Abstract
Peak width of skeletonized mean diffusivity (PSMD) is an emerging diffusion-MRI based marker to study subtle early alterations to white matter microstructure. We assessed PSMD over the clinical continuum in Dutch-type hereditary CAA (D-CAA) and its association with other CAA-related MRI-markers and cognitive symptoms. We included (pre)symptomatic D-CAA mutation-carriers and calculated PSMD from diffusion-MRI data. Associations between PSMD-levels, cognitive performance and CAA-related MRI-markers were assessed with linear regression models. We included 59 participants (25/34 presymptomatic/symptomatic; mean age 39/58 y). PSMD-levels increased with disease severity and were higher in symptomatic D-CAA mutation-carriers (median [range] 4.90 [2.77–9.50]mm2/s × 10−4) compared with presymptomatic mutation-carriers (2.62 [1.96–3.43]mm2/s × 10−4) p = <0.001. PSMD was positively correlated with age, CAA-SVD burden on MRI (adj.B [confidence interval] = 0.42 [0.16–0.67], p = 0.002), with number of cerebral microbleeds (adj.B = 0.30 [0.08–0.53], p = 0.009), and with both deep (adj.B = 0.46 [0.22–0.69], p = <0.001) and periventricular (adj.B = 0.38 [0.13–0.62], p = 0.004) white matter hyperintensities. Increasing PSMD was associated with decreasing Trail Making Test (TMT)-A performance (B = −0.42 [−0.69–0.14], p = 0.04. In D-CAA mutation-carriers microstructural white matter damage is associated with disease phase, CAA burden on MRI and cognitive impairment as reflected by a decrease in information processing speed. PSMD, as a global measure of alterations to the white matter microstructure, may be a useful tool to monitor disease progression in CAA.
Keywords
Introduction
Cerebral amyloid angiopathy (CAA) is a common type of cerebral small vessel disease (SVD) characterized by amyloid-beta deposition in the leptomeningeal and cortical cerebral vessels.1 –3 In addition to (recurrent) lobar intracerebral hemorrhages (ICH) and transient focal neurological episodes, many patients with CAA experience cognitive symptoms or even dementia independently from commonly co-occurring Alzheimer’s disease (AD).4 –7 While ICH is a contributor to the development of cognitive symptoms, it is not considered to be the main cause, as cognitive impairment often precedes ICH.8,9
Recent studies have shown that subtle but widespread damage to white matter properties invisible on conventional MRI sequences, so-called normal appearing white matter, is associated with cognitive dysfunction in CAA.10 –13 A key method to detect this white matter disruption in vivo is diffusion tensor imaging (DTI). 14 The DTI derived “peak width of skeletonized mean diffusivity” (PSMD) measure has been shown to be sensitive to subtle early changes of white matter properties. PSMD is an index of the dispersion of mean diffusivity (MD) values across the white matter skeleton.14,15 Moreover, PSMD in CAA is strongly correlated with information processing speed, executive functioning and outperforms conventional SVD markers on MRI in their relation to cognition.13,15 –21 However, while we know that white matter properties are affected in CAA it is still unclear how the integrity in the white matter microstructure changes with increasing disease severity, both clinically and radiologically.
Although most cases of CAA are sporadic, some rare hereditary autosomal dominant forms exist. Dutch-type hereditary CAA (D-CAA) is a monogenic variant of CAA caused by a mutation in the APP gene. Clinical and radiological manifestations of D-CAA are similar to sporadic CAA, but D-CAA has an approximately 20-year earlier onset and a more rapidly progressive disease course. This allows us to study both early preclinical and advanced stages of a relatively pure form of CAA with limited age-related confounders such as co-existing AD and hypertensive pathology. There is limited information on cognitive impairment in D-CAA; in presymptomatic mutation carriers cognitive abnormalities are thought to be seldom, while symptomatic patients perform worse across all cognitive domains. 22
In this study, we aimed to assess microstructural white matter integrity levels measured by the PSMD on MRI over the clinical continuum from early presymptomatic to severe symptomatic D-CAA. In addition, we investigated the value of PSMD as a marker of disease severity in patients with CAA by assessing its association with CAA disease burden on MRI (CAA-SVD score), global cognitive performance, processing speed and executive functioning. 23
Methods
We included presymptomatic and symptomatic D-CAA mutation carriers who participated in our ongoing prospective natural history study on D-CAA (the AURORA study) between February 2018 and April 2021. All participants were recruited via the (outpatient) clinic of the Leiden University Medical Center (LUMC). Inclusion criteria were age >18 years and a DNA proven APP mutation. Symptomatic D-CAA was defined by a history of at least one symptomatic ICH (sICH). Participants in whom no 3 T MRI with DTI sequences was performed were excluded from the current study.
This study was in accordance with the Helsinki protocol and approved by the ethics committee of the Leiden University Medical Center (reference number NL62670.058.17 (AURORA). Written informed consent was obtained from all participants. This manuscript is reported against the STROBE guidelines.
Clinical evaluation
We obtained data on demographics, medical history and clinical symptoms for all D-CAA mutation carriers via standardized questionnaires. Educational level was categorised according to the Verhage scale. 24 Montreal Cognitive Assessment (MoCA), 25 Trail Making Test part A (TMT-A) and part B (TMT-B) 26 were administered by trained researchers to assess global cognitive functioning, processing speed and executive functioning. Performance on TMT-A and TMT-B was transformed into sex-, age-, and education-adjusted t-scores using published normative data. 27
MRI protocol, image processing and assessment
MRI was performed on a 3.0 Tesla MRI scanner (Philips Achieva, Best, the Netherlands) and data were acquired using a standard 32-channel head coil. Three-dimensional T1 weighted images were acquired with the following parameters: repetition time (TR)/echo time (TE) = 9.7/4.6 ms, flip angle 7 degrees, 130 slices with no interslice gap and a field of view (FOV) of 217 × 172 × 156 mm with a voxel size of 1.2 × 1.2 ×1.2 mm, resulting in a scan duration of 2:48 min. T2 weighted images were acquired with the following parameters: TR/TE = 4744/80 ms, flip angle 90 degrees, 48 slices with no interslice gap and a FOV of 220 ×176 × 144 mm with a voxel size of 0.5 × 0.6 × 3 mm, resulting in a scan duration of 2:13 min. Three dimensional Fluid Attenuated Inversion Recovery (FLAIR) images were acquired with the following parameters: TR/TE = 4800/280 ms, inversion time of 1650 ms, 321 slices with no interslice gap and a FOV of 250 ×250 × 180 mm with a voxel size of 1.1 × 1.1 × 0.6 mm, resulting in a scan duration of 4:43 min. Susceptibility weighted images (SWI) were acquired using the following parameters: TR/TE = 31/7.2 ms, flip angle 17 degrees, 130 slices and a FOV of 230 × 190 ×130 mm with a voxel size of 0.6 × 0.6 × 1 mm resulting in a scan duration of 3:31 min. Diffusion images were acquired using the following parameters: TR = 8194 ms, TE = 76 ms, voxel size 1.72 × 1.72 × 2.5 mm, flip angle = 90 degrees. 48 slices and FOV of 220 × 220 ×120 mm, 45 gradient directions with a b-value of 1200 s/mm2 and one baseline image with b-value 0 s/mm2; scan duration 6:33 minutes, no interslice gap.
Diffusion MRI (dMRI) data was visually inspected to evaluate presence of major artefacts for which reason patients were excluded from further analysis and then the raw diffusion images were pre-processed using the MRtrix v3.0 package 28 (http://mrtrix.org) and the Functional Magnetic Resonance Imaging of the Brain software library (FSL), v6.0.5. 29 Noise and Gibbs ringing artefacts were removed (‘dwidenoise’, ‘mrdegibbs’; 30 MRtrix) and subject motion and eddy current induced distortions were corrected (‘eddy’; FSL). Following pre-processing we calculated PSMD v1.8.2 using the fully automated script (http://www.psmd-marker.com). 15 PSMD is a measure of the dispersion of mean diffusivity (MD) values across the white matter skeleton. To calculate PSMD, diffusion data of each subject was first skeletonized using Tract-Based Spatial Statistics procedure (TBSS), 31 part of FSL, using the FMRIB 1-mm fractional anisotropy (FA) template) thresholded at an FA-value of 0.2. Mean diffusivity (MD) images of that same subject were then projected onto the skeleton. To avoid contamination through cerebrospinal fluid (CSF) partial volume effects, the MD skeletons were further masked with the template skeleton thresholded at a fractional anisotropy (FA) value of 0.3 and a custom mask (provided with PSMD script), designed to exclude regions close to the ventricles. Finally, histogram analysis was performed on this masked MD-skeleton and PSMD was calculated as the difference between the 95th and 5th percentiles of the MD values within the skeleton.
For patients with an ICH, we masked out the voxels in the skeleton that were affected by ICH and surrounding gliosis. ICH masks and gliosis masks were manually delineated on T1-weighted images by M.S and N.V. under the supervision of an experienced neuroradiologist (M.A.A.v.W.), taking into account the appearance on FLAIR and T2 weighted sequences. The T1-weighted image was registered to diffusion space using FLIRT 32 (part of FSL) and the resulting transformation matrix was applied to the ICH masks. These masks were then provided as lesion mask into the PSMD calculation, by which the ICH was masked out of the skeleton.
The following MRI markers were scored in accordance with the Standards for Reporting Vascular changes on neuroimaging (STRIVE) criteria: 33 cerebral microbleeds (CMB), macrobleeds, cortical superficial siderosis (cSS), white matter hyperintensities (WMH) and enlarged perivascular spaces in the centrum semi-ovale (CSO-EPVS). Micro- and macrobleeds were counted on SWI images. The presence of cSS was also scored on SWI. cSS was scored as focal, defined as restricted to three or fewer sulci, or disseminated, defined as affecting four or more sulci, and the cSS focality score was calculated subsequently according to previously published methods.34,35 Fluid-attenuated inversion recovery (FLAIR) images were used to assess both deep and periventricular WMHs. Extent of WMH was scored according to the Fazekas rating scale. 36 CSO-EPVS were assessed on T2-weighted MRI and classified into four categories according to a validated, visual rating scale, ranging from none to 10 PVS in the first category to over 40 in the fourth category. 37 The CAA-SVD score was calculated for each participant. 23 The CAA-SVD score consisted of lobar CMBs (2-4: 1 point, ≥5: 2 points), cSS (focal: 1 point, disseminated: 2 points), CSO-EPVSs (>20: 1 point), and WMHs (deep WMH Fazekas score 2 or 3 and/or periventricular WMH Fazekas score 3: 1 point), with a higher score reflecting a more severe disease burden. Scoring of each marker was performed by a single observer with over 5 years of experience in the field and discussed with a neuroradiologist with over 15 years of experience in the field in case of uncertainties.
Statistics
Normality of all variables was assessed with Shapiro-Wilk tests. Descriptive statistics were performed for baseline characteristics. We performed a Mann Whitney U test or independent t-test to assess whether there was a difference in PSMD, CAA-SVD score, MoCA score, TMT-A and TMT-B score between presymptomatic and symptomatic D-CAA carriers, as appropriate. CMB count and PSMD were log10 transformed to obtain normal distributions for further analyses. All predictor and outcome variables were standardized (z-scores) to ease interpretation.
We used linear regression models with correction for sex and age to assess the correlation between PSMD, the CAA-SVD score and individual CAA related MRI markers (CMB count, cSS presence, deep and periventricular WMH and the CSO-PVS score). For cSS presence, which is a dichotomous outcome variable, we reported the unstandardized beta coefficients. For all other variables standardized coefficients are reported.
We assessed the association between PSMD and cognitive scores using linear regression analyses, adjusting for age and sex. MoCA was already adjusted for years of education (≤12 or >12), TMT-A and TMT-B -scores for age, sex an educational level, thus these variables were not included in the models. In this population age and disease severity are considered to be interconnected, the above mentioned analyses with age as a covariate were therefore repeated adjusting only for sex to facilitate interpretation of the magnitude of a potential age-effect.
To assess if PSMD is related to cognition in presymptomatic stages of the disease, we performed a sensitivity analysis between PSMD and cognitive scores in the presymptomatic group only.
We used a significance level of α = 0.05. All statistical tests were performed using IBM SPSS statistics version 25.
Results
From the 62 participants enrolled in the AURORA study in whom an MRI had been performed, one had to be excluded due to movement artefacts on MRI and two because no DTI sequences were available. We, therefore, included 59 participants in our study; 25 presymptomatic (mean age, 39 years) and 34 symptomatic (mean age, 58 years) (Table 1). Cognitive performance was measured in all presymptomatic D-CAA mutation carriers and in 30 (88%) symptomatic D-CAA mutation carriers (three were too exhausted after the MRI to perform the tests and one refused).
Baseline characteristics.
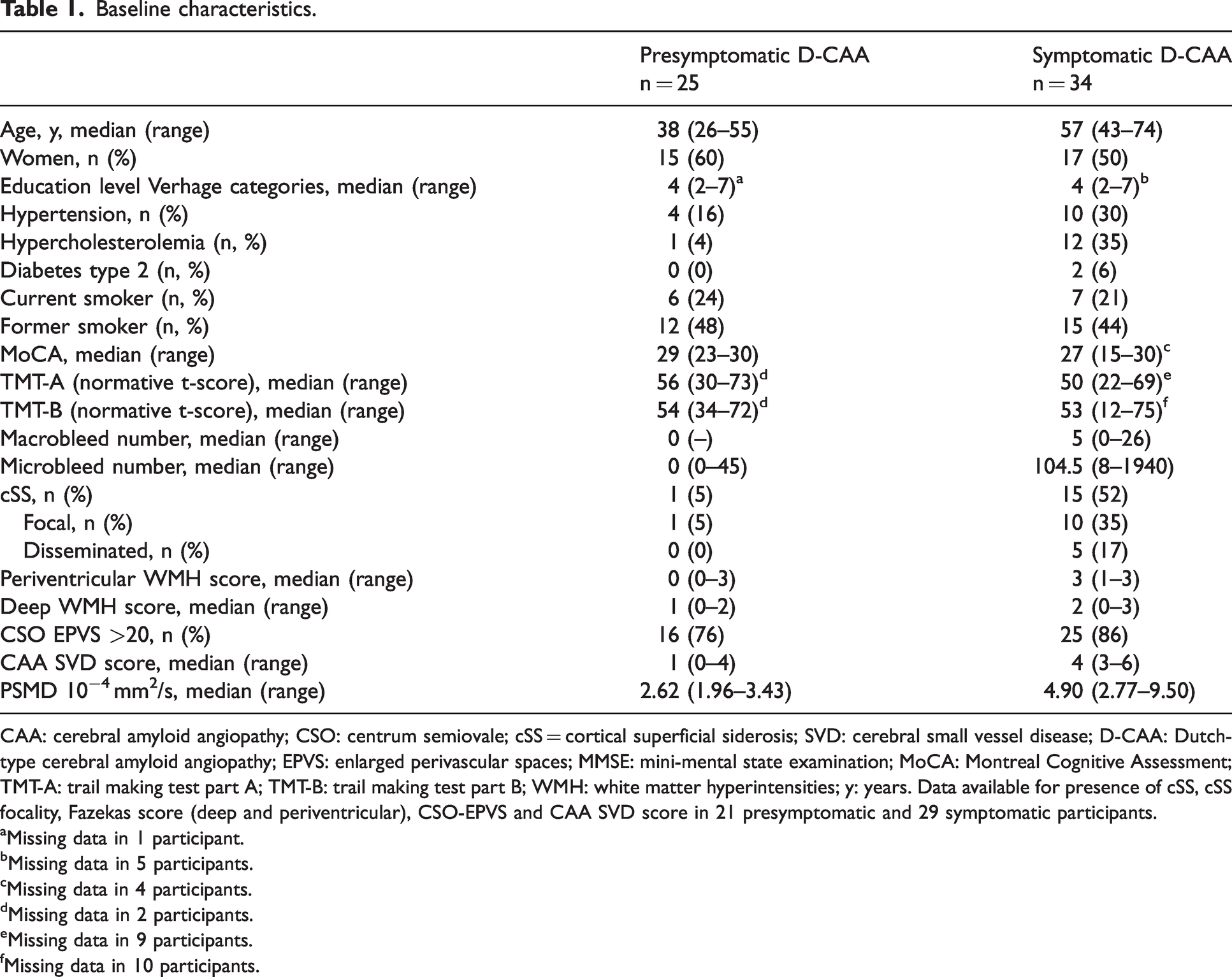
CAA: cerebral amyloid angiopathy; CSO: centrum semiovale; cSS = cortical superficial siderosis; SVD: cerebral small vessel disease; D-CAA: Dutch-type cerebral amyloid angiopathy; EPVS: enlarged perivascular spaces; MMSE: mini-mental state examination; MoCA: Montreal Cognitive Assessment; TMT-A: trail making test part A; TMT-B: trail making test part B; WMH: white matter hyperintensities; y: years. Data available for presence of cSS, cSS focality, Fazekas score (deep and periventricular), CSO-EPVS and CAA SVD score in 21 presymptomatic and 29 symptomatic participants.
Missing data in 1 participant.
Missing data in 5 participants.
Missing data in 4 participants.
Missing data in 2 participants.
Missing data in 9 participants.
Missing data in 10 participants.
PSMD levels over the clinical continuum in D-CAA
PSMD levels were higher in symptomatic D-CAA carriers compared to presymptomatic D-CAA carriers (median [range] presymptomatic: 2.62 [1.96–3.43] (mm2/s 10−4), median symptomatic: 4.90 [2.77–9.50] (mm2/s 10−4); p = <0.001; Figure 1(a)). Furthermore, PSMD was positively correlated with age (regression coefficient [95% confidence interval]: B = 57.76 [46.64–68.88], p = <0.001; Figure 1(b)), also within the separate groups (presymptomatic carriers: B [95%CI] = 11.86 [5.33–18.39], p = 0.001; symptomatic carriers: B [95%CI] = 4.75 [1.53–7.98], p = 0.005).
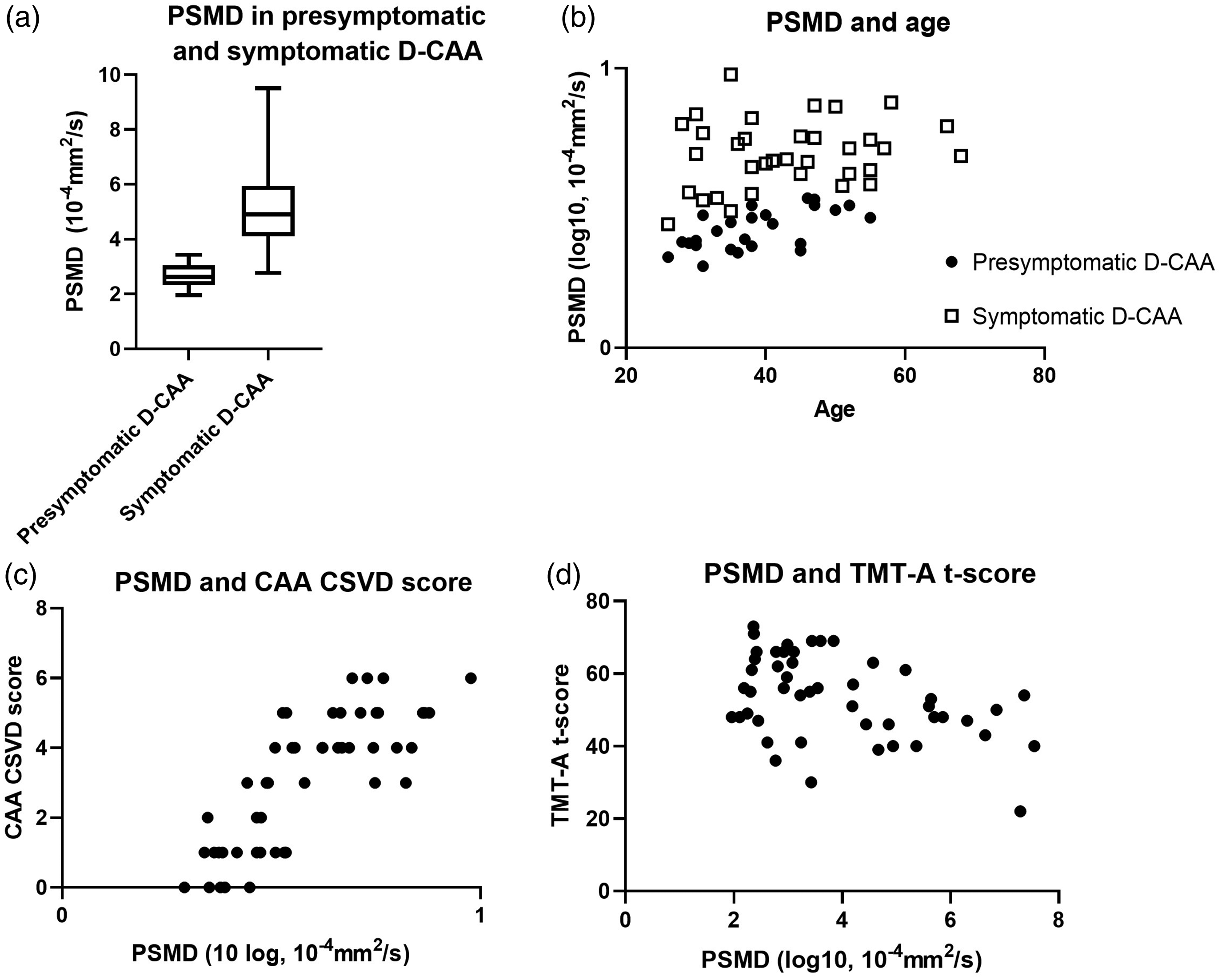
PSMD and disease phase (a), age (b), CAA SVD score (c) and TMT-A score (d).
PSMD and CAA-SVD score on MRI
CAA-SVD score was higher in symptomatic than in presymptomatic participants (median [range] presymptomatic: 1 [0–4], median symptomatic: 4 [3–6], p < 0.001). PSMD correlated positively with CAA-SVD score (adjusted B = 0.42 [0.16–0.67], p = 0.002; Figure 1(c)). PSMD was positively associated with number of CMBs (adj. B = 0.30 [0.08–0.53], p = 0.009), and with both deep (adj. B = 0.46 [0.22–0.69], p = <0.001) and periventricular (adj. B = 0.38 [0.13–0.62], p = 0.004) Fazekas scores. PMSD was not associated with presence of cSS and CSO-EPVS scores (Table 2). Age as a covariate had a significant effect on the association between PSMD and all CAA MRI-markers (Supplementary data Table 1.).
Association between PSMD and CAA related MRI markers in D-CAA mutation carriers.
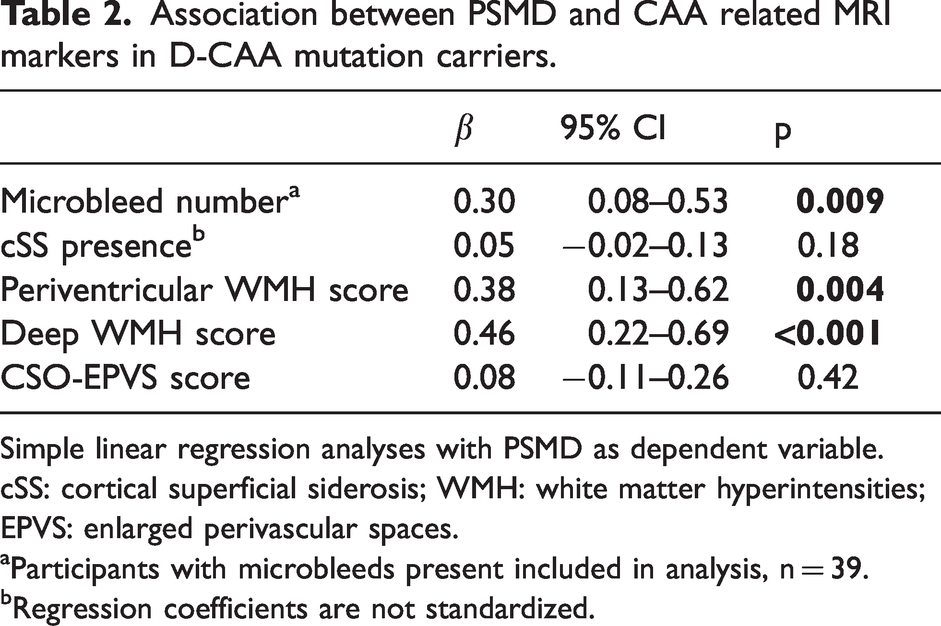
Simple linear regression analyses with PSMD as dependent variable.
cSS: cortical superficial siderosis; WMH: white matter hyperintensities; EPVS: enlarged perivascular spaces.
Participants with microbleeds present included in analysis, n = 39.
Regression coefficients are not standardized.
We found no association between CAA-SVD score and MoCA, TMT-A or TMT-B score.
PSMD and cognition
Presymptomatic D-CAA carriers performed better on the MoCA and TMT-A than symptomatic D-CAA patients, not on TMT-B (see Table 3). Increasing PMSD levels were associated with decreasing MoCA scores (B = −0.29 [−0.55–−0.032], p = 0.03), although significance was lost after adjusting for age and sex. Increasing PSMD, however, was correlated with decreasing performance on TMT-A (B = −0.42 [−0.69–−0.14], p = 0.04), but not on TMT-B (B = −0.29 [−0.59−0.06], p = 0.054) Figure 1(d)). In the sensitivity analyses in which only the presymptomatic D-CAA carriers were included, we found no associations between PSMD and MoCA (adj. B = 0.43 (−0.47–1.32), p = 0.33), TMT-A (adj. B = −0.28 (−1.31–0.74), p = 0.57) or TMT-B (adj. B = −0.29 (−1.11–0.54), p = 0.48).
Cognitive scores in presymptomatic and symptomatic D-CAA carriers.
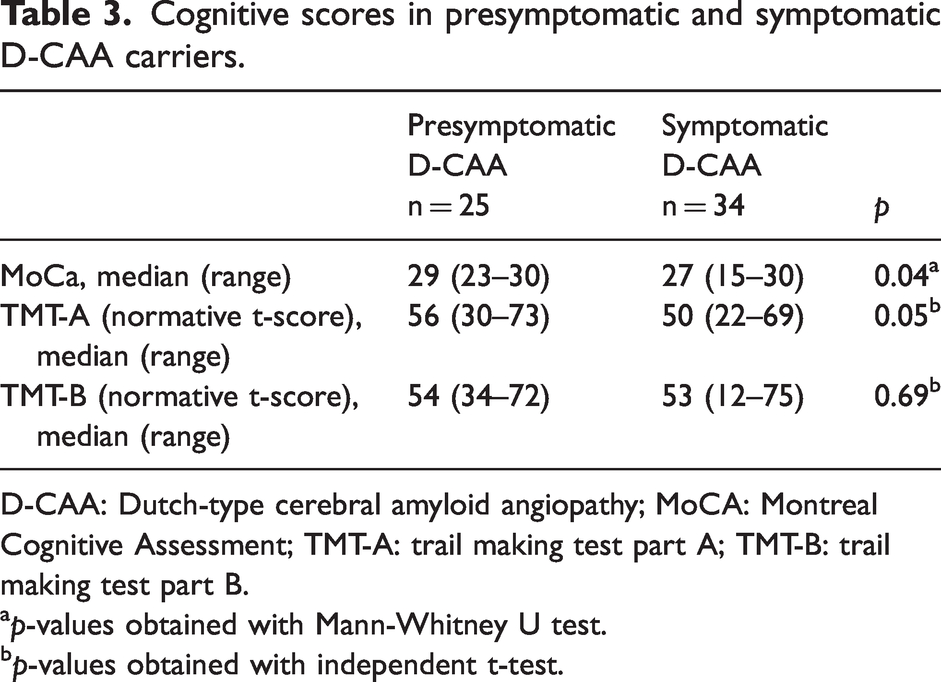
D-CAA: Dutch-type cerebral amyloid angiopathy; MoCA: Montreal Cognitive Assessment; TMT-A: trail making test part A; TMT-B: trail making test part B.
p-values obtained with Mann-Whitney U test.
p-values obtained with independent t-test.
Discussion
Our study showed that microstructural white matter integrity damage increases with disease severity of CAA, which is reflected by higher PSMD values in symptomatic versus presymptomatic D-CAA mutation carriers. Moreover, PSMD values are correlated positively with the CAA-SVD score and age. We also found that PSMD, but not CAA-SVD score was associated with a decrease in processing speed. These results combined suggest that PSMD can capture white matter damage and is a promising marker that adequately reflects disease severity in D-CAA.
Our findings expand a previous study from our group that found significant differences in PSMD values between symptomatic D-CAA patients and controls in another cohort of D-CAA mutation carriers, but not between presymptomatic D-CAA mutation carriers and controls. 38 This might have been caused by the small sample size. Another recent small study on D-CAA was able to detect white matter damage measured by PSMD in the presymptomatic phase of D-CAA while also showing increasing PSMD over time. 39 These studies did not look into the relation between PSMD and CAA-related MRI markers, except for WMH, nor did they investigate cognitive function.
In sporadic CAA, PSMD is increasingly recognized as a valuable marker for cerebral white matter microstructural damage. Increased PSMD values were detected in patients with sporadic CAA compared with controls and participants with mild cognitive impairment (MCI) or AD.16 –18 In accordance with our current study, PSMD was positively correlated with both WMH volume16 –18 and CAA SVD score.17,18 In contrast, no relation was found between PSMD and number of CMBs, which might be attributable to the relatively lower number of CMBs in sporadic CAA or to a lack of statistical power.
Damaged microstructural white matter is thought to be one of the main causes of cognitive decline, since it affects the efficiency of the brain to process information in CAA.13,23 We found no relation between PSMD values and global cognitive function (measured using the MoCA score), which is in line with previous research in sporadic CAA.17,18 This might be the result of the rather unspecific nature of the MoCA score, while cognition in CAA appears to be more specifically affected in the domains of executive functioning and processing speed.19,40,41 Although our findings on the relationship between PSMD and processing speed are in line with several recently published studies,16 –18,21 we did not find an association for PSMD and performance in executive functioning. Our cohort consists of relatively young D-CAA mutation carriers, who might have more reserve capacity to compensate on an executive functioning task than the relatively older cohorts of sporadic CAA patients as described in earlier mentioned studies.
Our study has limitations that need to be discussed. First, because of the cross-sectional design and the unavailability of follow-up data so far, no conclusions on causality or predictive value of PSMD can be made. 42 Second, we did not have a control population to formally test if PSMD was indeed affected in patients, specifically presymptomatic patients. However, a recent study assessing PSMD values in a life-span study, included two cohorts of healthy volunteers within the same age range as the D-CAA patients within this study. When comparing the PSMD values from the current study with the mean PSMD values reported within those two cohorts, PSMD seems to be already increased in our presymptomatic D-CAA group. 42 Furthermore, due to the occurrence of bilateral ICH in our cohort, we decided to mask out ICH affected voxels rather than calculating PSMD on the contralateral hemisphere and thereby changed the original PSMD mask. Earlier studies showed that this did not significantly affect PSMD values.16,18 Third, standardized questionnaires on anxiety and depression symptoms were administered in only a part of the study population. Therefore, we could not adjust for potential negative influence of mood disorders on cognitive performance. Lastly, our study participants did not undergo a complete neuropsychological test battery. We instead choose a global cognition screening tool commonly used in the clinic and the TMT-A and TMT-B test as a surrogate for processing speed and executive functioning respectively. Also, in order to assess true executive functioning, TMT-B score should be corrected for performance on TMT-A. However, to enable comparison of our results with other work in the field we choose to report uncorrected TMT-B results. As expected, in our study age as a covariate appeared to significantly influence the association between PSMD, the individual CAA-related MRI markers and the MoCA score. Age and disease severity are intertwined in this cohort with relatively young onset CAA, which is also reflected by the positive correlation found between age and PSMD levels. However, an underlying increase of PSMD values by healthy aging in these participants could not be ruled out and therefore adjusting for age seemed justifiable.
Strengths of our study include the relatively large cohort of D-CAA mutation carriers comprising the whole CAA disease continuum from the early presymptomatic to the symptomatic disease phase. Because first clinical symptoms in D-CAA occur on average 20 years earlier than in sporadic CAA, we were able to investigate PSMD in this cohort of participants with a relatively pure form of CAA with a low degree of age-related comorbidities such as Alzheimer’s disease or hypertension. In addition, the added value of our study is that we are the first to be able to relate PSMD values not only to CAA burden on MRI but also to cognitive function, an important and relatively under-studied clinical symptom of D-CAA.
Our results strengthen previous reports that PSMD is a marker for early brain changes in CAA. While CAA-related injury can be visualized on MRI in the form of lesion markers such as WMHs, CMBs and cSS, these MRI markers of CAA relate generally only weakly to cognition. 43 In our study no association was found between CAA-SVD score and performance on MoCA, TMT-A or TMT-B.
Furthermore we know that the damage is more extensive than captured by these lesions visible on standard MRI. Diffusion-based measures, such as PSMD, can capture both the visible and invisible damage to the white matter 44 and have been shown to relate to cognition more strongly than conventional lesion markers, 13 as also found in the current study.
A disadvantage of PSMD is that it can be influenced by ICH. However, in this hereditary D-CAA group we found that the use of ICH and gliosis masks during DTI processing for patients with a history of ICH, did not significantly change the PSMD values when compared to calculations without these masks (unpublished data). We, therefore, expect that also in sporadic CAA patients, in whom in general the incidence of (recurrent) ICH is considerably lower, the time-consuming step of mask construction for ICH might no longer be required.
In conclusion, in D-CAA mutation carriers, microstructural white matter damage is associated with disease phase, CAA severity as measured by CAA related SVD markers on MRI and with cognitive impairment as reflected by a decrease in processing speed. This indicates that PSMD, as a global measure of microstructural white matter integrity, may be used as a tool to monitor disease progression in CAA in clinical trials. Future longitudinal studies and prospective studies with repeated DTI-measurements are necessary to investigate whether PSMD may also act as prognostic marker of cognitive decline in CAA.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241261771 - Supplemental material for Microstructural white matter damage on MRI is associated with disease severity in Dutch-type cerebral amyloid angiopathy
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241261771 for Microstructural white matter damage on MRI is associated with disease severity in Dutch-type cerebral amyloid angiopathy by Ingeborg Rasing, Naomi Vlegels, Manon R Schipper, Sabine Voigt, Emma A Koemans, Kanishk Kaushik, Rosemarie van Dort, Thijs W van Harten, Alberto De Luca, Ellis S van Etten, Erik W van Zwet, Mark A van Buchem, Huub AM Middelkoop, Geert Jan Biessels, Gisela M Terwindt, Matthias JP van Osch, Marianne AA van Walderveen and Marieke JH Wermer in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Prof. Wermer is supported by a clinical established investigator grant from the Dutch Heart Foundation (2016 T086) and a VIDI (91717337) and ASPASIA grant from the Netherlands Organisation for Health Research and Development.
Data availability statement
Data will be shared upon reasonable request after signing a data transfer agreement. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: N. Vlegels reports support by the Netherlands CardioVascular Research Initiative–the Dutch Heart Foundation (CVON 2018–28 & 2012–06). M.R. Schipper reports independent support from the TRACK D-CAA consortium, consisting of Alnylam, Biogen, the Dutch CAA foundation, Vereniging HCHWA-D, and researchers from Leiden, Boston, and Perth. A. De Luca reports independent support from Alzheimer Nederland (WE-03-2022-11) as well as from ZonMW. R. van Dort is funded by the TRACK D-CAA consortium, consisting of Biogen, Alnylam, the Dutch CAA foundation, Vereniging HCHWA-D, and researchers from Leiden, Boston, and Perth. G.M. Terwindt reports independent support from the Dutch Research Council (NWO), European Community, the Dutch Heart Foundation, the Dutch Brain Foundation, and the Dutch CAA foundation.
M.J.P. van Osch reports support by a NWO-VICI grant (016.160.351) and a NWO-Human Measurement Models 2.0 grant (18969) as well as support from the Dutch Research Council (NWO), European Community, the Dutch Heart Foundation, and the Dutch Brain Foundation.
M.J.H. Wermer reports independent support from de Nederlandse Organisatie voor Wetenschappelijk Onderzoek ZonMw (VIDI grant 91717337), the Netherlands Heart Foundation, and the Dutch CAA foundation. The others report no conflicts.
Authors’ contributions
I. Rasing: Substantial contributions to conception and design, analysis and interpretation of data. Drafting the article and revising the article critically for important intellectual content. Final approval of the version to be published.
N. Vlegels: Substantial contributions to conception and design, analysis and interpretation of data. Drafting the article and revising the article critically for important intellectual content. Final approval of the version to be published.
M.R. Schipper: Substantial contributions to conception and design, analysis and interpretation of data. Drafting the article and revising the article critically for important intellectual content. Final approval of the version to be published.
S. Voigt: Substantial contributions to acquisition of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
E.A. Koemans: Substantial contributions to acquisition of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
K. Kaushik: Substantial contributions to acquisition of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
R. van Dort: Substantial contributions to acquisition of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
T.W. van Harten: Substantial contributions to acquisition of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
A. De Luca: Substantial contributions to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
E.S van Etten: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
E.W. van Zwet: Substantial contributions to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
M.A. van Buchem: Substantial contributions to conception and design. Revising the article critically for important intellectual content. Final approval of the version to be published.
H.A.M. Middelkoop: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
G.J. Biessels: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
G.M. Terwindt: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
M.J.P. van Osch: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
M.A.A. van Walderveen: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
M.J.H. Wermer: Substantial contributions to conception and design and to analysis and interpretation of data. Revising the article critically for important intellectual content. Final approval of the version to be published.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
