Abstract
Small acute diffusion-weighted imaging (DWI) lesions can accompany intracerebral hemorrhage due to cerebral amyloid angiopathy (CAA). We therefore examined the occurrence of such lesions in the context of CAA-related convexal subarachnoid hemorrhage (cSAH) both in a cross-sectional and longitudinal manner. DWI lesions were noted in 14/29 (48%) patients at their index cSAH and 12/21 patients (57%) showed acute small DWI lesions at follow-up MRI. Forty-four of 71 (62%) DWI lesions were spatially related to areas of cortical superficial siderosis. Clarification of the implications of our finding needs the investigation of larger patient groups.
Keywords
Introduction
Cerebral amyloid angiopathy (CAA) is a common small vessel disease of the brain characterized by progressive vascular amyloid-ß deposition in the cerebral cortex and leptomeninges. CAA is a major cause of intracerebral hemorrhage (ICH) in the elderly with lobar ICH location and cortico-subcortical cerebral microbleeds (Mbs) as the key neuroimaging manifestations. Convexal subarachnoid hemorrhage (cSAH) has been identified as an additional source and marker of bleedings related to active/progressive CAA.1–3 More recently, attention has also been focused on ischemic events in CAA which is not unexpected given other signs of microangiopathic brain damage associated with the disease. 4 Several previous studies reported on the presence of DWI lesions in patients with ICH.5,6 DWI lesions were noted in 15–23% of ICH patients, lesions occurred predominantly at cortico-subcortical brain areas, were commonly small, and have been related to markers of cerebral small vessel severity.5,6 It has therefore been suggested that small DWI-positive lesions in CAA patients with ICH reflect small vessel disease-related ischemic infarctions. The significance and the pathomechanism underlying DWI-positive lesions, however, remain poorly understood and also non-ischemic etiologies of DWI lesions appear possible. 7
Based on these data, we speculated that acute DWI lesions should also occur in association with CAA-related cSAH. We therefore carefully searched for the presence of DWI lesions in a group of patients with acute CAA-related cSAH and attempted to define possible characteristics of their appearance and distribution. We also looked for their associations with clinical and morphologic features, both in a cross-sectional and longitudinal manner.
Materials and methods
This study was performed on patients which we identified as having suffered from cSAH and had undergone MRI at an academic primary and tertiary care hospital over a nine-year period. 3 At the time of the acute event, 29 of a total of 45 such patients fulfilled the imaging criteria of CAA according to the modified Boston criteria 8 and served for this analysis. Clinical and imaging characteristics and the work-up of these patients have been previously reported in detail. 3 MRI of the brain at 1.5 T (94%) and 3.0 T included axial fluid-attenuated inversion recovery, diffusion-weighted imaging, and gradient echo T2*-weighted sequences (slice thickness 5 mm) for detection of intracranial bleedings. A detailed description of the MRI hardware and acquisition parameters used for these examinations is given in the online data supplement.
Two experts (C.E., F.F.) blinded to the clinical data evaluated all baseline MRI scans and assessed the presence, number, exact location and the diameter of focal DWI abnormalities. DWI lesions were identified as bright areas on DWI sequences and dark areas on the corresponding apparent diffusion coefficient (ADC) maps. Possible DWI lesions were defined as bright areas on DWI and isointense signal on ADC maps. DWI lesions in close proximity (<20 mm) to acute intracranial bleedings were not considered to avoid misinterpretation of artefactual signal changes and of secondary phenomena due to hematoma expansion. For all DWI-positive lesions, we also reviewed their spatial relationship to areas of cSS and of meningeal enhancement which was defined as a proximity of ≤ 20 mm. Other morphologic abnormalities, i.e. white matter hyperintensities, old infarcts/lacunes, Mbs, cSAH and cortical superficial siderosis (cSS) were rated in a standardized manner.8,9 All patients underwent intracranial time of flight angiography as part of the MRI protocol and an evaluation of the extra- and intracranial vessels with Doppler ultrasound.
Follow-up clinical and CT and/or MRI data over a median period of 24 ± 20 (range 6–78) months were available in all patients. A total of 32 follow-up MRIs was performed in 18 patients because of new clinical symptomatology, i.e. transient focal neurological episodes (TFNE) or a stroke syndrome, and 3 patients underwent pre-planned MRI. We retrieved all brain CT/MRI scans from MEDOCS 3 and reviewed them for recurrent ICH or cSAH, new ischemic infarcts and DWI lesions. Patients with evidence of a recurrent ICH or cSAH were defined as having ‘active CAA’. Patients’ outcome at follow-up was graded according to the modified Rankin Scale (mRS) as “favorable” (mRS scores 0–2) or “unfavorable” (mRS scores 3–6). The hospital institutional review board and the ethics committee of the Medical University of Graz approved the study (29–340 ex 16/17) and waived requirement for informed consent. Analyses of patient data were performed in accordance with the Declaration of Helsinki.
Statistical analysis
Besides descriptive statistics, we performed comparisons between patients with active versus non-active CAA and patients with versus those without DWI lesions. The Mann–Whitney U test was used for continuous data and frequencies were compared with the Fisher exact test. A more detailed description of the statistical methods is given in the online data supplement.
Results
Among the 29 cSAH patients, 19 had probable and 10 had possible CAA. 8 Their demographic, clinical and MRI characteristics are shown in the Table (online data supplement).
Prevalence of DWI lesions at baseline cSAH
At baseline, we observed DWI lesions in 14/29 (48%) patients. DWI lesions in acute cSAH
patients were mostly small (≤10 mm) as shown in Figure 1. Three of the 14 subjects also had larger
ischemic infarcts. Images from an 81-year-old patient with acute convexal
subarachnoid hemorrhage (cSAH) who experienced recurrent transient focal
neurological episodes. (a) Linear signal hyperintensity (arrow) on fluid-attenuated
inversion recovery (FLAIR) MRI with corresponding intrasulcal signal loss on the
gradient echo T2* sequences (not shown) consistent with acute cSAH. Also, note
cortical swelling of neighboring gyri. (b–d) Diffusion-weighted imaging revealed
multiple small (2–5 mm), cortico-subcortical, round-ovoid, bright lesions (arrows)
in the right parietal lobe.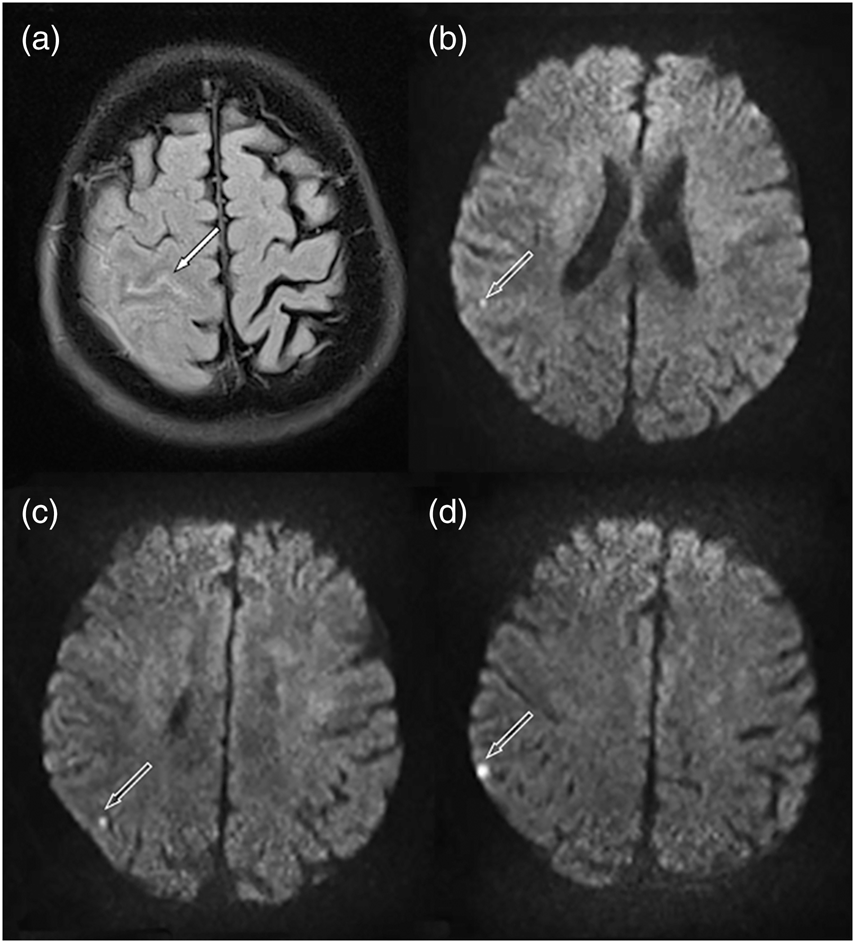
Prevalence and associations of DWI lesions during follow-up
During follow-up, 14/29 (48%) patients suffered a recurrent cSAH and 12/29 (41%) a new symptomatic ICH with 9 having both. These patients were defined as having active CAA (n = 17). Patients without new intracranial bleedings either scanned for transient symptoms or because of pre-planned follow-up defined the non-active CAA group (n = 12). DWI lesions were detected in 12/21 subjects (57%). Nine of 14 patients (64%) with recurrent cSAH and 8 of 12 patients (67%) with new symptomatic lobar ICH had DWI lesions on the follow-up MRI. None of the three patients with preplanned follow-up MRI had DWI lesions. Of 36 new DWI lesions, 35 were small (≤10 mm). Nine patients had DWI lesions on both baseline and follow-up scans. Presence of DWI lesions at baseline was significantly associated with the recurrence of DWI lesions (baseline DWI positive versus baseline DWI negative: 9/14 vs. 3/15; p = 0.025).
Characteristics of DWI lesions
Combining baseline and follow-up scans (n = 64), we identified a total of 71 small DWI lesions (including 30 possible lesions, 42%) in overall 17/29 (58%) of cSAH patients. These lesions were uniformly round to ovoid. Fifty-seven lesions (80%) were ≤ 5 mm and 14 (20%) were 6–10 mm (mean diameter 4 mm ± 1.9 SD). Larger ischemic infarcts occurred in overall four patients, three at baseline and one at follow-up (two had possible and two had probable CAA). In three patients, the larger infarct (axial diameter 1.4–3.5 cm) was present in the territory of the middle cerebral artery together with small DWI lesions in the same vascular supply area. One patient with small DWI lesions in the frontal lobe also had a cerebellar infarction (axial diameter 2 × 3 cm).
The 71 small DWI lesions comprised 66 (93%) supratentorial and 5 (7%) infratentorial lesions. The majority of the supratentorial lesions were located cortico-subcortically (55/66; 83%) and occurred in the frontal lobe (38/66; 58%). All 5 infratentorial DWI lesions occurred in the cerebellum. Forty-four of the 71 (62%) small DWI lesions occurred in close spatial relation to areas of cSS (Supplemental Figure 1). On the available 13 post-contrast MRIs, 13 of 23 (57%) DWI lesions were closely related to meningeal enhancement (Supplemental Figure 2) but remote from the acute cSAH.
Active CAA and other associations
Seventeen patients (58%) had active CAA (recurrent cSAH (n = 14) or subsequent ICH (n = 12)). Active CAA was significantly associated with the presence of lobar Mbs at baseline (p = 0.008), and TFNE (p = 0.01) (Supplemental Table). The presence of DWI lesions at the index cSAH, however, was not associated with recurrent intracranial bleedings.
Twenty-two of 29 cSAH patients (76%) experienced TFNE (supplemental Table) and 14 of
these 22 patients (64%) had small DWI lesion. Figure 2 shows serial MRIs of such a patient. Four of
the 7 TFNE negative patients (57%) had DWI lesions. Serial diffusion-weighted imaging
(DWI) in a 68-year-old patient with acute cSAH (not shown) during repeated transient
focal neurological episodes. (a) DWI reveals multiple, cortico-subcortical, bright
lesions in the frontal and parietal lobe (arrows) which were present at every
follow-up MRI. (b) Corresponding apparent diffusion coefficient (ADC) maps show
decreased diffusivity (arrows) consistent with acute ischemic
damage.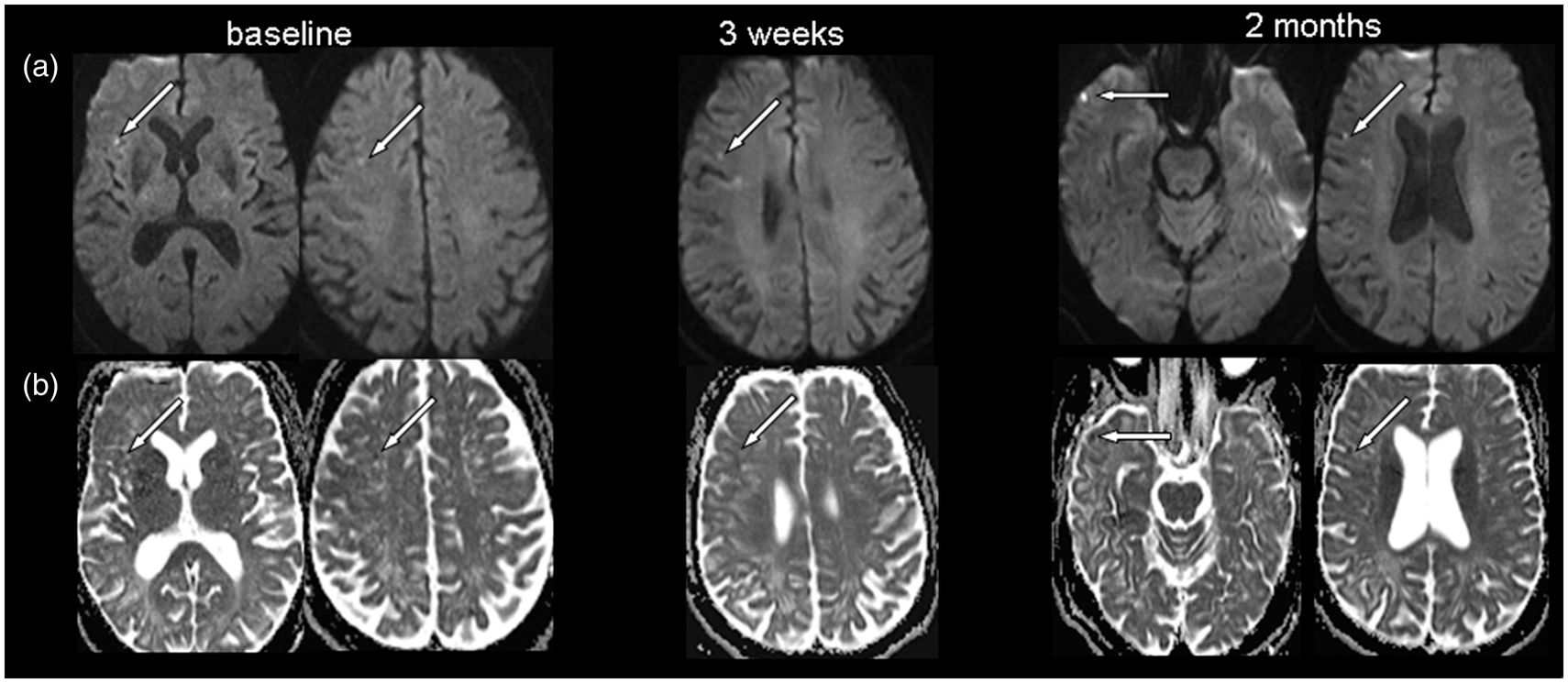
There was no difference in the cerebrovascular risk profiles of patients with active versus non-active CAA or of patients with or without DWI lesions including atrial fibrillation (AF) (Supplemental Table). Presence of DWI lesions in multiple vascular territories was also not associated with AF (DWI lesion in multiple territories vs. DWI lesion in singular territory 1/5 vs. 3/12; p = 0.67). Eleven patients received antithrombotic treatment at the time of their index cSAH (antiplatelets (n = 6); oral anticoagulation (n = 5)) and 7 (64%) of them had DWI lesions at baseline. Antithrombotic treatment was generally stopped and not restarted after the index cSAH. Six patients, however, were restarted with antithrombotic treatment by external physicians. Three of them suffered a new ICH, two with a concurrent cSAH and one of those patients also had small corticosubcortical DWI lesions. There was no evidence of occlusive carotid artery disease in any of the cSAH patients. Presence of DWI lesions was not related to outcome defined by the mRS.
Discussion
We found evidence of DWI-positive lesions in about half of patients with acute CAA-related cSAH. Lesions were mostly small, located in cortico-subcortical brain areas and frequently recurred on repeated MRI.
The observation of small DWI-positive lesions in CAA patients with ICH has been commonly interpreted as coexisting ischemic brain injury probably related to cerebral small vessel disease. Our study is the first to extend this observation to acute focal bleeding into the subarachnoid space in the context of CAA, i.e. so-called cSAH. Consistent with ICH patients,5,6 DWI lesions in our well characterized cSAH patients were small, round to ovoid, clinically silent and predominately located in cortico-subcortical brain areas. Appearance, distribution and the high prevalence of DWI lesions in acute cSAH patients also point to mechanisms related directly to the acute bleeding event and possibly promoted by cerebral small vessel disease rather than independent events. Also, the observation that new DWI lesions occurred frequently in patients with recurrent intracranial bleedings (i.e. active CAA) is in line with such considerations. Moreover, the high proportion of small DWI lesions spatially related to areas of cSS and evidence of DWI lesions in close association to meningeal enhancement on post-contrast MRI indicating small vessel leakage possibly due to CAA 3 support the idea that these small DWI lesions may be associated with focally active CAA. Additional – rather complementary than competing – factors promoting microthrombus formation in the setting of an acute cSAH might be the activation of the coagulation cascade or impaired fibrinolytic activity. 10 Likewise, endothelium-related processes causing vasoconstriction or activation of inflammation could be involved.
There is only scarce information about the relationship between small ischemic infarcts and clinical findings – other than cognitive dysfunction. Many of our cSAH patients who also had DWI-positive lesions presented with TFNE. Stereotyped TFNE are, however, a rather frequent clinical presentation of CAA-related cSAH. 11 It thus remains speculative whether this observation has some functional background or merely represents the expression of an overall higher grade of CAA severity. Otherwise, recurrent TFNE in the context of cSAH have been linked to partial seizure activity or clusters of cortical spreading depolarizations.12,13 which in turn might propagate disturbed vasomotor function. Along these lines, a possibly non-ischemic origin of DWI lesions as seen in transient global amnesia can also not be excluded. 7
Retrospective design and small sample size can be viewed as limitations of our study. An additional limitation comes from the fact that our follow-up observations are heavily weighted towards patients with active CAA, i.e. follow-up MRIs were primarily performed in patients who presented with new symptoms and not in a systematic manner. This prohibits a valid interpretation of the difference in the frequency of DWI-positive lesions between patients with active versus non-active CAA during follow-up. Also that clinical MRI was routinely performed at 1.5 Tesla (94% of scans) and not at higher magnetic field strengths could be viewed as a limitation of the study. We attempted to compensate for these limitations by comprehensive cross-sectional and longitudinal analyses as well as detailed workup of neuroimaging and clinical data from well-documented observations. Consequently, we found a surprisingly high prevalence of DWI-positive lesions, evidence of new DWI lesions on MRI follow-up scans, and some spatial relation of DWI-positive lesions and cSS in cSAH patients. If DWI lesions in the context of CAA-related cSAH represent just an intriguing epiphenomenon or constitute a useful imaging marker of progressing/ongoing cerebral small vessel disease as suggested by our study will need further investigation of larger patient groups.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Markus Beitzke: concept and design, acquisition of data, analysis and interpretation of data, critical revision, drafted the article. Christian Enzinger: acquisition, analysis and interpretation of data, critical revision. Alexander Pichler: analysis and interpretation of data, critical revision. Gerit Wünsch: acquisition and analysis of data, critical revision. Franz Fazekas: acquisition, analysis and interpretation of data, concept and design, critical revision, study supervision.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
