1 segment of the right MCA (b, c, arrows). The right M1 stenosis was felt to be most consistent with ICAD, and the patient underwent further evaluation with vessel wall imaging (d–f). A coronal T2-weigthed image demonstrates the right M1 stenosis (d, arrow). Post-contrast black-blood vessel wall imaging demonstrates mild enhancement of the arterial wall localizing to the region of stenosis, which is shown in the coronal (e, arrow) and sagittal (f, arrow) planes. The inset images (e, f) highlight this enhancement (e, f, arrowheads).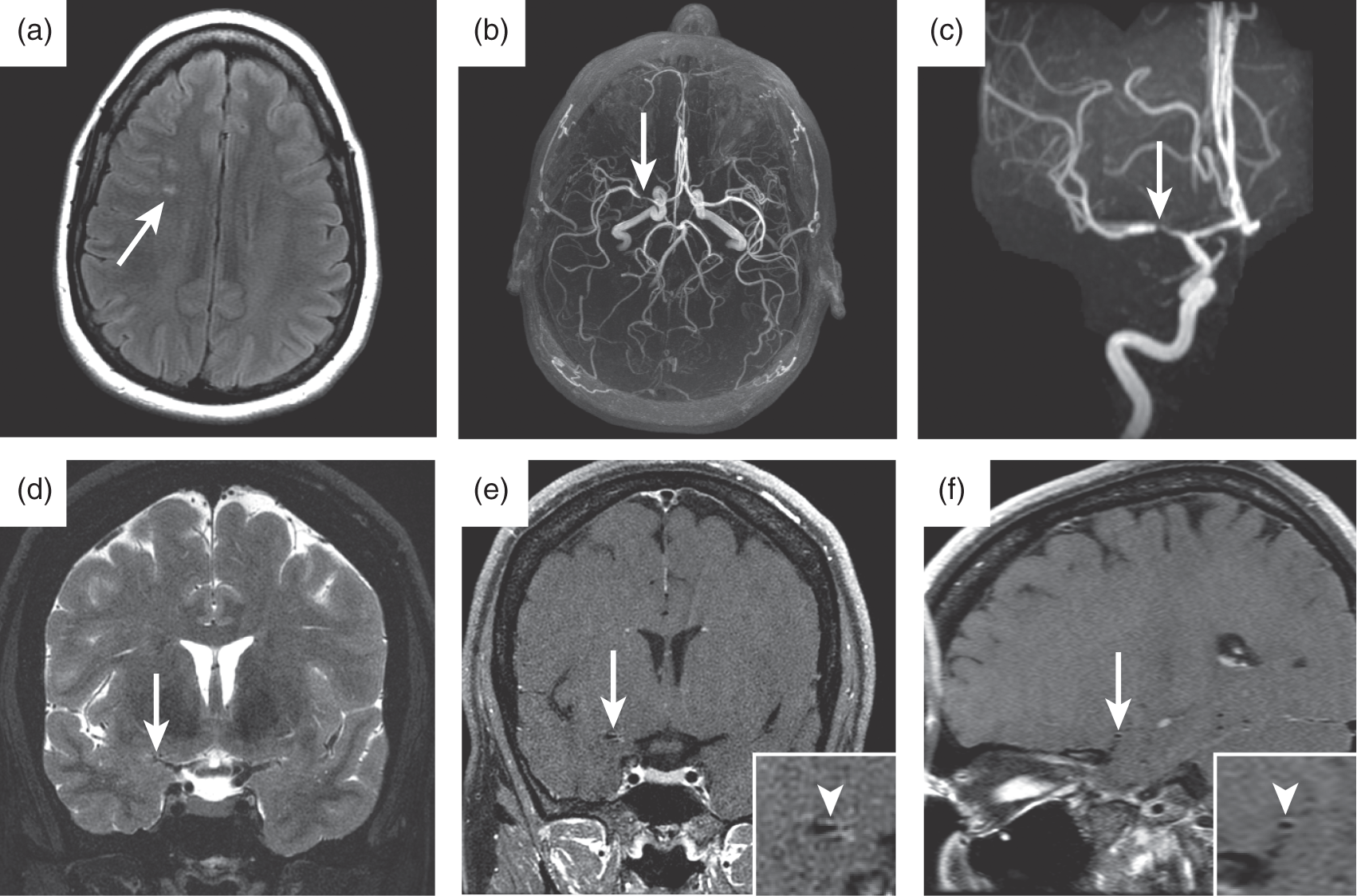
Future directions in atherosclerotic plaque and vessel wall imaging
Cervical and intracranial atherosclerotic plaque and vessel wall imaging hold great promise in the evaluation and risk stratification of patients with acute ischemic stroke. The preliminary studies summarized above should be validated with larger prospective trials. If IPH, vessel wall enhancement, and plaque ulceration are validated as risk markers for stroke, it will be of interest to determine whether these findings may be successfully used as imaging surrogates in medical or interventional studies in the emerging era of personalized medicine. Furthermore, we envision how individual patients' plaque imaging evaluation may lead to change in medical management to reduce the risk of ischemic stroke.
Ischemic stroke treatment advances
There have been significant advances in endovascular stroke treatment by mechanical thrombectomy over the past several years, although these advances were preceded by significant setbacks in earlier trials. In 2013, the Interventional Management of Stroke III (IMS-III), Mechanical Retrieval and Recanalization of Stroke Clots Using Embolectomy (MR RESCUE), and Local Versus Systemic Thrombolysis for Acute Ischemic Stroke (SYNTHESIS) trials were published, and these trials all found no benefit of endovascular stroke therapy compared to medical management in the treatment of acute ischemic stroke.98–100 These data generated much discouragement in the stroke neurology and neurointerventional community, and many of these physicians struggled to understand these trial results when they had so often seen superior outcomes with endovascular stroke therapy in their own practices.
Critical analyses of IMS-III, MR RESCUE, and SYNTHESIS identified several design limitations of these trials that principally included: (1) a lack of vascular imaging prior to randomization and (2) a paucity of third generation stent retriever devices used for mechanical thrombectomy in the endovascular arms of the studies.2,101 For example, patients in the IMS-III were randomized without performing a CTA or MRA in all patients, and 19% of patients in the endovascular arm did not receive endovascular treatment because no target arterial blockage was identified.99,101 Stent retrievers result in significantly higher rates of revascularization compared to older generation devices,102,103 but stent retrievers were used in only 1% of the IMS-II, 13% of the SYNTHESIS, and 0% of the MR RESCUE endovascular arms.98–100 These, and other, design limitations likely accounted for the failure of these trials to show a benefit of endovascular stroke therapy compared to medical management.
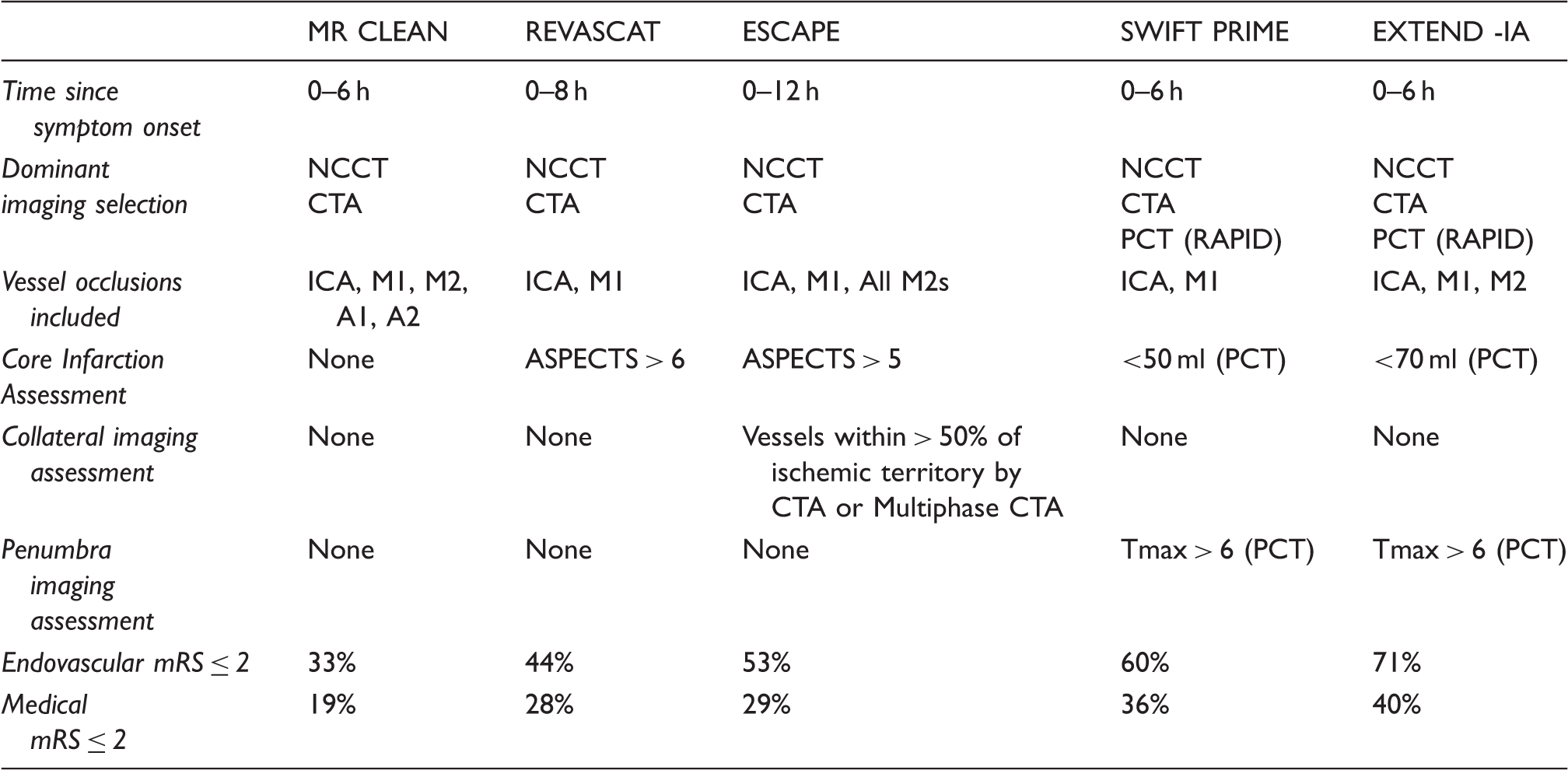
Clinical and imaging selection and outcomes in recent randomized endovascular stroke trials.
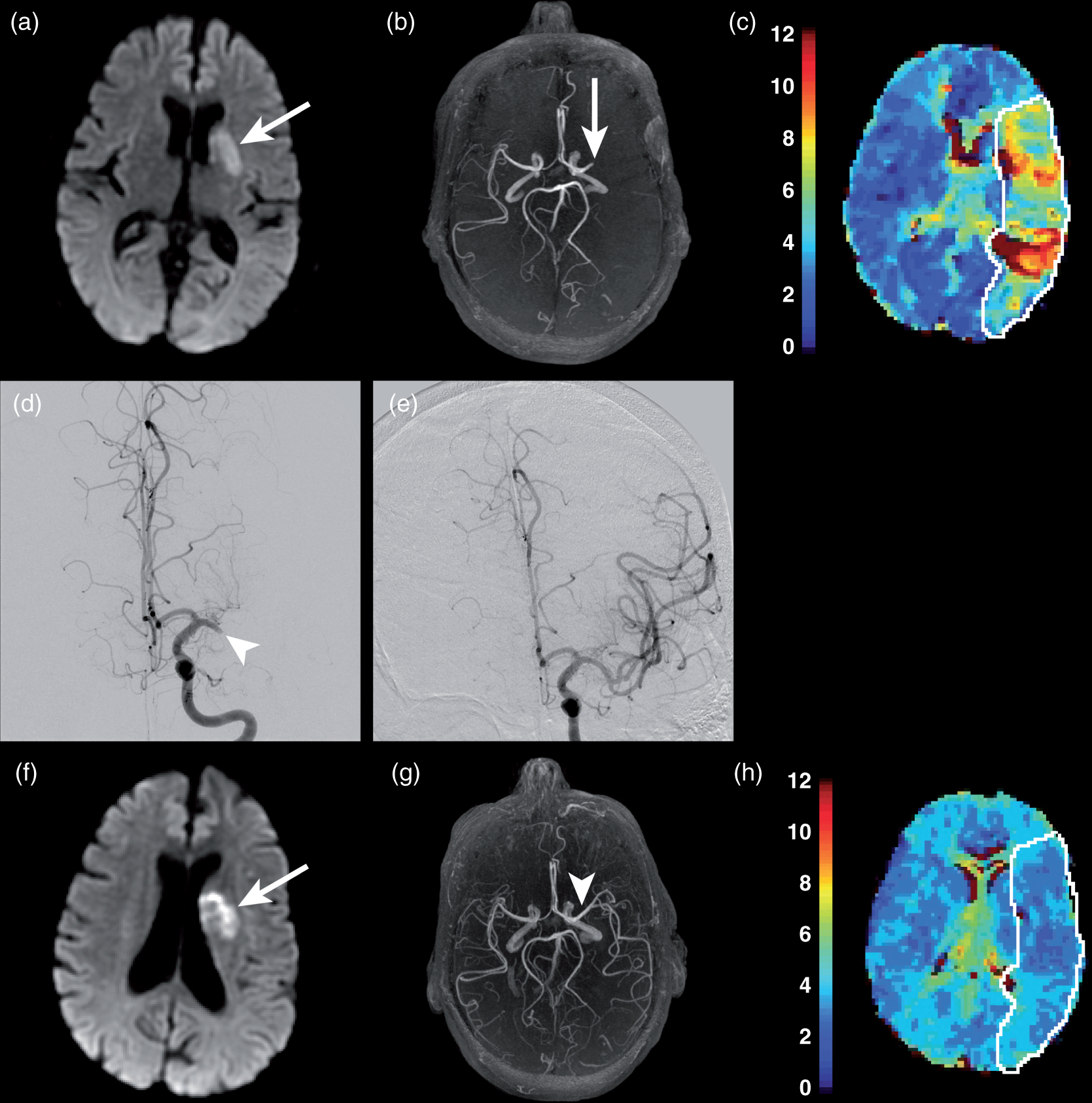
Endovascular stroke therapy in a patient with acute ischemic stroke due to occlusion of the M1 segment of the left middle cerebral artery. An elderly patient with an acute ischemic stroke due to occlusion of the M1 segment of the left MCA. (a–c) MRI obtained prior to endovascular treatment shows a small core infarction on DWI (A, arrow), occlusion of the M1 segment of the left MCA on a MRA maximum intensity projection image (b, arrow), and a large penumbra on the PMRI Tmax map (c, white outline). (d, e) The patient underwent endovascular stroke therapy, and a digital subtraction angiogram prior to treatment demonstrates occlusion of the M1 segment of the left MCA (d, arrow), which was completely recanalized after mechanical thrombectomy (e). An MRI obtained on the day after endovascular treatment shows an unchanged region of core infarction on DWI (f, arrow), complete recanalization of the left MCA (g, arrowhead), and normalization of cerebral perfusion on the PMRI Tmax map (h, white outline).
Neuroimaging selection of endovascular stroke candidates
The neuroimaging evaluation of acute ischemic stroke patients has several goals that include establishing the ischemic stroke diagnosis, excluding stroke mimics, identifying contraindications to intravenous or intra-arterial thrombolysis, and identifying patients who may benefit from endovascular treatment. 2 Patients most likely to benefit from endovascular stroke treatment are those with a relatively small volume of core infarction, a large vessel occlusion (cervical or intracranial internal carotid artery, M1 or M2 segment of the MCA), and salvageable brain tissue.2,104 The five randomized trials all addressed these three factors, but there was variation in the imaging techniques used in the neuroimaging evaluation.4–8 These three principal components of acute ischemic stroke imaging are now discussed further.
Ischemic core imaging
The ischemic core, which we define as irreversibly infarcted brain tissue, should be measured or estimated in all acute ischemic stroke patients to guide subsequent treatment. Diffusion-weighted imaging (DWI) that demonstrates restricted diffusion in brain tissue remains the gold-standard for core infarction identification (Figure 5) However, not every patient can undergo MRI, and not every stroke center has unrestricted access to MRI. In fact, very few patients among the five randomized trials underwent pre-endovascular treatment evaluation by MRI.4–8,105 These limitations of MRI have led to the widespread adoption of CT-based techniques to assess the core infarction.
CT-, PCT-, and MRI-based measures of core infarction in a patient with an acute ischemic stroke. Non-contrast head CT images from a patient with an acute ischemic stroke due to occlusion of the M1 segment of the left MCA (a, b). These images demonstrate hypodensity of the left lentiform nucleus (a, arrow; b, arrowhead), left insular ribbon (a, arrowhead), and the left caudate head (b, arrow). No abnormal hypodensity was identified in the cortical regions, and this patient was assigned a favorable ASPECTS of 7. By contrast, PCT images (c, d) estimated a much larger area of core infarction in this patient. CBF imaging (c) showed a large region of decreased blood flow within the left MCA territory (C, arrow), and CBV imaging showed a slightly less large region of decreased cerebral blood volume (d, arrow). The patient underwent successful revascularization, and DWI obtained after treatment (e, f) demonstrated a core infarction (e, f, arrows) that corresponded to the core infarction estimated by the CBV and CBF PCT images.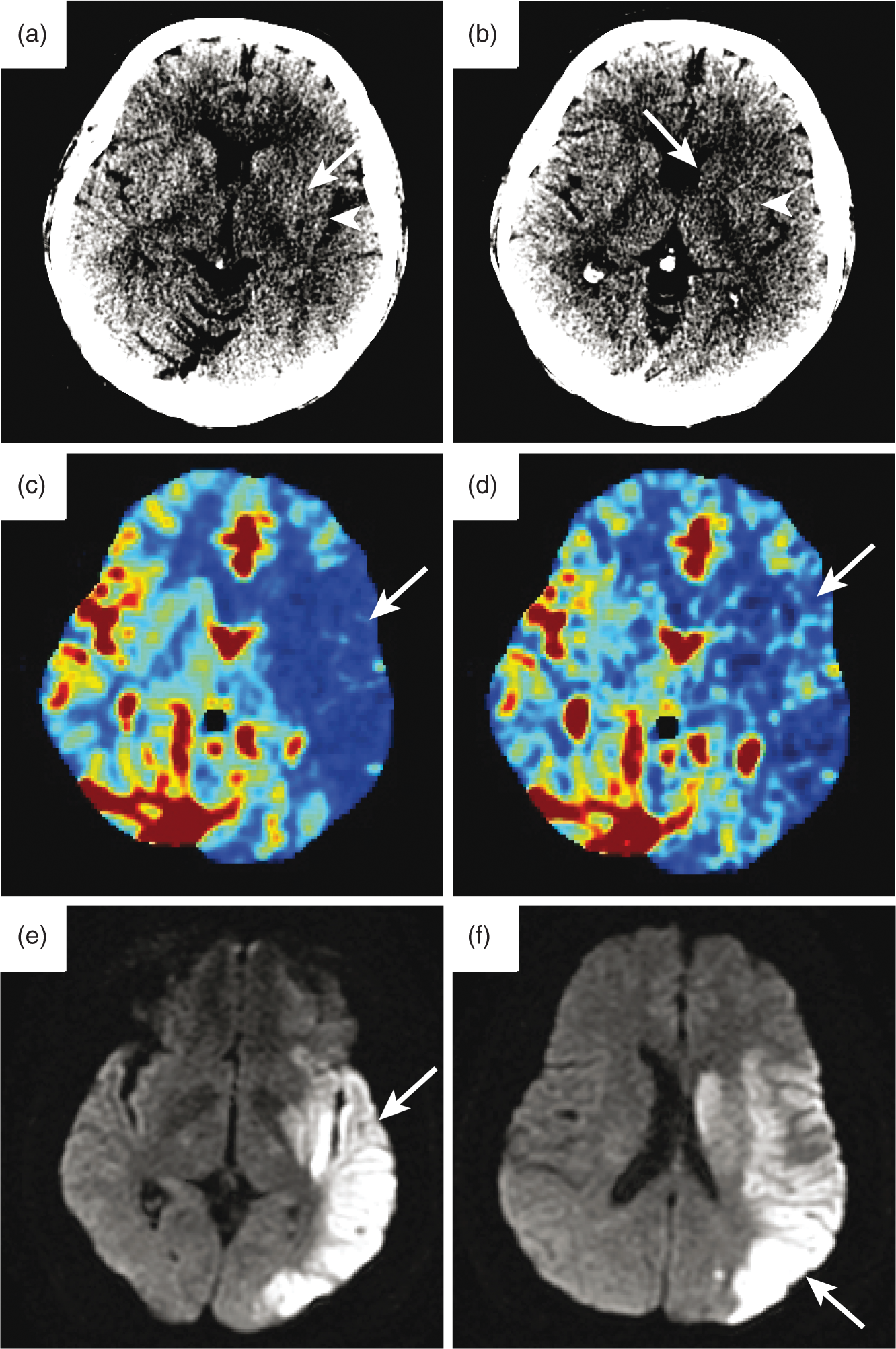
The simplest CT measure of core infarction is a volumetric estimation of abnormal hypodensity. Most centers currently use the more quantitative 10-point Alberta Stroke Program Early CT Score (ASPECTS) as an estimate of the size of core infarction on non-contrast head CT. 106 The ESCAPE, REVASCAT, and SWIFT PRIME trials required an ASPECTS of 6-10 for inclusion, and patients with ASPECTS of 0-5 excluded under the assumption that endovascular treatment would offer no benefit in the setting of a large volume of core infarction and that treatment of these patients would lead to an increased risk of reperfusion hemorrhage.4,7,8 By contrast, the MR CLEAN trial did not require any estimation of the core infarct size, and a non-contrast head CT was only used to exclude intracranial hemorrhage. 6 A recent subgroup analysis of the MR CLEAN trial determined patient outcomes as a function of pre-treatment ASPECTS, which were grouped into large infarction (ASPECTS 0–4), moderate infarction (ASPECTS 5–7), and small infarction (ASPECTS 8–10). 107 This study found no significant difference in outcome or adverse events among these groups, which suggests that patients with ASPECTS less than six may be considered for treatment. 107
Core infarction may also be estimated by perfusion CT (PCT) as a significant reduction in either CBF or cerebral blood volume (CBV) relative to the contralateral normal hemisphere (Figure 5).104,108 The EXTEND-IA and SWIFT PRIME trials both estimated the core infarction as a 70% reduction in CBF relative to the normal cerebral hemisphere, and the volume of core infarction was processed using commercially available automated software.4,5 Patients with a core infarction greater than 50 ml (SWIFT PRIME) 4 or 70 ml (EXTEND-IA) 5 were excluded from treatment.
These results demonstrate that the size of core infarction is an important consideration in the selection of endovascular treatment candidates, but much uncertainty exists as to the most optimal imaging method to estimate the ischemic core size. Furthermore, whether patients with core infarctions larger than 70 ml (especially in non-dominant hemispheres) may benefit from treatment remains uncertain. Some studies have found that the size of presenting core infarction is a strong determinant of patient outcome.109–111 The finding that patients with low ASPECTS still benefit from endovascular treatment 107 and the increasingly observed phenomenon of reversal of diffusion restriction after endovascular therapy 112 underscores the need for additional research into how the core infarction influences patient outcome after endovascular treatment.
Vascular imaging to localize vessel occlusion
The use of non-invasive vascular imaging to identify the presence of a large vessel occlusion is critical in the appropriate selection of patients for endovascular stroke treatment. The five recent endovascular stroke trials all used CT or MR angiography to localize the level of vascular occlusion prior to randomization to endovascular or medical treatment.4–8 The ESCAPE, REVASCAT, and SWIFT PRIME trials were the most restrictive in their selection criteria, and these trials required occlusion of the internal carotid artery or M1 segment of the MCA for consideration of treatment.4,7,8 By contrast, the EXTEND-IA and MR CLEAN trials allowed for more distal occlusion of the M2 segment of the MCA.5,6 Anterior cerebral artery occlusion was also allowed in the MR CLEAN trial, but only three patients with anterior cerebral artery occlusions were included in this trial. 6
The use of vascular imaging in the evaluation of stroke patients who are being considered for endovascular treatment ensures that only patients with a target for treatment undergo triage to the neuroendovascular suite.2,104 The IMS-III trial did not use vascular imaging prior to triage for endovascular therapy, and, as described previously, 19% of patients undergoing endovascular therapy did not have a vascular occlusion that could be intervened upon. 99 Given the significant resources required for endovascular stroke therapy, appropriate patient triage with vascular imaging is likely to be very cost effective.
Salvageable brain tissue and penumbral imaging
A fundamental goal of endovascular stroke therapy is to limit the size of cerebral infarction through the timely recanalization of the occluded vessel. The patient most likely to benefit from endovascular recanalization is one with a small volume of core infarction and a large volume of brain tissue at risk of infarction if CBF is not restored in a timely manner (the so called “target mismatch”) (Figure 6).2,104,113
CT and MRI examples of the target mismatch in acute ischemic stroke. Three different patients presenting with acute ischemic stroke and target mismatch neuroimaging profiles. Patient 1 (a–c). Non-contrast head CT demonstrates a small core infarction in a hypodense right lentiform nucleus (a, arrow). PCT demonstrates a prolonged time-to-maximum (Tmax) within the right MCA territory (b, arrow). Maximum projection images from CTA demonstrate occlusion of the M1 segment of the right MCA (c, arrow). Patient 2 (d–f). PCT head demonstrates a small core infarction in the right corona radiata as an area of markedly reduced CBF (d, arrow) and a prolonged Tmax within the right MCA territory (e, arrow). Maximum projection images from CTA demonstrate occlusion of the M1 segment of the right MCA (f, arrow). Patient 3 (g–i). DWI identifies a small core infarction in the left lentiform nucleus (g, arrow). There is prolonged Tmax within the left MCA territory on PMRI (h, arrow). Maximum projection images from MRA demonstrate occlusion of the M1 segment of the left MCA (i, arrow).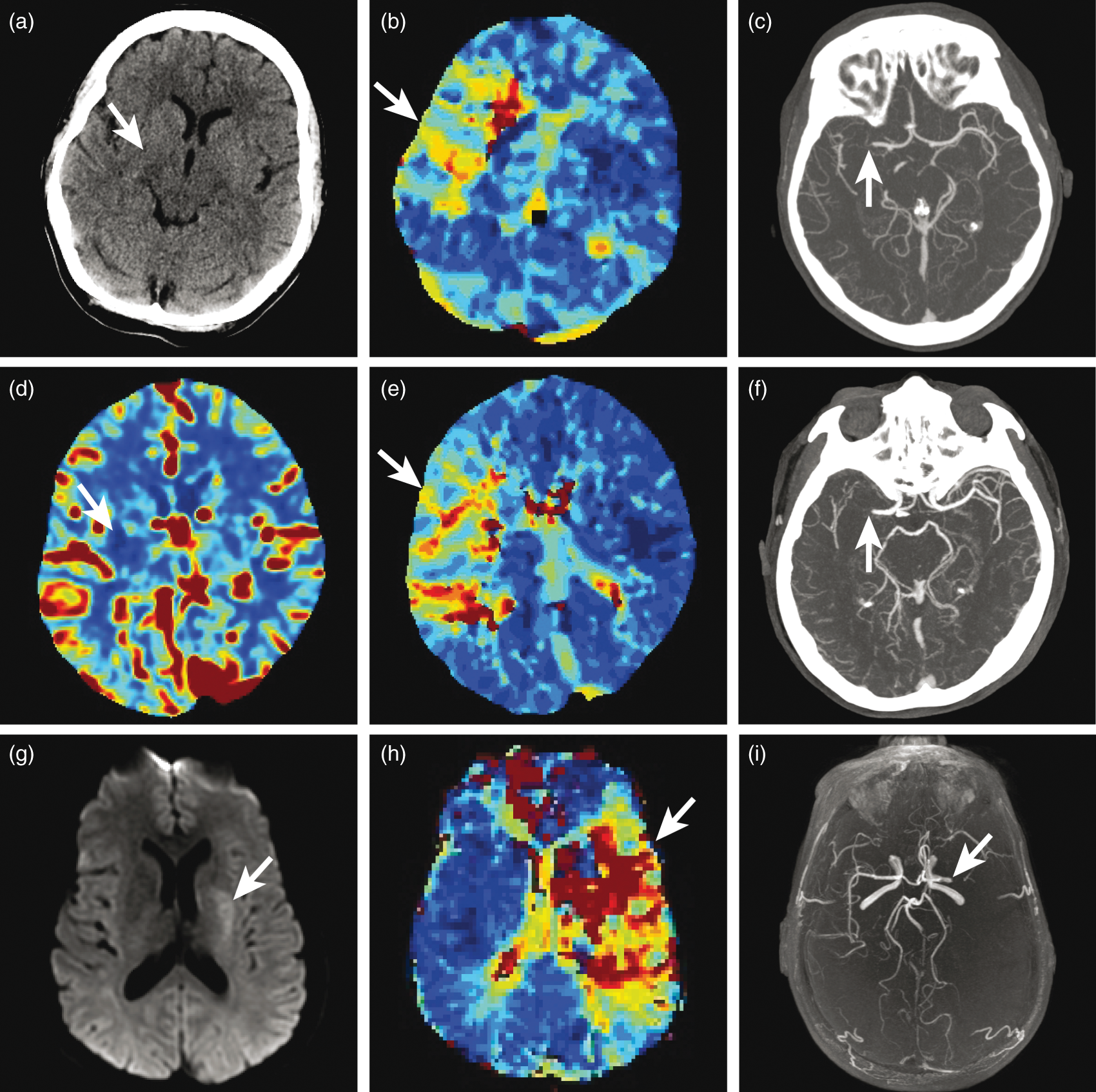
There remains significant variation and controversy surrounding the best method by which the volume of salvageable tissue should be estimated. There are some who argue that time is of the essence in acute ischemic stroke, and that additional imaging designed to characterize the salvageable brain tissue (“penumbra”) results in unnecessary delays to treatment. This philosophy fits within the imaging selection criteria of the MR CLEAN and REVASCAT trials in which only a non-contrast head CT or CTA were required before consideration of treatment.6,8 In this imaging selection model, the size of the penumbra is estimated as a mismatch between the estimated core of infarction on the non-contrast head CT and the clinical stroke severity, which is most often measured by the National Institutes of Health Stroke Scale (NIHSS) score. Patients without a large core infarction who have a high NIHSS must have salvageable tissue and should be considered for endovascular stroke treatment. This strategy was effective in MR CLEAN and REVASCAT, and it resulted in a good clinical outcome in 33% (MR CLEAN) and 44% (REVASCAT) of patients who underwent endovascular therapy compared with only 19% (MR CLEAN) and 28% (REVASCAT) of patients in the medical management arms.6,8
By contrast, others have argued for a more stringent neuroimaging inclusion criteria prior to consideration for endovascular treatment. The two techniques most commonly performed for additional evaluation of endovascular stroke candidates include (1) collateral imaging using multiphase CTA, PCT, or PMRI and (2) penumbral imaging using PCT or perfusion MRI (PMRI) techniques. Robust collateral vessels allow for increased blood flow to ischemic brain tissue, which limits the size of the core infarction and maintains the viability of the penumbra. The ESCAPE trial included patients with moderate-to-good collaterals, which were assessed by CTA or multiphase CTA. 7 In ESCAPE, a good clinical outcome was achieved in 53% of the endovascular arm compared to 29% of the medical management arm, 7 and a higher rate of good outcomes in the endovascular arm were achieved compared to the MR CLEAN and REVASCAT trials.6,8 These data suggest a benefit of collateral imaging in selecting patients for endovascular therapy.
The SWIFT PRIME and EXTEND-IA trials performed PCT or PMRI and quantified the ischemic penumbra as the volume of tissue with a time-to-maximum delay of greater than six seconds using the automated RAPID software.4,5,114 The use of penumbral imaging in these trials resulted in a good clinical outcome in 60% (SWIFT PRIME) and 71% (EXTEND-IA) of patients who underwent endovascular therapy compared with only 35% (SWIFT PRIME) and 40% (EXTEND-IA) of patients in the medical management arms.4,5 Thus, the results of SWIFT PRIME and EXTEND-IA also suggest that a more rigorous neuroimaging selection leads to better outcomes when patients undergo endovascular stroke treatment.
The results of the recent five randomized stroke trials underscore both the importance of neuroimaging in selecting patients for treatment and the need for ongoing research to determine the most optimal imaging algorithms for patient triage and to eliminate futile procedures. Patients being considered for endovascular therapy at minimum require an assessment of the size of core infarction and non-invasive vascular imaging to confirm the presence of a large vessel occlusion. Additional imaging designed to understand better the patient's cerebral perfusion using collateral or penumbral imaging may offer additional benefit in selecting patients who are most likely to benefit from endovascular stroke therapy. Among the recent trials, there was a trend toward better outcomes in studies with more rigorous neuroimaging selection for enrollment, although other confounding variables might also explain this difference. Future studies should seek to determine the best neuroimaging algorithm for the selection of endovascular stroke candidates. These studies would be bolstered by studies designed to estimate the cost-effectiveness of collateral and penumbral imaging based upon the recent trial results.
Neuroimaging prediction of outcome after ischemic stroke
The importance of non-invasive brain imaging is not restricted to the acute evaluation of patients with ischemic stroke. Imaging studies obtained both before and after ischemic stroke treatment provide prognostic information and the likelihood of achieving a good clinical outcome. As the number of patients undergoing endovascular stroke treatment increases, we anticipate an increased role of neuroimaging in the prediction of patient outcome. Here we briefly review emerging areas of stroke outcomes imaging research.
The size of cerebral infarction has long been known to predict patient outcome, and patients with large cerebral infarctions have a worse prognosis, which may be measured at presentation or within days of presentation.109,115–117 More recent studies focusing on structural imaging after ischemic stroke have found that the location of infarction is important in predicting patient outcome. Patients with infarction involving eloquent tissue (motor and language regions in particular) are more likely to have a poorer clinical outcome than those with infarctions involving less eloquent tissue. 118 Similarly, preservation of the corticospinal tract has been shown to be an important predictor of motor function recovery. 119
Diffusion-tensor imaging (DTI) continues to evolve as a prognostic tool for recovery after an ischemic stroke. DTI measures fractional anisotropy in the major neuronal tracts of the brain, and it may characterize disruption of these tracts following ischemic stroke. Injury to the corticospinal tract from direct ischemia or due to Wallerian degeneration correlates with poor recovery of motor function when measured by DTI weeks to months after presentation.119–123 Interestingly, similar injury to the corticospinal tract on DTI in the acute setting does not predict motor outcome, which suggests some recovery of the corticospinal tract is possible. 124 Similar to the corticospinal tract, decreased fractional anisotropy in the superior longitudinal and arcuate fasciculi correlates with language deficits following ischemic stroke. 125 Large prospective studies validating neuronal tract and connectivity network disruption as predictors of patient outcome are needed.
The functional connectivity between neural networks may also be assessed by resting-state functional-MRI (rs-fMRI). This technique measures changes in regional CBF using blood oxygen level dependent (BOLD) imaging, and it may be used to demonstrate activation of compensatory networks after ischemic stroke. 126 rs-fMRI shows promise as prognostic tool after ischemic stroke, but additional studies are needed to understand the significance and prognostic importance of changes in neural networks. It will also be of interest to learn whether changes in resting-state functional networks may reveal activation of inhibitory pathways that may impair recovery after stroke. 127 The identification and understanding of such inhibitory pathways would represent additional targets for therapies designed to improve patient recovery after stroke.128,129
We expect that these and other neuroimaging techniques will become more established predictors of outcome after ischemic stroke. 56 Prospective and randomized trials are needed to validate these techniques for prognostication. Additionally, validation of imaging biomarkers that predict outcome after stroke may also be used as surrogate endpoints for trials designed to test new therapeutic approaches, which has the potential to make clinical trials more efficient and require less lengthy and expensive patient follow up.
Future directions in ischemic stroke treatment
The recent endovascular stroke trials have led to a rapid change in ischemic stroke treatment in a short period of time, and the results of these trials have generated many important questions that require additional study. Future studies to compare and refine imaging selection algorithms for endovascular stroke treatment are needed. The stroke healthcare provider and research community must continue to discuss and debate the practical, and cost-effectiveness of various acute ischemic stroke imaging protocols, which has important implications for patients and the increasingly expensive USA healthcare system.
The recent endovascular stroke trials showed an overwhelming benefit of treatment for patients presenting within 6 h of symptom onset. The REVASCAT and ESCAPE trials also enrolled patients presenting up to 8 and 12 h after onset, respectively, but these studies were not powered to determine endovascular treatment effectiveness in these later time windows.7,8 Therefore, it remains uncertain whether patients presenting in later time windows may also benefit from endovascular treatment. The Endovascular Therapy Following Imaging Evaluation for Ischemic Stroke 3 (DEFUSE 3) and the Trevo and Medical Management Versus Medical Management Alone in Wake Up and Late Presenting Strokes (DAWN) trials are two randomized trials investigating this question. Both trials are randomizing patients to endovascular stroke therapy versus medical management 6 to 16 h (DEFUSE 3) or 6 to 24 h (DAWN) after symptom onset, and both trials are using vascular imaging and PCT or PMRI with a target mismatch profile for patient inclusion. Similarly, the randomized WAKE-UP trial is asking whether patients with an unknown time of symptom onset benefit from intravenous thrombolysis. 130 Patients eligible for enrollment in the WAKE-UP trial have an MRI that demonstrates a core infarction to fluid attenuation inversion recovery (FLAIR) signal mismatch, and patients with this profile are randomized to intravenous thrombolysis with tPA compared to placebo. 130
The results of these trials will be of significant interest given the potential to extend significantly the treatment window for patients with ischemic stroke. Additional studies have correlated PCT and clinical data in an effort to characterize better the penumbra in late time windows. 131 Thus, the ongoing randomized stroke trials and efforts to combine imaging and clinical data are testing a personalized medicine approach for ischemic stroke treatment triage by using physiologic PMRI or PCT imaging in their inclusion criteria, and it will be of interest to see if this imaging approach is validated in these later time windows.
Conclusions
The endovascular stroke trials of 2015 have led to a sea change in the treatment of patients with acute ischemic stroke in a very short period of time. Modern endovascular stroke therapy coupled with the recent advances in stroke prevention and stroke imaging advances promises to continue to decrease the morbidity, mortality, and frequency of ischemic stroke. Ongoing and future studies will optimize the imaging selection of stroke patients for endovascular therapy and possibly extend the time window for treatment based upon a personalized medicine imaging approach.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
