Abstract
The glymphatic-lymphatic fluid transport system (GLFTS) consists of glymphatic pathway and cerebrospinal fluid (CSF) lymphatic outflow routes, allowing biological liquids from the brain parenchyma to access the CSF along with perivascular space and to be cleaned out of the skull through lymphatic vessels. It is known that increased local pressure due to physical compression of tissue improves lymphatic transport in peripheral organs, but little is known about the exact relationship between increased intracranial pressure (IICP) and GLFTS. In this study, we verify our hypothesis that IICP significantly impacts GLFTS, and this effect depends on severity of the IICP. Using a previously developed inflating balloon model to induce IICP and inject fluorescent tracers into the cisterna magna, we found significant impairment of the glymphatic circulation after IICP. We further found that cerebrovascular occlusion occurred, and cerebrovascular pulsation decreased after IICP. IICP also interrupted the drainage of deep cervical lymph nodes and dorsal meningeal lymphatic function, enhancing spinal lymphatic outflow to the sacral lymph nodes. Notably, these effects were associated with the severity of IICP. Thus, our findings proved that the intensity of IICP significantly impacts GLFTS. This may have translational applications for preventing and treating related neurological disorders.
Keywords
Introduction
The brain is a sophisticated organ that requires mechanical and molecular protection from traumatic brain injury (TBI). The cerebrospinal fluid (CSF) in subarachnoid space (SAS) is one of the most crucial support layers by providing buoyancy to maintain fluid homeostasis of the brain and spinal cord. 1 It is generally accepted that the CSF is predominantly produced by the choroid plexus or other tissues (such as the blood-brain barrier) and absorbed by the arachnoid projections (villi or granulations). However, a new CSF flow direction has been proposed recently that a significant portion of CSF enters the brain along with the perivascular spaces and flush through the brain parenchyma, and the pathway was termed the “glia-lymphatic” or “glymphatic” system.2,3 The most fundamental function of the glymphatic system is lymphatic clearance that removes extracellular metabolites and waste products from the parenchyma into the CSF. Studies show that glymphatic system is centrally involved in the clearance of interstitial lactate and β-amyloid (Aβ).2,4,5 Moreover, traumatic brain injury, Alzheimer's disease, type 2 diabetes, subarachnoid hemorrhage, and multiple microinfarctions impair the glymphatic circulation and reduce CSF-interstitial fluid (ISF) exchange, which leads to the accumulation of toxic metabolites in the brain, such as Aβ and other biomarkers of brain injury, and neurobehavioral deficits.2,3,6–12 These adverse outcomes undoubtedly demonstrate the importance of the lymphatic clearance of the glymphatic system.
In addition to the classic model of subarachnoid CSF reabsorption into the dural sinuses via arachnoid projections within the skull, studies have suggested that CSF outflow can also occur through lymphatic reabsorption.13–17 The meningeal lymphatics (MLVs) are considered the direct communication pathway between CSF and peripheral lymphatics, and recent studies showed that CSF mainly flowed out of the cranial cavity via the MLVs.18,19 These lymphatic vessels are located close to the superior sagittal and transverse sinuses on the dorsal side of the dura and are more widely distributed along the skull base. 20 In recently published work, it was shown that MLVs drainage CSF components, Aβ and extracellular tau, cellular debris, erythrocytes and immune cells from the brain and SAS to the deep cervical lymph nodes (dCLN).19–29 The glymphatic system and CSF outflow are not isolated and share interrelated anatomical structures and functions; macromolecules and antigens from the brain parenchyma can access the CSF and then drain into the cervical lymph nodes via the meningeal lymphatic vasculatures and additional routes. 3 Thus, they were named the glymphatic-lymphatic fluid transport system (GLFTS) by some scholars.3,30
Understanding the physiological regulators and pathological changes of GLFTS will contribute to developing new strategies for treating neurologic disorders such as TBI. Elevated intracranial pressure (ICP) is a widespread pathological phenomenon in many neurological disorders, including TBI, stroke, infection, or neoplasia. In peripheral organs, the lymphatic reabsorption of solutes in the ISF is mediated by periodic changes in extracellular volume, known as extrinsic pumping. When peripheral lymphatic clearance of ISF solutes is slowed, lymphatic drainage can be rescued by increasing local pressure through physical tissue compression. 31 In the brain, the effects of this pressure may be more different because of the uniquely sealed anatomical environment of the intracranial space. However, there is little direct research evidence of the relationship between IICP and GLFTS.
In this study, we hypothesized that IICP might disrupt brain function by decreasing glymphatic flow and cervical lymphatic efflux of CSF. We assessed the effect of IICP on circulation in the glymphatic system and lymphatic efflux by injecting two fluorescent tracers into the cisterna magna and detecting tracers coverage in brain tissue, lymph nodes, and MLVs. Laser speckle imaging and two-photon in vivo imaging were used to assess whether IICP affects arterial pulsation and cerebral blood flow (CBF). Three IICP groups with different elevated ICP were established to assess the impact of severity of IICP.
Materials and methods
All experiments were conducted in accordance with, and approval of the National Institutes of Health guidelines for the Animal Care and Use Committee of Tianjin Medical University General Hospital. The research adheres to the Transparency and Openness Promotion Guidelines. Appropriate measures were taken to ensure minimal pain or discomfort in animals. The manuscript was prepared in accordance with the ARRIVE guidelines. 32
Animals
Both mice and rats were used to detect the effect of IICP on CSF-ISF exchange and lymphatic efflux. For nine to ten weeks male Sprague-Dawley rats were only used for the acute glymphatic influx, evaluation of CBF, and evaluation of lymphatic efflux. In contrast, 8–10 week-old male C57BL/6 mice were used for all studies (acute glymphatic influx; evaluation of CBF; in vivo assessment of cerebral vessels by two-photon fluorescence microscopy; evaluation of Lymphatic efflux and Dorsal meningeal Lymphatic function). Mice and rats were housed in specific pathogen-free conditions under standard 12-h-light/dark cycle conditions in rooms equipped with control for constant temperature and humidity levels. All animals (mice and rats) were acquired from Beijing Vital River Laboratory Animal Technology Inc. In all experiments, mice and rats were anesthetized intraperitoneally with 7% chloral hydrate (at a dosage of 5 μl/g for both rats and mice) and intraperitoneally injected with 0.2% morphine (at a dosage of 0.1 ml/10 g) to relieve pain.
Induction of IICP and measurement of ICP in rats and mice
This IICP paradigm was adapted from the previously published inflating balloon model.33,34 A Fogarty catheter balloon (Edwards Laboratories Inc, cat.# 12TLW803F) was inflated to compress the brain tissue to induce IICP (Figure 1(a)). This model is ideal for studying the various degree of IICP because it can adjust the ICP and keep it within the target range.
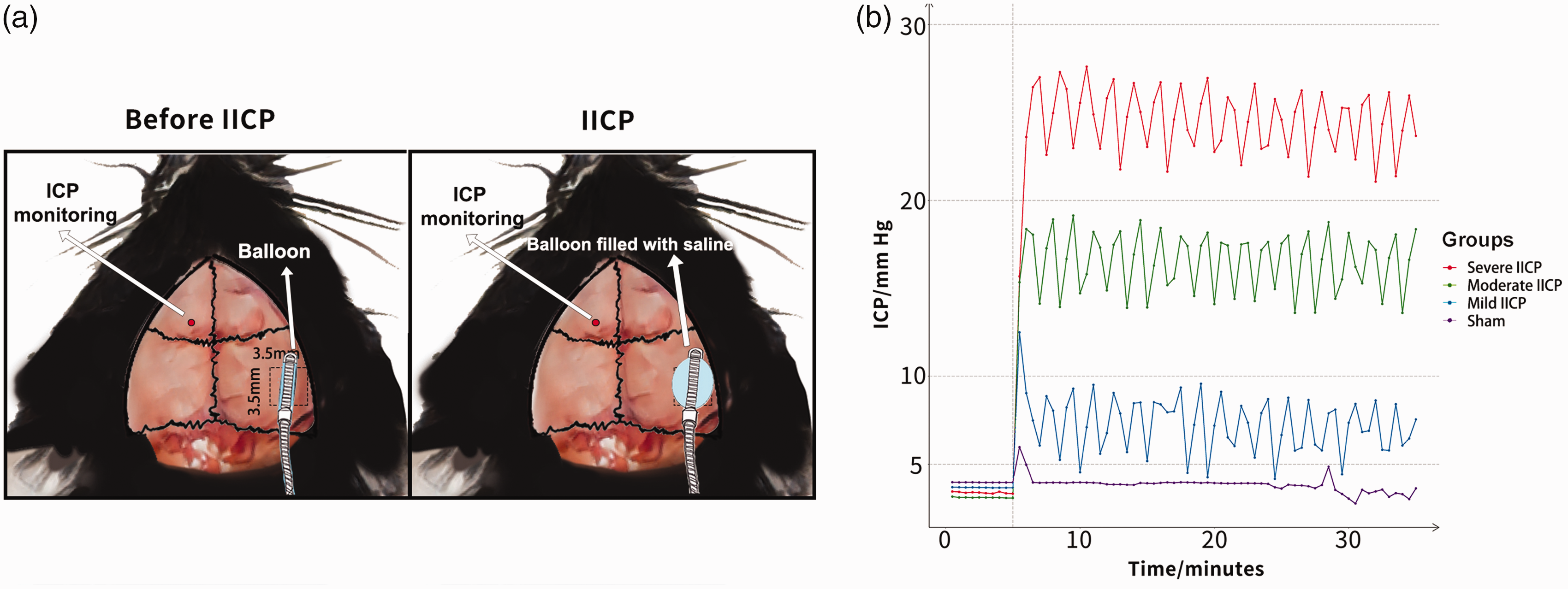
Schematic diagram and the three groups of IICP. (a) Schematic diagram of Surgical preparation and establishment of IICP model in mice; skull window preparation (shown by black dashed line) and (b) ICP monitoring data were obtained from three levels of IICP groups: mild IICP (5 mm Hg–10 mm Hg), moderate IICP (10 mm Hg–20 mm Hg), severe IICP (20 mm Hg–30 mm Hg).
ICP was measured according to previously published protocols with modification.19,35,36 Animals were anesthetized, fixed in a stereotaxic frame, and shaved their hair. After the scalp was sterilized with 70% ethanol, a midline scalp incision was made, and the periosteum was reflected. A bone hole with a diameter of 0.5 mm was drilled over the left frontal cortex (+1.5 mm anterior-posterior [AP], −1.5 mm medial-lateral [ML] for mice; +2.0 mm AP, −2.0 mm ML for rats). The dura was pricked with a sharp needle, and a pressure sensor catheter (transonic, cat#FTH-1211B-0018, USA) was inserted perpendicularly into the left white matter at a depth of 1 mm to record ICP continuously (Figure 1(b)). After stabilizing the ICP signal, a rectangular craniectomy (3.5 mm × 3.5 mm for mice; 7.0 mm X 7.0 mm for rats) was created over the right temporoparietal cortex using a high-speed drill with constant saline irrigation to prevent cortical heating by the drill bit. The bone flap was lifted away, and then a modified 3 F Fogarty arterial embolectomy catheter was carefully inserted into the left temporoparietal epidural space. The balloon at the tip of the catheter had been adjusted in advance to fit the mouse and rat bone window to cause minimal damage to brain tissue and dura mater. IICP was induced by inflating the intracranial balloon. The inflation volume was adjusted during the experiment period to maintain IICP at the range required. During the procedure, bone wax and bio gum were used to hold the balloon in place and prevent it from slipping out. ICP was recorded throughout the experiment.
Three levels of IICP were investigated in this study: 1) mild IICP (5 mm Hg–10 mm Hg), 2) moderate IICP (10 mm Hg–20 mm Hg), and 3) severe IICP (20 mm Hg–30 mm Hg). The classification standard was based on a previous study, 37 which illustrated that ICP elevation to critical levels (i.e., >20 mm Hg) was clinically significant in human TBI and reflected severe TBI in experimental TBI. Sham group received the incision and thinned skull window preparation, and their ICP was recorded continuously in the same ways as the IICP groups. However, they did not undergo craniectomy and balloon compression. Animals with visible brain damage after the increase of ICP were excluded from the study (n = 5).
Fluorescent tracers
Three fluorescent tracers were used in this experiment. Rhodamine B isothiocyanate (RITC) conjugated dextran (molecular weight 70 kD, Sigma, 2.5% in artificial CSF) was injected into the cisterna magna to observe effects of acute elevated ICP on CSF penetration into the brain parenchyma through perivascular space and CSF lymphatic outflow. 0.5 μm fluorescent beads 505/515 (Invitrogen, cat#F8813) were injected into the cisterna magna to compare the influence of IICP on the clearance of dCLN and sacral lymph nodes (sLN), and dorsal meningeal lymphatic function following the previous protocol. 35 Fluorescein isothiocyanate (FITC) labeled dextran (molecular weight 2000 kda, Sigma, 2.5% dissolved in normal saline) was injected intravascularly to visualize the brain vessels in two-photon imaging.
Intracisternal tracer infusions
In order to avoid the influence of the force of balloon dilation on CSF circulation, we chose to perform a cisternal injection 10 minutes after the induction of IICP, when the IICP was usually maintained stable, and further balloon dilation was not required. CSF tracer injections were performed as described.12,26,35,38 After which the mice or rats were anesthetized, their heads were fixed on a stereotaxic frame while an incision in the skin was made at the midline, and the posterior atlanto-occipital membrane covering the cistern was surgically exposed.
Using a 100-μl syringe (Gastight #1710, 1.46-mm inner diameter; Hamilton) and 33 g needle, the volume of the desired fluorescent trace was injected into the CSF-filled cisterna magna compartment. For the bead distribution experiments in mice, 4 μl of fluorescent beads 505/515 were injected at a 0.5 μL/min rate. For mice's RITC-conjugated dextran distribution experiments, 10 μl of RITC-dextran was injected at a 1 μl/min rate. For RITC-dextran experiments in rats, 40 μl of RITC-dextran was infused at 4 μl/min. The tracers were allowed to circulate for an additional 30 minutes following the injection. Throughout the injection, the needle inserted into the cisterna magna stayed in place until the circulate ends to prevent tracer leakage and backflow upon needle removal, and bio gum was also applied from the occipital bone to the base of the wound to secure the needle within the cisterna magna and prevent leakage. Then, the animal was euthanized, transcardially perfused with phosphate-buffered saline (PBS, pH = 7.4, Solarbio), and the tissues needed were collected and postfixed for 12 hours in 4% paraformaldehyde in PBS (Solarbio) overnight after which tissues were transferred to the PBS for long-term preservation. A mild ICP increase was observed in both beads and dextran experiments during the injection process, but these effects persisted only for the duration of tracer infusion.
Tissue collection
Mice and rats were euthanized with anastatic overdose and then transcardially perfused with PBS and 4% paraformaldehyde. dCLN were dissected carefully and fixed in 4% paraformaldehyde in PBS overnight at 4°C and then incubated with DAPI (dilution 1:1000, Invitrogen) for 2 h at room temperature (RT) before being placed on glass slides for fluorescence imaging. For meningeal whole-mount specimen collection, skin and muscle were stripped from the outer skull, and the skullcap was removed with surgical scissors and fixed in 4% paraformaldehyde overnight at 4°C. Then the meninges (dura mater and arachnoid mater) were carefully dissected from the skullcap. Meningeal whole-mounts specimens were then moved to PBS for further immunofluorescence staining. Brains were removed and kept in 4% paraformaldehyde in PBS overnight and dehydrated with 30% sucrose for 2 days before being frozen in tissue plus tech OCT (Thermo Fisher Scientific) and sliced into 100 μm coronal sections using a cryogenic microtome (Leica Microsystems). A total of 8 or 2 sections representative of the craniocaudal axis between +2.50 AP and −3.50 AP were acquired and then stained with DAPI for 30 minutes before being placed on coverslips for fluorescence imaging in vitro.
Immunofluorescence staining
For immunofluorescence staining, meninges were washed in PBS and then blocked with either 2.5% goat serum or 2.5% bovine serum albumin, with 0.1% triton, 0.05% Tween 20 in PBS for 1.5 h at RT. This blocking step was followed by incubation with appropriate dilutions of primary antibodies anti-Lyve-1 (Abcam, cat#ab14917, dilution 1:200) overnight at 4°C. Meningeal whole-mount samples were then washed three times for 15 min at RT in PBS, followed by incubation with the appropriate secondary Alexa Fluor 555 donkey anti-rabbit IgG (1:500 dilution, Invitrogen) for 2 h at RT in PBS. The whole-mount specimens were then washed 3 times for 15 min at RT. The tissue was then placed on glass slides with coverslips for further microscopy imaging.
Ex-vivo imaging of fluorescent CSF-tracers
Fluorescence microscopes with ×1.2 or ×4.0 magnifications were used to perform epifluorescence microscopy. Exposure times were kept constant across all experimental groups and time points. ImageJ was used for image quantification. The percentage of area above a predetermined threshold level was measured in each slice or whole-mount samples. The mean percentages of the covered area were calculated using Microsoft Excel software.
According to previously published protocol modification, the tracer permeation of the brain parenchyma was visualized in ex vivo studies by an inverted fluorescence microscope (ix73, Olympus). 12 CSF penetration was defined by the percentage of the area covered by fluorescence in brain slices. 8 representative slices were averaged together for each mouse, and 2 representative slices were averaged together for each rat. The lymph nodes were placed in glass slides, and images were taken using an upright fluorescence microscope (BX51, Olympus) as described previously.12,35 The percent volume of beads or RITC-dextran coverage in dCLN or sLN was assessed by calculating the proportion of area covered by beads divided by the total area of the node using Image J software. The right and the left dCLN or sLN percent coverage were averaged together for each mouse. Images of meningeal whole mounts were acquired using the upright fluorescence microscope. The percentage of area covered with Lyve-1 dye was used to determine the coverage of the lymphatic vessels. The percent volume of beads coverage in meninges was assessed by calculating the proportion of area covered by beads divided by the total area of the lymphatic vessels using Image J software.
Analysis of cerebral blood flow
A laser speckle flow imager (PeriCam PSI, Perimed, Stockholm, Sweden) was utilized to observe CBF changes in rats and mice at different levels of IICP. Specifically, rats and mice were anesthetized and placed on a stereotactic frame. After the head hair was subtracted and disinfected with 70% ethanol, a 2 cm midline scalp incision was made with a scalpel, and the skull was exposed. The skull on the right temporal top was thinned with a drill. The CBF was then recorded as the baseline at the thinning skull before IICP, and then the IICP model was made according to the above methods. Blood flow was measured at the contralateral thinning skull after IICP. After measurement, a stable CBF interval of 30 seconds was selected for inter-group comparative analysis using PIMsoft, and the results were expressed as the ratio of pre-modeling to post-modeling.
Measurement of cerebrovascular pulsatility in vivo 2-photon fluorescence imaging
Cerebral vascular pulsatility was evaluated by in vivo 2-photon microscopy as described previously.12,38 Animals were anesthetized, and their body temperature was maintained at physiological parameters by placing them on a heating pad (37°C). Next, a midline scalp incision was made, and the periosteum was reflected. The skull was fixed using a custom metallic frame. A 3-mm-diameter thinned skull window was performed over the cerebral cortex, 1 mm lateral and 0.5 mm posterior to bregma for cerebrovascular observation. The surgery is meticulous to ensure the dura was not ruptured. To visualize the vasculature, 0.15 ml BBB impermeable FITC -dextran was immediately injected into the tail vein. The cerebral vasculature was first imaged with 512 × 512 pixel frames from the surface to a depth of 240 μm with a 5 μm z-step. After imaging, vessels were identified as surface arteries and veins, penetrating arteries, or ascending veins based on morphology as previously described:12,38,39 surface arteries passing superficially to surface veins and exhibiting less branching at superficial cortical depths.
To measure vessel diameters, 3000 ms X–T line scans were first acquired orthogonally to the vessel axis in surface arteries and veins. Then, penetrating vessel line scans were acquired exactly 100 μm below the cortical surface to avoid differences in pulsatility that may arise due to cortical depth. In each animal, two vessels of each type were arbitrarily selected for pulsation assessment through the thinned skull. In total, 12 vessels of each type were assessed throughout the study. Image analysis was conducted with ImageJ software and Matlab, and vessel diameter was extracted from X–T plots and plotted versus time. Steady-state vessel diameters were calculated as the mean value over the 3000 ms epoch. Vessel wall pulsatility (derived units μm × s) was calculated as the absolute value of area under the diameter–time plot, integrated about the running average over the 3000 ms epoch.
Statistical analysis
All statistical analyses were performed using “GraphPad Prism 8” GraphPad software. The Kolmogorov-Smirnov test was used to assess normal distribution of the data. All data were presented as the mean ± SD. Differences were considered to be significant at a level of p < 0.05. All studies were evaluated by two-way ANOVA followed by Tukey's post hoc test for multiple comparisons.
Results
ICP-associated decrease of glymphatic recirculation of CSF and ISF
To evaluate whether the severity of the IICP plays a role in the glymphatic CSF penetration into the brain parenchyma, we employed a balloon compression model of IICP that uses an inflating catheter balloon to compress brain tissue to increase ICP acutely. After 10 minutes of IICP, the tracer molecule RITC- dextran (70 kDa) was slowly infused into the subarachnoid CSF of the cisterna magna as well as in a cohort of sham mice (Figure 2(a)). Thirty minutes after the end of CSF tracer infusion, animals were perfused and PBS and then fixative, the brains were dissected and sliced, and tracer penetration was evaluated by whole-slice fluorescence imaging in vitro fluorescence microscopy. CSF penetrance into CNS structures was measured using the percentage of area occupied by RITC- dextran in eight representative slices per one mouse in the sham and IICP groups. The fluorescence area coverage from each mouse's eight representative brain slices was averaged to define CSF penetration. Compared to mice that underwent a sham procedure, CSF tracer penetration into the brains of animals with mild IICP, moderate IICP, and severe IICP was significantly reduced (Figure 2(b) and (c)). Moreover, there was a noticeable trend towards decreased dextran influx in the mice that had received more oppression, and IICP is rising even higher, suggesting that glymphatic circulation is also likely affected by the severity of IICP. We also assessed the circulation of the glymphatic system in IICP rats, and the results were similar (Figure 2(d) and (e)).
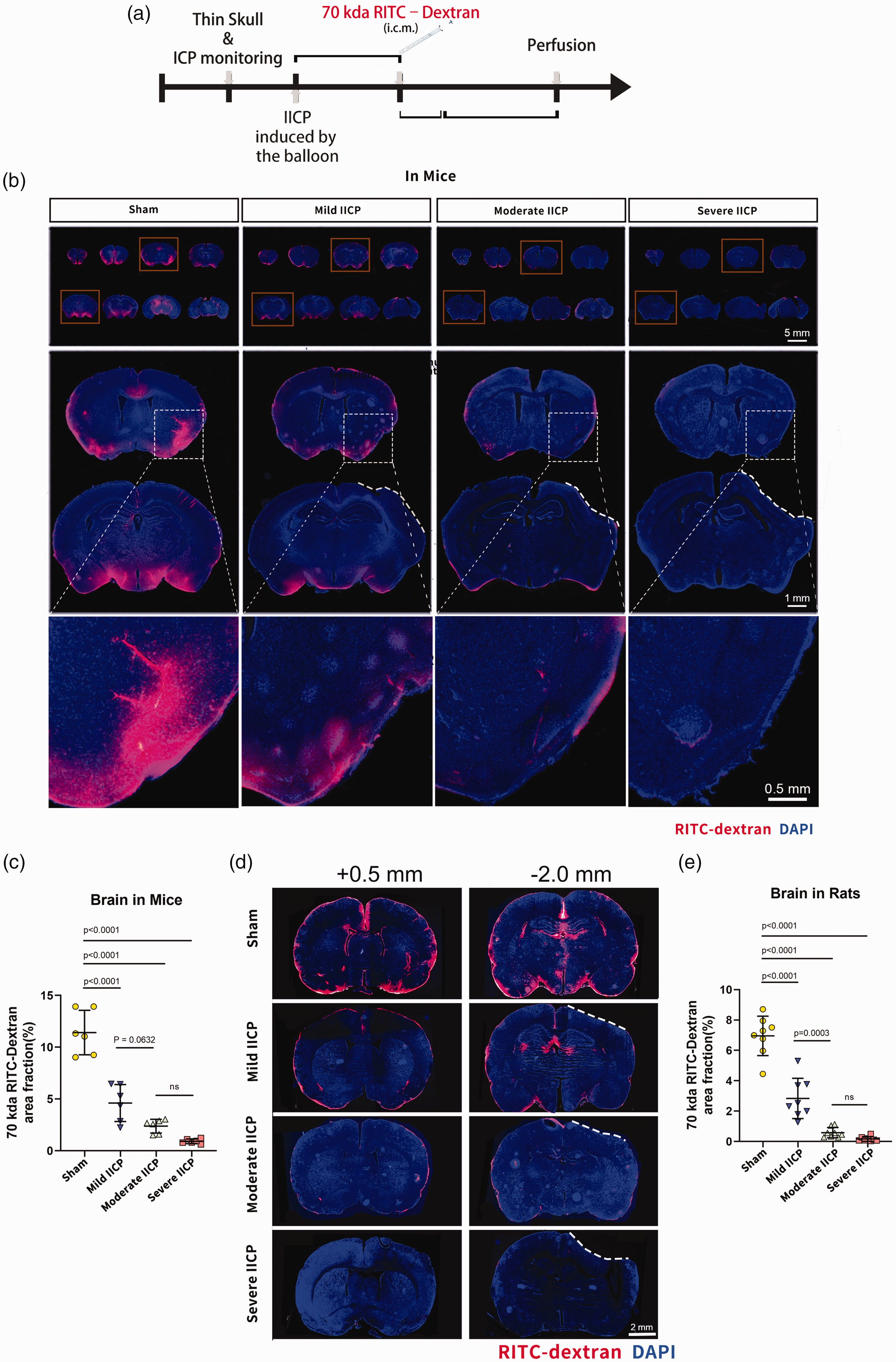
Assessment of CSF drainage and dextran influx in the brain of animals with IICP. (a) Plan of experiment with a timeline. (b) Representative brain sections stained for nuclei (DAPI; blue) and RITC-dextran (red) influx into the brain parenchyma of mice with control (sham) and IICP (scale bar, 5 mm; inset scale bar, 1 mm). The compressing site is indicated with the white dashed line. The higher magnification area is indicated with the white dotted box. (c) Quantification of the percentage of RITC-dextran covered area fraction in brain sections shows a significant decrease in the IICP groups compared to the control group (sham vs. mild IICP vs. moderate IICP vs. severe IICP). (d) Representative brain sections stained with DAPI (blue) and RITC-dextran (red) influx into the brain parenchyma of rats for control (sham) and IICP groups (scale bar: 2 mm). The compressing site is indicated with the white dashed line. (e) Quantification of the percentage of RITC-dextran covered area in brain sections shows a significant decrease in the IICP groups compared to the control (sham) group (sham vs. mild IICP vs. moderate IICP vs. severe IICP). In (c), (e), the data is shown as mean ± SD (n = 6 animals per group for the mouse study; n = 8 animals per group for the rat study; one-way ANOVA with Tukey's multiple comparison post-hoc test).
Effects of increased intracranial pressure on cerebral blood flow and cerebrovascular pulsation
Glymphatic circulation is critically dependent on cerebrovascular dynamics, and arterial pulsation is the primary driving force for CSF recirculation into the brain parenchyma along paravascular pathways.12,38–40 So we next sought to determine whether IICP impairs CBF and cerebrovascular pulsation. To do so, we used a laser speckle flow imager. We established the IICP model on the right side of the skull and used a laser speckle on the left side of the skull to detect CBF changes at baseline and during IICP (Figure 3(a) and (b)). We found that CBF decreased significantly after IICP, and there was a noticeable trend toward decreased CBF in the mice with ICP (Figure 3(c) and (d)), which is consistent with previous research.41,42 We also assessed the CBF in IICP rats, and the results were similar to those in mice (Suppl. Figure 1, Supplementary data).
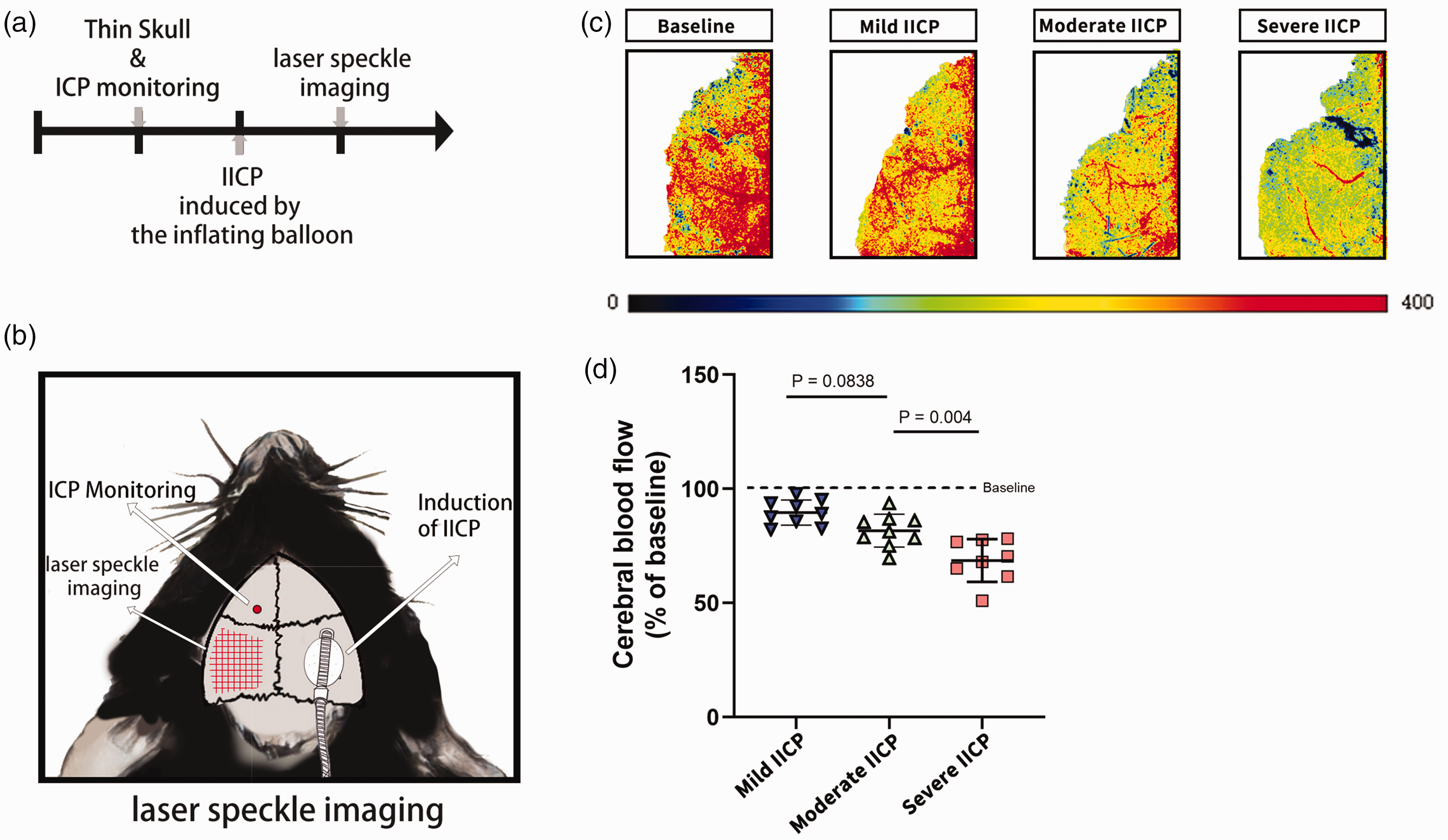
Assessment of CBF and vascular pulsation during IICP. (a, b) Experimental timeline and schematic diagram for CBF experiments. (c) Representative images show the cortical perfusion measured by laser speckle photography. (d) Quantification of CBF shows a significant decrease in the IICP groups compared to sham groups (mild IICP vs. moderate IICP vs. severe IICP). In (d), data are presented as mean ± SD (n = 8–9 animals per group; one-way ANOVA following Tukey's multiple comparison post-hoc test).
Next, we used a two-photon microscope to further observe the cerebral vessels in vivo and evaluate the influence of IICP on arterial pulsation. We compared the changes in cerebrovascular vessels in vitro pre-IICP and post-IICP (Figure 4(a)). Following intravascular injection of a fluorescently labeled dextran (FITC-dextran, 2000 kDа), vascular imaging was performed through the left thinned skull window. Subsequently, the IICP model was established. After 10 minutes, the mouse was returned to the two-photon microscope for repeat imaging of the same cortical vessels. We observed in these conditions that partial vascular occlusion occurred after the increase of ICP, and surface arteries and veins showed remarkably less pulsation (Figure 4(b)). We then quantified the pulsation of the cerebrovascular wall of the no-occluded blood vessels using in vivo 2-photon microscopy according to previously validated techniques with modification12,38,39 (Figure 4(c)). The morphology identified vessels as surface arteries and veins, penetrating arteries, or ascending veins. Surface arteries were located superficially to surface veins exhibiting less branching at superficial cortical depths (Figure 4(d)). Quantitative changes in vessel diameter over a 3-second epoch were measured by high-frequency XT line scans orthogonal to the vessel axis. The pulsatility index was calculated as the summation of the absolute difference between the instantaneous diameter and the 3000-msec running average of the diameter over the scanning period (Figure 4(e)). In contrast to other studies where penetrating arteries were more easily affected,12,38,43 we observed a significant decrease in all types of vascular pulsatility following IICP compared with the control (sham) group (Figure 4(f)). Consequently, these data suggest that after IICP, partial occlusion occurred, and the pulsatility of no-occluded cerebrovascular was reduced. These changes indicate immediate and global impairment of glymphatic CSF influx into cerebral tissues with IICP.
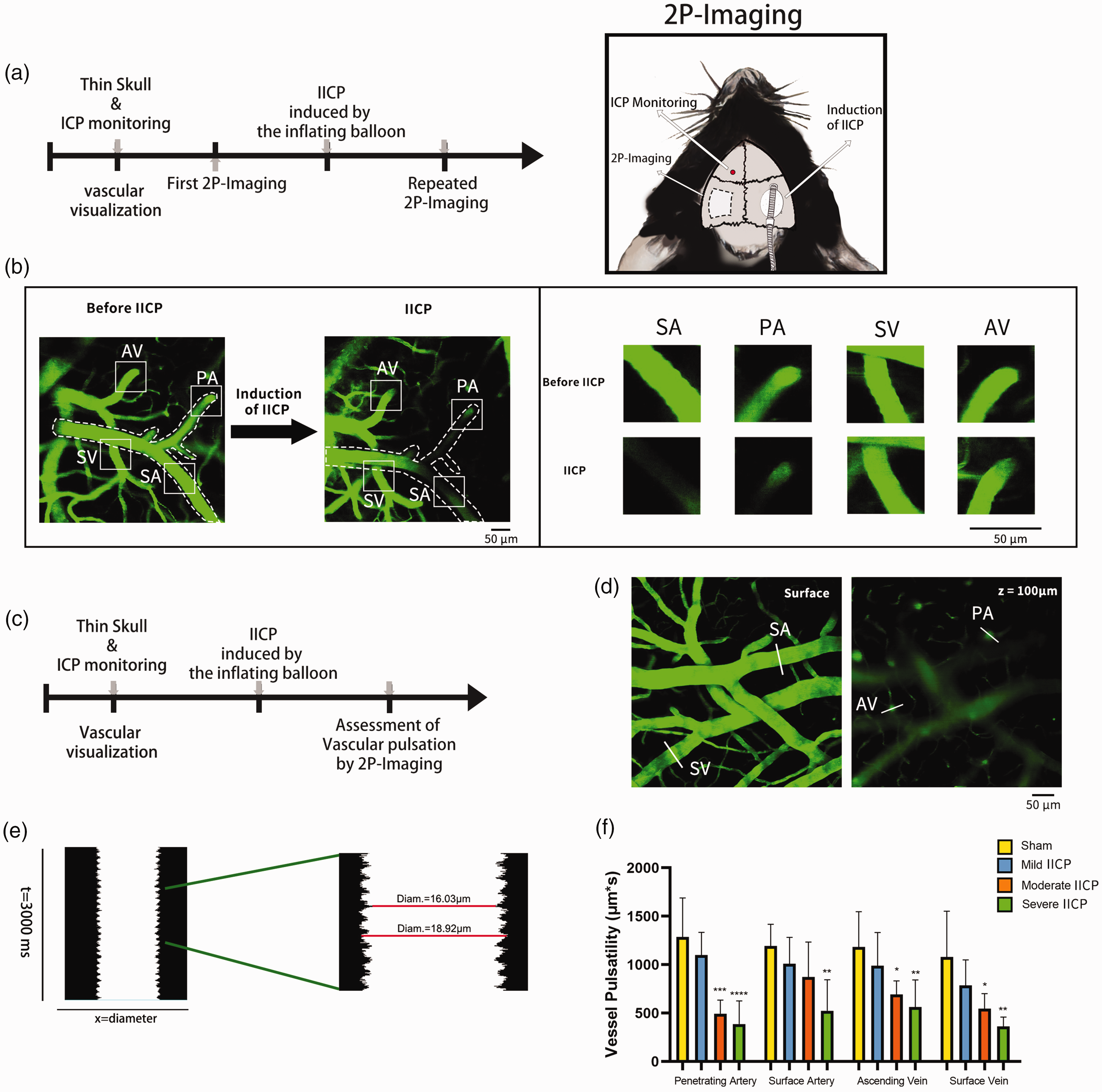
Analysis of vascular pulsation during IICP. (a) Experimental timeline for in vivo observation of vascular changes before and after IICP, and schematic diagram for two-photon in vivo imaging. (b) The cerebrovascular column is visualized by injecting 2000 kD RITC-dextran intravenously (left image: collapsed 100 mm XYZ stack, magnification: X20). Cerebrovascular measurements at the same site were repeated before and after IICP (Representative picture: moderate IICP). Reduced or even occluded blood flow and significantly reduced arterial pulsation in superficial arteries and superficial veins can be observed in this image. Superficial vein (SV), superficial artery (SA), penetrating artery (PA), and ascending vein (AV) were classified according to morphology (shown by a solid white box). A white dotted line shows an almost occluded artery vessel. (c) Experimental timeline for quantitating vascular pulsation in the IICP and SHAM groups in vivo. (d) Three-second XT line scans orthogonal to the vessel axis, capturing the dynamic change in vessel diameter across time, are acquired at the brain's surface or a depth of 100 mm below the surface (shown by a solid white line). (e) vessel diameter was extracted from X–T plots by Matlab and plotted versus time. Steady-state vessel diameters were calculated as the mean value over the 3000 ms epoch. Vessel wall pulsatility (derived units µm × s) was calculated as the absolute value of area under the diameter integrated about the running average over the 3000 ms epoch and (f) Quantification of vascular pulsation shows a significant decrease in the IICP groups when compared to sham groups for all four types of vessels (vs. sham; *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001). In (f), the data is presented as mean ± SD (n = 6 animals per group, with one-way ANOVA followed by Tukey's post-hoc test).
IICP results in cervical lymphatic dysfunction but enhanced spinal lymphatic function
Three major anatomical pathways of CSF outflow were proposed: 1) routes through foramina in the skull along cranial nerves (Olfactory route through the cribriform plate, optic route, the peripheral pathways of the glossopharyngeal, vagus, and accessory nerves at the jugular foramina, and other crucial cranial nerves);15,18,44–48 2) drainage through the meningeal or dural lymphatic vessels; 3) spinal lymphatic drainage. In this study, The effect of IICP on dCLN drainage was observed as dCLN is the primary outflow pathway of meningeal lymphatic vessels, and is also involved in the other CSF outflow routes along the olfactory nerve, the trigeminal nerve, the glossopharyngeal nerve, vagus nerve, and other cranial nerves routes.18,20,49 Furthermore, the effect of ICP on spinal lymphatic drainage was also investigated. A recent study elucidated the pathways of circulation and clearance of CSF solutes in the spine. 50 According to this study, the spinal outflow of CSF tracers occurred predominantly from intravertebral regions of the sacral spine to lymphatic vessels, leading to sacral lymph nodes. Therefore, sacral lymph nodes were collected in this study to detect changes in CSF efflux through spinal lymphatic absorption under IICP.
After the balloon model of IICP was employed, two fluorescent tracers 70 kDa RITC- dextran and fluorescent beads were separately injected into cisterna magna to investigate whether IICP impacts cervical lymphatic drainage and spinal lymphatic drainage. The dCLN, sLN, and meninges were harvested 30 minutes after the end of injection to assess the presence and distribution of fluorescent tracers (Figure 5(a)). When we examined lymph nodes using the fluorescence microscope, we observed a substantial decrease in fluorescent tracers drainage to the dCLN but increased drainage to the sLN in IICP mice compared with the control (sham) group. In addition, the severity of the IICP also plays an essential role in tracer drainage. There was a remarkable trend toward decreased dCLN drainage and increased sLN drainage in the mice that had received more oppression (Figure 5(b) to (d)). We also assessed the cervical lymphatic drainage and sacral lymphatic drainage in IICP rats by injection of RITC-dextran, and the results were similar to those in mice (Suppl. Figure 2, Supplementary data).
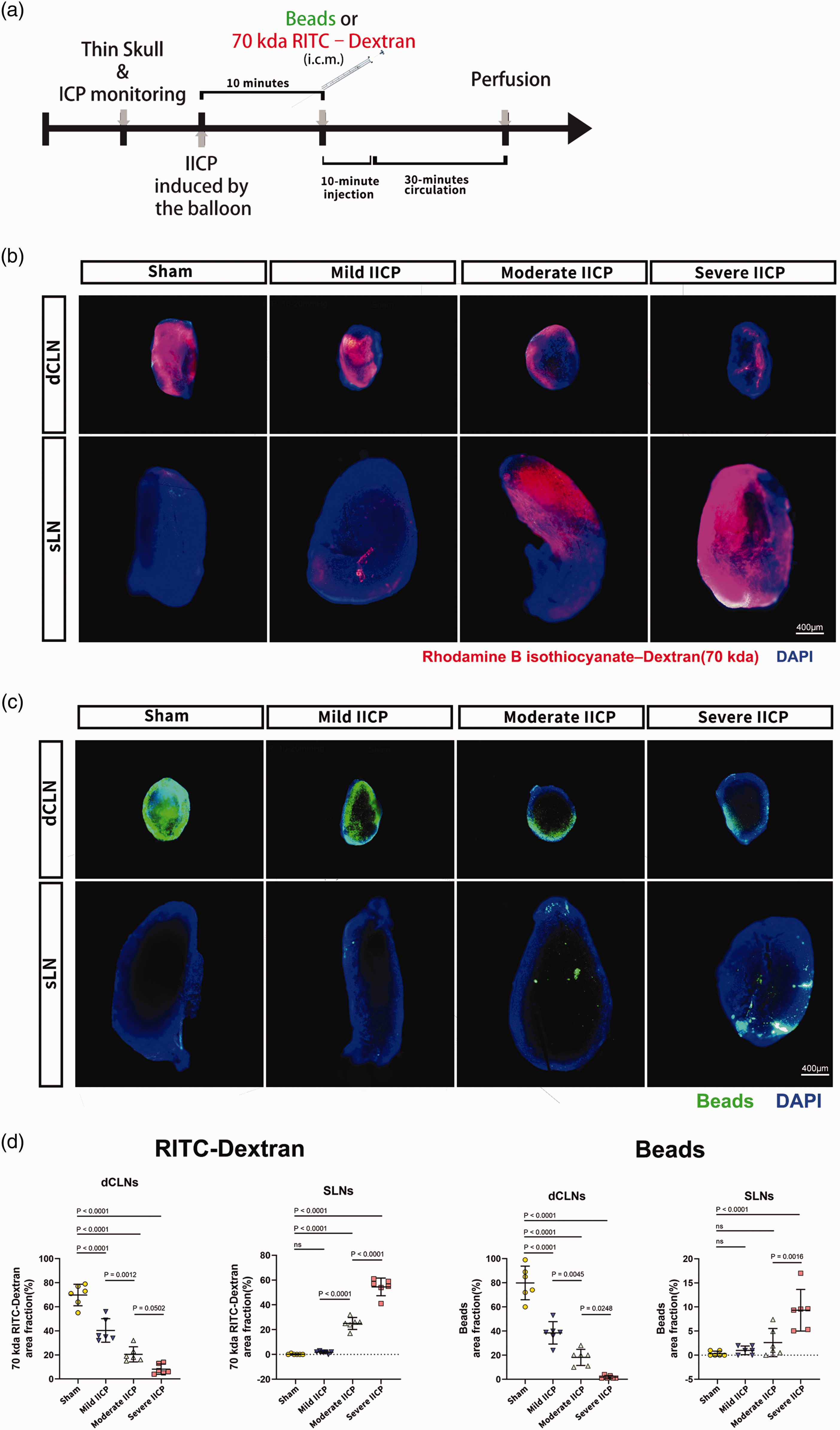
Analysis of dCLN and sLN drainage during IICP. (a) Schematic diagram of experimental timeline. (b) Representative LN stained for nuclei with DAPI (blue) and 70 kDa RITC- dextran (red) shows clearance into the dCLN and sLN of mice from sham and IICP groups (scale bar: 400 µm). (c), Representative LN stained with DAPI (blue) and 0.5 µm fluorescent beads 505/515 (green) shows clearance into the dCLN and sLN of mice from sham and IICP groups (scale bar: 400 µm) and (d) Quantification of the percentage of RITC- dextran covered area (left panel) in whole-mount samples of LN shows a significant decrease of dCLN drainage in the IICP groups when compared to the control (sham) group (sham vs. mild IICP vs. moderate IICP vs. severe IICP) and a significant increase in sLN drainage in the IICP groups when compared to control (sham) group (sham vs. mild IICP vs. moderate IICP vs. severe IICP). The percentage of the fluorescent bead-covered area (right panel) in LN whole-mount samples shows the same trend as the RITC-dextran experiments. In (d), the data is presented as mean ± SD (n = 6 animals per group for each experiment with one-way ANOVA followed by Tukey's multiple comparison post-hoc test).
IICP perturbs absorbance of CSF by the dorsal meningeal lymphatic vessels
To further explore the effect of IICP on CSF lymphatic outflow, we examined the drainage function of the dorsal lymphatic vessels, which do not have lymphatic valves as the lymphatic vessels at the base of the skull and could be more vulnerable to pressure changes20,24 To determine whether the uptake of CSF into the meningeal lymphatic vasculature was altered during IICP, Beads were injected into the SAS of cisterna magna, and the CSF uptake of meningeal lymphatic vessels along with the transverse sinus was examined according to a previously published protocol. 35 The meninges were harvested to examine the uptake of CSF contents in the lymph vessel 30 min post-injection (Figure 6(a)). Analysis of the dorsal meningeal whole-mount samples revealed that the lymphatic uptake of fluorescent beads was impaired immediately after IICP. There was also a trend toward decreased meningeal lymphatic function in the mice after severe IICP (Figure 6(b) and (c)). Taken together, these data indicate that even mild IICP can result in cervical lymphatic drainage disorder and disorder of dorsal MLVs, and the extent of these impairments is closely related to the severity of IICP.
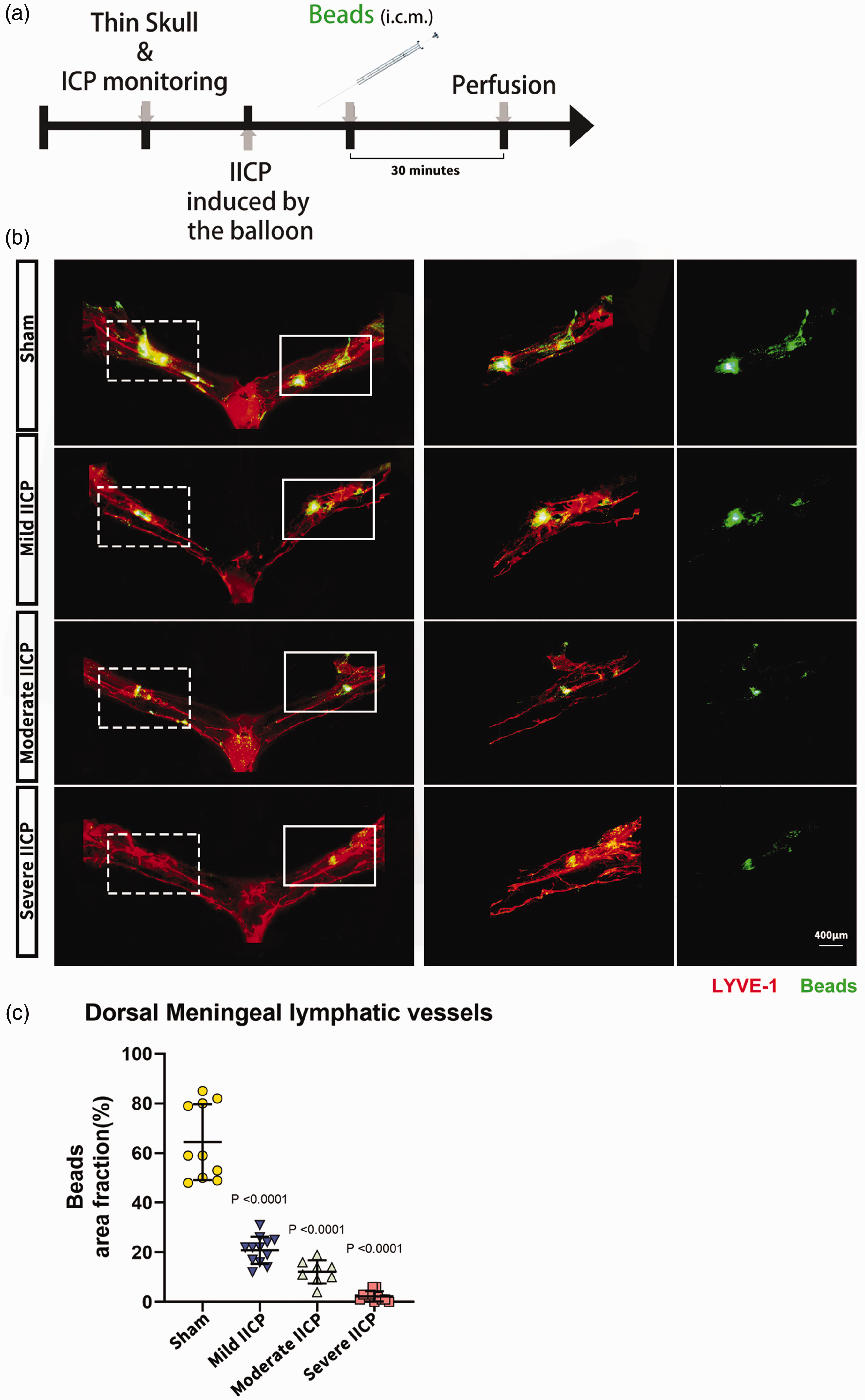
IICP leads to impairments in dorsal meningeal lymphatic drainage. (a) Schematic diagram of experimental timeline. (b) Representative images of dorsal meningeal whole mounts stained for Lyve-1 dye (red color) shows the uptake of beads in CSF by dorsal meningeal lymphatic vessels from sham and IICP groups of animals (scale bar: 400 µm). The main fluorescence accumulation area along the transverse sinus on the compression side is shown by solid line boxes. The main fluorescence accumulation area along the transverse sinus on the opposite side is shown by dashed line boxes and (c) Quantification of the percentage of fluorescent bead covered area in dorsal meningeal whole-mount samples demonstrates a significant decrease in the IICP groups when compared to the control (sham) groups (vs. sham). In (c), the data is presented as mean ± SD (n = 8–12 animals per group with one-way ANOVA followed by Tukey's multiple comparison post-hoc test).
Discussion
Some scholars have proposed the influence of blood or blood and pressure on GLFTS using the model of subarachnoid hemorrhage, subdural hematoma, and intracerebral hemorrhage.6,28,29,51–53 Despite that, our understanding of the pathologic processes that promote defective lymphatic drainage in the CNS is still limited. Elevated ICP is a prevalent factor for many neurological disorders, and its relationship with GLFTS remains unclear. Bolte et al. established a transient ICP elevation model by ligating bilateral internal jugular veins in mice, which saw IICP peak (up to 10 mmHg) within 3 hours and return to normal ICP within 24 hours after ligation. By injecting fluorescent beads into the cisterna magna, they found significantly less drainage to the dCLN 3 h after ligation and that there was reduced bead accumulation around the meningeal lymphatic vasculature of ligated mice. The authors concluded that IICP might provoke impaired drainage of dCLN and dorsal meningeal lymphatic dysfunction. 35 Plog et al. found in the mice with craniotomy (without pathological injury) a decreased level of glymphatic flow as well as reduced arterial pulsation, suggesting that lower ICP may also affect glymphatic circulation. 38 Vinje et al. used physical-mathematical models to demonstrate that the distribution of CSF flow to distinct clearance pathways depends on ICP, with the arachnoid granulations as the main contributors to this outflow. 54 However, direct evidence of the effect of IICP on GLFTS is still lacking, while the effect of severity of IICP on GLFTS remains entirely unclear. In our study, we demonstrated that glymphatic inflow decreases after IICP. At the same time, cerebrovascular occlusion and the decrease in cerebrovascular pulsation may be the main reason for GLFTS impairment. Moreover, we found weak tracer drainage to dCLN and the dorsal dural lymphatics and significant drainage to sLN following IICP. The above results in this study occurred immediately after IICP and were all related to the severity of IICP.
The epidural balloon dilation model is one of many IICP models widely used to investigate the hemodynamic response in the past.55–60 While other investigators include subdural balloon model,33,34,56 lumbar artificial CSF infusion, 42 cisterna-magna artificial CSF infusion. 61 The epidural model was chosen because it has minimal damage to the dura compared to the subdural model, and the CSF infusion model was excluded as it artificially accelerated CSF circulation. The IICP classification in this study was based on the severity of TBI. This disease corresponds better to balloon dilation models since the elevated pressure in balloon dilation models and raised ICP in TBI are both regional and not always transmitted equally in all directions due to the inhomogeneity of brain structures. 62 However, no matter which IICP model is used, these are substantially different from IICP in the actual pathological process of diseases. In future experiments, the influence of IICP on GLFTS should be based on a specific disease model.
According to the literature, CSF flow is pulsatile and driven primarily by arterial wall motion (cerebrovascular pulsatility) caused by the cardiac cycle. 63 Thus, it is reasonable to assume that the pulsing of blood vessels requires not only the walls, but also the filling of blood. When intracranial pressure increases, cerebrovascular vessels become ischemic, and blood does not fill up in the blood vessels. As a result, the heart beats are difficult to transmit to the cerebrovascular vessels, so the cerebrovascular pulsatility decreases. In addition to reduced cerebrovascular pulsatility, our results suggest that cerebral vascular occlusion occurs when IICP occurs. These two factors may together lead to the impairment of glymphatic circulation.
IICP often occurs after TBI and is a significant driver of mortality.64,65 Management of IICP is critical for the treatment. However, convincing evidence that monitoring ICP improves the outcome of TBI patients is still lacking. 66 ICP and cerebral perfusion pressure monitoring remain necessary for neurological disease intensive care.66–68 Monitoring ICP could be used as a prognostic factor of TBI to prevent cerebral hernia.69,70 Uncontrolled IICP could cause a cerebral hernia, leading to adverse clinical consequences, including death.71–74 For TBI, inadequate control of IICP is the leading cause of death, and it is strongly associated with poor clinical outcomes.64,65 Traditional antihypertensive methods often include decompressive craniectomy, hypertonic saline, and mannitol.73–76 Recent studies have also suggested that manual craniocervical lymphatic drainage could be used to reduce ICP in patients with severe brain diseases. 77 Our study found that IICP impacts the drainage of GLFTS, suggesting that these antihypertensive methods may improve neurological functions by relieving damage to the CNS lymphatic system. However, low IICP caused by decompressive craniectomy also leads to CSF dynamics disorder and impairs functions of glymphatic system. 38 We hypothesize that GLFTS fully play their normal physiological functions only when ICP is in its normal physiological range.
In addition to removing CSF from the cranial cavity, CSF is also absorbed through spinal lymph vessels. In a study of healthy volunteers, spinal CSF absorption of 0.11–0.23 ml/min was found, roughly half of the mean CSF production rate of 0.34 ml/min. 78 Several studies reported that increased absorption of spinal lymphatic drainage is associated with IICP-related diseases: In a study of GL261 glioma mouse, 19 CSF tracers were redirected into the spinal space, and the impaired CSF clearance from the cranium was compensated by a lymphatic outflow from the sacral spine. Another study on the hydrocephalus model suggests that spinal outflow pathways in non-communicating hydrocephalus increased and compensated for reduced CSF outflow through normal pathways.79,80 Notably, a common feature of these two disorders is elevated ICP. Our current study demonstrates that spinal lymphatic drainage is enhanced in IICP, suggesting that modulation of spinal lymphatic drainage may provide a new therapeutic option for numerous neurologic diseases associated with IICP. Compared to cervical lymphatic drainage, the lymphatic pathway in the spine may be a safer and more convenient place to be controlled because of its anatomical structure. Exploring this pathway also has implications for treating neurodegenerative diseases. In a study of older mice, the contribution of the nasal route to CSF outflow was reduced significantly, but not for the spinal routes. 81 These data suggest that this pathway may still play an essential role in aging patients. Finally, we were studying this pathway in rodent models in this study. In humans, because our species spend a significant amount of time in an upright position, the outflow in the spine (primarily the caudal end) would perhaps be even more critical than in rodents.50,79 To sum up, a better understanding of spinal lymphatic drainage leads to a better understanding of the metabolite removal of the entire CNS and contributes to treating many neurologic diseases. Further investigation is needed to study the neuropathological role of spinal lymphatic outflow and its relationship with intracranial lymphatic drainage.
The meningeal lymphatic system consists of the dorsal MLVs and the MLVs at the base of the brain, and there is still a contradiction regarding pathways that are critical for CSF outflow.20–22,82 At the core of this debate is the existence of a blockade between SAS and the dural mater, the arachnoid barrier layer, 83 which prevents the dura from directly contacting the CSF. Louveau et al. injected tracer into the cisterna magna and found that the tracer accumulated quickly in lymphatic vessels along the transverse dural sinus. Thus, the authors hypothesized the “hot spots” where liquids can be directly transferred to lymphatic vessels from the SAS. Nevertheless, this has yet to be assessed and proved on an anatomical level.19,21 In contrast to study by Louveau et al., Ma et al. have observed tracers in SAS that left jugular foramen along with the nerve draining to the dCLN; They considered this path as a critical CSF outflow site of meningeal lymphatic vessels. 18 Similarly, Ahn et al. also found that tracers in SAS were taken by the dural lymphatic vessels at the base of the skull and then left the cranial cavity through the petrosquamous fissure and the jugular and stylomastoid foramina. 22 Our study examined the effect of IICP on the drainage function of the dorsal meningeal lymphatic vessels since it does not have lymphatic valves. However, we also believe that IICP affects the lymphatic drainage of the skull base because the distribution of valves on the lymphatic vessels at the base of the skull was relatively scarce. 20 Further experimentation is still needed to test this hypothesis.
Conclusion
This study demonstrated that IICP impaired the circulation in the glymphatic system by inducing vessels occlusion and reducing cerebrovascular pulsation. IICP also perturbed cervical lymphatic drainage and dorsal meningeal lymphatic function, but enhanced spinal lymphatic outflow to the sLNs, which may be a compensatory mechanism for impaired cervical lymphatic drainage. All results were closely correlated with the severity of ICP. Even though the intracranial lymphatic system concept was reported in 2015, no effective therapeutic strategy has been developed yet based on this concept. We believe that the results of our study will provide pivotal clues for exploring new therapeutic strategies for the treatment of many types of neurological disorders.
Supplemental Material
sj-jpg-1-jcb-10.1177_0271678X221119855 - Supplemental material for Effects of increased intracranial pressure on cerebrospinal fluid influx, cerebral vascular hemodynamic indexes, and cerebrospinal fluid lymphatic efflux
Supplemental material, sj-jpg-1-jcb-10.1177_0271678X221119855 for Effects of increased intracranial pressure on cerebrospinal fluid influx, cerebral vascular hemodynamic indexes, and cerebrospinal fluid lymphatic efflux by Tangtang Xiang, Dongyi Feng, Xinjie Zhang, Yupeng Chen, Hanhua Wang, Xuanhui Liu, Zhitao Gong, Jiangyuan Yuan, Mingqi Liu, Zhuang Sha, Chuanxiang Lv, Weiwei Jiang, Meng Nie, Yibing Fan, Di Wu, Shiying Dong, Jiancheng Feng, Eugene D Ponomarev, Jianning Zhang and Rongcai Jiang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The staff received salary support from the General Hospital of Tianjin Medical University and The Neurological Institute of Tianjin. This study was supported by The National Natural Science Foundation of China (via grant no.82001323 to C Gao, grant no. 82071390 to R Jiang), The Tianjin Research Program of Application Foundation and Advanced Technology (via grant no. 19YFZCSY00650 to R Jiang) and The Clinical Study of Tianjin Medical University (2017kylc007 to R.J).
Acknowledgements
The authors wish to thank the individuals and their guardians for participating in this study. Additional thanks to the Tianjin Institute of Neurology staff; Huijiao Chen, the head nurse of NICU of Neurosurgery department; and Professor Hui Shen in the institute of Biomedical Engineering and Technology.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
RJ, JZ, EP, and TX conceived and designed the study and analyzed the data. TX, DF, XZ, HW developed the methodology. TX, YC, XL, ZG, ML, ZS, JY, CL, WJ and MN carried out the experiments. YF, DW, SD, and JF analyzed the data and interpreted the results. XT wrote the manuscript. XJ, DF, and HW provided technical support. RJ and JZ reviewed and revised the manuscript and supervised the study. All authors have read and approved the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
