Abstract
A potential two-way passage of cells and substances between the brain and skull bone marrow may open for new insights into neurological disease. The arachnoid membrane was traditionally considered to restrict cells and larger molecules in CSF from entering the dura and bone marrow directly. However, new data on exchange between brain and skull bone marrow have recently emerged. Here, we conducted a systematic literature to answer the question: What is the current evidence regarding the movement of cells and molecules between the brain and skull bone marrow, spanning CSF and meninges? We excluded studies related to head or skull trauma, cranial fractures or defects, cancer invasion, CSF leakage, spontaneous intracranial hypotension, spinal dura mater, and studies solely focusing on meningeal lymphatic vessels or the passage of substances from CSF to meningeal lymphatic vessels. The review identified 16 studies that provide evidence of communication between the brain, meninges and skull bone marrow. Cells (such as B and T cells and neutrophils), bacteria, and substances (tracers, drug compounds) have been reported to pass between the brain and skull bone. However, most studies are performed in rodents, emphasizing the need for translation to humans.
Introduction
Leveraging groundbreaking discoveries made in 2015,1,2 which unveiled the presence of functional meningeal lymphatic vessels capable of transporting substances from the cerebrospinal fluid (CSF) to extra-cranial lymph nodes, a profound resurgence of interest has emerged regarding the organization and functional properties of the brain meninges and skull bone marrow. There is a growing interest how the brain may interact via CSF with the peripheral and central immune system at brain borders (subarachnoid CSF, brain meninges and skull bone marrow), and its role in immunosurveillance of the central nervous system (CNS) and clearance of waste substances from the CSF. At the same time, there is an ongoing debate about the anatomical and molecular organization of CNS barriers, the compartmentalization of the subarachnoid space, and the extent to which meninges function as impermeable or semipermeable barriers for cells and solutes.3 –6
Traditionally, the arachnoid and dural meninges were deemed largely impermeable barriers to passage of substances and cells.4,7,8 However, experimental studies have reported the passage of immune cells from the CSF to the meninges,1,2,9 or vice versa,10,11 with CSF governing immune cell migration from skull bone marrow to the dura. 12 Critics argue that these studies may overlook considerations of the properties of the arachnoid mater, forming a barrier between the subarachnoid space and the dura mater. 4
From a clinical perspective, human studies utilizing intrathecal magnetic resonance imaging (MRI) contrast agent (gadobutrol) as a CSF tracer have revealed brain-wide extravascular tracer enrichment, 13 as well as tracer enrichment in parasagittal dura along the superior sagittal sinus, 14 skull bone marrow, 15 and extracranial lymph nodes 16 (see Figure 1). These observations suggest the passage of the tracer from the subarachnoid CSF space across the meninges to the skull bone marrow and extracranial lymph nodes, challenging the previous notion of arachnoid impermeability. Critics, on the other hand, may contend that tracer enrichment in various tissues after intrathecal administration is due to recirculation of the CSF tracer through the vascular compartment. An argument against this is that the tracer is administered in a low dose (0.25–0.50 mmol), with very limited passage into the vasculature over the intact blood-brain-barrier, and tracer concentrations in blood at µmol level 17 are unlikely detected at MRI.
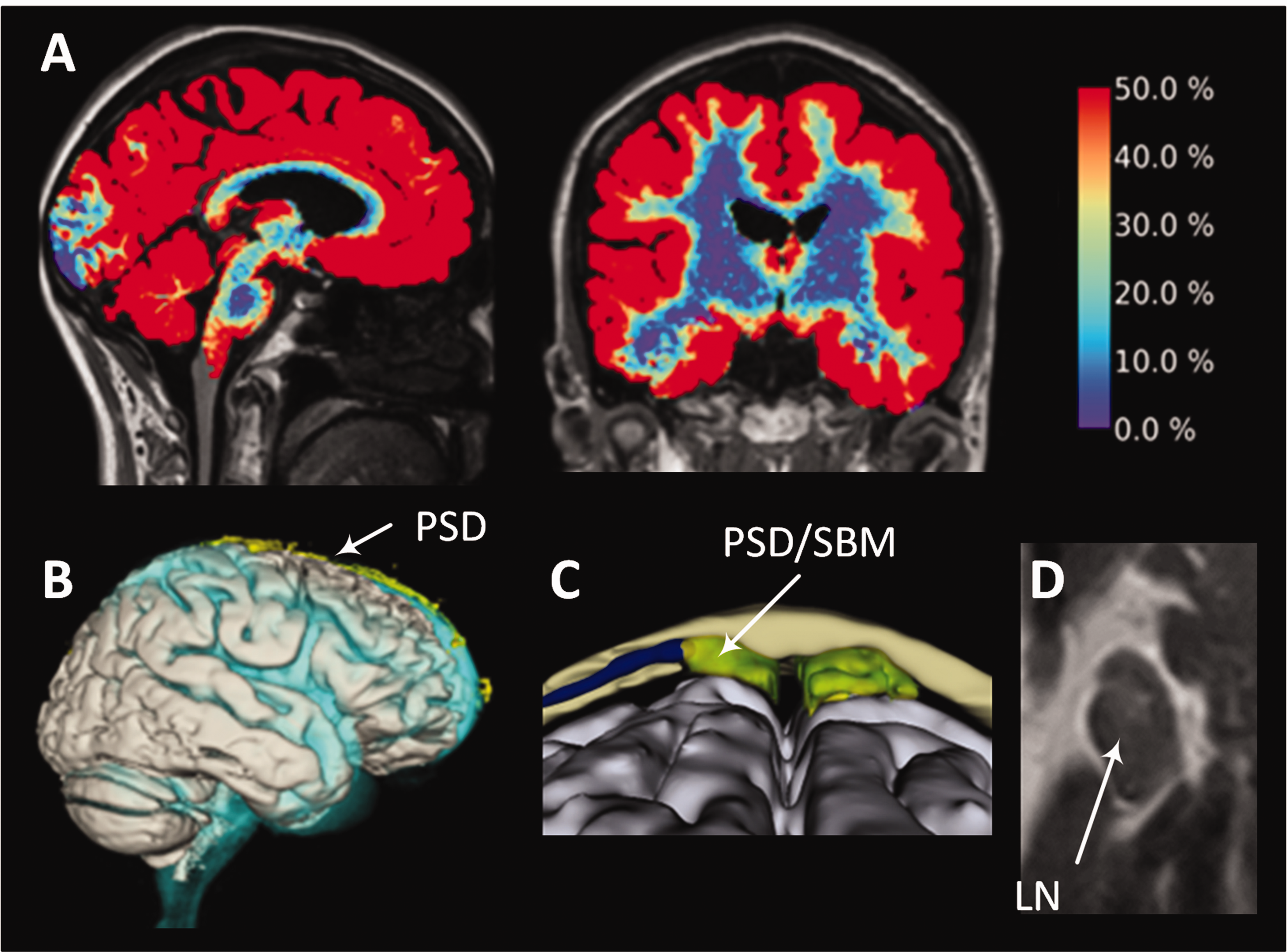
An intrathecal tracer distributes within brain, parasagittal dura, skull bone marrow and extracranial lymph nodes in humans. Following the intrathecal injection of an MRI contrast agent (gadobutrol, with a molecular weight of 604 Da) as a cerebrospinal fluid (CSF) tracer, the distribution is observed in (A) the entire brain 13 (color bar on the right indicating the percentage increase in tracer enrichment after about 24 hours). Additionally, the tracer disperses within (B) the parasagittal dura (PSD) near the superior sagittal sinus, 14 (C) PSD within skull bone marrow (SBM), 15 and tracer enrichment was seen within (D) extracranial lymph nodes (LN).16 These tracer studies consistently reveal that the peak tracer enrichment in various compartments typically occurs around 24 hours, suggesting a prolonged process.
Given the present controversy, this study addressed the question about the transport of cells and solutes between the brain and the skull bone marrow across the meninges. We systematically reviewed the literature, asking: What is the current evidence for passage of molecules and cells between the brain, CSF of subarachnoid space, brain meninges, and the skull bone marrow? For this purpose, we excluded studies addressing traumatic head injuries, skull fractures, or spontaneous cerebrospinal fluid (CSF) leaks, all potentially causing abnormal passage of cells and molecules between the brain and skull bone marrow.
Materials and methods
Data sources
A systematic search was conducted utilizing the following bibliographic databases: EMBASE (Ovid), MEDLINE (Ovid), The Cochrane Library. The search was limited to English articles and included publications from any date. Unpublished studies, gray literature (e.g., reports, white papers, and newsletters), abstracts and conference presentations were excluded.
We formulated the following research question initiating the literature search: What is the current evidence for passage of molecules and cells between brain, CSF and skull bone marrow across dura mater in the absence of traumatic or non-traumatic dura leakage?
Inclusion criteria were: English language, human and animal studies, and studies addressing the crosstalk of cells and substances between the brain, CSF, and cranial structures across the meninges.
Exclusion criteria were: Head and/or skull trauma, head injury, post-traumatic brain injury leakage, cranial fracture, cranial and/or calvarial defect, cranial defect repair, dura-bone formation, dural impact on bone formation and healing, cranial fracture healing, cancer invasion, CSF/dura leakage, spontaneous intracranial hypotension, spinal dura, and vertebral lymphatic vessels. Studies addressing the passage of cells between anatomic structures during embryogenesis were not considered. Additionally, studies solely focusing on meningeal lymphatic vessels or the role of meningeal lymphatic drainage failure in neurological disease were excluded.
The full search strategy is presented in the Supplementary Table 1.
Study selection
The search team comprised all four authors. The first author (HET) conducted the initial search and selection, which was subsequently reviewed by the other authors RE, PKE, and GR. A digital library was established using EndNote and shared among the authors.
Data extraction
The data extracted included 1) Methods used (including species studied, method for estimating crosstalk between the brain, CSF, dural meninges and skull bone marrow, 2) Anatomical evidence for cross-talk between CSF and skull bone marrow at meninges. 3) Functional evidence for cross-talk, i.e. evidence for passage of either cells or molecules between the brain, CSF and skull bone marrow across the meninges. We distinguished between animal and human studies.
Results
Study selection
Figure 2 presents a PRISMA flow diagram 18 of the review (http://www.prisma-statement.org/PRISMAStatement/PRISMAStatement.aspx). The final search was conducted on January 11th, 2024. After eliminating duplicates, 663 publications were initially identified. Following the application of criteria, 647 publications were excluded, resulting in 16 selected publications. Among these, 10 were animal studies, 3 were human studies, and 3 encompassed both animals and humans. The search strategy employed in Medline, Embase, and the Cochrane Library is outlined in Supplementary Table 1, and the sixteen included references are provided in Supplementary Table 2.
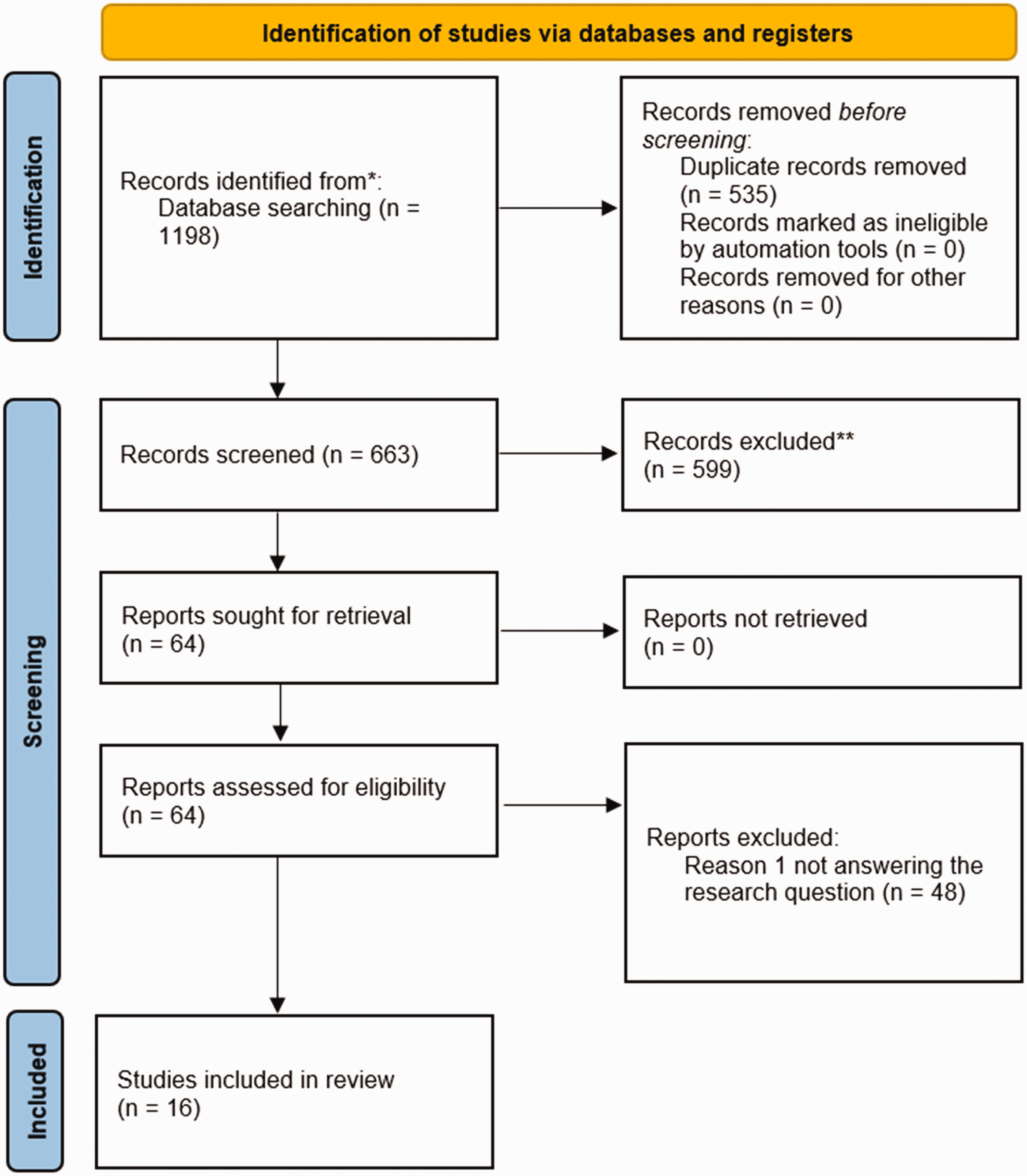
PRISMA. Flow diagram of included studies. 18
Results from animal studies are detailed in Table 1, while human study outcomes are presented in Table 2. Additionally, an overview of the results is depicted in Figure 3.
Evidence from animal studies of cellular and solute passage between brain, CSF and skull bone marrow across meninges.

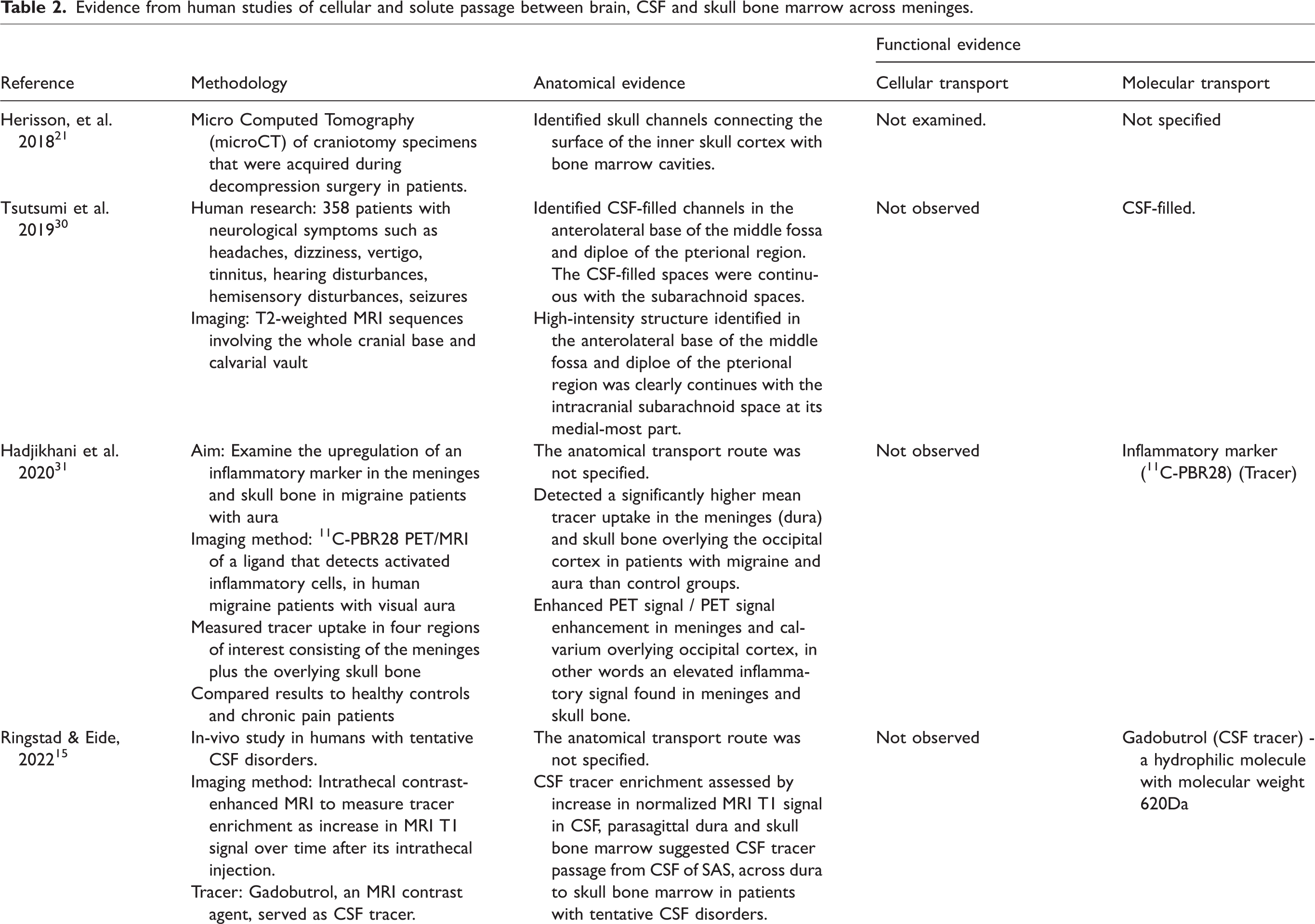
Evidence from human studies of cellular and solute passage between brain, CSF and skull bone marrow across meninges.
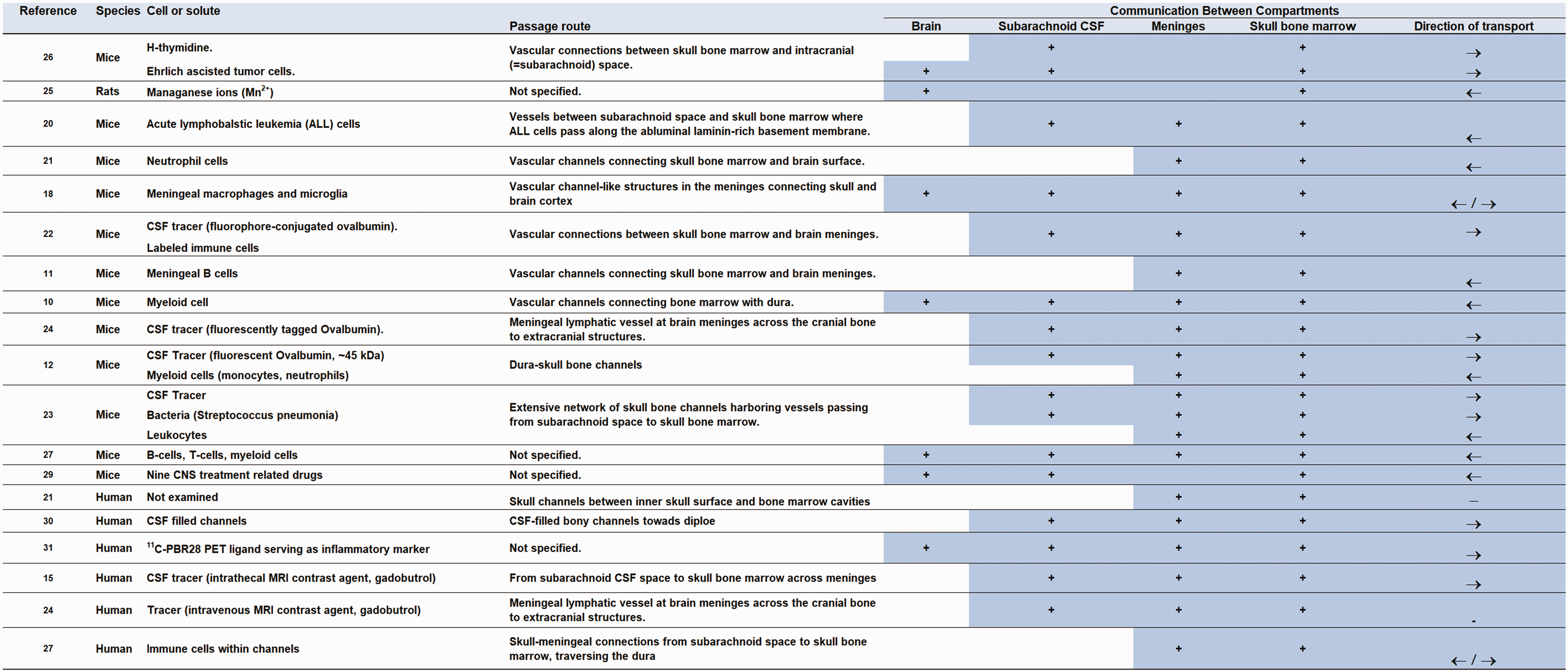
Overview of studies included in the systematic review. The literature review differentiated between animal and huma studies. → indicates transport in direction from subarachnoid space towards skull bone marrow. ← indicates transport in direction from skull bone marrow towards subarachnoid space.
Animal studies
Anatomical evidence
Regarding the anatomical route for potential cell and solute passage, studies in rodents have demonstrated skull-meningeal channels, acting as bridges between meninges, the subarachnoid space, and skull bone marrow.10 –12,19 –24 Moreover, channels in the calvarial bone have been shown to harbor vessels crossing from the dural meninges to the skull bone marrow.20,23,24 Pulous et al. 23 estimated that in an adult mouse, over 1,000 bone channels reach into the cranial vault. Experimental evidence suggests that cells and solutes in the CSF pass outside vessels traversing the bone marrow cavities, meninges, and the subarachnoid space.12,19,20,23,24 Notably, CSF tracers with molecular weights ranging from 66 kDa to 2,000 kDa appeared along a subset of dural vessels, providing evidence of passage of large molecules between subarachnoid CSF spaces and skull bone marrow through an extensive skull channel network. 23 Among the identified studies, eight10 –12,21 –23,25,26 provided evidence for cell and solute passage between the meninges and skull bone, five12,20,23 –25 between the subarachnoid space and the skull bone, and three between the brain cortex, meninges, and skull bone.10,24,26
To the best of our knowledge, a study in 1993 24 first demonstrated meningeal vessels passing through openings or channels in the skull bone that expand into sinusoids within the bone marrow cavity. In this study, histological images of the brain with attached sphenoid bone showed the existence of vascular connections or canals between what the authors denote as “intracranial” space and the skull bone marrow. 24 However, the microvessels claimed to bridge the subarachnoid space and skull bone marrow were not directly visualized. More recently, others utilized a light-sheet microscopy methodology to identify “vascular-channel-like structures” in the meninges connecting the brain cortex with the skull bone, 19 and meningeal lymphatic vessels penetrating the skull via openings at the skull base. 25 In vivo imaging provided evidence that cellular and tracer transport between CSF and skull bone marrow in the dura-skull channels occur along perivascular spaces of the bridging vessels.12,23 One study observed meningeal B-cell migration through skull vascular channels from the calvaria to the meninges. 11 Another investigation showed neutrophil migration through channels connecting the skull bone marrow with the dura. 21 A third study reported myeloid cell infiltration from skull bone marrow to brain meninges and brain parenchyma during CNS injury through skull-dural channels. 10 While not demonstrated, these authors speculated that myelomonocytic cells pass along vessels residing within channels connecting skull bone marrow with dura.
The methods used to study the passage of substances between the CSF and skull bone marrow included injections of tracers into the CSF12,22,23,25 and administering bacteria to the cisterna magna. 23 A common tracer is fluorescent-tagged ovalbumin, which has a molecular weight about 45 kDa. Moreover, mouse models of brain disease or injury10,19,21,22 were applied to study the migration of leukocytes from skull bone marrow to the meninges and/or brain parenchyma. Another approach was in vivo cell labeling of bone marrow cells in the mouse skull and confocal plus electron microscopy of channels connecting the skull and dura. 21 Two studies performed histological cross-sections of the mice brain with adjacent skull bone.20,24 Specific nano-labeling was used to enhance signal of fluorescent proteins, thereby allowing a high degree of transparency through the mouse skull and the opportunity to visualize subcellular details, including vascular channels between the skull and brain meninges. 22
In summary, several studies provide evidence for channels between the dura mater and skull bone marrow that harbor vessels where the bony perivascular space may serve as a conduit for cells and solutes.
Functional evidence
Studies examining the functional aspects of crosstalk between brain, CSF, meninges and skull explored the transport of molecules and cells. Srebro et al. 24 reported that H-thymidine, injected intrathecally in mice, penetrated into the sphenoid bone marrow from the arachnoid space and labeled leukocytes in the skull bone marrow. 24 More recently, two studies12,23 showed that a CSF tracer administered to the cisterna magna passed into the skull bone marrow. Administering a tracer to the CSF revealed tracer uptake in meningeal lymphatic vessels that penetrated the skull through bony openings. 25 On the other hand, external application of manganese chloride (MnCl2) to an exposed skull bone of rats, showed diffusion of manganese (Mn2+) ions through the skull bone into the brain, detectable by MRI. 26 However, this study did not define the anatomical route other than that the manganese ions penetrates the meninges to enter the cerebral cortex.
Several studies reported the exchange of cells between CSF and the skull bone at the meninges. Six studies observed myeloid and lymphoid cell migration from the skull bone marrow to the brain meninges in mice, including meningeal B-lymphocytes,11,27 T-lymphocytes, 27 monocytes,10,12 macrophages and neutrophils. 12 Cugurra et al. 10 provided evidence that the skull bone marrow supplies monocytes to the meninges and brain parenchyma both in health and disease. They reported that following brain injury, monocytes derived from skull bone marrow infiltrate the brain through channels in skull bone marrow and dura. In another study, meningeal B-cells migrate from the calvaria to the meninges through vascular channels. 11 In addition, neutrophil cells were found to migrate through direct vascular channels, which cross the inner skull bone, connecting the skull bone marrow spaces with the dura mater; 21 however, passage of cells between dura mater and subarachnoid space was not addressed. Okonogi et al. 28 reported that cranial irradiation could induce bone marrow derived microglia (BMDM) in adult mouse brain. The exact passage route was not defined in this study, and the authors speculated about passage via a disrupted blood-brain-barrier. One of the included articles reported migration of bacteria (streptococcus pneumonia) from the meninges to the skull bone marrow after the injection of bacteria in the cisterna magna of mice. 23
Yao et al. 20 reported migration of acute lymphoblastic leukemia (ALL) cells into the brain along vessels passing directly between the skull bone marrow and the subarachnoid space. The vessels were found to cross through bony channels of the skull, connecting dura-skull bone, enabling direct communication between skull bone marrow and the subarachnoid space. 20 It is well established that ALL cells fail to breach the blood-brain-barrier (BBB), and that metastasis from ALL seldom involve the brain parenchyma but restricted to the leptomeninges. It is therefore notable that the basement membrane of the bridging vessels was found rich in laminin, and that the perivascular passage of cells was mediated by α6 integrin-laminin interactions. This could become a therapeutic target since most ALL cases express laminin receptor α6 integrin and blockade of this receptor was found to inhibit the perivascular tumor cell transport.
Another approach to study the possible therapeutic potential of connections between skull bone marrow and CSF of the subarachnoid space was explored by Kang et al. 29 who administered nine different drugs within the skull bone marrow (intracalvariosseous), measured brain concentrations and performed quantitative pharmacokinetic analysis. The nine tested drugs gave substantially higher brain concentrations (minimum 2-fold to maximum 342-fold) than the corresponding intravenous administration. The authors concluded that the transport route was extravascular rather than intravascular via vascular channels/connections between the skull bone marrow and the meninges. 29
Human studies
Anatomical evidence
Four of the human studies provided anatomical evidence for a connection between the skull bone marrow and the meninges or subarachnoid space21,25,27,30 (Table 2). Tsutumi et al. 30 observed CSF filled channels continuing with the subarachnoid spaces in the middle fossa, and Herrison et al. 21 identified in both mice and humans skull channels crossing the inner skull cortex from the skull bone marrow to the dural meninges. Jacob et al. 25 observed what they interpreted as perforating meningeal lymphatic vessels and slow-flow channels crossing the skull, thus providing a link between the subarachnoid space, dura and calvarial bone. A recent study characterized skull-meninges-connections at the cellular level, bridging the dura mater with the skull bone marrow. The study was conducted on human skull-dura postmortem samples. They observed that the skull-meninges-connections transverse the dura mater, opening to the subdural space underneath to arachnoid granulations in humans. 27 These authors quantified over 500 skull-meningeal connections, and found that they are 40–90 µm wide, and occasionally >150 µm wide. They further found that PDGFR-B signal was present at both the vessels and the skull-meningeal connection lumen, indicating that the lumen of the skull-meningeal connections is lined by fibroblastic cells that are known antigen-presenting cells. In addition to the fibroblastic cell layer, immune cells were present.
Functional evidence
Four human studies provided functional evidence of passage between the skull bone and the subarachnoid space; three studies examining tracer transport,15,25,31 and one revealing channels harboring cells 27 (Table 2). These studies primarily provide evidence for transport of solutes.
The first study to demonstrate efflux of CSF into skull bone marrow of any species utilized an MRI contrast agent as CSF tracer, enabling visualization of tracer enrichment between subarachnoid CSF and the skull bone marrow across the meninges in a patient group. 15 Another study examined upregulation of an inflammatory marker in meninges and skull bone in human patients with migraine with visual aura. Using 11C-PBR28 PET/MRI with the PET ligand 11C-PBR28 serving as an inflammatory marker, they found that migraine patients with aura had significantly higher tracer uptake (i.e. increased inflammatory signal) in the meninges and the adjacent skull bone overlying occipital cortex than control groups. 31 The authors speculated that the meningeal/calvarial inflammation was caused by direct connections between the cerebral cortex and meninges and skull bone, but the study did not address the substances or passage route involved.
Discussion
This systematic literature review revealed studies providing evidence for 1) anatomic bony channels between the dura and the skull bone marrow, 2) blood vessels residing within these bony channels linking the subarachnoid space with the skull bone marrow, 3) passage of cells and solutes along the outside of these vessels, and 4) evidence for bidirectional transport, for example, CSF moving from subarachnoid space to the skull bone marrow and cells moving from the skull bone marrow to the subarachnoid space. Most data are derived from animal experiments, emphasizing the need for confirmation in humans.
The research field is young, as most of the literature has emerged since 2015. An exception is the 1993 study by Srebro et al., 24 providing the first evidence of direct vascular connections between skull bone and the brain.
Transport capacity
An unresolved question is how much cells and solutes exchange between subarachnoid CSF spaces, dura and skull bone marrow – that is, what is the transport capacity? This likely depends on the region. Conversely, considering that CSF is continuously presented to the skull bone marrow and possesses the ability to instruct cellular responses, 12 the skull bone marrow may play a pivotal role in regulating CNS immune responses. Consequently, previously the importance of CSF as a messenger to instruct inflammatory responses of the CNS seem to have been underestimated. CSF may thus be essential for CNS immunosurveillance and waste clearance. In this regard, waste clearance via meningeal lymphatic structures may be of importance in Alzheimer’s disease (clearance of amyloid-β and tau)32 –34 and in Parkinson’s disease (clearance of α-synuclein).35,36
Concerning translation to humans, a major outstanding question revolves around methods for visualizing the passage of substances across the brain borders. Currently, the use of MRI to identify lymphatic vessels in human dura mater remains highly debated.37 –40 Caution is recommended to avoid over-interpretation of structures interpreted as “lymphatic vessels” at MRI, 25 as they may instead represent various types of structures in the dura, such as veins, a dense network of capillaries, liquid lakes, intradural arachnoid granulations, and dural stroma.41,42
Efflux routes
A critical inquiry pertains to how cells and solutes traverse the arachnoid, given prior evidence of arachnoid barrier cells forming an impermeable obstacle to substances within the subarachnoid CSF. 4 Smyth et al. 43 recently identified gaps in the arachnoid barrier cell layer where bridging veins from cerebral cortex enter the dura mater. These structures, termed arachnoid cuff exits (ACE), encircle veins that penetrate the arachnoid barrier cell layer, providing a conduit for cells and substances between subarachnoid CSF and the dura mater. Notably, arachnoid border cells were labeled by Prox1-eGFP, which also marked the so- called subarachnoid lymphatic-like membrane (SLYM), posited as a fourth brain meninges alongside the pia mater, arachnoid mater and dura mater. 3 Prox1 was also recently found to label a fibroblast layer within the subarachnoid space,4,44 though conflicting evidence has been provided regarding the existence of the SLYM and a compartmentalized subarachnoid space.3,4 On the other hand, the hypothesis of a non-compartmentalized subarachnoid space 4 is challenged by human in vivo observations of a perivascular subarachnoid spaces (PVSAS) that facilitates the passage of tracer within the subarachnoid space and along the arteries toward the brain. 6
Emerging evidence indicates that substances within CSF may traverse along vessels from the subarachnoid space to the dura mater. The molecular composition of the abluminal part of vessels, currently emphasizing laminin and α6 integrin interactions, appears crucial for the perivascular passage of cells and substances from CSF towards the dura and skull bone marrow (refer to Figure 4). α6 integrin-laminin interactions for abluminal passage of ALL cells between the skull bone marrow and subarachnoid CSF. 20 In this regard, it is noteworthy that laminin staining was intense around the ACE points, 43 and the authors proposed that laminin and α6 integrin interactions are pivotal for trafficking cells, including myeloid cells, to the subarachnoid space at ACE points. This perspective aligns with observations of intrathecal CSF tracer passing from the subarachnoid space to the dura along vessels (perivascular), through skull bone channels to skull bone marrow cavities. 23
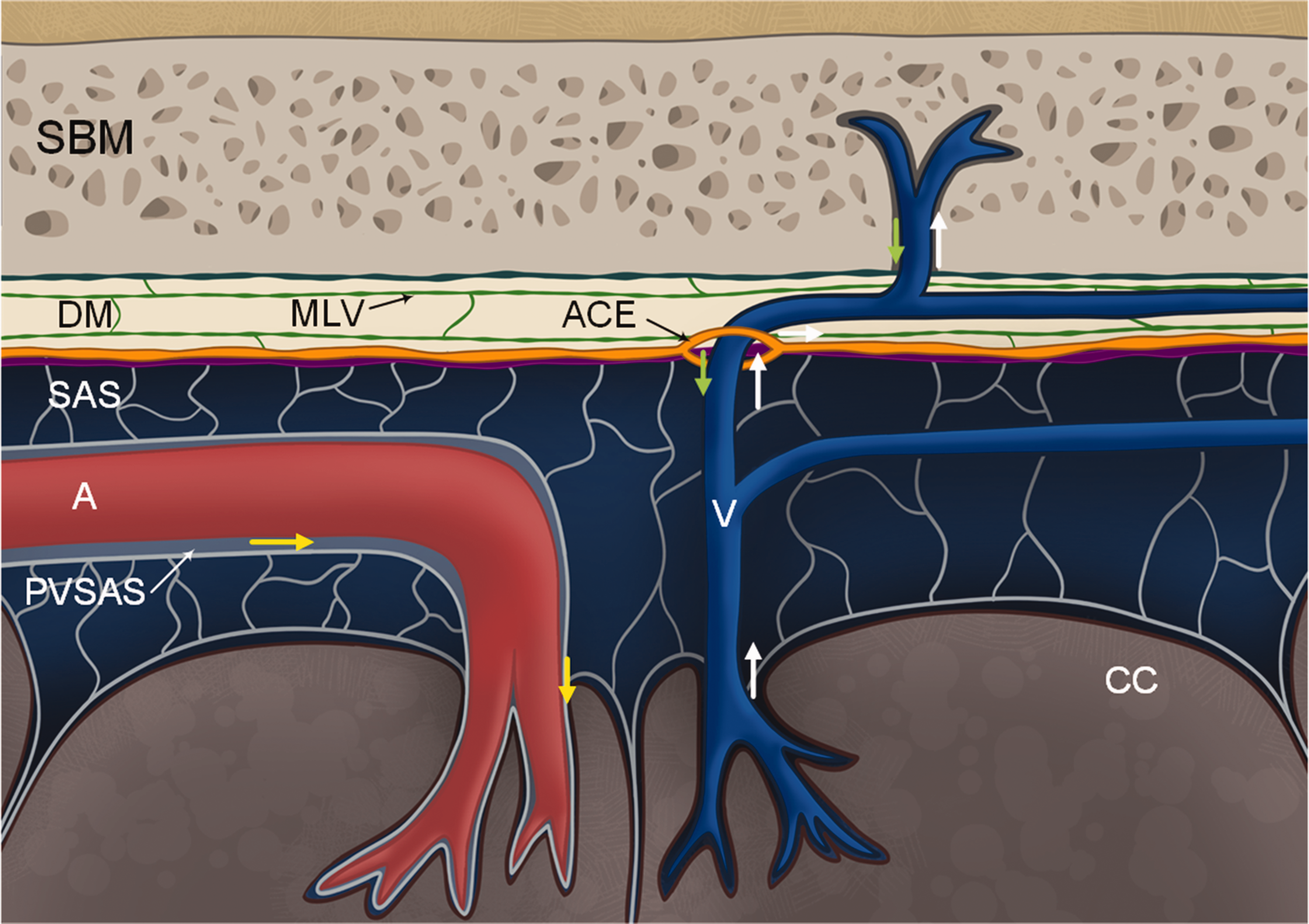
Cartoon illustrating current evidence derived from the literature review regarding the potential route for passage of cells and solutes from subarachnoid CSF space to skull bone marrow across meninges. The review highlights numerous studies demonstrating the existence of vascularized bone channels in the skull bone, extending from the skull bone marrow (SBM) to the dura mater (DM).10 –12,19 –24,27 Blood vessels within these bony channels traverse the dura mater and extend across the arachnoid membrane to reach the subarachnoid space (SAS).20,23 Recent findings revealed the presence of arachnoid cuff exits (ACE) forming cuffs around vessels, 43 enabling the passage of cells and substances along the arachnoid barrier cells adjacent to the dura border cells. Research indicates that the basement membrane of vessels serves as a transport pathway for cells and substances.20,43 This basement membrane is notably rich in laminin, which may interact with α6 integrin, enabling transport of ALL cells from skull bone marrow to the subarachnoid space. 20 Remarkably, molecules of diverse mass and properties, including dextrans with a molecular weight of 70,000 kDa, can traverse this pathway. 43 Within the dura mater, meningeal lymphatic vessels are present, and notably, there is no blood-brain-barrier within the dura. It is further indicated that CSF influx (yellow arrows) occurs along perivascular subarachnoid space (PVSAS) 6 towards cerebral cortex (CC), CSF efflux (white arrows) along veins, and via ACE to dura and skull bone marrow. From skull bone marrow cells and substances (green arrow) may pass along vessels within bone channels, across dura to the subarachnoid space. Illustration: Øystein Horgmo, University of Oslo.
Diverse molecules and cells have been shown to pass between brain and skull bone marrow across meninges. These include MRI contrast agents (e.g., gadobutrol, molecular weight 604 Da), 15 large-molecular size compounds such as ovalbumin (45k Da),23,25 dextrans (up to 70,000 kDa), 23 and smaller sized substances like manganese ions (molecular weight about 55 Da), 26 which is comparable to water in size, and H-thymidine (molecular weight about 242 Da). 24 The various cell types studied include myeloid cells,10,12 meningeal B-cells 11 (only from skull bone marrow to the meninges), neutrophils 21 (migrating through channels from skull bone marrow to the dura), macrophages (only from meninges to brain). 19 One study observed bacterial cell migration to the skull bone from brain, 23 and passage of tumor or leukemia cells.20,24 With regard to the recently discovered ACE points, substances of variable molecular masses and properties (including dextrans of 10, 70 and 2,000 kDa, ovalbumin 45 kDa, and 0.5 µm polystyrene beads) passed via these points, indicating bulk flow properties. 43
Understanding and treatment of neurological diseases
Another question is the implications the new knowledge may have for understanding and treating neurological diseases. Since immune cells and substances can be released directly from the skull bone marrow and dura into the CSF, skull bone marrow-derived cells and molecules may participate in brain immune surveillance at the meninges and also be carried into the brain. 15 Additionally, communication between the intracranial compartment and bone marrow may enhance our understanding of common symptoms such as headache during influenza – since viruses can directly infect bone marrow, and now can be hypothesized to inflict an associated inflammation in adjacent meninges or periosteum. 15 Moreover, Hadjikhani et al. 31 showed an elevated inflammatory signal in the overlying meninges and skull bone of the occipital lobe in migraine patients with aura, suggesting that meningeal inflammation contributes to the pathophysiology of photophobia. The authors proposed that the inflammatory signal migrates from the underlying brain to the meninges and bone marrow, indicating cross talk between bone marrow and brain. 31 The skull bone marrow may function as a hub for immune progenitor cells, which, upon contact with antigens from bacteria, may develop, proliferate, and migrate to the meninges and brain parenchyma to perform their functions during infections or brain injuries. A direct pathway may have great significance in neural inflammation. The skull bone channels may function as ducts for bacteria, antigen, and cytokine migration into the skull bone marrow, which then can induce proliferation of white blood cells in the calvarial bone marrow. 23 Additionally, myeloid cells originating from the local skull bone marrow rather than from the blood may serve as a “myeloid” reservoir for meninges and brain. Experimental evidence indicates that CSF may instruct cell activation within the bone marrow, allowing for subsequent entry of leukocytes in the dura and brain tissue during brain infections or after CNS injury. 12
The study conducted by Kolabas et al. 27 observed skull inflammation subsequent to brain inflammation in patients with neurological diseases. They identified a disease-specific increase in TSPO-PET-signal in calvarial regions in patients with Alzheimer’s disease and other neurological diseases. TSPO is a protein significantly upregulated in the brain during neuroinflammation, showing a strong correlation between changes in the skull and brain translocator protein-PET signal in patient with Alzheimer’s disease. These novel findings may establish a link between the skull and neurological diseases in humans, suggesting that TSPO-PET imaging of the skull can reveal distinct signal patterns in inflammatory and degenerative CNS conditions. Could it be that these skull-brain communication channels play an important role in neurodegenerative diseases including Alzheimer’s disease? And could it be that the bone marrow of skull could serve as a novel target in diagnosis of brain diseases?
Another aspect emerging from this review is the potential for future therapeutic approaches. For instance, the study documenting manganese diffusion from the skull into the brain 26 suggests a novel and less invasive route for drug delivery to the brain. A recent study hypothesized that the direct vascular channels between the skull bone marrow and the meninges may also enable drug delivery from the skull bone marrow to the brain. 29 The study reported that selected compounds reached the brain tissue several times higher by intracalvariosseus administration than by systemic application. These findings suggest that administration of CNS drugs into the skull bone could be a new pathway or approach for effective treatment of brain diseases. Moreover, the study conducted in migraine patients with aura, showing an enhanced inflammation signal in the skull bone overlying the occipital cortex, 31 might suggest alternative treatment options for migraine and neuro-inflammatory diseases. Additionally, the findings of a direct communication between bone marrow and CSF enlighten our understanding of the mechanisms underlying autoimmune and neurodegenerative brain disorders and may lead to the development of new therapeutic approaches for diseases in the central nervous system.
Outlook and conclusions
While our primary focus in this review is the interaction between the brain and the skull bone marrow, it is important to note that the vertebral bone marrow also plays a significant role in influencing the spinal cord. Given that CSF circulates throughout the entire craniospinal compartment, this interplay not only affects the brain but also the entire CNS. A recent review highlights that spinal cord trauma in mice results in alterations in CSF composition, which subsequently affects the adjacent vertebral bone marrow. 45 In essence, it appears that CSF carries signals to the vertebral bone marrow, stimulating myelopoiesis and supplying cells to the underlying meninges and spinal cord. These findings offer functional evidence for the interaction between vertebral bone marrow and the spinal cord, although further research is warranted to explore the anatomical evidence of this interaction.From the present literature review, a picture emerges that the brain borders, including the meninges and skull bone marrow, play an essential role in both immune surveillance and waste clearance. Instead of being regarded as separate anatomical entities, the CNS-meninges-skull organ-system should rather be viewed as an anatomical and functional continuum. Since an organ is defined as a collection of specialized tissues that structurally form a functional unit, specialized to perform a particular function, the brain meninges may in this respect by definition be considered as a separate organ.
Taken together, emerging evidence from the last decade has demonstrated existence of vascular bone channels extending from the skull bone marrow to the dura mater. These channels may host vessels that bridge the previously considered impermeable barriers, including meninges, between the brain, subarachnoid space and the skull bone marrow. Recent research has revealed the existence of cuffs or openings around bridging veins, facilitating the passage of even large mass substances from the subarachnoid space to the dura mater. Current evidence suggests that the transport route occurs along the vessel (abluminal), specifically at the basement membrane, where the molecular composition is crucial. This emphasizes the significant role of the abundant expression of laminin within the basement membrane and its interactions with α6 integrin. While several questions persist, the brain meninges now emerge as a specialized structure with distinct functions, particularly in waste clearance and central nervous system (CNS) immunosurveillance.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X251316392 - Supplemental material for Evidence for cellular and solute passage between the brain and skull bone marrow across meninges: A systematic review
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X251316392 for Evidence for cellular and solute passage between the brain and skull bone marrow across meninges: A systematic review by Helena Eide Therkelsen, Rune Enger, Per Kristian Eide and Geir Ringstad in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Ethics approval and consent to participate
Not applicable.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
The authors would like to thank Hilde Iren Flaatten, librarian at the Medical Library at Rikshospitalet – University of Oslo, for developing the search strategy and performing the database searches.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceptualization and Design: HET, RE, PKE GR. Systematic review: HET, RE, PKE GR. Original Draft: HET. Writing, Review and Editing: HET, RE, PKE GR. All authors (HET, RE, PKE GR) approved the final manuscript. Correspondence: PKE.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
