Abstract
Functional brain changes such as altered cerebral blood flow occur long before the onset of clinical symptoms in Alzheimer’s disease (AD) and other neurodegenerative disorders. While cerebral hypoperfusion occurs in established AD, middle-aged carriers of genetic risk factors for AD, including APOE ε4, display regional hyperperfusion due to hypothesised pleiotropic or compensatory effects, representing a possible early biomarker of AD and facilitating earlier AD diagnosis. However, it is not clear whether hyperperfusion already exists even earlier in life. Here, 160 young and cognitively healthy participants from the Chinese PREVENT cohort underwent 3 T arterial spin labelling and T1 MRI and genetic testing for APOE and MAPT rs242557 status. Using FSL, we performed a whole brain voxel-wise analysis and a global mean grey matter analysis comparing for the effects of both risk genes on cerebral perfusion. No significant alterations were seen for APOE genotype, but in MAPT rs242557 A carriers, we observed a significantly hyperperfusion in the left anterior cingulate cortex and left insular cortex. There were no effects of APOE or MAPT status on the global perfusion. These results are novel and may suggest that MAPT genotypes demonstrated a distinct hemodynamic profile in a very young age.
Keywords
Introduction
Alzheimer’s disease (AD) is the most common cause of dementia globally, 1 and among the most burdensome diseases in older populations, accounting for more disability years in those aged over 60 than stroke, musculoskeletal conditions, cardiovascular disease and all cancers 2 and its prevalence is continuing to grow due to ageing populations. AD is heterogeneous in its presentation but it typically presents with progressive memory loss and a combination of other symptoms such as confusion, personality changes and deterioration of speech and vision. 1 However, research in recent years has suggested that AD pathogenesis including neuroinflammation and vascular changes start many years before any symptoms occur. 3 The two main pathological events leading to AD are the formation of extracellular amyloid beta (Aβ) plaques and intracellular hyperphosphorylated tau neurofibrillary tangles in the brain 4 and both of these events have been shown to precede the development of cognitive symptoms. 5 In addition to age, there are many other risk factors of AD ranging from lifestyle and vascular factors to family history and genetic polymorphisms.
The apolipoprotein E (APOE) ε4 allele (APOE ε4) is the most common genetic risk factor for AD, 6 with genome-wide association studies (GWAS) showing it accounts for as much as 23.52% of all AD heritability compared to the less than 1% contributed by most other genetic risks.7,8 It has been proposed that a mechanism by which APOE ε4 increases the risk of developing AD is related to Aβ aggregation. 9 As part of its metabolic role, the APOE protein has been shown to break down Aβ and thus prevent plaque formation, 10 however the ε4 isoform displays impaired Aβ clearance and thus increases the chance of plaques developing leading to AD. 9 APOE ε4 has also been suggested to have roles in other aspects of AD pathology including lipid metabolism, temporal lobe atrophy and altered cerebral blood flow (CBF). 11 Another gene associated with neurodegeneration involving tau pathology is the microtubule associated protein tau (MAPT) gene, which occurs in two haplotypes, H1 and H2, with the H1 haplotype being associated with increased risk of AD and other tauopathies via differential expression of various exons relevant to tau aggregation.12,13 The sub-haplotype H1c has been suggested to be predominantly responsible for this association. 14 MAPT is not specifically associated with AD, but with other neurodegenerative disorders that involve tau pathology such as Frontotemporal dementia (FTD) and Progressive Supranuclear Palsy.8,15
This study focuses on a specific single-nucleotide polymorphism (SNP) of the MAPT gene, rs242557. This SNP possesses 2 alleles, the major allele G, and the minor allele A 14 and is thought to be significant in defining the H1c sub-haplotype. 16 The general consensus is that the A allele is associated with an increased risk of tauopathy14,17 with a meta-analysis finding this to follow a dominant genetic model 18 and GWAS results showing an association between the A allele and increased plasma tau levels. 19 However, there is some uncertainty regarding the role of the rs242557 SNP in neurodegeneration, with some suggesting it has no significant association with late onset AD 20 and some even suggesting an opposite effect in Chinese and other Asian populations with the G allele increasing AD risk while the A allele is protective.21,22 Nevertheless, the association between MAPT genotype and broader pathological processes containing tau pathology means it is of interest across several neurodegenerative disorders including AD.
Historically, AD diagnosis has been based on symptoms leading to later presentation when irreversible pathological changes have already occurred. In order to detect the disease earlier, other observable biomarkers are required. Functional changes in the brain visible on neuroimaging have been observed to occur prior to structural changes and so could be used to detect AD before excessive structural damage occurs. 23 Reductions in cerebral perfusion have consistently been shown to occur in AD and it is thought that such changes may contribute to AD pathology through mechanisms including destruction of neurons and glial cells and metabolic disruption.6,24 Furthermore, there is a wealth of evidence suggesting that these changes can be observed long before central pathophysiological mechanisms of AD become consolidated,6,25 and variations in CBF have been demonstrated to occur as early as puberty. 26 This in combination with the ease of observation of these perfusion changes with modern imaging techniques such as arterial spin labelling (ASL) MRI 27 makes them a promising biomarker for early AD similar to other established techniques such as fluorodeoxyglucose positron emission tomography. 28
This study investigated the impact that APOE and MAPT status on cerebral perfusion as measured with ASL MRI in a cohort of cognitively healthy young Chinese adults around 20 years of age. There were three main reasons for carrying out this research in such a young cohort. Firstly, we aimed to determine if perfusion differences were already present at this young age. Secondly, we sought to find the effect that these genetic risk factors had in the CBF of participants in good health as by using a young cohort, we controlled for other causes of CBF alterations associated with age and comorbidities (for example cardiovascular disease), thus increasing the chance that any observed perfusion differences were related to the genetic factors being investigated. Investigating the correlation between cerebral perfusion and risk factors for neurodegeneration in a cohort of young Chinese participants would also provide a novel finding, filling a gap in the evidence base as there is very little literature assessing this effect in participants of this age or ethnic group, in particular relating to MAPT. Finally, we hoped to see if perfusion differences observed in carriers of these genetic risk factors in previous studies also held true in a Chinese population. We hypothesised that we would observe areas of increased CBF in young APOE ε4 carriers in keeping with previous findings across the literature, and that increased perfusion may also be seen in carriers of other genetic risk factors for neurodegeneration such as MAPT.
Material and methods
Participants
Recruited from Southwest University in Chongqing, 160 cognitively healthy Chinese undergraduate students aged from 17 to 22 underwent 3 T MRI brain scans, genetic and cognitive testing. All of them gave written informed consent for their participation in the study. Research ethics was provided by Southwest University’s local ethics committee (2017-09-10) following ethical standards of the Helsinki Declaration of 1975 (and as revised in 1983).
Procedures
Genetic testing was performed via MassARRAY system (Agena iPLEX assay, San Diego, CA, United States) and included tests for APOE and MAPT rs242557 status. All participants had 3 T Siemens MRI scans including T1-weighted and PASL sequences. The imaging protocol followed that in the PREVENT dementia study as described by McKiernan et al. 29 The T1 sequence used was Magnetization Prepared Rapid Acquisition Gradient Echo (MPRAGE) and had 160 slices (repetition time = 2.3 s, voxel size = 1 × 1 × 1 mm). The PASL acquisition was by Proximal Inversion with Control of Off-Resonance Effects (PICORE) QT2 with 50 pairs of tag/control images, single delay and 2D echo-planar imaging (EPI) read-out (repetition time = 2.5 s, inversion time = 1.8 s, bolus duration = 0.7 s, voxel size = 3 ×3 × 7.5 mm). Six of the participants who underwent MRI did not complete the imaging protocol either due to technical issues with the scanner or the participants decided to end the session prematurely. This left 154 useable MRI data sets which were included in the subsequent analysis.
Image processing
The MRIs of each participant were processed and analysed using the Functional Magnetic Resonance Imaging (FMRIB) Software Library (FSL) version 6.0. 30 To process the images in preparation for perfusion mapping, the calibration image (M0) of each ASL image was separated using FSL’s fslroi tool, and the brain was extracted from each T1 image using the Brain Extraction Tool (BET). 31 One participant was excluded from the analysis at this stage due to incomplete ASL resulting in low signal to noise ratio (SNR).
Quantitative perfusion mapping was then run using the Bayesian Inference for Arterial Spin Labelling MRI (BASIL) toolbox. 32 BASIL outputs perfusion maps with perfusion measured in units of ml/100 g/min. 32 Fields used in BASIL included spatial regularisation 33 and partial volume correction. 34 BASIL provides an estimation of voxel-wise perfusion as well as incorporates data from other images (such as corresponding T1 images) thus allowing for spatial regularisation and correction for partial volume effects (PVE).
Following on from this, perfusion maps were normalised to the Montreal Neurological Institute (MNI) 152 standard space to allow for group comparison. This was done by creating a standard template for this dataset by registering the grey matter (GM) segmented T1 image of each participant to MNI 152 space using FSL’s optimised voxel-based morphometry (VBM) protocol, FSL-VBM, 35 and subsequently registering each participants’ ASL data to T1 and then template space with FMRIB’s Linear Image Registration Tool (FLIRT). 36 This was done for partial volume corrected (PVC) and non-PVC perfusion maps, as it is recommended to analyse PVC images alongside their uncorrected counterparts. 37 Additionally, the low resolution of ASL data makes it vulnerable to a PVE in which any individual voxel is unlikely to be comprised entirely of GM or white matter (WM), which can result in errors when estimating GM perfusion. 37 This effect was corrected for using BASIL, and furthermore PVE typically does not affect young adults. 37
Images were systematically checked for quality and the presence of errors or artifacts at each stage of the analysis. These checks included ensuring there were no structural abnormalities, motion artifacts, downstream signal losses in higher slices due to excessive label decay, or excessive perfusion differences between cerebral hemispheres (labelling asymmetry), which in such a young cohort with no known diseases would most likely be due to labelling errors, variations in the sensitivity of the receiver coil or processing errors. Six participants were excluded after running BASIL due to the presence of significant labelling asymmetry, which left 147 remaining participants with data suitable for the final analysis. See Figure 1 for the PASL labelling sequence and an example of labelling asymmetry errors.
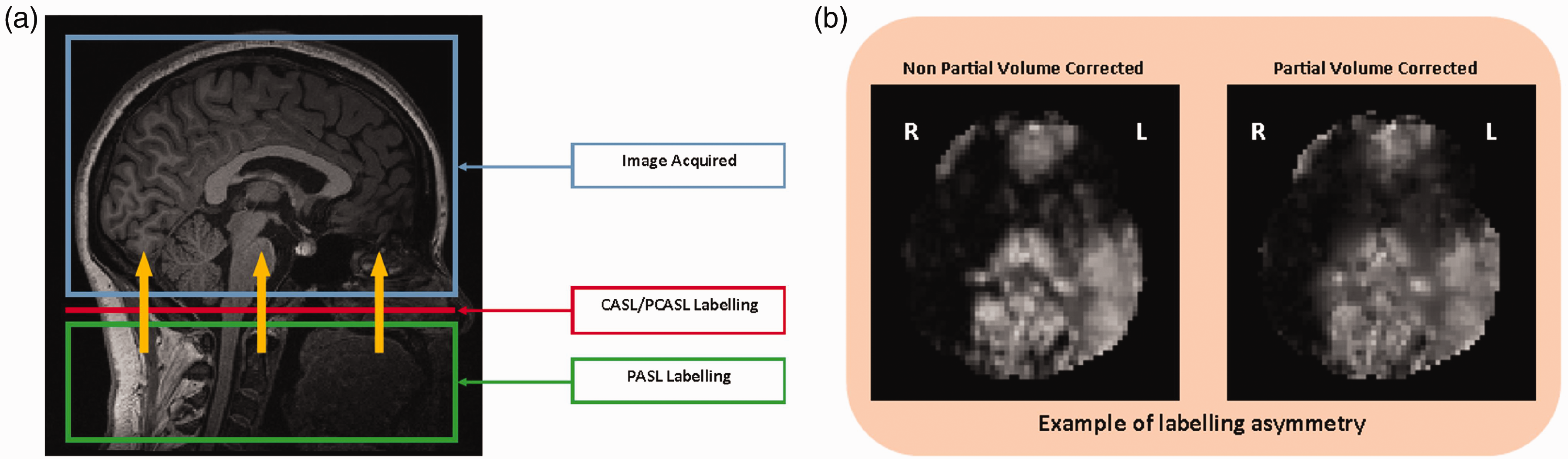
Panel a – The labelling sequence used in this project was PASL. The yellow arrows represent the flow of labelled blood. Adapted from Alsop et al., 27 T1 weighted MRI taken from this study. Panel b – Examples of possible causes of labelling asymmetry include labelling errors, variations in receiver coil sensitivity or processing errors.
Statistical analysis
For each statistical model, participants were grouped either based on APOE or MAPT rs242557 status, with MAPT A allele carriers (genotypes AA and AG) being grouped together in order to increase statistical power, as there is evidence suggesting that the A allele has a dominant effect on AD risk regardless of specific genotype. 18 Differences in demographic data between carriers and non-carriers of each risk allele were compared in R studio. 38 Continuous variables (age and years of education) were compared via two sample unpaired T tests and categorical variables (sex and APOE/MAPT carrier status) were compared via Chi-squared tests.
The statistical analysis for whole brain perfusion was carried out using “randomise”, FSL’s nonparametric permutation inference tool. 39 Randomise uses general linear modelling to output p-value images of MRI data. Two sample unpaired T tests were run to compare for the presence of APOE ε4, presence of MAPT rs242557 A allele, and effect of being homozygous for MAPT rs242557 A allele. Two-way analysis of variance (ANOVA) was also run to assess the interaction between these two risk genes. These included the covariates of age, sex, and years of education to control for basic demographic factors hypothesised to influence the functional properties of the neural and vascular tissue. Further T tests were also run to investigate the effects of age, education years and sex while controlling for APOE and MAPT status. Randomise was run with 5000 permutations and a GM mask derived from the study specific template was supplied. Threshold-free cluster enhancement (TFCE) was chosen as the output method because it has been shown to be more sensitive than other methods. 40 Randomise also runs family-wise error (FWE) correction to account for multiple group comparisons.
Global mean GM perfusion was also compared between genetic risk groups. The PVC and non-PVC mean GM perfusion of each participant were derived by BASIL. These were then analysed in R studio. 38 Mean GM perfusion was compared between carriers and non-carriers of both APOE and MAPT genetic risk factors via two sample unpaired T tests. Violin plots were also generated to visualise any differences in distribution of mean GM perfusion by APOE/MAPT status using the ggplot2 package for R studio.
Results
Demographics
Demographics are shown in Table 1. The APOE and MAPT groups did not differ significantly for age, years of education and sex. Additionally, groups were not different for the other genetic risk factor with no significant correlation observed between the presence of APOE and MAPT risk factors (χ2 = 0.78, p = 0.38).
Table of participant demographic data across the cohort and by genetic risk factor group.
Whole brain voxel-wise analysis
Figure 2 shows the averaged ASL data of the trial cohort used in the analysis prior to thresholding for perfusion changes as a visual demonstration of imaging quality used.
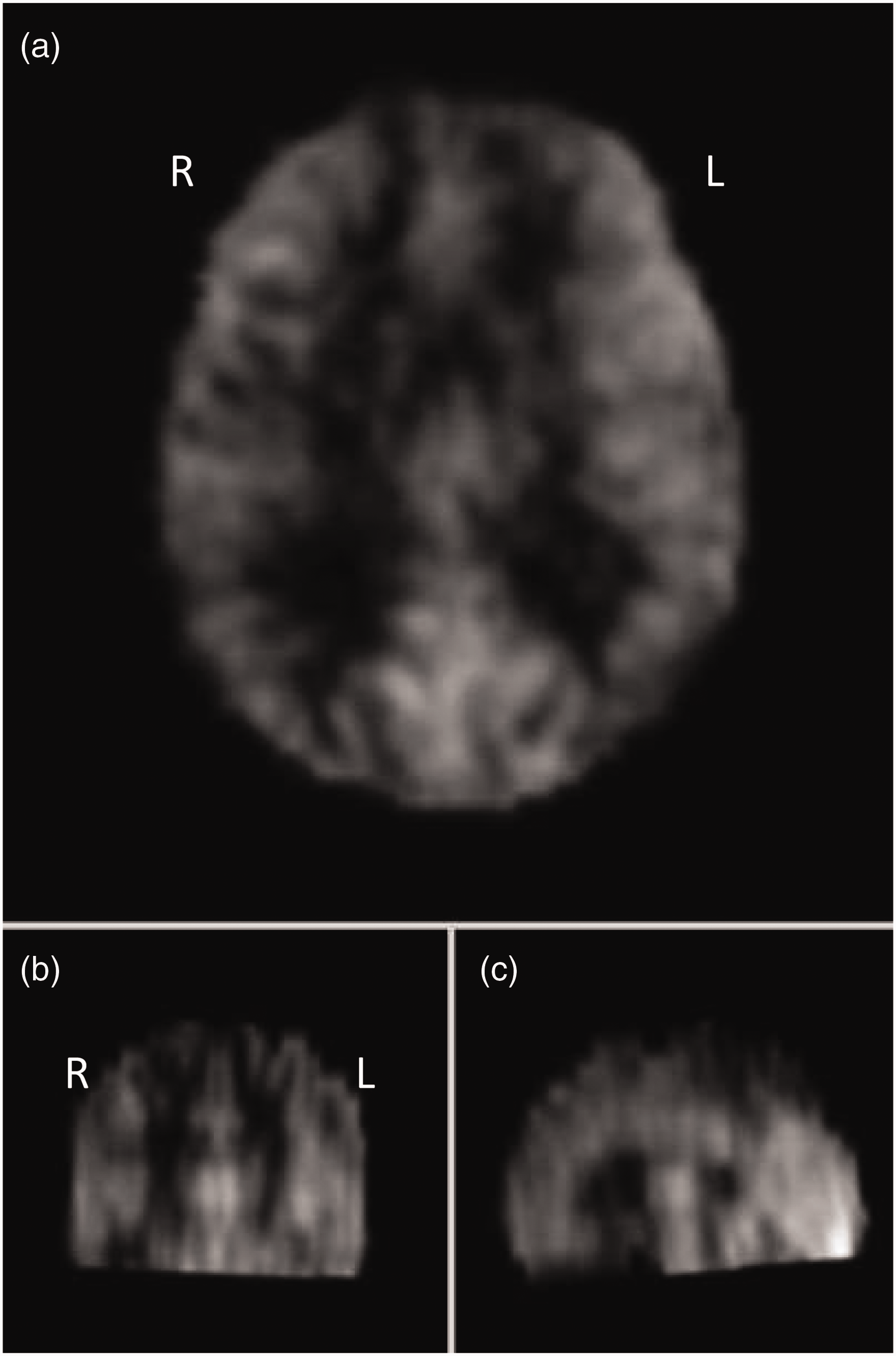
Averaged non-PVC imaging combining all cohort MRIs mapped to standard space before thresholding for areas of increased/decreased perfusion. Axial (Panel a), coronal (Panel b) and sagittal (Panel c) views shown.
The APOE analysis ultimately yielded no significant perfusion findings after FWE correction. Findings were largely consistent between non-PVC and PVC results. APOE analysis was also run while controlling for the additional variable of MAPT status. This did not change the results.
The main MAPT analysis compared cerebral perfusion between rs242557 A allele carriers (genotypes AA and AG) and non-carriers (genotype GG). The MAPT rs242557 A allele carrier group was associated with widespread increases in CBF prior to FWE correction; see Figure 3(a) for non-PVC results. After FWE correction, A allele carriers were found to still have significantly increased perfusion (p ≤ 0.05) in the left anterior cingulate cortex (ACC) in both the non-PVC and PVC analyses, and in the left insular cortex in the non-PVC analysis only (Figure 3(b)).
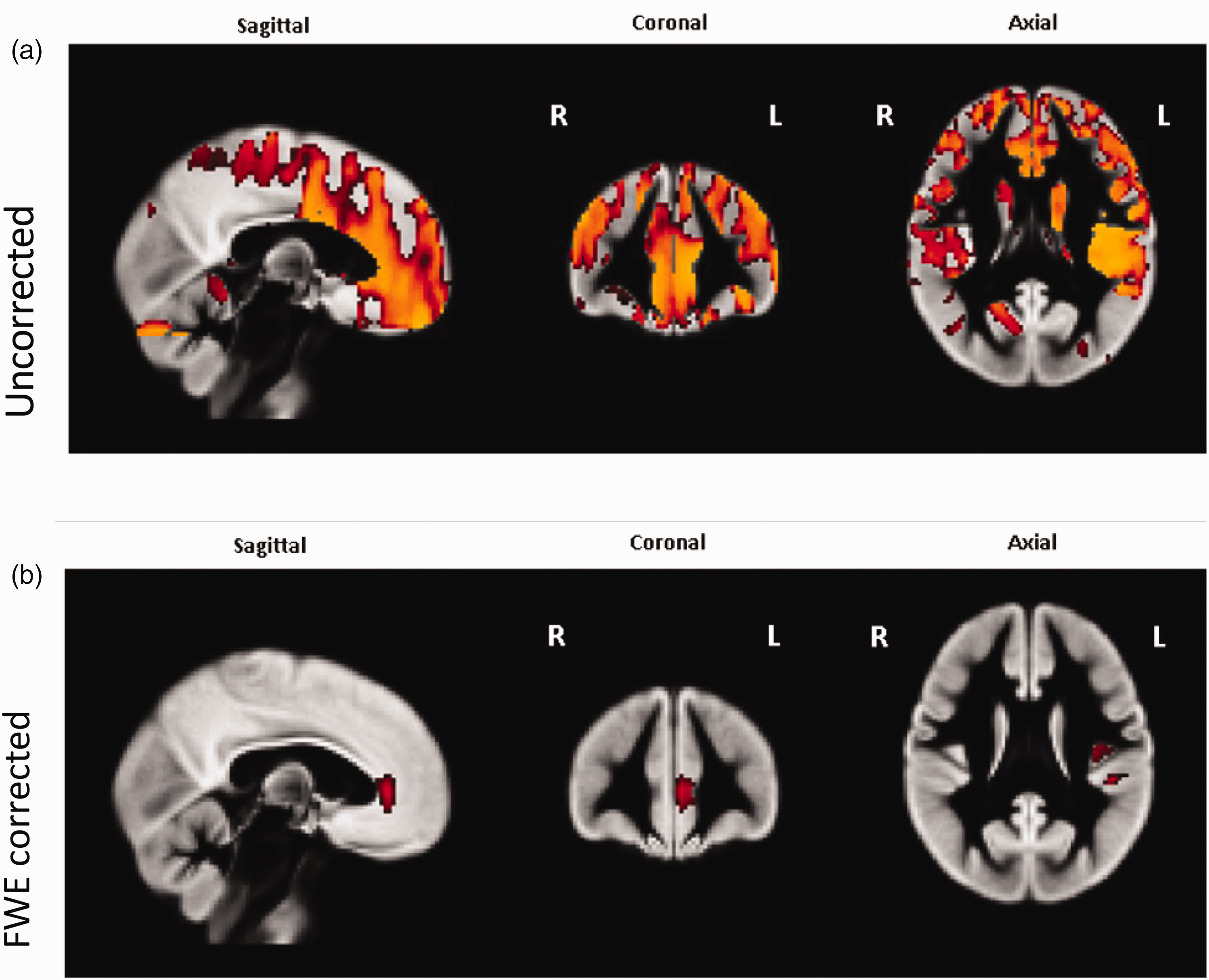
Areas of significantly increased perfusion in MAPT rs242557 A allele carriers when compared with non-carriers, all data shown from–non-PVC analysis. Panel a – uncorrected for FWE. Widespread perfusion increases were seen bilaterally in the superior and inferior frontal lobe. Panel b – Areas of significantly increased perfusion in MAPT rs242557 A allele carriers when compared to non-carriers, corrected for FWE. Increased perfusion was seen in the left anterior cingulate cortex (ACC) in both PVC and non-PVC analyses whereas increased perfusion in the left insular cortex was only seen in the non-PVC analysis.
Further analyses were carried out to investigate the influence of being homozygous for the rs242557 A allele (genotype AA) compared with those with at least one copy of the G allele (genotypes AG and GG). No significant perfusion effects were seen in this analysis. Finally, voxel-wise perfusion analysis for MAPT was repeated while controlling for the additional variable of APOE status. Changes in perfusion associated with A allele status were still observed in the same regions but with slightly reduced significance, no longer achieving significance after FWE correction. ANOVA assessing the effect of the interaction between APOE and MAPT rs242557 on voxel-wise cerebral perfusion found no significant results. This shows that there was no influence from the interaction between these two genetic factors on CBF in this young cohort.
Additional analyses were performed to investigate the impact of other demographic factors on cerebral perfusion in healthy participants at this age. The correlation between CBF and age, years of education, and sex were each investigated but none of these achieved any significant results. The only factor to be associated with any increased perfusion before FWE correction was age (see Figure 4), which showed increased perfusion predominantly in the right superior frontal gyrus correlated with increasing age but none of these clusters survived FWE correction.
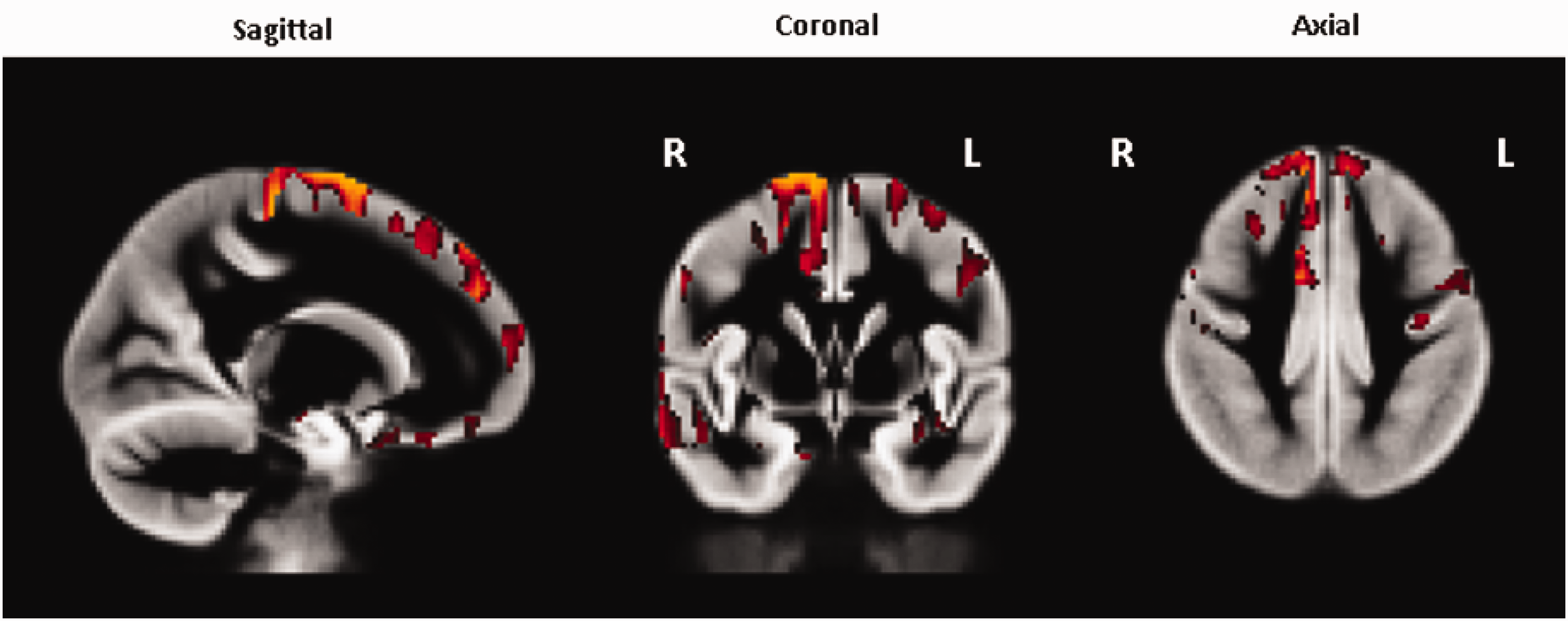
Areas of significantly higher perfusion correlated with older age, uncorrected for family-wise error, non-PVC results. Higher perfusion was seen predominantly in the right superior frontal gyrus. None of these findings retained significance after FWE correction.
Global mean grey mater perfusion analyses
In addition to whole brain voxel-wise perfusion analyses, the global mean perfusion across the entire cerebral GM of each participant was also compared between carriers and non-carriers of APOE and MAPT genetic risk factors. Again, this was carried out for both non-PVC and PVC data, 37 with PVC mean GM perfusion being higher than non-PVC for all participants. There was no significant difference in mean GM perfusion between APOE groups for either the non-PVC (T = 0.12, p = 0.90) or PVC (T = 0.062, p = 1.0) data. MAPT rs242557 status also had no significant effect on mean GM perfusion with either non-PVC (T = 0.98, p = 0.33) or PVC (T = 0.98, p = 0.33) data; see Figure 5. This lack of any generalised perfusion changes that may have signified no potential atrophy is to be expected in this young, healthy cohort.
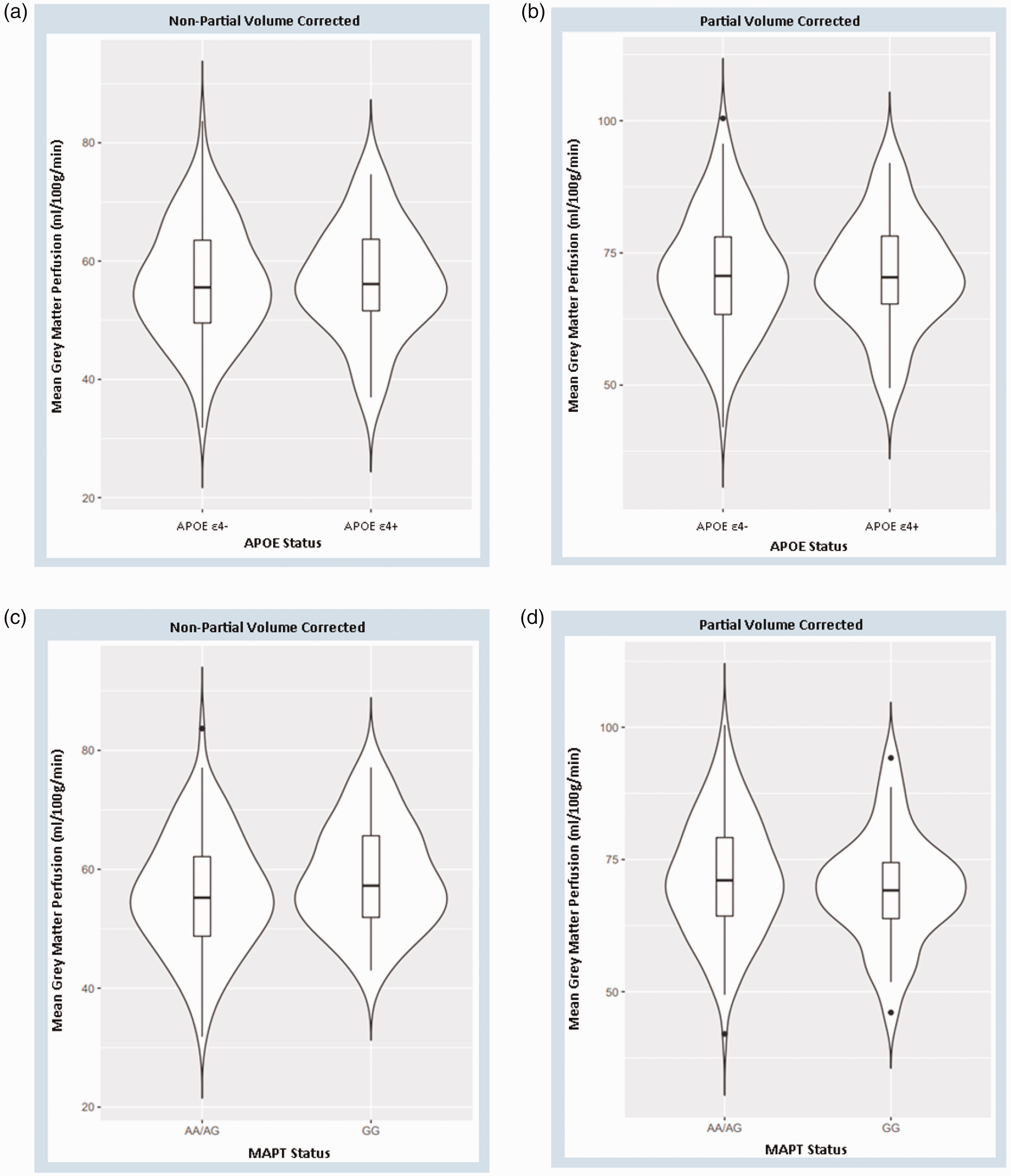
Violin plots of mean grey matter perfusion by genetic risk status showing median and interquartile range. Non-PVC plots shown as little difference in data distribution between APOE (Panel A & B) and MAPT (Panel C & D) groups was observed in either non-PVC or PVC plots.
Discussion
To our knowledge this study has the largest cohort of any investigation of cerebral perfusion changes in young adults to date. In this ASL MRI study, we did not find a significant association between APOE ε4 carrier status and cerebral perfusion in our cohort of young cognitively healthy adults. This was the case in both the voxel-wise analysis and the global mean GM perfusion analysis, as well as with both non-PVC and PVC data. This is consistent with two previous studies that have investigated whole brain perfusion based on APOE status in young participants (mean age below 30 years) via PASL and have similarly found no significant changes.41,42 However, other existing research has demonstrated significant perfusion changes in young APOE ε4 carriers.43,44 The aforementioned studies used similar small or moderately sized cohorts (minimum 15 and maximum 31 in the APOE4 carrier group) and all had majority female participants apart from one. 41
It is noteworthy that even among research that has found a correlation between APOE status and cerebral perfusion in young participants there is still inconsistency in their findings, with both increased perfusion 44 and decreased perfusion 43 being observed. Furthermore, even trials designed with the purpose of replicating earlier research have shown inconsistent results. 45 Differences in the applied sequences, methodological choices (e.g. image processing tools, calibration method, correction or no correction for partial volume effects and with what method) and quality control protocols which varied between these studies, could potentially explain such differences. These different results in small/moderately sized cohorts may also suggest that APOE ε4 exhibits considerable heterogeneity in its presentation at this age group or alternatively that no consistent perfusion effect is seen in these populations due to their young age and lack of cerebral atrophy, both of which could explain why we found no cerebral perfusion changes in this study. This emphasizes the need for larger longitudinal cohort studies starting at this age group.
The absence of significant changes in perfusion associated with APOE ε4 status means that, with caution, one may consider the implication of the null hypothesis in relation to the APOE status. This may indicate that, in this cohort, abnormalities in cerebral perfusion do not develop in early adulthood. The importance of this is three-fold. First, this highlights the need for longitudinal studies to determine when perfusion deficits develop in different populations – previous literature may suggest this time is around midlife as altered perfusion has been observed in cognitively healthy middle-aged and older APOE ε4 carriers.6,29,44,46 –48 Second, changes in perfusion may not be the first detectable pathological process and other early identifiable biomarkers may be required. Third, the development of perfusion abnormalities may occur alongside other pathological processes that occur in middle to later life. However, it is also worth noting that our findings in a Chinese cohort may not be reflective of differing demographics, as previous research has shown differing progression to AD in APOE ε4 carriers of different ethnic backgrounds. 49
We also included an analysis of effect of MAPT genotypes on CBF as an alternative marker of neurodegeneration. There is currently very little evidence linking MAPT status to CBF in pre-symptomatic carriers, making our finding of increased CBF in the ACC in young MAPT rs242557 A allele carriers a novel one. A previous study observed an inverse relationship between cerebral perfusion and estimated years to onset of symptoms in MAPT mutation carriers, 50 while another found no significant correlation between MAPT status and CBF but suggested this may be due to the small sample size used resulting in low statistical power. 51 Much of the existing research investigating the link between MAPT and CBF focuses on FTD rather than AD.50,51 Another study has previously demonstrated an early link between MAPT pathology and the ACC, 52 which alongside our results may suggest the ACC is a brain region that is sensitive to MAPT mediated vulnerabilities, however there were significant differences between our study and this earlier paper which instead focussed on a MAPT mutation, P301L, and its primary observation was ACC atrophy not perfusion changes. There has been research in both young 44 and old 53 APOE ε4 carriers that has shown altered CBF in the ACC which also highlights this as a region of interest for CBF changes in carriers of genetic risk factors for neurodegenerative disease, a correlation that makes logical sense due to the ACC’s role in the limbic system. 54
Although these findings appear to support our hypothesis of genetic risk factors for neurodegenerative disease correlating with increased CBF in young cognitively healthy carriers, there exist alternative interpretations of these results due to the nature of the young, cognitively healthy cohort and the cohort’s lack of diversity. Assuming that the MAPT rs242557 A allele increases risk of tau mediated neurodegeneration14,17 –19 then our results could be suggestive of a biphasic perfusion response akin to that seen in APOE ε4 carriers, in which early hyperperfusion precedes hypoperfusion.44,55 However, as our research involved a Chinese population and as it has been suggested that the G allele may instead confer increased risk in this ethnic group,21,22 then it is possible that the increases in perfusion we observed with the A allele (and corresponding decrease with the genotype GG) could signify an early neuroprotective effect of the A allele that may exist in Chinese populations. However, yet another possibility is that this is an incidental finding, as there is no proof that CBF changes in such a young cohort would correlate with potential neurodegenerative disease that would occur decades later. Furthermore, these findings may not be representative of populations of different demographic makeup to our entirely Chinese cohort.
Analysing effects of both APOE and MAPT status allowed us to also assess for any interaction between the two. We found no statistically significant interaction between the two genetic risk factors. Controlling for the effect of the other genetic risk factor did affect the significance of the results in the individual APOE and MAPT analyses but the effect was small, so one should not over-interpret this evidence. Additionally, there was no correlation between APOE and MAPT status in the demographic analysis, potentially suggesting that these two genetic factors are inherited independently. There is some evidence that suggests APOE and MAPT interact to affect AD pathology. MAPT rs242557 status has been shown to have a stronger association with late onset AD in a Chinese population in the presence of the APOE ε4 allele 21 and further evidence of this additive effect has been demonstrated by APOE ε4 exacerbating MAPT pathology. 56 However contrasting evidence exists showing that MAPT does not have a significant effect on AD risk in APOE ε4 carriers, 15 and a trial in a Hungarian Caucasian population found no interaction between MAPT and APOE in AD development, 57 so overall the current literature on this topic is variable and thus inconclusive.
Our analysis revealed no significant correlations between cerebral perfusion and age, years of education or sex. These findings were unsurprising in such a young cohort and especially one comprised entirely of undergraduate students with little variability in age and years of education. Increasing age is the main risk factor for neurodegenerative diseases such as AD and it has been correlated with decreases in cerebral perfusion as observed by ASL. 58 Additionally, both fewer years of education 59 and female sex 60 have also been associated with increased risk of AD and decreased cerebral perfusion. However, all of these associations have only been shown to exist in older populations. Our results suggest none of these factors interact with the genotypes we tested to influence perfusion changes in a young cognitively healthy population.
One major strength of the study is that it has more samples than most other similar studies to date.41 –44 Additionally, the use of a young and cognitively healthy cohort controls for other causes of perfusion changes that are prevalent in older populations such as hypertension, dyslipidaemia, metabolic syndrome, and age-related cerebral atrophy, increasing the likelihood that the observed differences in perfusion are a result of the genetic factor being investigated rather than other factors. Using a younger cohort also reduces the influence of PVE on ASL data, as PVE is exacerbated by cerebral atrophy which is very rare in healthy young adults. 37
The study was however limited by using a young cohort that potentially was not being representative of AD as established AD does not begin until many decades later and is not guaranteed to occur in carriers of genetic risk factors. As all participants were of the same ethnic group (Chinese), it is possible that our findings may not generalise to other populations, especially in light of the evidence regarding MAPT rs242557 potentially having a different effect in Chinese to other ethnicities.21,22 Furthermore, because the entire cohort was characterised by a higher educational attainment and showed little age-related variability, it is impossible to determine any effect of significant education or age differences on cerebral perfusion at this age from our data. Although ASL has a number of advantages over other imaging modalities, it does however also have limitations including low resolution, vulnerability to motion and relatively short lifetime of the magnetically labelled blood water. 27 In this particular study we used a low SNR single time-point ASL variant, PICORE, which is not in line with the primary recommendation by the ASL community but is still within the recommended pulsed ASL implementations.
Conclusions
This was a novel study assessing CBF changes in a young, cognitively healthy Chinese cohort with a larger sample size than previous similar research. Our results showed no significant correlation between APOE status and cerebral perfusion, an interesting negative finding to contrast to previous smaller cohort papers that have observed areas of both increased and decreased CBF. While we observed a novel significant finding of increased perfusion in the left ACC and left insular cortex in carriers of the MAPT rs242557 A allele, these findings are not consistent with the wider literature. Taken in isolation these results could suggest that MAPT rs242557 related vulnerability begins at early adulthood, and these perfusion changes could be a result of MAPT’s involvement in synaptic plasticity or compensatory mechanisms, however it is very unlikely that amyloid and tau related pathology could begin at this age and without similar findings across the literature these perfusion changes in a young healthy cohort may simply be incidental. Further longitudinal studies investigating perfusion changes are needed to establish the effect these genes have during the life course, to assess if any observed hyperperfusion in young cohorts is reproducible and if so to determine whether this correlates with areas of hypoperfusion in later neurodegeneration.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LS’s participation is funded by Alzheimer’s Research UK Senior Research Fellowship (ARUK-SRF2017B-1) and NIHR Sheffield Biomedical Research Centre. JZ is funded by China Ministry of Education’s Humanity and Social Sciences Project (23YJA190001). SSB is supported by the Association of British Neurologists (Stroke Association/Berkeley Foundation). JOB is supported by the NIHR Cambridge Biomedical Research Centre.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JOB has acted as a consultant for TauRx, Novo Nordisk, Biogen, Roche, Lilly and GE Healthcare and received grant or academic support from Avid/Lilly, Merck and Alliance Medical. CR is the founder of Scottish Brain Sciences, and acted as a consultant for Biogen, Eisai, MSD, Actinogen, Roche, and Eli Lilly, and received payment or honoraria from Roche and Eisai in the past.
Authors’ contributions
SKB conducted the analyses. SKB and LS drafted the manuscript. JZ collected the data. CR, LS and JZ set up the extension of the PREVENT-Dementia study. CR, MED, AM, SSB, MDM and JOB provided feedback on the manuscript. LS provided overall supervision for the study.
