Abstract
Disruptions to the gut-brain-axis have been linked to neurodegenerative disorders. Of these disruptions, reductions in the levels of short-chain fatty acids (SCFAs), like butyrate, have been observed in mouse models of Alzheimer’s disease (AD). Butyrate supplementation in mice has shown promise in reducing neuroinflammation, amyloid-β accumulation, and enhancing memory. However, the underlying mechanisms remain unclear. To address this, we investigated the impact of butyrate on energy metabolism in mouse brain slices, primary cultures of astrocytes and neurons and in-vivo by dynamic isotope labelling with [U-13C]butyrate and [1,2-13C]acetate to map metabolism via mass spectrometry. Metabolic competition assays in cerebral cortical slices revealed no competition between butyrate and the ketone body, β-hydroxybutyrate, but competition with acetate. Astrocytes favoured butyrate metabolism compared to neurons, suggesting that the astrocytic compartment is the primary site of butyrate metabolism. In-vivo metabolism investigated in the 5xFAD mouse, an AD pathology model, showed no difference in 13C-labelling of TCA cycle metabolites between wild-type and 5xFAD brains, but butyrate metabolism remained elevated compared to acetate in both groups, indicating sustained uptake and metabolism in 5xFAD mice. Overall, these findings highlight the role of astrocytes in butyrate metabolism and the potential use of butyrate as an alternative brain fuel source.
Introduction
The gut-brain axis is a complex network of bidirectional signalling between gastrointestinal tract bacteria and the central nervous system (CNS). 1 This communication occurs through a combination of immune, neuroendocrine, or vagal signalling and is central to homeostasis. 2 As research into the gut microbiome expands, it is being associated with a range of psychiatric and neurodegenerative disorders such as depression, Parkinson’s disease (PD) and Alzheimer’s disease (AD).3,4 Gut microbiota produce a variety of metabolites which are absorbed into peripheral circulation as well as the brain, yet the extent to which these metabolites reach the brain and directly target specific cells remains unclear.2,5 Of the metabolites produced in the gut, short chain fatty acids (SCFAs) are the most abundant and display neuroactive properties. 6
The SCFAs acetate, propionate and butyrate are derived predominantly from bacterial fermentation of dietary fibre and saccharides in the gut. 7 These SCFAs play a pivotal role in modulating the body’s immune response, regulating appetite, and acting as the main energy substrate of colonocytes.8 –10 In the brain specifically, SCFAs preserve blood-brain barrier (BBB) integrity 11 regulate microglia function, 12 activate free fatty acid receptors and affect gene expression through histone deacetylase inhibition (HDACi).13 –15 Among these SCFAs, butyrate, has gained significant attention as a neuroprotective agent due to its metabolic, HDACi and anti-inflammatory properties.16,17 Studies in Alzheimer’s disease (AD), wild-type (WT) and germ free (GF) mice have further demonstrated the transmission of AD pathogenesis and cognitive decline via microbiota.18,19 Recent studies exploring the link between dysbiosis, and AD have revealed that reductions in the levels of butyrate producing bacteria and subsequently lower butyrate levels are associated with AD.20 –22 Increasing butyrate levels in-vivo and in vitro has demonstrated a potential therapeutic impact on AD by reducing the levels of amyloid-β peptide (Aβ) and tau proteins in the brains of mice.23 –25 The HDACi activity of butyrate and its neuroprotective effects has been studied in detail, however, evidence suggests that these benefits could also be through gut-derived butyrate modulating brain glucose metabolism.26,27 In addition to this, mouse studies on butyrate supplementation, probiotic treatment, and high fibre diets have shown increases in brain butyrate concentrations,28,29 but the direct metabolic effects of butyrate on the brain remains mostly unexplored. Under normal conditions glucose is the primary fuel of the brain, but a progressive decrease in glucose uptake and metabolism is observed in AD patients prior to cognitive decline.30,31 Fortunately, the brain can use alternative fuels such as ketone bodies, amino acids, and fatty acids.32,33 Butyrate may serve as an energy source if it reaches the brain at adequate and safe levels, restoring energy homeostasis, and ameliorating the symptoms of AD.5,34
The purpose of this study was to enhance comprehension of butyrate brain metabolism at both the cellular and whole organism levels. To accomplish this, we mapped butyrate metabolism by stable isotope tracing in the presence and absence of other known cell specific brain metabolites (acetate and β-hydroxybutyrate). 35 The metabolic mapping was performed in acutely isolated cerebral cortical slices, primary cultured cortical astrocytes, and neurons and finally in-vivo in the 5xFAD mouse model of AD using 13C isotopically enriched butyrate with gas chromatography-mass spectrometry (GC–MS) analysis. Our results reveal that butyrate is oxidatively metabolised in brain slices and more specifically astrocytes, supporting energy supply and glutamine synthesis for use in neurons. In the in-vivo model, we show that both wild-type control and 5xFAD mouse brains absorb and metabolise [U-13C]butyrate, providing new avenues for understanding and developing metabolic interventions for AD.
Materials and methods
Materials
The stable 13C isotopes [1,2-13C]acetate (CLM-440-SP, sodium salt, 99%), [U-13C]D-β-hydroxybutyrate (CLM-3853-PK, sodium salt, 97%) and [U-13C]glucose (CLM-1396-5, 99%) were all obtained from Cambridge Isotope Laboratories (Andover, MA, USA) and [U-13C]butyrate (488380, sodium salt, 99%) was purchased from Sigma Aldrich (St. Louis, MO, USA). D-glucose (16301), acetate (S5636, sodium salt 99%), butyrate (303410, sodium salt 97%) and (R)-β-Hydroxybutyric acid-βHB, (54920) were purchased from Sigma Aldrich (St. Louis, MO, USA). All other chemicals used were of the purest grade available from commercial sources.
Animals
Male NMRI mice were purchased from Envigo (Cambridgeshire, United Kingdom) and were used for experiments at 11 weeks of age. Male mice were used for the brain slice study to minimise sex-specific metabolism variation as changes in sex hormones can affect brain mitochondrial function. 36 The NMRI mice were acclimatised for two weeks before experiments and were single housed in individually ventilated cages. 7-day old pups from NMRI mice were used for the preparation of cultured cortical astrocytes and cultured cortical neurons were produced using day 15 gestation NMRI mouse foetuses. Female transgenic 5xFAD mice (TG(APPSwFlLon, PSEN1*M146L*L286V)6799Vas, Jax strain: 034840) and wild-type (WT) female mice (Jax strain: 100012) were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and used for experiments at 4 months old which correlates with increased β-amyloid 42 deposition, gliosis and cognitive decline. 37 The 5xFAD mice express five familial AD mutations in the amyloid precursor protein (APP) and presenillin1 (PSEN1) genes under the neuron-specific Thy1 promoter, leading to rapid brain amyloid deposition. 37 The mice were housed together in individually ventilated cages. Female mice were chosen for the AD and WT in-vivo experiments as plaque pathology and neuroinflammation is more severe in the hippocampus and cortex of female 5xFAD mice compared to males.38 –40 Changes to brain glucose metabolism are also reported to be greater in female 5xFAD, in closer resemblance to human AD. 41 The transgenic and WT mice were genotyped from ear clippings by a standard PCR protocol (Jax protocol: 23370) as previously described. 42 All the mice were bred and maintained in a specific pathogen-free, temperature and humidity-controlled environment with 12 h light/dark cycle and free access to water and chow at the Department of Drug Design and Pharmacology, University of Copenhagen. Experiments were approved by the Danish National Ethics Committee and performed according to the European Convention (ETS 123 of 1986). The experiments using animals have been reported in compliance with the ARRIVE guidelines. The experimental sets including stable-isotope injections and microwave brain fixations were made in compliance with the Ministry of Food, Agriculture and Fisheries of Denmark, permit number: 2020-15-0201-00441.
Brain slice incubations
The incubation procedure for brain slices acutely isolated from mice followed established protocols. 43 Euthanasia was performed using cervical dislocation, followed by decapitation. The brain was removed and transferred to slushed ice-cold artificial cerebrospinal fluid (ACSF) with the following composition in mM: NaCl 128, NaHCO3 25, D-glucose 10, KCl 3, CaCl2 2, MgSO4 1.2, KH2PO4 0.4, pH = 7.4. Subsequent dissection focused on extracting cerebral cortices from the mouse brain, with the remaining tissue discarded. The isolated mouse cerebral cortices were sliced into 350 μm sections using a McIlwain tissue chopper (The Vibratome Company, O’Fallon, MO, United States), and the slices were separated under a microscope. Two cortical slices per condition were placed in each chamber, positioned just beneath the surface of 10 mL 37°C oxygenated (5% CO2/95% O2) ACSF and pre-incubated for 60 minutes to allow for recovery from slicing in a custom-made incubation apparatus. 43 The media were later replaced with ACSF containing stable 13C-enriched compounds: 5 mM [1,2-13C]acetate, 1 mM [U-13C]butyrate or 1 mM [U-13C]BHB (all supplemented with 5 mM unlabelled D-glucose) and incubated for an additional 60 minutes. For the competition assay, slices were incubated with [1,2-13C]acetate in the presence of unlabelled 12C-butyrate or 12C-BHB, [U-13C]butyrate in the presence of 12C-acetate or 12C-BHB and [U-13C]BHB in the presence of 12C-acetate or 12C-butyrate for 60 minutes. All incubations were concluded by transferring slices to ice-cold 70% ethanol. The slices were then sonicated and centrifuged (4000 g × 20 mins); the supernatant was extracted and lyophilized before analysis using gas chromatography–mass spectrometry (GC–MS).
Preparation of primary cultures of cortical astrocytes and neurons
The primary cultures of cortical neurons and astrocytes were prepared according to Walls et al., 2014.44 To prepare astrocytes, 7-day old pups from NMRI mice were decapitated, and cortices were isolated and passed through a nylon filter with a pore size of 80 µm, by mechanical dissociation into DMEM containing 2.5 mM glutamine, 6 mM glucose, 26.2 mM NaHCO3, 100 IU/mL penicillin and 20% fetal calf serum. Then the suspension was triturated thrice using a 13 G cannula, and the cells were then seeded into 25 cm2 flasks, at a density of 2.5 × 106 cells/ml. The astrocytes were incubated in a humid atmosphere with 5% CO2 at 37°C and the medium was changed twice a week and after two weeks of culturing, 0.25 mM dibutyryl cyclic AMP (dBcAMP) was added to the medium for maturation. After three weeks the astrocytes were used for experiments.
Cortical neurons were prepared from foetuses of 15-day gestation NMRI mice quickly and cortices were collected in a 35 mm Petri dish containing a solution of Krebs buffer, bovine serum albumin (BSA) and MgSO4 stock solution (150 mM). The cortices were transferred to a Teflon plate and cut into small cubes (0.5 mm × 0.5 mm) using a razor blade and homogenised using a Pasteur pipette. The tissue was gently centrifuged and stopped after reaching 500 g. The supernatant was removed, and the tissue was suspended in a solution containing trypsin and incubated for 15 minutes at 37°C after which trypsinisation was inhibited. 44 The tissue suspension was centrifuged (500 g × 5 mins), the supernatant was discarded, and the pellet was triturated in a solution of DNAase, trypsin inhibitor and MgSO4 stock solution (Walls et al., 2014). The suspension was transferred to a modified DMEM mixture 44 and seeded onto poly-D-lysine coated 6-well plates at a density of 2.75 × 106 cells/ml. After 2 days of culturing, 20 μM cytosine arabinoside was added to inhibit proliferation of astrocytes and 12 mM glucose was added after days 2. After 1 week of culturing, the neurons were used for experiments.
Cortical astrocytes and neurons incubations
For the primary cultures of cortical astrocytes and neurons to acclimatise to lower concentrations of glucose, they were pre-incubated with DMEM culture media containing 2.5 mM D-glucose at 37°C, 5% CO2 for 2 hrs. After pre-incubation the astrocytes and neurons were washed with 37°C phosphate-buffered saline (PBS), before being incubated with DMEM containing 13C labelled substrates for 60 minutes at 37°C, 5% CO2. The astrocytes were incubated with [1,2-13C]acetate (2.5 mM) or [U-13C]butyrate (250, 500 or 1000 µM, concentrations reported to increase mitochondrial function 34 with and without competing unlabelled 12C substrates, 12C-acetate (2.5 mM) or 12C-butyrate (250, 500 or 1000 µM). For the neurons, incubation conditions of [U-13C]glucose (2.5 mM) or [U-13C]butyrate (250, 500 or 1000 µM) with and without competing unlabelled 12C substrates, 12C-glucose (2.5 mM) or 12C-butyrate (250, 500 or 1000 µM). All conditions, except [U-13C]glucose, were further supplemented with 2.5 mM unlabelled D-glucose. After incubation, the medium was collected, and metabolism halted by washing the cells with ice-cold PBS. Cells were lysed and extracted with 70% ethanol, then centrifuged at 4°C (4000 g × 20 mins). The supernatant containing soluble cell extracts were lyophilised and kept at −20°C before being analysed by GC-MS.
In-vivo metabolism and brain tissue microwave fixation
WT and 5xFAD female mice were injected (intraperitoneal) with [1,2-13C]acetate (504 mg/kg, 0.6 M) or [U-13C]butyrate (684 mg/kg, 0.6 M).45 –48 15 minutes after injection, metabolic brain activity was instantly stopped via microwave fixation at 4 kW for 1.10 seconds (model GA5013, Gerling Applied Engineering). The mice were then decapitated, and the cerebral cortex, hippocampus and cerebellum were dissected, then quickly transferred dry ice until extraction. The tissue was sonicated with 70% ethanol and centrifuged (4000 g × 20 mins). The supernatant was collected, lyophilised, and kept at −20°C before being analysed by GC-MS. The experimenter was blinded to the genotype of the injected mice before data analyses were performed.
Metabolic mapping using gas chromatography coupled to mass spectrometry (GC–MS)
The 13C-enrichment of TCA cycle metabolites and amino acids was determined using GC–MS according to a method previously described. 44 Brain slice, primary culture and microwave fixation cell extracts were reconstituted in water, acidified, and extracted twice with ethanol. The metabolites were then extracted into an organic phase with 96% ethanol/benzene and derivatized using N-tert-butyldimethylsilyl-N-methyltrifluoroacetamide. The samples were analysed by GC (Agilent Technologies 7820 A chromatograph, J&W GC column HP-5MS, parts no. 19091S-433) linked to MS (Agilent Technologies 5977E mass spectrometer). The 13C-enrichment was corrected for the natural abundance of 13C by analysing standards of the unlabelled metabolites of interest. Data are presented as percentage of labelling of the isotopologue M + X, where M corresponds to the molecular weight of the unlabelled molecule and X is the number of 13C-enriched carbon atoms in the molecule.
Statistical analysis
Data are presented as means ± standard deviation (SD), with individual data points shown. 8 mice (n = 8) were used for the brain slice incubations and 4–5 mice (n = 4–5) mice were used for the in-vivo brain metabolism experiments. One outlier in Figure 1(b) ([U-13C]butyrate with unlabelled acetate) and in Figure 3(a) (1000 μM [U-13C]butyrate with unlabelled acetate) was excluded using a Rout test Q = 1% on Graphpad. Each individual data point represents a biological replicate (obtained from an individual animal). The values from the cell cultures were obtained from a minimum of eight technical replicates (n = 8) from three cell culture batches (N = 3). Data were first tested for normality using the Shapiro–Wilk test (α = 0.05). Unpaired data sets were then compared using either one-way ANOVA with Dunnett’s T3 or Bonferroni comparisons or Kruskal-Wallis test with Dunn’s post-hoc test. Unpaired two-way ANOVA tests were undertaken with Fisher’s least significant difference (LSD) for multiple comparisons. For data comparing two groups, unpaired data were compared using either two-tailed Welch’s t-test or Mann–Whitney test. The significance level was set at p < 0.05 and is indicated as *p < 0.05, **p < 0.01, ***p < 0.001 ****p < 0.0001.
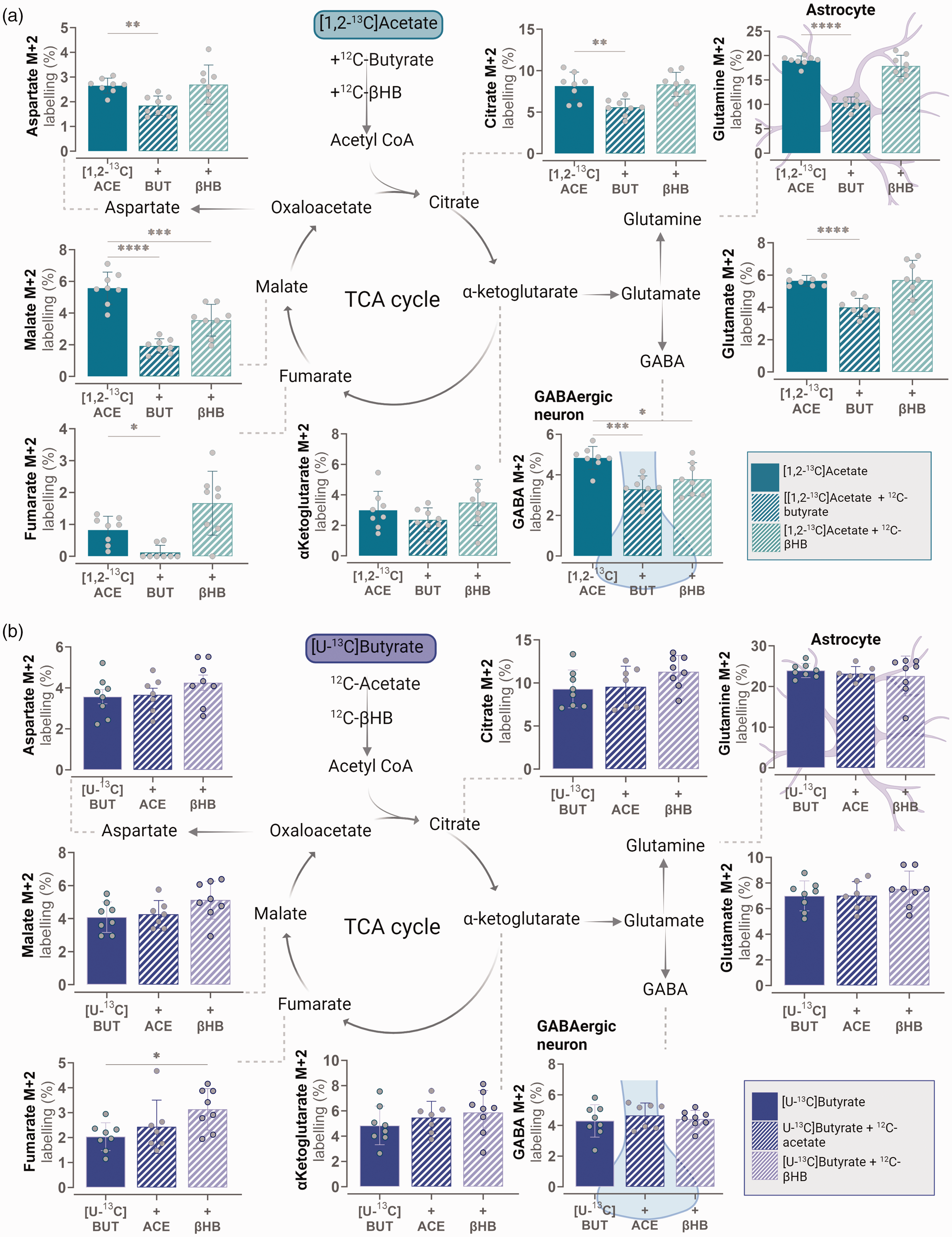
Butyrate is preferred over acetate as a metabolic substrate in NMRI mouse cerebral cortex slices. (a) Metabolic competition assay between [1,2-13C]acetate in the absence and presence of unlabelled 12C-butyrate or 12C-βHB and (b) metabolic competition assay between [U-13C]butyrate in the absence and presence of unlabelled 12C-acetate or 12C-βHB. Concentrations: 5 mM Acetate, 1 mM Butyrate and 1 mM βHB, all incubations with addition of 5 mM D-glucose. ACE: Acetate, BUT: butyrate βHB: β-hydroxybutyrate. Mean ± SD, n = 7–8 from individual animals, repeated measures one-way ANOVA with Bonferroni post hoc test or Kruskal-Wallis test with Dunn’s post-hoc test., *p < 0.05, **p < 0.01, ***p < 0.001 ****p < 0.0001.
Results
Butyrate is a preferred substrate over acetate in mouse cerebral cortex slices
To investigate the metabolic preference between acetate, butyrate and β-hydroxybutyrate (βHB), we performed metabolic competition experiments using isotope tracing in isolated cerebral cortical slices from NMRI mice. 49 The cortical slices were incubated in the presence of either [1,2-13C]acetate or [U-13C]butyrate together with a competing unlabelled (i.e. 12C) metabolic substrate (Figure 1). Both the labelled and unlabelled metabolic substrates will enter cellular metabolism as acetyl CoA units, giving rise to subsequent 13C/12C enrichment in TCA cycle intermediates and connected amino acids (Figure 1). Metabolism of an unlabelled 12C substrate will dilute the 13C labelling, showing which metabolic substrate is preferred. When the slices were incubated with [1,2-13C]acetate, a substrate primarily metabolised in astrocytes 50 and exposed to unlabelled 12C-butyrate, we found consistent reductions in the 13C enrichment of most measured metabolites (Figure 1(a)). In contrast, when the slices were incubated with [U-13C]butyrate and exposed to unlabelled 12C-acetate, no change in 13C enrichment was observed in any of the metabolites (Figure 1(b)). These results demonstrate that metabolism of butyrate is preferred over that of acetate in the brain slices when the metabolic substrates are provided together. Intriguingly, metabolism of [U-13C]butyrate resulted in the highest 13C enrichment in glutamine (Figure 1(b)), which was also the case for [1,2-13C]acetate (Figure 1(a)). Since glutamine is selectively synthesised in astrocytes, 51 this may indicate that butyrate is primarily metabolised in astrocytes. In addition, we included the ketone βHB in the competition experiment. βHB also enters cellular metabolism as acetyl CoA but is oxidised to a larger extent in neurons than astrocytes.52,53 We found that unlabelled 12C-βHB had little to no effect on [1,2-13C]acetate and [U-13C]butyrate metabolism (Figure 1(a) and (b)). Moreover, no changes were observed in any of the measured metabolites when slices were incubated with [U-13C]βHB and exposed to either 12C-acetate and 12C-butyrate (Supplementary Figure 1). The lack of substrate competition between butyrate and βHB, combined with the observation that butyrate is preferred over the astrocytic substrate acetate (Figure 1), and the high 13C enrichment in glutamine from [U-13C]butyrate metabolism, may suggest that butyrate metabolism is primarily taking place in astrocytes.
Butyrate competes with acetate as a metabolic substrate in astrocytes but does not compete with glucose in neurons
The metabolic competition experiments of the NMRI mouse brain slices suggested that butyrate metabolism may primarily take place in astrocytes. To further investigate the potential differences in astrocytic and neuronal metabolism of acetate compared to butyrate, incubation experiments were conducted in primary cultures of cortical mouse astrocytes and neurons separately. As acetate is primarily metabolised in astrocytes 50 a competition assay between 2.5 mM of [1,2-13C]acetate and a range of unlabelled 12C-butyrate concentrations (250, 500 and 1000 μM) could give an indication of metabolic substrate preference within the astrocytic compartment (Figure 2(a)). Glucose is the main energy substrate of the brain, and metabolism of [U-13C]glucose provides an overview of general metabolic function in the brain slices. 43 Therefore, a second competition assay between 2.5 mM [U-13C]glucose and 12C-butyrate (250, 500 and 1000 μM) was performed to assess preferential substrate metabolism in neurons, as they have been estimated to account for 80% of total brain energy expenditure 54 (Figure 2(b)). Astrocytes incubated with [1,2-13C]acetate in the presence of 1000 μM 12C-butyrate resulted in a significant reduction in the M + 2 labelling across most of the metabolites measured (citrate, glutamine, glutamate, fumarate, malate and aspartate). M + 2 labelling of α-ketoglutarate upon incubation with [1,2-13C]acetate and 1000 μM 12C-butyrate was not reduced when compared to the [1,2-13C]acetate control (Figure 2(a)). Interestingly, even at the lowest concentration of added 12C-butyrate tested (250 μM), the metabolite glutamate showed a 44.9% reduction in M + 2 labelling compared to the control of [1,2-13C]acetate metabolism in the absence of unlabelled 12C-butyrate (Figure 2(a)). These results indicate that butyrate is a highly competitive substrate for oxidative metabolism in cultured astrocytes compared to acetate. However, in neuronal cultures, the addition of unlabelled 12C-butyrate to [U-13C]glucose did not affect the level of 13C incorporation into any TCA cycle intermediates and amino acids except for fumarate (Figure 2(b)). Increasing concentrations of unlabelled 12C-butyrate were unable to reduce M + 2 labelling of the other TCA cycle metabolites from [U-13C]glucose, suggesting that glucose is a preferred metabolic substrate in neurons when compared with butyrate.
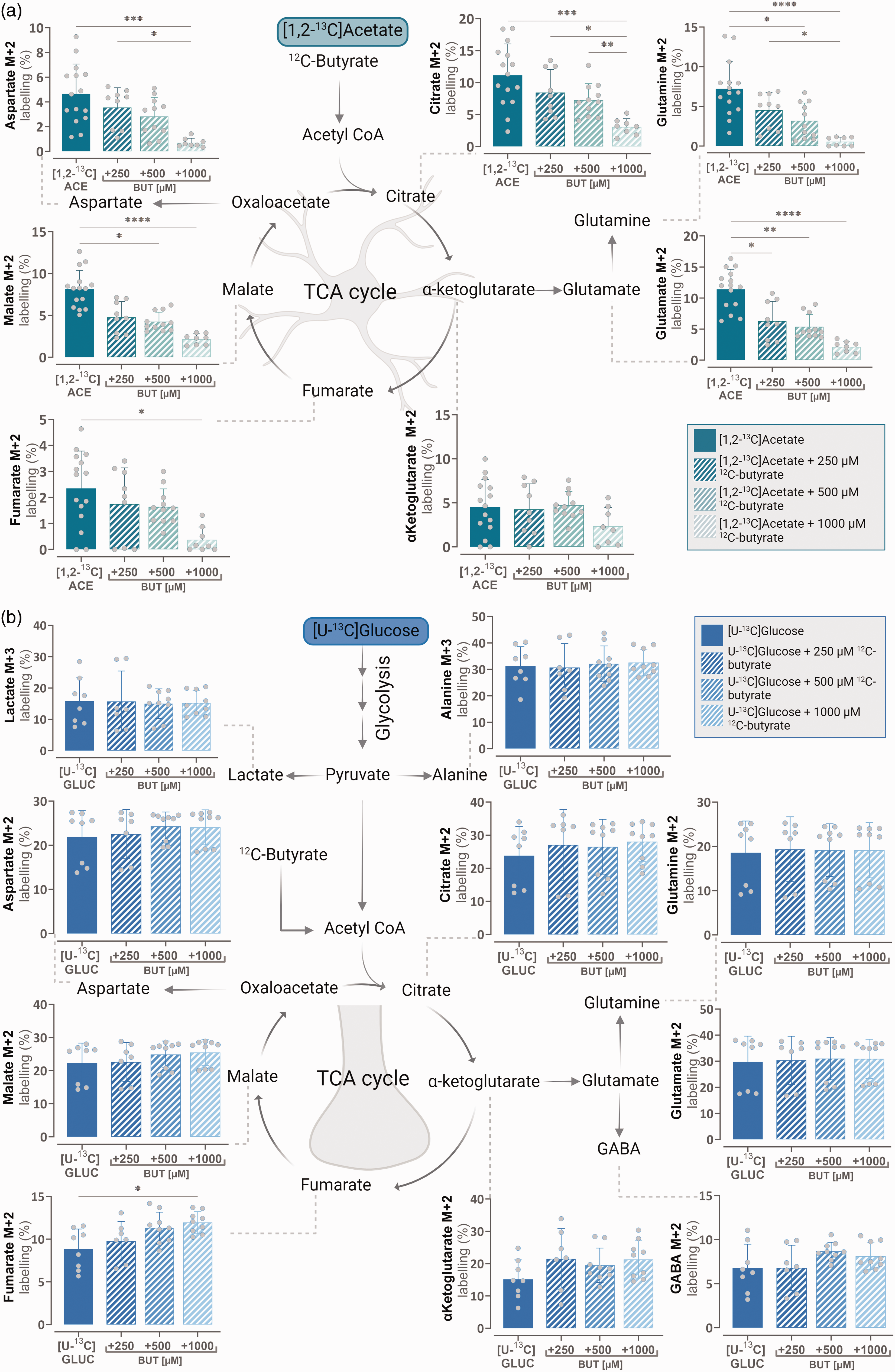
Butyrate competes with acetate as a metabolic substrate in astrocytes but does not compete with glucose in neurons. (a) Intracellular M + 2 carbon labelling of TCA cycle intermediates and amino acids in cultured cortical mouse astrocytes after incubation Continued.with [1,2-13C]acetate (2.5 mM) in the absence and presence of unlabelled 12C-butyrate (250, 500 or 1000 μM) and 2.5 mM 12C-glucose and (b) intracellular M + 3 carbon labelling of glycolytic products and M + 2 carbon labelling of TCA cycle intermediates and amino acids in cultured cortical mouse neurons after incubation with [U-13C]glucose (2.5 mM) in the absence and presence of unlabelled 12C-butyrate (250, 500 or 1000 μM). ACE: acetate, BUT: butyrate, GLUC: glucose. Mean ± SD, minimum N = 3, n = 8, one-way ANOVA with Bonferroni post hoc test or Kruskal-Wallis test with Dunn’s post-hoc test,*p < 0.05, **p < 0.01, ***p < 0.001 ****p < 0.0001.
Butyrate is preferred over acetate as a metabolic substrate in astrocytes and is metabolised to a lesser extent in neurons
In order to confirm results described above and explore whether there was a metabolic preference for butyrate over acetate in cultured astrocytes, metabolism of [U-13C]butyrate with and without 12C-acetate was tested. Competition assays between [U-13C]butyrate (250 and 1000 μM) and unlabelled 12C-acetate (2.5 mM) in astrocytes further confirmed that butyrate was preferred over acetate as a metabolic substrate (Figure 3(a)). Metabolism of 250 μM [U-13C]butyrate with the addition of unlabelled 12C-acetate was found to reduce M + 2 labelling in glutamate by 17.5% when compared to the 250 μM [U-13C]butyrate control (Figure 3(a)). By contrast, 1000 μM of unlabelled 12C-butyrate was able to reduce metabolism of 2.5 mM [1,2-13C]acetate in the majority of measured metabolites in the range of 72.5- 92.3% (Figure 2(a)). When the astrocytes were incubated with 1000 μM [U-13C]butyrate, unlabelled 12C-acetate reduced M + 2 labelling in glutamate, whereas 13C enrichment of TCA cycle metabolites and amino acids glutamine, α-ketoglutarate, fumarate, malate and aspartate remained unchanged. This demonstrates the ability of acetate to compete with butyrate for astrocytic metabolism but to a much lower extent. Overall 13C labelling percentages of TCA cycle metabolites were also greater from [U-13C]butyrate than from [1,2-13C]acetate in astrocytes (Figures 2(a) and 3(a)).
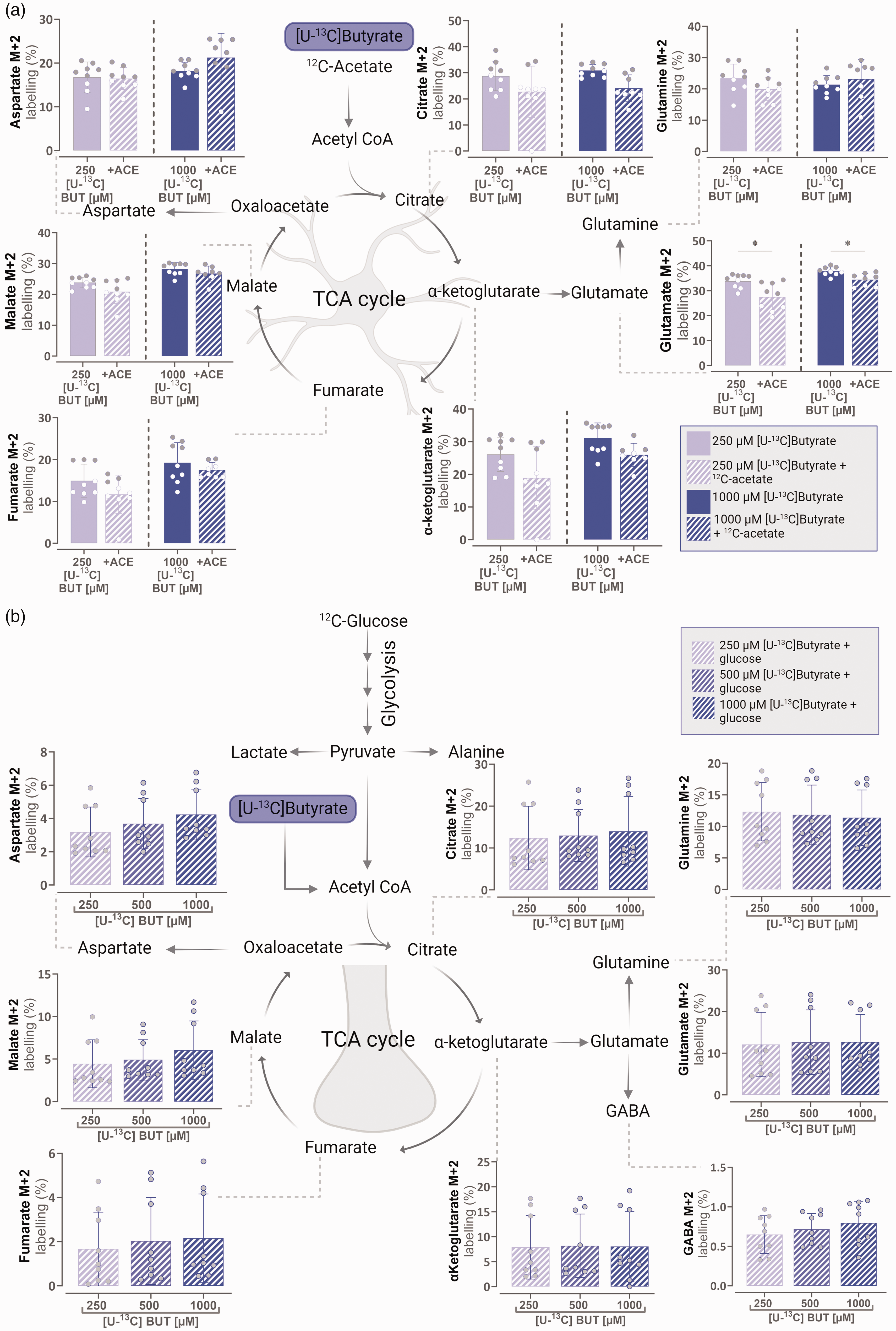
Butyrate is preferred over acetate as a metabolic substrate in astrocytes and is metabolised to a lesser extent in neurons. (a) Intracellular M + 2 carbon labelling of TCA cycle intermediates and amino acids in cultured cortical mouse astrocytes after Continued.incubation with [U-13C]butyrate (250 or 1000 μM) in the absence and presence of unlabelled 12C-acetate (2.5 mM) and (b) intracellular M + 2 carbon labelling of TCA cycle intermediates and amino acids in cultured cortical mouse neurons after incubation with [U-13C]butyrate (250, 500 or 1000 μM) in the presence of unlabelled 12C-glucose (2.5 mM). ACE: acetate, BUT: butyrate; GLUC: glucose. Mean ± SD, minimum N = 3, n= 8, (a) unpaired Welch’s t-test or Mann–Whitney test, (b) one-way ANOVA with Bonferroni post hoc test or Kruskal-Wallis test with Dunn’s post-hoc test,*p < 0.05, **p < 0.01, ***p < 0.001 ****p < 0.0001.
In neuronal cultures, competition assays between [U-13C]butyrate (250, 500 and 1000 μM) and unlabelled 12C-glucose (2.5 mM) were performed (Figure 3(b)). The TCA cycle intermediates and amino acids measured all showed 13C enrichment from [U-13C]butyrate in the presence of 12C-glucose. However there was no dose dependent difference in metabolism between 250, 500 and 1000 μM [U-13C]butyrate + 2.5 mM 12C-glucose (Figure 3(b)). M + 2 labelling of all the TCA cycle metabolites measured from [U-13C]butyrate (250, 500 or 1000 μM) + 2.5 mM 12C-glucose (Figure 2(b)) was less than M + 2 labelling from 2.5 mM [U-13C]glucose + 12C-butyrate (250, 500 or 1000 μM) (Figure 3(b)). This shows that neurons are capable of metabolising butyrate, but to a very low extent. Additionally, comparing metabolism of labelled [U-13C]butyrate in astrocytes and neurons (Figure 3(a) and (b)) showed that 13C enrichment was far greater in astrocytes across all measured metabolites than in neurons. Further suggesting that astrocytes are the main compartment for butyrate metabolism.
In-vivo metabolism of butyrate across different brain regions, is greater than acetate metabolism in both WT and 5xFAD mice
To assess the degree to which butyrate was taken up by the brain and metabolised, mice were administered either [1,2-13C]acetate or [U-13C]butyrate via intraperitoneal injection (I.P.) and after 15 minutes brain metabolism was immediately stopped by focussed microwave fixation. Both wild-type (WT) and the 5xFAD model of AD mice of the same genetic background were included to explore possible differences in acetate and butyrate brain metabolism under physiological and pathophysiological conditions. Following injection with [1,2-13C]acetate or [U-13C]butyrate, no differences were observed in 13C labelling of TCA cycle metabolites between WT and 5xFAD mice (Figure 4). 13C labelling of TCA cycle metabolites across different brain regions (cortex, hippocampus, and cerebellum) revealed no apparent differences, indicating sustained metabolism of acetate and butyrate in brain areas associated with metabolic decline in AD. Furthermore, higher M + 2 labelling of all TCA cycle intermediates and amino acids was evident in the cortices and hippocampi of mice receiving [U-13C]butyrate (Figure 4(a) and (b)). In the cerebellum, greater 13C enrichment of metabolites was observed from [U-13C]butyrate than from [1,2-13C]acetate for all but citrate (Supplementary Figure 2). These data show a large capacity for in-vivo brain butyrate metabolism and suggest sustained butyrate metabolism during the early development of AD.
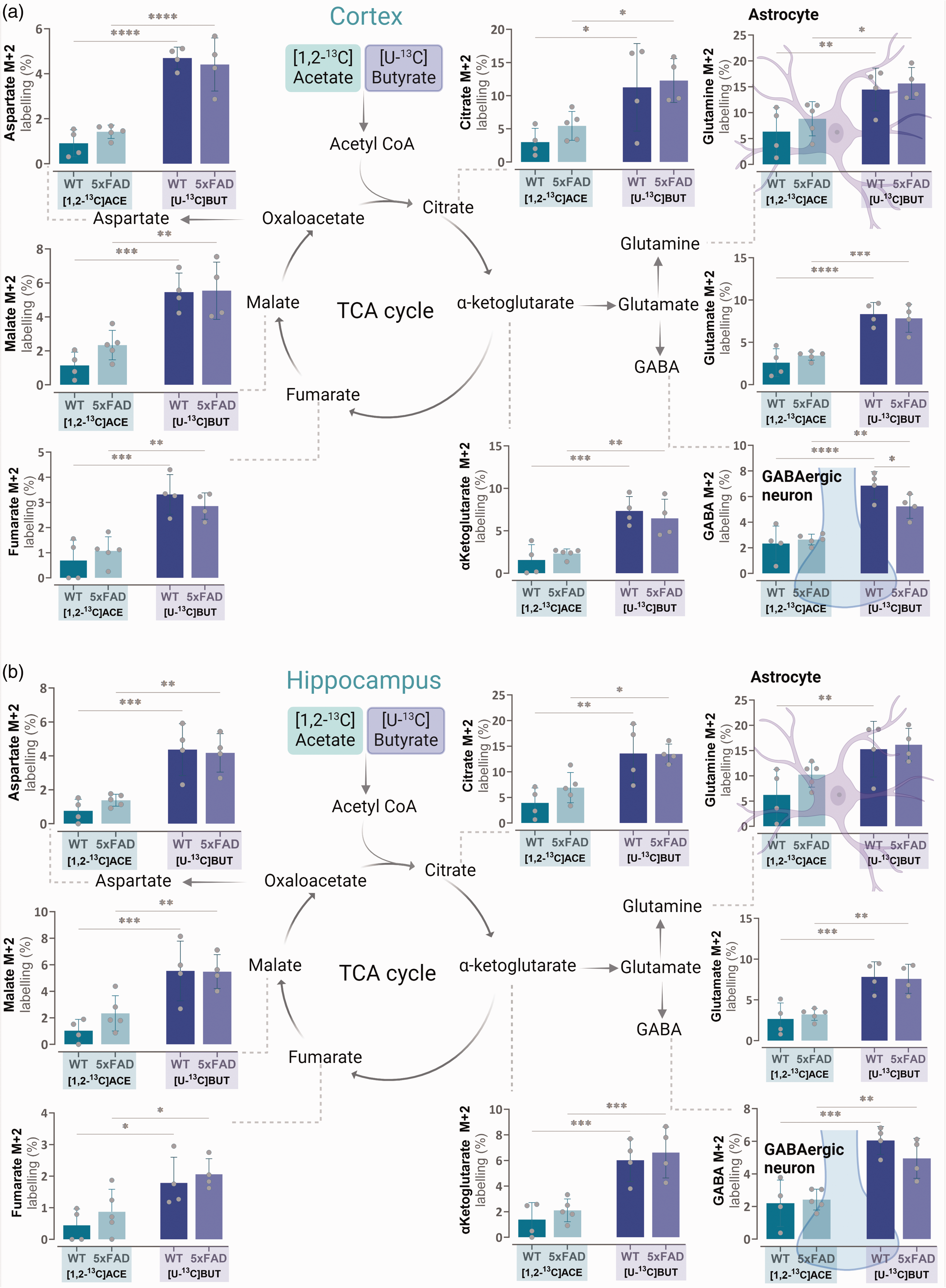
In-vivo metabolism of butyrate across different brain regions, is greater than acetate metabolism in both WT and 5xFAD mice. Intracellular M + 2 13C enrichment of TCA cycle intermediates and amino acids 15 minutes after I.P. injection of [1,2-13C]acetate (green bars) or [U-13C]butyrate (purple bars) in 4 month old WT and 5xFAD mice. (a) cortex, (b) hippocampus. ACE: acetate, BUT: butyrate. Mean ± SD, n = 4–5 individual animals, 2-way ANOVA with Fisher’s least significant difference (LSD) post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001 ****p < 0.0001.
Discussion
Metabolic preference between SCFAs butyrate and acetate
Fatty acids have long been recognised as substrates capable of reaching the brain and being oxidised to produce energy.7,55 While much is known about the metabolism of the SCFA acetate in the brain,12,43,56 studies on the direct metabolism of butyrate in the brain are limited as are comparisons in metabolism between SFCAs. The preferential metabolism of butyrate over acetate in cerebral cortical slices, as demonstrated in our experiments (Figure 1), aligns with and extends previous findings on the uptake of SCFAs. 57 When cerebral cortical slices and cultured astrocytes were incubated with [1,2-13C]acetate and unlabelled 12C-butyrate, a reduction in 13C enrichment of a majority of the TCA metabolites occurred. This suggests a competitive inhibition by butyrate, indicative of its preferential uptake and metabolism. 35 Conversely, the absence of change in 13C enrichment with [U-13C]butyrate in the presence of unlabelled 12C-acetate underscores metabolic dominance of butyrate. A possible reason for why 13C enrichment from butyrate is higher than acetate could be the difference in the number of labelled carbon atoms available for exchange (as butyrate contains four carbon atoms in comparison to acetate containing two). To mitigate this difference in carbon atom number, varying concentrations of butyrate were tested, for example, 500 µM 12C-butyrate compared to 2.5 mM [1,2-13C]acetate in cultured astrocytes resulted in lower competition however a statistically significant reduction in labelling for some metabolites was still observed. Longer chain length could again explain the metabolic preference for butyrate over acetate as this property increases membrane permeability and diffusion to access mitochondria. 58
Astrocytes as the main cellular compartment for butyrate brain metabolism
In the cerebral cortical slices a notable observation was the high 13C enrichment in glutamine observed from [U-13C]butyrate metabolism, suggesting a significant astrocytic involvement in butyrate metabolism. 51 Although viability and functionality in terms of metabolism was not immediately assessed prior to incubation, this has been extensively assessed and established previously in our system investigating the effect of acetate and glucose as metabolic substrates in mouse brain slices. 43 The addition of unlabelled 12C-butyrate reduces labelling of TCA intermediates coming from [1,2-13C]acetate. When conditions are reversed by incubating with [U-13C]butyrate in the presence and absence of unlabelled 12C-acetate, a reduction in metabolic activity is not observed. Rather, both labelled substrates show very similar metabolic activity to each other, suggesting that butyrate is not reducing the metabolic activity or impairing the viability of these cerebral cortical slices. Our findings are further reinforced by the results from our competition experiments involving β-hydroxybutyrate (βHB). The negligible impact of unlabelled 12C-βHB on the metabolism of acetate and butyrate suggests distinct metabolic pathways or preferences for these substrates, which is consistent with the existing literature on neuronal and astrocytic energy metabolism.52,53
The distinction between astrocytic and neuronal metabolism was further explored through separate incubation experiments with cortical mouse astrocytes and neurons. The significant reduction in M + 2 labelling in astrocytes incubated with [1,2-13C]acetate (a known marker of astrocytic metabolism) 59 and various concentrations of unlabelled 12C-butyrate reinforces the notion that butyrate is a highly competitive substrate for oxidative metabolism in astrocytes. In contrast, neuronal cultures exhibited a preference for glucose over butyrate, highlighting the metabolic differences between these two cell types. The data of this study furthermore aligns well with our previous observation of preferential astrocyte metabolism of medium-chain fatty acids (MCFAs) in brain slices. 60 Our findings are consistent with the known metabolic characteristics of astrocytes as the primary site of fatty acid β-oxidation and underscores the importance of considering cell-specific metabolism in brain energy dynamics.28,34,61
Sustained in-vivo brain metabolism of butyrate in WT and 5xFAD mice
The in-vivo aspect of our study, involving both WT and 5xFAD mice, provides further context to our in vitro findings. The observed sustained metabolism of butyrate in various brain regions, irrespective of the AD pathology, suggests a robust capacity for butyrate metabolism in the brain. This is particularly noteworthy given the metabolic decline associated with AD.31,62,63 Our previous study of [U-13C]glucose metabolic mapping in 5xFAD acutely isolated cerebral slices, revealed disrupted glucose metabolism in both the cortex and hippocampus predominantly at 4 months of age between wild-type and 5xFAD mice. 64 This could imply a potential therapeutic relevance for butyrate in metabolic interventions for neurodegenerative diseases. Furthermore, dysfunctional astrocyte metabolism is gaining attention as a driver of AD pathology. 65 It must be stated that with our approach, we cannot determine that both substrates reach the same concentration in plasma or brain tissue although the substrates were initially administered at equal molar concentrations (0.6 M). Still, studies suggest 15 minutes after administration the plasma levels of these substrates are within a similar range.66 –68 What we can evaluate however, is whether it is possible to detect 13C-enrichment derived from butyrate metabolism in different brain regions and if such labelling patterns of metabolism were affected in the 5xFAD mouse model to potentially assess the implications of cerebral butyrate metabolism in AD pathology. Another important point that could be argued is that differences in WT and 5xFAD brain metabolism could be due to differences in brain blood flow, however a recent study reports that there are no brain-wide nor regional differences in cerebral blood flow between WT and 5xFAD mice. 69 Given the astrocytic metabolism of butyrate demonstrated above, it may be suggested that butyrate supplementation could provide an alternative fuel to restore astrocyte function in AD,24,27,70 but further studies are needed to explore this.
In conclusion, our study delineates a clear metabolic preference for butyrate over acetate in the cerebral cortex, primarily mediated through astrocytic metabolic activity. This preference is sustained in-vivo across different brain regions and under pathophysiological conditions, such as in AD models. These findings open new avenues for exploring metabolic therapies targeting specific substrates and pathways, especially in the context of neurodegenerative diseases where metabolic dysfunction is a hallmark feature. Future research should aim to elucidate the molecular mechanisms underpinning this preferential metabolism and its implications for brain health and disease.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241270457 - Supplemental material for Metabolic preferences of astrocytes: Functional metabolic mapping reveals butyrate outcompetes acetate
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241270457 for Metabolic preferences of astrocytes: Functional metabolic mapping reveals butyrate outcompetes acetate by Aishat O Ameen, Sebastian W Nielsen, Martin W Kjær, Jens V Andersen, Emil W Westi, Kristine K Freude and Blanca I Aldana in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Independent Research Fund Denmark [grant number 1030-00285B] for AA PhD stipend; and by Hørslev Fonden (MWK research assistant stipend) [grant number NA].
Acknowledgements
The authors would like to acknowledge the excellent technical assistance of laboratory technician Heidi Marie Nielsen.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceptualisation, AA and BIA; experimental design and work AA, MWK, SN, JVA; writing, original draft preparation, AA, SN; review and editing, AA, BIA, JVA, EWW and KF. All authors have read and agreed to the final version of the manuscript. Data can be provided upon reasonable request by contacting the authors.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
