Abstract
Cerebral amyloid angiopathy (CAA) is frequently found post mortem in Alzheimer’s dementia, but often undetected during life especially since in vivo hallmarks of CAA and its vascular damage become overt relatively late in the disease process. Decreased neurovascular coupling to visual stimulation has been put forward as an early MRI marker for CAA disease severity. The current study investigates the role of neurovascular coupling in AD related dementia and its early stages. We included 25 subjective cognitive impairment, 33 mild cognitive impairment and 17 dementia patients and 44 controls. All participants underwent magnetic resonance imaging of the brain and neuropsychological assessment. Univariate general linear modeling analyses were used to assess neurovascular coupling between patient groups and controls. Moreover, linear regression analyses was used to assess the associations between neurovascular coupling and cognition. Our data show that BOLD amplitude is lower in dementia (mean 0.8 ± 0.2, p = 0.001) and MCI patients (mean 0.9 ± 0.3, p = 0.004) compared with controls (mean 1.1 ± 0.2). A low BOLD amplitude was associated with low scores in multiple cognitive domains. We conclude that cerebrovascular dysfunction, most likely due CAA, is an important comorbidity in early stages of dementia and has an independent effect on cognition.
Keywords
Introduction
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by Aβ-containing extracellular plaques and tau-containing intracellular neurofibrillary tangles. 1 It is often considered the most common cause of dementia in aging. In dementia patients older than 65 years, AD pathology rarely occurs isolated. Concurrent cerebrovascular disease is a common neuropathological finding in post mortem AD. 2 Moreover, vascular pathology may even precede the development of AD pathology, since cerebrovascular dysregulation is one of the earliest pathological events in the disease process of late-onset AD. 3
In this respect, cerebral amyloid angiopathy (CAA) is frequently found in clinically diagnosed AD. CAA is a common type of cerebral small vessel disease (SVD) that is caused by deposition of amyloid β (Aβ) in small to medium-sized cerebral blood vessels and leptomeningeal arteries. 4 Post-mortem studies have shown that up to 98% of AD patients show moderate to severe CAA. 5 The presence of advanced CAA in AD is associated with greater cognitive impairment 6 and/or faster cognitive decline 7 and could exacerbate AD pathology. 8
Still, CAA is often undetected in patients with AD, especially since in vivo hallmarks of CAA and its vascular damage only become overt relatively late in the disease process. Cerebrovascular Aβ deposition and alterations of cerebrovascular physiology precede hemorrhagic brain lesions by decades. 9
Decreased neurovascular coupling to visual stimulation has been put forward as an early, robust and sensitive marker for disease severity in CAA. Neurovascular coupling is decreased in sporadic CAA patients,10,11 and decreases further over a 1-year period. 12 Patients with Dutch-type CAA, a hereditary variant of CAA, show a decreased neurovascular coupling already in the presymptomatic phase of the disease before classic radiological hallmarks of CAA become overt. 13 Neurovascular coupling in Dutch-type CAA decreases further without an increase of hemorrhagic lesions in the brain over a 4-year period. 14 Decreased neurovascular coupling to visual stimulation is therefore thought to reflect early vascular damage due to cerebrovascular Aβ deposition. 15
The use of this marker opens the possibility to study early, or even subtle, manifestations of CAA related cerebrovascular dysfunction as comorbidity, or even cause of dementia. This offers increased insight into etiology of dementia, and is expected to be helpful in the development of efficient therapies.
The current study aimed to investigate whether neurovascular coupling is decreased in AD related dementia and its early stages. Secondly, it aimed to investigate whether neurovascular coupling is independently associated with cognitive deficits. The hypothesis was that neurovascular coupling would be decreased in memory clinic patients when compared to controls. We expected the largest decrease in neurovascular coupling in patients with AD related dementia followed by those with mild cognitive impairment (MCI). Moreover, we hypothesized that a decreased neurovascular coupling is associated with poorer cognitive function.
Materials and methods
Study design and participants
This cross-sectional case-control study was performed between September 2019 and December 2021 at the Department of Radiology of the Leiden University Medical Center (Leiden, the Netherlands). For this study we included 25 SCI patients, 33 MCI patients, 17 dementia patients and 44 control subjects. Patients were recruited from the memory clinic of the Leiden University Medical Center (Leiden, the Netherlands) and the memory clinic of the Haaglanden Medical Center (The Hague, the Netherlands). All patients were referred to the memory clinic by their general practitioner or a medical specialist based on their cognitive complaints. At the memory clinic they were examined according to a standardized protocol, comprising a general medical and neurological examination, a neuropsychological assessment and a CT or whole brain MRI. A diagnosis was made in a multidisciplinary consensus meeting using the National Institute of Neurological and Communicative Disorders and Stroke and Alzheimer’s Disease and Related Disorders Association (NINCDS-ADRDA) criteria. 16 Patients not demonstrating cognitive deficits on neuropsychological testing were classified as SCI. Patients with a diagnosis SCI, MCI or dementia (probable AD or mixed type dementia) were selected for inclusion to this current study. Patients with other causes of dementia, e.g. frontotemporal dementia or Lewy body dementia, were not selected for this study. Control subjects were recruited via various advertisements. All participants were screened for neurological diseases and cardiovascular risk factors, i.e. hypertension, hyperlipidemia, diabetes and smoking. Exclusion criteria for control subjects were a history of stroke or cognitive complaints. Control subjects were screened with the same neuropsychological assessment as the memory clinic patients and excluded from this study if they showed performance indicative of cognitive impairment. Exclusion criteria for all groups were MRI contra-indications, specific contra-indications to fMRI such as seizure within prior year and non-correctable visual impairment, incapacitated to give informed consent, and age above 90 years. All participants were categorized according to the modified Boston MRI criteria for probable CAA or not. 17 This allowed us to control in analyses for the radiological presence of CAA to ensure that our finding were not driven by the participants with more advanced CAA. The medical ethics committee of Leiden Den Haag Delft approved the study and written informed consent was obtained from all subjects. The procedures followed were in accordance with institutional guidelines and the World Medical Association (WMA) Declaration of Helsinki.
Image acquisition
All participants underwent an MRI of the brain at the LUMC on a 3-Tesla scanner (3 T Achieva Philips Medical Systems, Eindhoven, the Netherlands) using a standard 32-channel head coil. For each participant, three-dimensional (3 D) T1-weighted images (echo time [TE] 3.5 ms, repetition time [TR] 7.9 ms, flip angle 8°, field of view [FOV] = 250 mm × 199 mm × 170 mm, scan duration 257 s), 3 D fluid-attenuated inversion recovery (TE/TR: 1.65 s/4.8s, slices 326, FOV 250 x 250 x 182, matrix size 224 x 224, scan duration 273 s), T2*-weighted images (TE/TR/flip angle: 7.9 ms/31 ms/17°, slices 130, FOV 230 x 189 x 130, matrix size 384 x 315, scan duration 211 s) and visually stimulated blood-oxygen-level-dependent (BOLD) fMRI scans (TE/TR/flip angle: 30 ms/1500 ms/75°, FOV 220 × 220 x 75 mm, matrix 80 × 80 mm, slices 25, slice thickness 3 mm, 2 dummy scans before actual scans of 130 dynamics with scan duration 195 sec) were acquired. The visual stimulation task was presented on an MRI compatible BOLD screen (Cambridge Research Systems) which was visualized via a mirror on the head coil. The visual stimulus was presented in a block-design schedule with an 8 Hz flashing radial black and white checkerboard pattern for 20 sec (stimulus), alternated with 28 sec of gray screen (rest), as described previously. 11 Each scan consisted of four cycles of stimulus and rest conditions. A fixation dot was presented during both the checkerboard pattern as well as the grey screen. This fixation dot changed color (light red – dark red) and the participants were asked to press a button every time a color change was seen. Correct responses were monitored in order to check if attention was focused on the task.
Image analysis/processing
Before analysis, all MRI scans were submitted to a visual quality control check to ensure that no gross artifacts were present in the data. Lobar microbleeds (location as described by the Boston criteria 18 ) and deep microbleeds were scored on T2*-weighted images according to criteria as described previously. 19 Intracerebral hemorrhages were defined as parenchymal defects with evidence of hemosiderin in their wall. 20 The presence of cortical superficial siderosis was described as linear residues of blood in the superficial layers of the cortex seen on T2*-weighted images, and classified as focal (3 or less sulci) or disseminated (4 or more sulci). 21 Lacunar infarcts were defined as round or oval lesion with a diameter between 3 mm and 15 mm, hypointense with a surrounding rim of hyperintensity on fluid-attenuated inversion recovery images and hyperintense on T2-weighted images. 22 White matter hyperintensities were defined and analyzed using a semiautomated and validated method using fluid-attenuated inversion recovery images as described previously. 23 Gray matter volume was calculated using Structural Image Evaluation, using Normalization, of Atrophy, 24 part of the FSL. 25 FMRIB’s Integrated Registration and Segmentation Tool was used to determine the volumes of the hippocampus. This starts by registering all images to MNI152 templates. Second it fits models for all different structures (meshes) to the images and finally applies boundary correction for the volumetric output. BOLD fMRI scans using a checkerboard stimulus were analyzed to determine the neurovascular coupling parameters, i.e. BOLD amplitude, time to peak (TTP), and time to baseline (TTB), as described previously 11 (Figure 1). Additionally, we determined the time from the beginning of the checkerboard stimulus to the beginning of the BOLD signal change (time to signal activation) and the time from the end of the checkerboard stimulus to the beginning of the BOLD signal decline (time to signal deactivation).
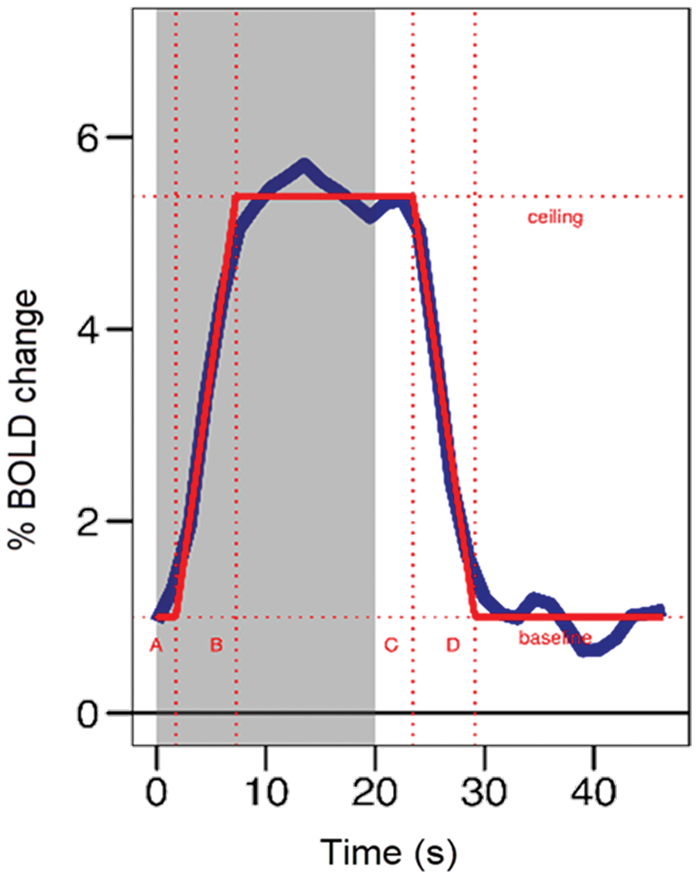
This figure shows an example of the shape of the hemodynamic response curves (blue line) in the occipital lobe after checkerboard stimulation (gray) and subsequent rest (white). The y-axis represents the change in BOLD amplitude in percentage. The x-axis represents time in seconds. The red line represents the individual fitted trapezoid from which the three BOLD parameters were derived. BOLD amplitude was defined as the distance from baseline to the peak response (ceiling). Time to peak was calculated by subtracting A (the beginning of the BOLD signal change) from B (onset of the trapezoid ceiling). Time to baseline was calculated by subtracting C (beginning of the BOLD signal decline) from D (onset of baseline). Additionally, time to signal activation was determined as the time from the beginning of the checkboard stimulation (gray) to A (the beginning of the BOLD signal change). Time to signal deactivation was calculated by subtracting 20 s (duration of the checkerboard stimulation) from C (beginning of the BOLD signal decline).
Cognitive function
All participants performed standardized neuropsychological testing assessing five cognitive domains: global cognitive functioning, memory, executive functioning, psychomotor speed, and language. Global cognitive functioning was assessed using the Mini Mental State Examination (MMSE) 26 and the Six Item Cognitive Impairment Test (6-CIT). 27 Memory was evaluated using subtest Visual Reproduction from the Wechsler Memory Scale IV (WMS IV), 28 the Visual Association Test (VAT), 29 the 8-words test 30 and subtest digit span from Wechsler Adult Intelligence Scale – IV (WAIS-IV). 31 Psychomotor speed was assessed using Trail Making Test (TMT) part A 32 and the D-KEFS Color Word Test part 1 and 2. 33 For testing of executive function TMT part B, D-KEFS Color Word Test part 3 and 4 and subtests Key Search Test and Rule Shift Cards Test from the Behavioral Assessment of the Dysexecutive Syndrome (BADS) were used. 34 Language was evaluated using letter and category fluency 35,36 and the Boston Naming Test (BNT). 37 Raw test scores were transformed into z-scores for each cognitive domain.
Statistical analysis
All statistical analyses were performed with the Statistical Package of Social Sciences (SPSS, version 25.0). Differences between memory clinic patients (SCI, MCI and dementia) and controls in demographic variables were analyzed using t-tests for the normally distributed continuous variables, Mann-Whitney U tests for non-normally distributed continuous variables and chi-square tests for categorical variables. Before analysis, data inspection was performed. No extreme outliers were present in any of the four participant groups with respect to the BOLD amplitude parameter.
To test our primary objective - to assess the neurovascular coupling parameters (BOLD amplitude, TTP and TTB) between the patient groups and controls, multiple univariate GLM analyses were used, with group and sex as fixed factors and age as covariate. Additionally, to assess if findings of group differences were independent of classic SVD markers, deep microbleeds and lacunar infarcts were dichotomized (present or not present) and added to the first model together with WMH volume and gray matter volume as fixed factors or covariate appropriately. WMH volumes were log transformed. To assess if group differences are driven by the probable CAA patients in our cohort, an additional adjustment for probable CAA was done. In order to reduce the risk of type I errors, Bonferroni corrections for multiple comparison were applied. With Bonferroni corrections – corrected for the three neurovascular coupling parameters - an alpha of < 0.017 (0.05/3) was used as cut-off value being statistically significant.
To assess the associations between neurovascular coupling parameters and cognitive domains, multiple linear regression analyses were performed. Our primary focus is on the associations between cognitive domains and the BOLD amplitude, however for completeness we will also show the associations with TTP and TTB. In the first model, we adjusted only for sex and age. Secondly, we added classic SVD markers (deep microbleeds, lacunar infarcts, WMH volume and gray matter volume) to the model. Additionally, to assess if the significant associations between cognition and neurovascular coupling are independent of AD, we added hippocampus volume (as marker for AD) to the model. Finally, to assess if the associations are driven by the probable CAA patients in our cohort, an additional adjustment for probable CAA was done. With Bonferroni corrections – corrected for the five cognitive domains - an alpha of < 0.01 (0.05/5) was used as cut-off value being statistically significant.
Results
Participants
Table 1 shows the demographic and clinical characteristics for each group. The MCI and dementia group were older than controls and the SCI and MCI group had more male participants. MMSE score was lower for MCI and dementia patients. In the patient groups there was a higher incidence of cardiovascular risk factors, i.e. higher incidence of diabetes mellites in SCI, higher incidence of hyperlipidemia in MCI, and higher incidence of cardiovascular disease in dementia. There were no differences in systolic or diastolic blood pressure and there were comparable distributions of hypertension and smoking. In the MCI and dementia group there was a higher incidence of CAA related hemorrhagic markers. There were no differences in the presence of lacunar infarcts and deep microbleeds. WMH volume was larger in dementia group. Gray matter volume was smaller in all patient groups and hippocampus volume was smaller for the MCI and dementia group.
Demographic and clinical characteristics.
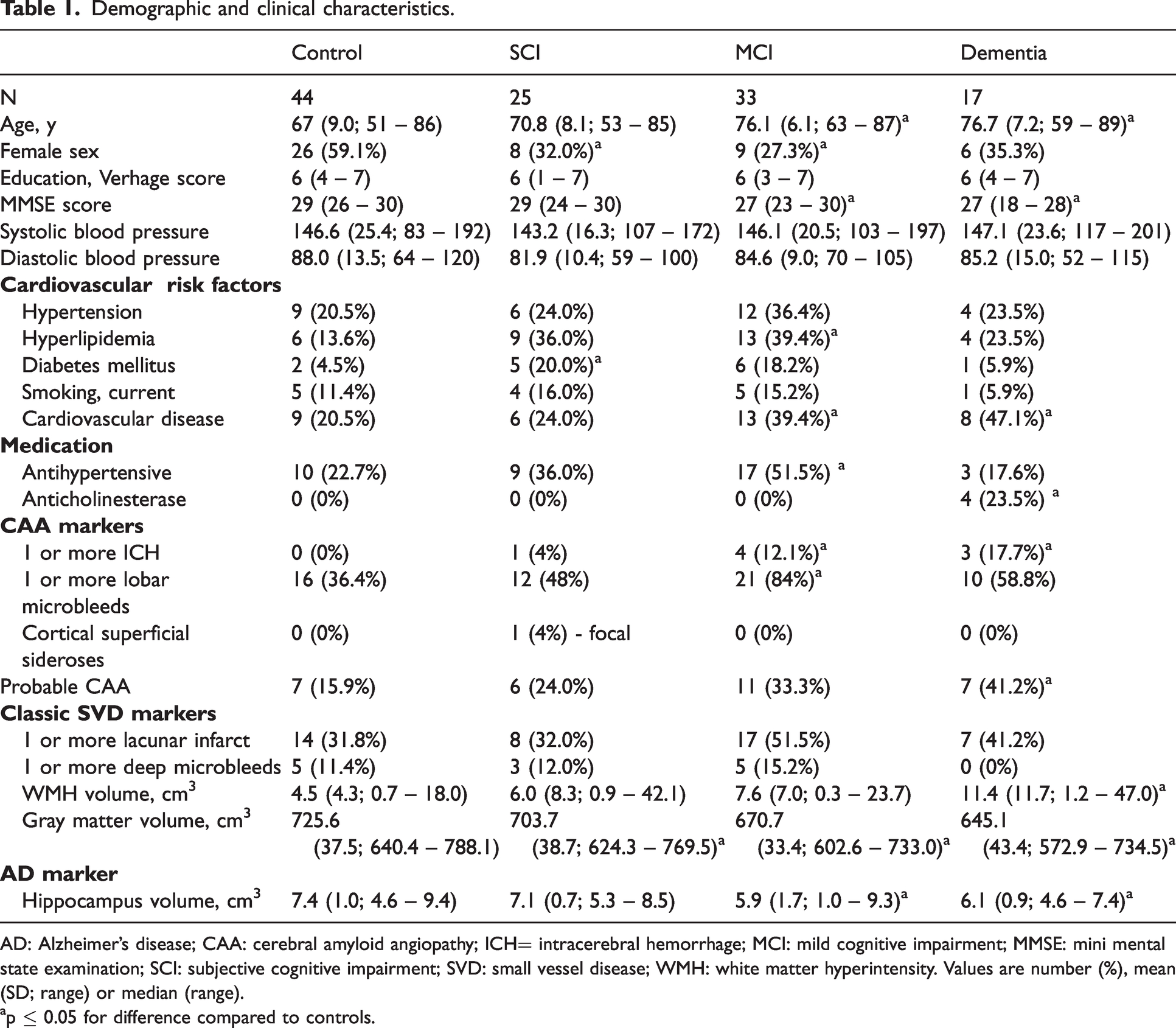
AD: Alzheimer’s disease; CAA: cerebral amyloid angiopathy; ICH= intracerebral hemorrhage; MCI: mild cognitive impairment; MMSE: mini mental state examination; SCI: subjective cognitive impairment; SVD: small vessel disease; WMH: white matter hyperintensity. Values are number (%), mean (SD; range) or median (range).
p ≤ 0.05 for difference compared to controls.
Neurovascular coupling between groups
Figure 2 shows a typical example of a BOLD activation pattern and BOLD time course of a single subject from each group. Figure 3(a) shows the distribution of the BOLD amplitude for each group separately. When adjusted for age and sex, the amplitude of the BOLD response after visual stimulation shows significantly lower values in dementia patients (mean amplitude 0.8 ± 0.2; p = 0.003) compared with controls (mean amplitude 1.1 ± 0.2) (Table 2). When additionally adjusted for classic SVD markers, i.e. deep MB, lacunar infarcts, WMH volume and gray matter volume, the BOLD amplitude also showed significantly lower values in the MCI patients (mean amplitude 0.9 ± 0.3; p = 0.001) when compared to controls. To assess if these group differences are driven by the probable CAA patients, an additional adjustment for probable CAA was done. The BOLD amplitude remained significantly lower in both dementia (p = 0.001) and MCI (p = 0.004). Additionally, to explore the potential impact of antihypertensive treatment and anticholinesterase drugs univariate GLM analysis was performed to assess the BOLD amplitude between participants with and without treatment, adjusted for sex and age. We found no differences in BOLD amplitude between groups for antihypertensive treatment (p = 0.144) or anticholinesterase treatment (p = 0.227). No differences in BOLD amplitude were found between the SCI patients and controls. When adjusted for age and sex, time to peak was longer for SCI patient when compared to controls (p = 0.006). After adjustment for SVD markers, this difference was no longer statistically significant (Figure 3(b)). No differences were found between MCI or dementia patients and controls regarding time to peak. For time to baseline, no differences between the patient groups and controls were found (Figure 3(c)). Moreover, no group differences were found for our additional parameters - time to signal activation and time to signal deactivation.
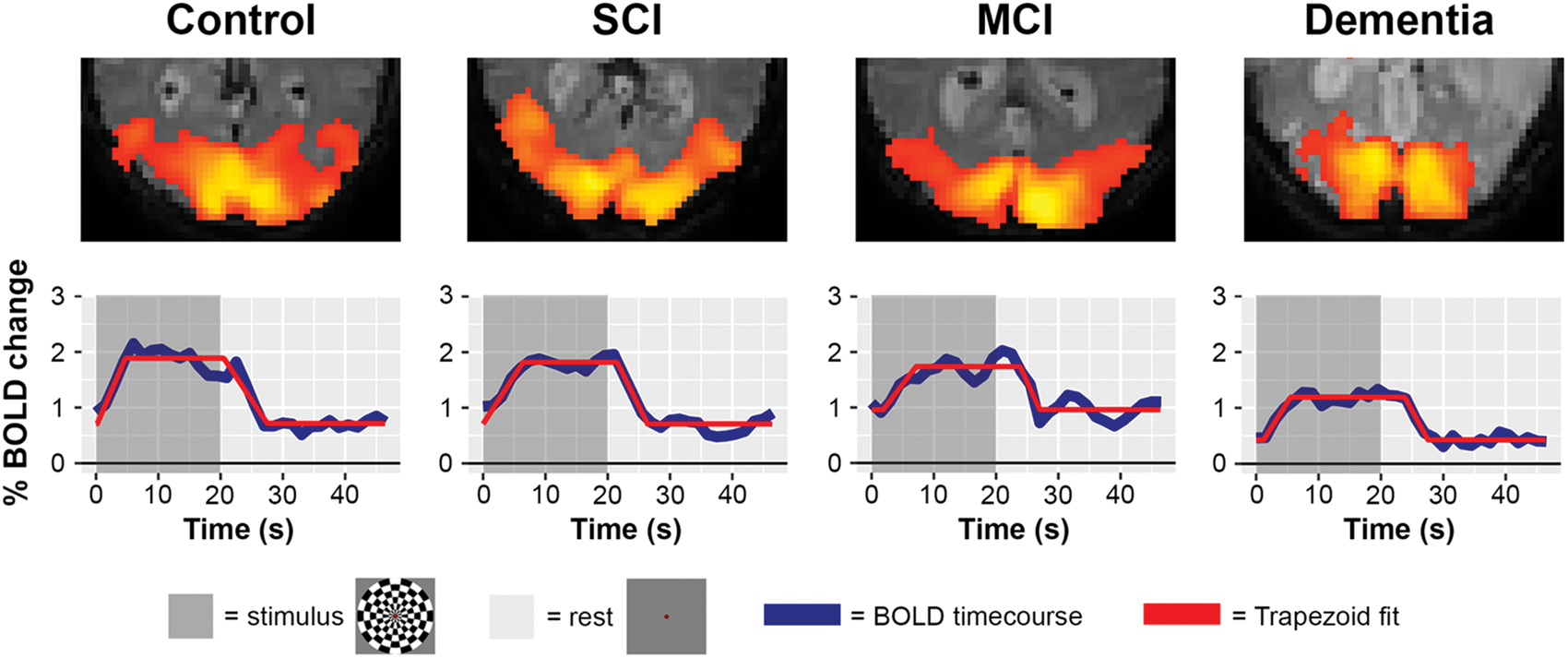
This figure shows a typical example of the BOLD activation map superimposed on an anatomical image and the BOLD time course for a random control (75 year old male, BOLD amplitude 1.2), SCI (74 year old male, BOLD amplitude 1.1), MCI (78 year old male, BOLD amplitude 0.8) and dementia patient (74 year old male, BOLD amplitude 0.7).
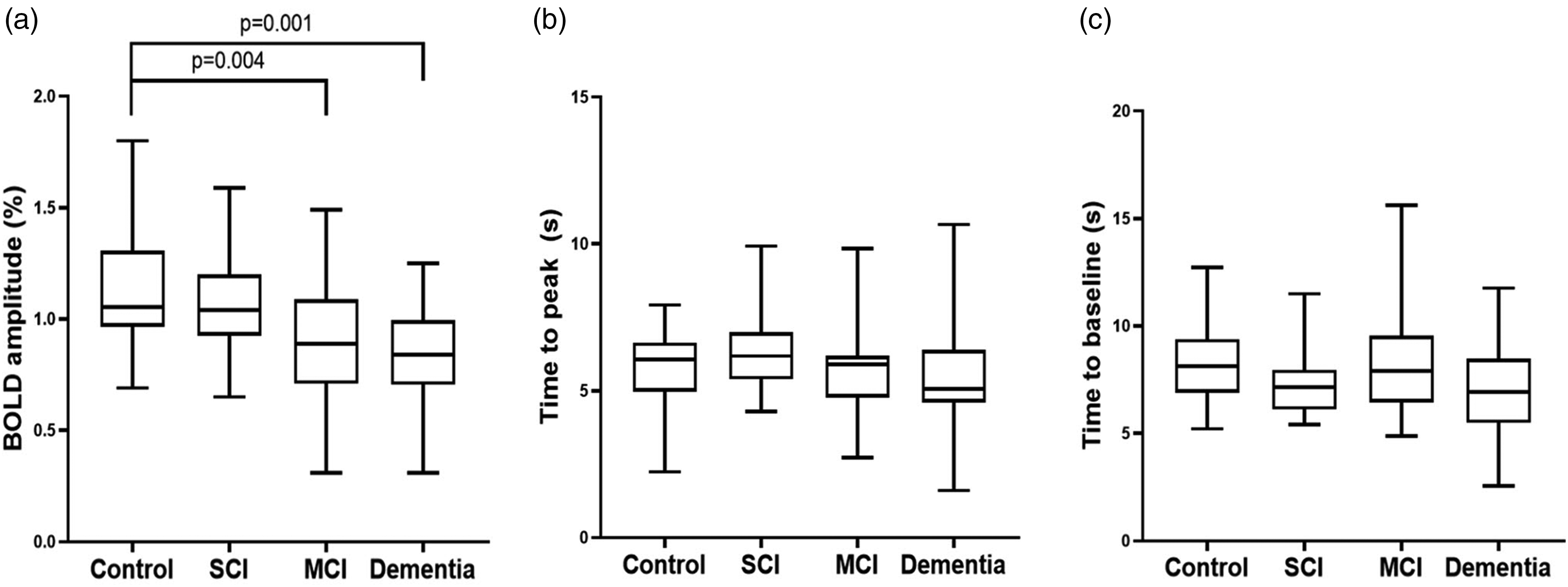
This figure shows the distribution of the BOLD amplitude in percentage change (a), time to peak in seconds (b) and time to baseline in seconds (c) for each group separately. Amplitude of the BOLD response show significantly lower values in dementia and MCI in comparison to controls.
Neurovascular coupling parameters between patient groups and controls using GLM.
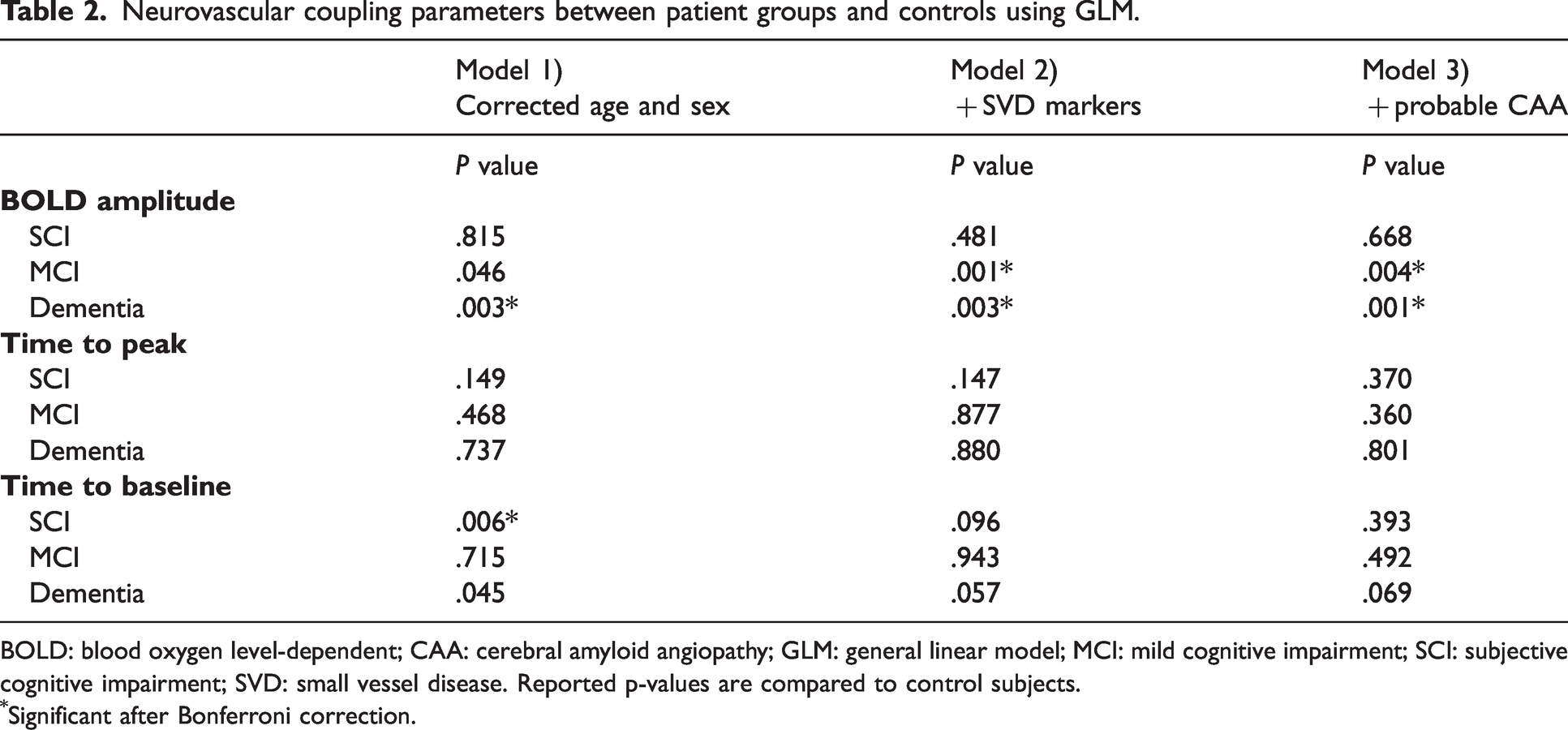
BOLD: blood oxygen level-dependent; CAA: cerebral amyloid angiopathy; GLM: general linear model; MCI: mild cognitive impairment; SCI: subjective cognitive impairment; SVD: small vessel disease. Reported p-values are compared to control subjects.
Significant after Bonferroni correction.
Neurovascular coupling parameters and cognition
The associations between the three neurovascular coupling parameters and five cognitive domains are shown in Table 3. When adjusted for age and sex a low amplitude of the BOLD response was associated with a low score on global cognitive functioning (p = 0.003), memory (p < 0.001), executive functioning (p = 0.009) and language (p < 0.001) (Figure 4). These associations remained significant when adjusted for SVD markers. To assess whether our findings were driven by AD pathology, hippocampus volume was added to the model. After this adjustment a low amplitude of the BOLD response remained associated with a low score on global cognitive functioning, memory, executive functioning and language (all p < 0.001). Moreover, the BOLD amplitude also showed an association with psychomotor speed (p = 0.005), i.e. a low BOLD amplitude was associated with a low speed. Finally, we added probable CAA to the model, to assess whether our findings were driven by the participants with probable CAA. All associations remained significant. With regard to time to peak and time to baseline we found no associations with any cognitive domains.
Associations between neurovascular coupling parameters and cognitive domains.
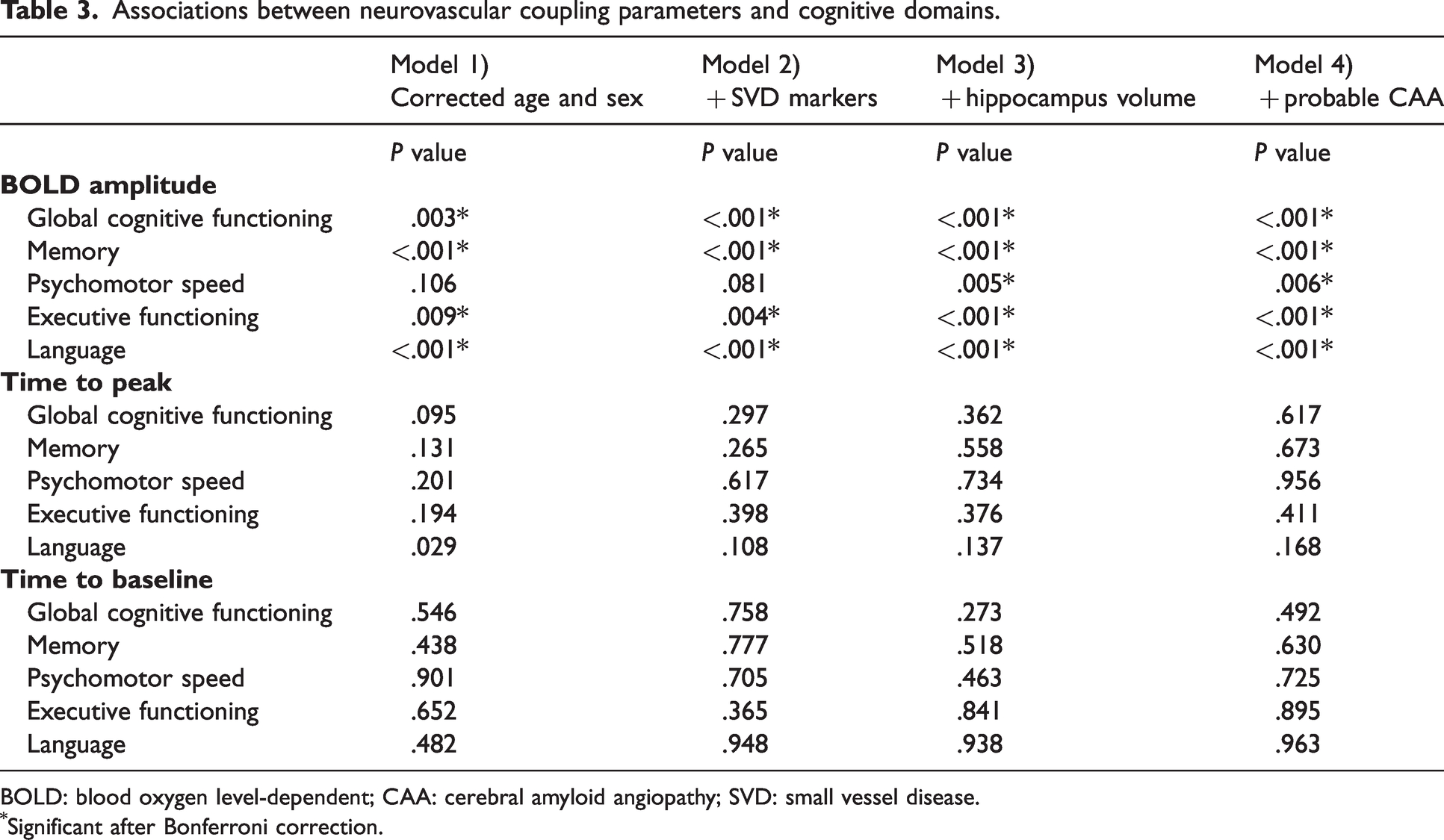
BOLD: blood oxygen level-dependent; CAA: cerebral amyloid angiopathy; SVD: small vessel disease.
Significant after Bonferroni correction.
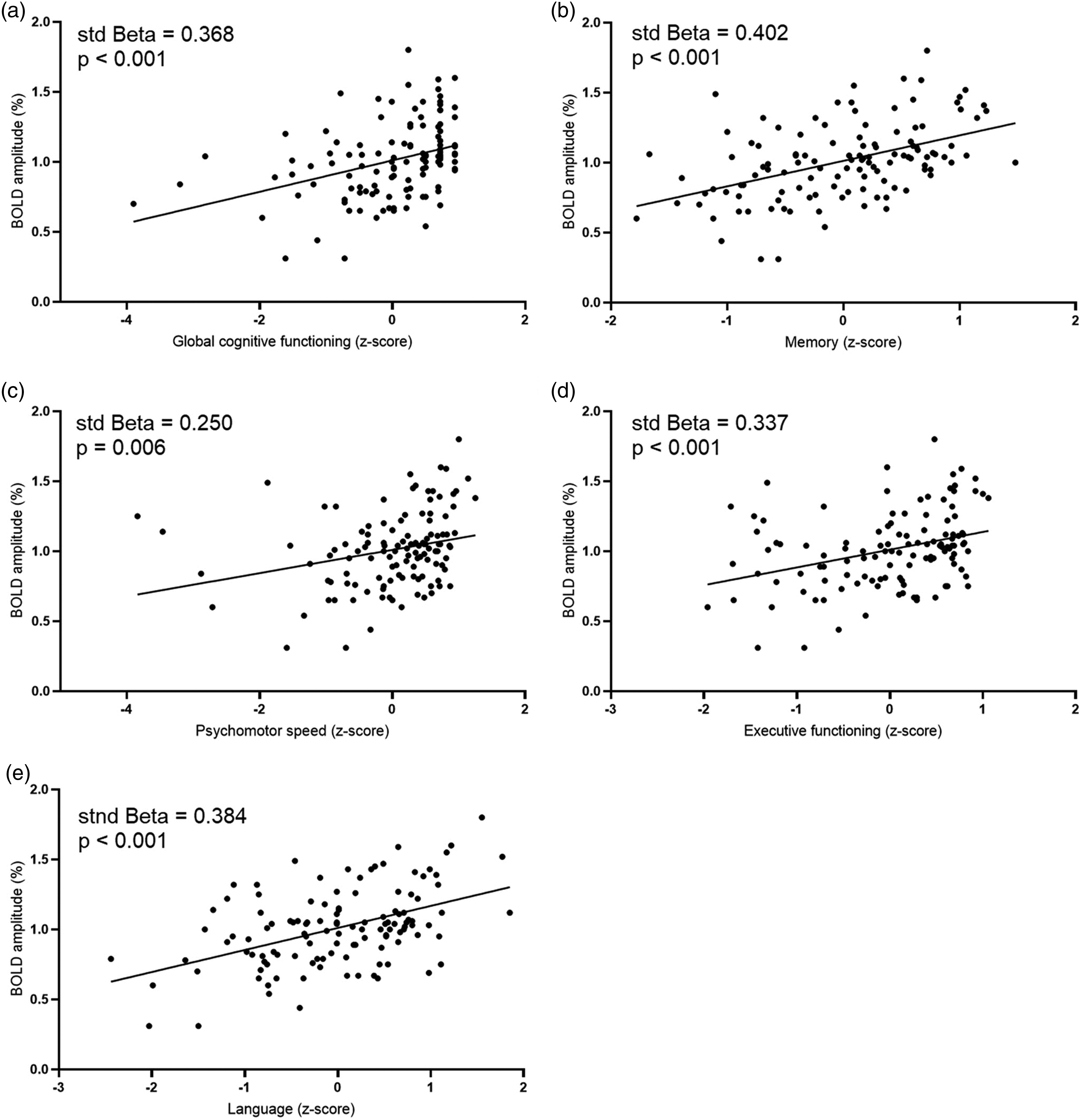
This figure shows the significant associations between BOLD amplitude and global cognitive function (a), memory (b), psychomotor speed (c), executive functioning (d) and language (e). on the x-axis the different cognitive domains in Z-scores and on the y-axis the amplitude of the BOLD response in percentage change.
Discussion
The primary objective of this study was to assess neurovascular coupling in AD related dementia and its early stages. Our data show that neurovascular coupling is decreased in both MCI and dementia patients. We also show that a decreased neurovascular coupling is independently associated with poorer cognitive function in multiple cognitive domains.
Our primary finding that neurovascular coupling is decreased in MCI and dementia is thought to reflect cerebrovascular dysfunction due to mild-to-moderate CAA. Hemorrhagic MRI markers, which are currently used to estimate the prevalence of CAA in AD patients during life,38,39 represent changes relatively late in the disease process and identify only the more severe CAA in this population. The prevalence estimates of CAA in AD patients using strictly lobar cerebral microbleeds (22%) underestimates the prevalence of CAA based on post mortem neuropathological examination (48%). 40 Thus leaving a great percentage of CAA in AD patients undetected. As a decrease in neurovascular coupling precedes the occurrence of hemorrhagic events,9,13 our finding of decreased neurovascular coupling is thought to represent not only the more severe CAA but also the early vascular damage due to cerebrovascular Aβ deposition. Our finding was independent of other classic SVD markers and was not driven by the participants with more advanced CAA.
Our findings add to the observations of a decreased cerebrovascular reactivity in MCI and AD patients as measured by transcranial doppler sonography 41 –45 and by BOLD MRI with CO2 as vasodilatory stimulus.46 –48 These studies may also have been detecting changes related to comorbid CAA although it was not mentioned specifically in these studies. A decreased neurovascular coupling is not always reported despite using a similar visual stimulus and BOLD MRI, 49 possibly due to differences in participant selection and exclusion criteria.
Our second important finding is that a decreased neurovascular coupling is associated with poorer cognitive function. Our data show that a low amplitude of the BOLD response is significantly associated with a low score on global cognitive functioning, memory, executive functioning, psychomotor speed and language. Similar associations between cerebrovascular reactivity and cognitive screening instruments were found using transcranial doppler sonography, 50 ASL MRI 51 and CO2 BOLD MRI. 48 Although reports on the association with specific cognitive domains are conflicting.48,52 Our findings are independent of sex, age, classic SVD markers and hippocampus volume, suggesting that decreased neurovascular coupling, possible due to CAA, may be directly associated with cognition and has an independent contribution to the cognitive decline in dementia. This adds to the findings from a large clinical-pathologic study that show that CAA is an independent contributor to AD dementia. 7
Neurovascular coupling to visual stimulation may be a useful marker for clinical trials and it has already been used for in clinical trials in CAA. 54 Vascular amyloid lowering strategies might prevent pathophysiological processes leading to vascular dysfunction 53 and CAA could therefore be a new target for treatment to slow down or even prevent cognitive decline in AD.
A limitation to the method used is that the observed BOLD effects may not only be caused by changes in the vessel wall, but also have a neuronal component. At present, we cannot exactly disentangle the extent to which the vascular and neural component contribute to the BOLD signal in this study. However, the finding in symptomatic CAA decreased neurovascular coupling in response to a stimulus was only observed in the occipital cortex, and not observed in the motor cortices in response to a motor task, 10 favors the ‘vessel wall pathway’. Another limitation of our study is that the current cross-sectional design does not allow us to determine whether neurovascular coupling is predictive for disease progression. Longitudinal studies are therefore necessary to further investigate whether neurovascular coupling and its change over time have predictive value for the disease progression of dementia.
In conclusion, our data show that neurovascular coupling, measured as changes in the blood oxygen level-dependent (BOLD) signal in the occipital cortex to visual stimulation, is lowered in dementia and more importantly, already in its early stage of MCI. Moreover, neurovascular coupling is independently associated with cognitive decline. These results indicate that cerebrovascular dysfunction, possible due to CAA, is an important comorbidity in early stages of dementia. Subtle damage to the vessels due to cerebrovascular Aβ deposition appears to play an important role in the disease process of dementia, even before structural damage to the vessels due to hemorrhagic lesions occur.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Alzheimer Nederland Nederland [WE.03-2018-12] - part of Deltaplan Dementie - and the Netherlands Organization for Scientific Research [Veni grant: 016.196.118].
Acknowledgements
We thank Caroline Jurgens for her consulting role. Also, we thank Jessie Lak, Francesca Baarzel, Jessy Kauffman and Esther Grin for their contribution to the data collection.
Authors’ contributions
SD, JG and SvR contributed to the concept and design of this study. SD, ND, GL, JG and SvR contributed to the data acquisition and SD, ND, AH and SvR to analysis of the data. SD, ND, MNW, GB, SR, JG and SvR contributed to the interpretations of the data and manuscript writing. All authors edited and commented on the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
