Abstract
Great uncertainty exists as to whether aging enhances the detrimental effects of tissue plasminogen activator (tPA) on vascular integrity of the ischemic brain. We hypothesized that tPA treatment would augment ischemic injury by causing increased blood-brain barrier (BBB) breakdown as determined by quantitative serial T1 and T2 magnetic resonance imaging (MRI), and the transfer constant for gadolinium-diethylenetriamine penta-acetic acid (Gd-DTPA) from blood to brain in aged (18 to 20 months) compared with young (3 to 4 months) Wistar rats after middle cerebral artery occlusion, mediated through the acute disassembly of claudin 5 and occludin. Increased T2 values over the first hour of postreperfusion were independently augmented following treatment with tPA (P < 0.001) and aging (P < 0.01), supporting a synergistic effect of tPA on the aged ischemic brain. Blood-brain barrier permeability for Gd-DTPA (KGd) was substantial following reperfusion in all animal groups and was exacerbated by tPA treatment in the elderly rat (P < 0.001). The frequency of hematoma formation was proportionately increased in the elderly ischemic brain (P < 0.05). Both tPA and age independently increased claudin 5 and occludin phosphorylation during ischemia. Early BBB permeability detected by quantitative MRI following ischemic stroke is enhanced by increased age and tPA and is related to claudin 5 and occludin phosphorylation.
Introduction
Stroke is the third most common cause of death and disability worldwide, and increases dramatically with advancing age (Hollander et al, 2003). Epidemiological evidence suggests that the elderly disproportionately suffer more disability and mortality following stroke, and arguably have the most to gain from stroke treatments (Barber et al, 2000; Paolucci et al, 2003). Thrombolytic therapy for acute ischemic stroke remains the only available treatment for acute disabling stroke but despite significant benefit its overall effect is limited by the small proportion of patients eligible (Barber et al, 2001) and by concerns regarding increased risk of fatal outcome and symptomatic intracranial hemorrhage (The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995; Wardlaw et al, 2009). Furthermore, we have very little information on the effects of age on stroke severity or of the biological effects of thrombolytic therapy on the elderly ischemic brain. This is because very few patients (0.5%) enrolled were aged over 80 years (Wardlaw et al, 2009), and the limited observational data available suggest an increased incidence of ICH, which is more frequently fatal in the elderly (Mateen et al, 2010; Lindley et al, 2011).
Rapid reperfusion following thrombolytic therapy, although desired, potentially contributes to secondary injury through a cascade of events considered to include factors such as hemodynamic disturbances, inflammatory processes, free radical formation, and breakdown of the blood-brain barrier (BBB) (del Zoppo, 1994; Heo et al, 2005). Specifically, the presence of enhanced BBB permeability has been identified as a major factor that increases the incidence of hemorrhagic transformation (Knight et al, 1998; Jiang et al, 2002; Ding et al, 2005). We have previously found that the elderly were more likely to have poor outcome and suffer more adverse effects from the therapy (Barber et al, 2000). The Canadian Stroke Effectiveness Study also made the important observation that any form of confluent hemorrhage, whether termed symptomatic or asymptomatic, conferred a greater risk of more severe outcome (Dzialowski et al, 2007). This suggests that any form of hemorrhage induced by tissue plasminogen activator (tPA) is not benign even if it does not produce a recognizable deterioration in neurologic score.
The brains barrier to paracellular diffusion is created by tight junction proteins, which are capable of rapid disassembly in response to extracellular stressors such as pain, inflammation, and hypoxia (Hawkins and Davis, 2005), and therefore may in part account for the early increase in BBB permeability we have observed previously (Kaur et al, 2009). Both claudin and occludin are critical for tight junction BBB function and paracellular diffusion (Furuse et al, 1998; Hawkins and Davis, 2005). Aging in experimental animals and humans is associated with significant structural functional alterations in the BBB (Mooradian et al, 2003). What is not known is whether such changes in tight junction expression in the elderly rat confer an increased BBB permeability during ischemia; furthermore, what may be the consequences of tPA on BBB permeability, specifically in relation to occludin and claudin 5 structure and function.
We have previously shown that quantitative magnetic resonance imaging (qMRI) can better define susceptible tissue in the development of early vasogenic edema (using T2) during ischemia reperfusion (Kaur et al, 2009). The apparent diffusion coefficient (ADC) has traditionally been considered to be sensitive to cytotoxic edema during ischemia (de Crespigny et al, 1993). However, the ADC provides no information regarding BBB integrity or stroke severity, and the predictive value of ADC changes for the recovery of ischemic tissue have been questioned (Oppenheim et al, 2001; Fiehler et al, 2002). This limitation is greatest in the reperfusion phase (Kaur et al, 2009) because the ADC moves relatively rapidly from decreased values through a pseudonormal to elevated values. Alternatively, the use of qMRI T2 measurements might provide information regarding the secondary injury to the BBB that occurs in the reperfusion phase, and how BBB permeability may be modified during stroke by age and tPA, and ultimately determine the risk of hemorrhagic transformation. The blood-brain transfer constant (KGd) for the transfer of gadolinium-diethylenetriamine penta-acetic acid (Gd-DTPA) from blood to brain can also be used as a measure of BBB dysfunction using MRI (Ewing et al, 2003). Thus, quantified MRI could potentially be used as a measure of vascular integrity and its modification following injury, repair, or treatment. In this study, we hypothesized that tPA administered after ischemic stroke in the elderly rat produces increased BBB permeability mediated through a derangement of tight junction structure and function.
Materials and methods
Middle Cerebral Artery Occlusion
All experiments and procedures were approved by the animal care committees of the University of Calgary and the National Research Council of Canada (NRC). All rats were anesthetized with isoflurane and a craniotomy (2 to 3 mm) was then made over the right middle cerebral artery (MCA) (2 to 3 mm anterior to the union of the zygomatic arch and the temporal bone). Cortical cerebral perfusion was estimated by a laser-Doppler flowmetry probe situated over the core and penumbral regions during preischemia, during MCA occlusion (MCAO), and during reperfusion, using Perisoft software (Version 1.3, Perimed Inc., North Royalton, OH, USA). After occlusion of the common carotid artery, a microaneurysm clip was placed on the right MCA to produce a reduction in cortical Doppler flow below 15% of its baseline. The microaneurysm clip and the sutures around the common carotid artery were removed after 90 minutes allowing for reperfusion (Kaur et al, 2009). The animal core body temperature was maintained at 37.4°C ± 0.2°C using a rectal biofeedback control system throughout the surgical procedure, during the imaging protocol, and recovery from anesthesia. Cannulation of the external jugular vein was performed using PE50 for administration of tPA or saline. All animals undergoing MCAO had tail artery cannulation for sampling of blood for arterial blood gas analysis. Also, in all animals, arterial blood pressure was monitored during the surgery via the tail artery using AcqKnowledge software (Biopac Systems Canada, Montreal, QC, Canada). Arterial blood gases were analyzed for PaO2, PaCO2, and pH during occlusion and after reperfusion using a blood-gas analyzer (Critical Care Express, Nova Biomedical, Waltham, MA, USA).
Experimental Groups and Drug Treatments
The experimental goal of the study was to determine the effects of intravenous tPA on reperfusion injury in the elderly rat brain. Eighty-four male Wistar rats (40 young, 200 to 250 g; 44 elderly, 600 to 950 g) were acquired from Charles River Canada (Senneville, QC, Canada). The randomization process was conducted by tossing a coin. Tissue plasminogen activator or saline was administered on average 30 minutes after reperfusion. This time delay represented the time required to transfer the animal from the surgical table into the MRI, and acquire pretreatment MRI sequences. The tPA (Activase; Genetech Inc., South Fransisco, CA, USA), which was reconstituted and made to a concentration of 10 mg/mL, and the dose administered to the animal was 10 mg/kg. Ten percent of dosing solution was given as IV bolus (10% of total dose) followed by IV infusion at the rate of 2.5 mL/h immediately after reperfusion in transient MCAO. The relatively high dose of tPA was chosen on the basis of the ∼10-fold difference in fibrin-specific activity between human and rodent systems (Korninger and Collen, 1981; Sumii and Lo, 2002). All analysis of MR images and histology was conducted blind to treatment allocation.
Magnetic Resonance Imaging
Sequential MR images were acquired at the time of treatment with either tPA or saline (time 0) and then at 1 hour (time 1 hour) and 24 hours after treatment. Rats were anesthetized with isoflurane and placed in a cradle positioned within the center of the bore of a Bruker 9.4 T MR system. Images were acquired using a 3.5-cm diameter quadrature volume radio frequency (RF) coil (3 × 3 cm2 field of view, 128 × 128 matrix size). Respiration and rectal temperature were monitored and the animals were kept normothermic.
Images for a T2 map consisted of a set of multislice multiecho T2-weighted images (TR = 2,500 milliseconds, echo spacing of TE = 10 milliseconds, 24 echoes). A T1 map was acquired in a single slice using an inversion recovery Snapshot-FLASH imaging sequence (TR = 4.25 seconds, TE = 1.8 milliseconds, with 12 inversion delays ranging between 234 and 9,226 milliseconds). An ADC map was acquired using a spin-echo sequence (TR = 1,500 milliseconds, TE = 40 milliseconds, three b values of 108, 356, and 768 s/mm2). Imaging software developed by the National Research Council of Canada (Marevisi, National Research Council of Canada, Ottawa, ON, Canada) was used to analyze the imaging data.
Changes in T1, T2, and ADC measures (an increase in the intensity of T1 and T2 maps and a decrease in ADC maps) were analyzed by investigators blinded to the experimental animal groups. Dynamic changes in quantitative MRI parameters were performed on a single slice through the striatum because of practical time constraints limiting the number of T1 slices. Quantitative measures were made within a slice through the striatum for regions of interest (ROIs) that included the infarct core (ROI-1), and a homotopic position contralaterally (ROI-2). The ROI was of predetermined size and its placement within the hemispheres was consistent in the same anatomical region within the stroke lesion, which was established at 24 hours. Absolute values (T1 and T2), and ADC within the areas of maximal increase in hyperintensity were measured. Area of infarction on a standard anterior striatal slice was calculated for T1, T2, and ADC rather than volume because of limitations arising from the single slice availability for the T1 sequence.
Assessment of Stroke Injury
Infarct areas and volumes were calculated using the multislice T2 MR images. The infarct area in each T2 slice was readily apparent as a region of increased T2. The calculation of the infarct area included a measure of the number of voxels in the ipsilateral hemisphere (with the exception of the ventricles) having a T2 value greater than a threshold of 2 s.d. above the mean T2 in the contralateral cortex. For each slice, the infarct area was corrected for edema (ipsilateral infarct area (uncorrected)/((ipsilateral hemisphere area/contralateral hemisphere area)), multiplied by the number of slices.
After MRI, animals were deeply anesthetized and transcardially perfused with saline followed by 4% paraformaldehyde. The heads of the animals were placed in formalin overnight before removing the brains and embedding in paraffin. Ten standardized coronal sections of 8 µm thickness spaced 1.0 mm apart were taken from the prefrontal cortex to the rostral pons. Following deparaffinization, brain sections were processed for standard hemotoxylin and eosin staining for assessment of edematous, hemorrhagic, and neuronal injury.
Measurement of Blood-Brain Barrier Dysfunction
Magnetic resonance imaging assessment using Patlak plots: Blood-brain barrier function in a selected slice was assessed by determining the transfer constant for Gd-DTPA (KGd) from the blood into the brain at ∼1 hour posttreatment and at 24 hours. This was calculated using T1 measures in the sagittal sinus and in the ROIs—that is the parietal cortex in the infarct core and the equivalent contralateral control cortex, using methods described previously (Ewing et al, 2003). Rapid quantitative estimates of brain tissue T1 values were obtained using an image adaptation of the Look-Locker method (Knight et al, 2005). Baseline T1-weighted spin-echo and Look-Locker scans were collected before Gd-DTPA infusion. After obtaining one or two baseline estimates, Gd-DTPA 0.1 mmol/kg was injected into the jugular vein in < 5 seconds. Using the Look-Locker T1 procedure, sets of maps of the longitudinal relaxation rate (R1 = 1/T1) were acquired sequentially at 3 minutes intervals for the total duration of 30 minutes (Ewing et al, 2003). Differences measured in the ischemic area of injury in the ipsilateral hemisphere were compared with the same region in the contralateral hemisphere.
Immunohistochemical assessment using IgG extravasation: IgG extravasation was assessed in 10 µm thick coronal sections (Bregma: 1.2 mm), with goat anti-rat IgG antibody conjugated to horseradish peroxidase. Diaminobenzidine was used for visualization of the antibody reactivity. Using an Olympus BX 61 (Olympus Canada, Markham, ON, Canada) light microscope at × 1.5 magnification, IgG extravasation was quantified by measuring the gray scale value using comparative ROIs in both cerebral hemispheres, and subtracting the gray scale intensity (in arbitrary units) of the contralateral and ipsilateral stroke regions, using comparatively similar ROIs. The results are presented as ratio of contralateral to ipsilateral gray scale values. With increasing BBB injury, darker staining was observed, indicative of greater IgG extravasation.
Measurement and localization of tight junction proteins: Western blot measurement of occludin and claudin 5.
The animals were anesthetized and transcardially perfused with ice-cold phosphate-buffered saline. The brains were removed, microdissected, and tissue samples obtained from the contralateral cortex, and the infarct region in the ipsilateral cortex. The tissues samples were homogenized in RIPA buffer (150 mmol/L NaCl, 10 mmol/L tris, 0.1% sodium dodecyl sulfate, 1.0% Triton X-100, 1% deoxycholate, 5 mmol/L EDTA; pH 7.4), containing phosphatase inhibitors (100 µmol/L sodium orthovanadate, 5 mmol/L sodium fluoride) and protease inhibitors (Protease Inhibitor Cocktail tablets, Roche Canada, Mississauga, ON, Canada; 1 tablet/10 mL of buffer). Thereafter, the homogenized tissue was centrifuged at 12,000 r.p.m. for 20 minutes at 4°C. The supernatant was collected, aliquoted, and stored at —80°C until use.
Immunoblot analysis of tight junction proteins occludin and claudin 5 was performed on protein extracts of parietal ipsilateral ischemic and contralateral cortex of rats at early and 24 hours time points after reperfusion. The protein concentration of extracts was determined using a Bio-Rad Assay (Lowry et al, 1951). Bovine serum albumin was used as standard. Electrophoresis of the total proteins (100 µg of protein/lane) was performed on a denaturing sodium dodecyl sulfate polyacrylamide gel electrophoresis (10% (w/v) acrylamide). The proteins were transferred overnight at 4°C onto a polyvinylidene fluoride (PVDF) membrane using Bio-Rad wet transfer unit. The membrane was washed with tris-buffered saline containing Tween-20 (20 mmol/L Tris-HCl, 100 mmol/L NaCl, 0.25% (v/v) Tween-20; 3 × 20 minutes), and incubated for 1 hour in blocking buffer (containing 7.5% (w/v) dry skim milk). Successively, the blots were incubated for 1.5 hours with monoclonal mouse anti-occludin (1:2,000; Zymed, San Francisco, CA, USA), mouse anti-claudin 5, and mouse anti-β-actin (1:2,000; Santa Cruz Biotechnologies, Santa Cruz, CA, USA). After washing (3 × 15 minutes), the blots were blocked and incubated for 2 hours with horseradish peroxidase conjugated and goat anti-mouse secondary antibodies (1:10,000; Upstate, Lake Placid, NY, USA). Protein bands were visualized by exposing the X-ray film (GE Healthcare Canada, Baie d'Urfe, QC, Canada) using enhanced chemiluminescence (GE Healthcare Canada). The X-ray films were processed by a Kodak machine (Kodak Canada, Verdun, QC, Canada). The processed X-ray films were scanned and the images were analyzed for densitometry using ImagePro Plus software (Media Cybernetics, Bethesda, MD, USA).
Statistical Analysis
The data are presented as mean ± s.d., and were analyzed using a two-way analysis of variance (ANOVA) with multiple comparisons (Dunn's test with comparison to a control group or Tukey's test) and one-way ANOVA for repeated measures using SigmaStat software (Systat Software Inc., San Jose, CA, USA). Fisher's exact test was used to determine proportional differences in hemorrhage between groups. Pearson's correlation was used to analyze the relationship between KGd and quantitative T2. Differences were considered significant at P < 0.05.
Results
Baseline Characteristics and Physiology
A total of 84 animals were used in the two age groups. Of the 40 young rats, 38 were randomized to tPA or saline 30 minutes after transient MCAO, and 2 surgical sham animals received saline. Six animals receiving tPA died because of hemorrhagic complications of the drug (systemic and intracranial), and as a result, no MRI data or histological samples could be obtained. Data were obtained from the remaining 32 animals (equally allocated to saline and tPA). Similarly, of the 44 elderly animals, 42 were randomized to tPA or saline, and 2 surgical sham animals received saline. In total, five elderly animals died (one saline related to surgical complications; and four tPA-treated animals related to bleeding complications), and as a result no MRI data or histological samples could be obtained. Of the remaining 37 elderly animals, 18 were allocated to saline and 19 to tPA. The mean weight of rats in the younger group was 219 ± 53 g, and in the elderly group 779 ± 114 g. All the animals successfully reached the criteria of decreased blood flow (< 15% of baseline) after placement of a microaneurysm clip on the MCA. The blood pressure measures, relative cerebral blood flow, and arterial blood gas analysis for each animal group are presented in Table 1.
Physiological parameters in elderly and young animals that underwent ischemia reperfusion
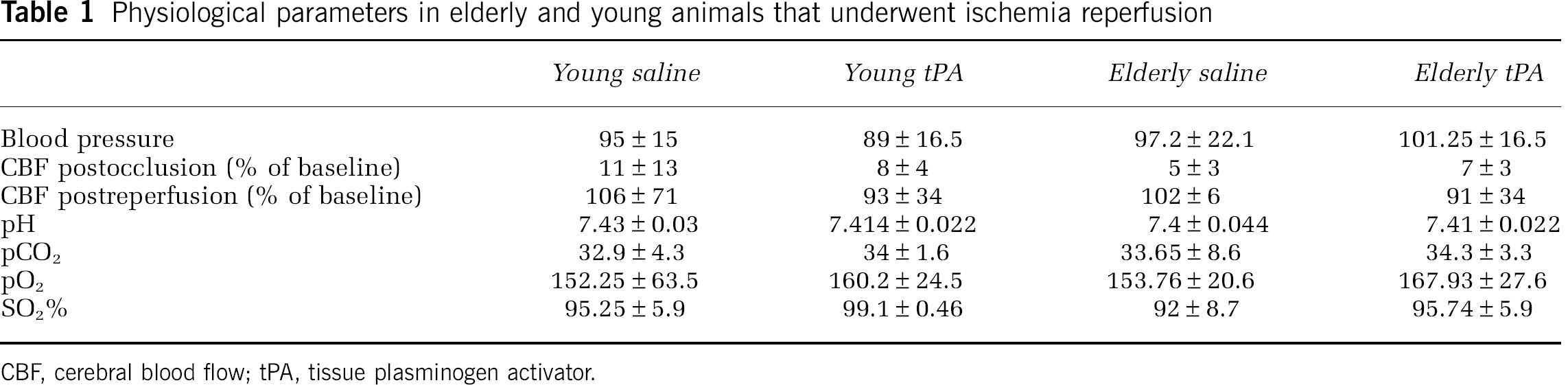
CBF, cerebral blood flow; tPA, tissue plasminogen activator.
Quantified magnetic resonance imaging tissue characteristics following ischemia/reperfusion: tissue plasminogen activator and age increase absolute T2 values: Ischemia-reperfusion increased T2 values in all animal groups within the ipsilateral cortex at time 0 when compared with contralateral T2 values (P < 0.001). Before treatment, the contralateral T2 values ranged from 44 ± 3 to 48.6 ± 1.53 milliseconds, which increased in the ipsilateral cortex of the various groups, to 57 ± 1.5 milliseconds (young saline), 54.1 ± 4.6 milliseconds (young tPA), 60 ± 7.7 milliseconds (elderly saline), and 61.16 ± 7.3 milliseconds (elderly tPA). The T2 values in the contralaterial cortex did not change over time. There were variations in the right-left differences in T2 values before and after treatment administration for each animal group (Figure 1). The most significant findings were that absolute T2 left-right differences between treatment times at 0 and 1 hour posttreatment independently increased both with tPA administration (P < 0.001) and with age (P < 0.01). In addition, quantified T2 measurements at 24 hours revealed an increase with tPA treatment (P < 0.001) and an interaction was identified between age and treatment (P < 0.05), implying the increased T2 in the elderly ischemic brain is deleteriously moderated by tPA when measured at this time point.
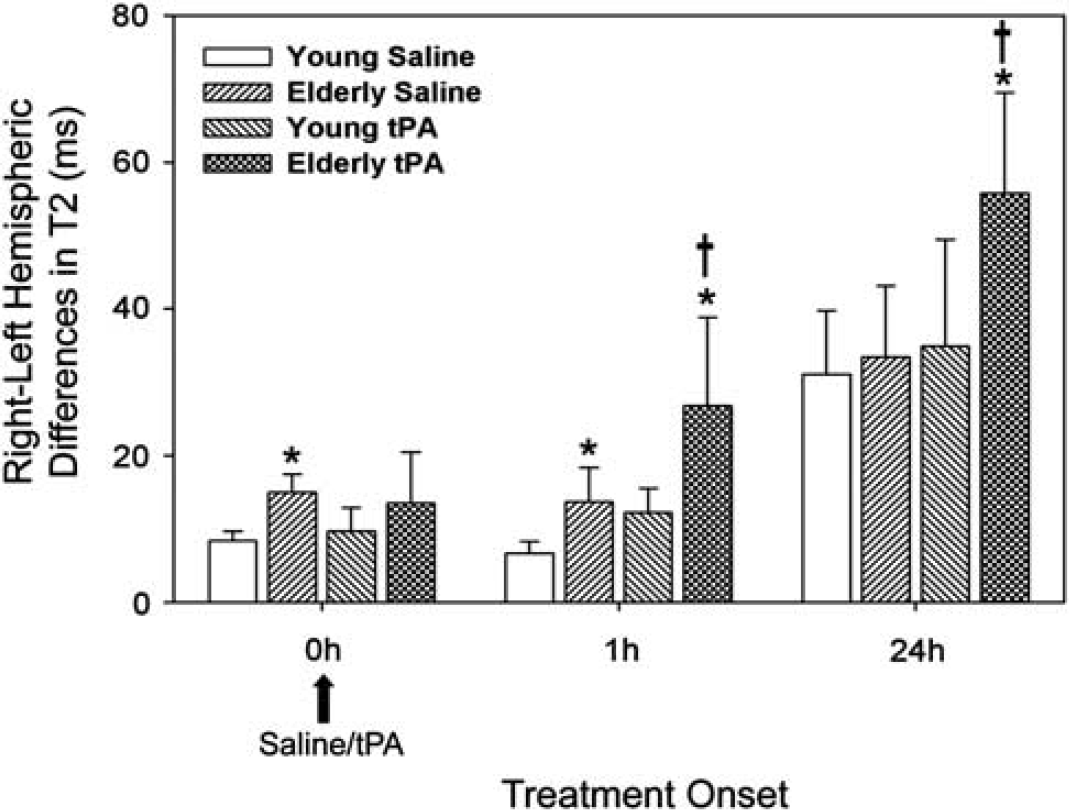
The elderly rat brain and tissue plasminogen activator (tPA) administration are associated with an increase in quantitated T2 in ischemia reperfusion. Two-way analysis of variance (ANOVA) revealed an independent effect of age (*P < 0.01) at all time points, and the enhanced T2 effect seen in the elderly brain is modified by tPA at 1 (†P < 0.001) and 24 hours (†P < 0.05). (*Young different from elderly; †saline different from tPA).
Absolute T1 was increased in all animal groups within the ipsilateral cortex at time 0 when compared with contralateral T1 values (P < 0.001). Before treatment, the contralateral T1 values ranged from 1,802 ± 34 milliseconds (elderly) to 2,316 ± 148 milliseconds (young), which increased in the ipsilateral cortex to 2,353 ± 81 milliseconds (young saline), 2,026 ± 109 milliseconds (young tPA), 2,129 ± 130 milliseconds (elderly saline), and 2,222 ± 172 milliseconds (elderly tPA). There was a change in the T1 value between time 0 and time 1 for elderly animals receiving tPA (2,222 ± 172 versus 2,319 ± 196; P = 0.04, one-way repeated measures ANOVA). No such an effect was observed in the young animals treated with tPA.
The ADC values were variably affected by ischemia reperfusion. The respective ipsilateral and contralateral values were not significantly different in the immediate reperfusion phase for young animals treated with saline (8.0 ± 9.9 × 10−5mm2/sec and 8.8 ± 1.4 × 10−4mm2/sec; NS, P = 0.2) or tPA-treated animals (8.25 ± 5.0 × 10−5mm2/sec and 8.2 ± 6.6 × 10−5mm2/sec; NS, P = 0.84). In elderly animals treated with saline, ipsilateral and contralateral values in the immediate reperfusion phase were significantly decreased (5.90 ± 0.2 × 10−3mm2/sec and 8.6 ± 3 × 10−5mm2/sec, respectively; P < 0.05). Such differences were not observed for animals allocated to the tPA group (6.7 ± 1.4 × 10−4 mm2/sec and 7.2 ± 1.0 × 10−4mm2/sec; P = 0.4).
Stroke lesion evolution: tissue plasminogen activator and age result in early increase in T2 lesion area: A one-way ANOVA revealed independent increased T2 lesion area growth related to tPA treatment within 1 hour following its administration in both young and elderly animals (P < 0.01) (Figure 2). T2 infarct volume analysis revealed larger T2 volumes at 24 hours in elderly animals exposed to tPA (P < 0.05) when compared with elderly saline and young tPA-treated animals. Similarly, tPA treatment increased T1 lesion area in both young (P < 0.05) and elderly rats (P < 0.001, repeated measures ANOVA) (Figure 2). There was no significant difference in T1 and T2 corrected lesion area, and T1 and T2 lesion area were larger than ADC at all immediate reperfusion time points for both pretreatment and posttreatment groups. There was no difference in the extent of the ADC lesion size pretreatment or posttreatment for either treatment or age.
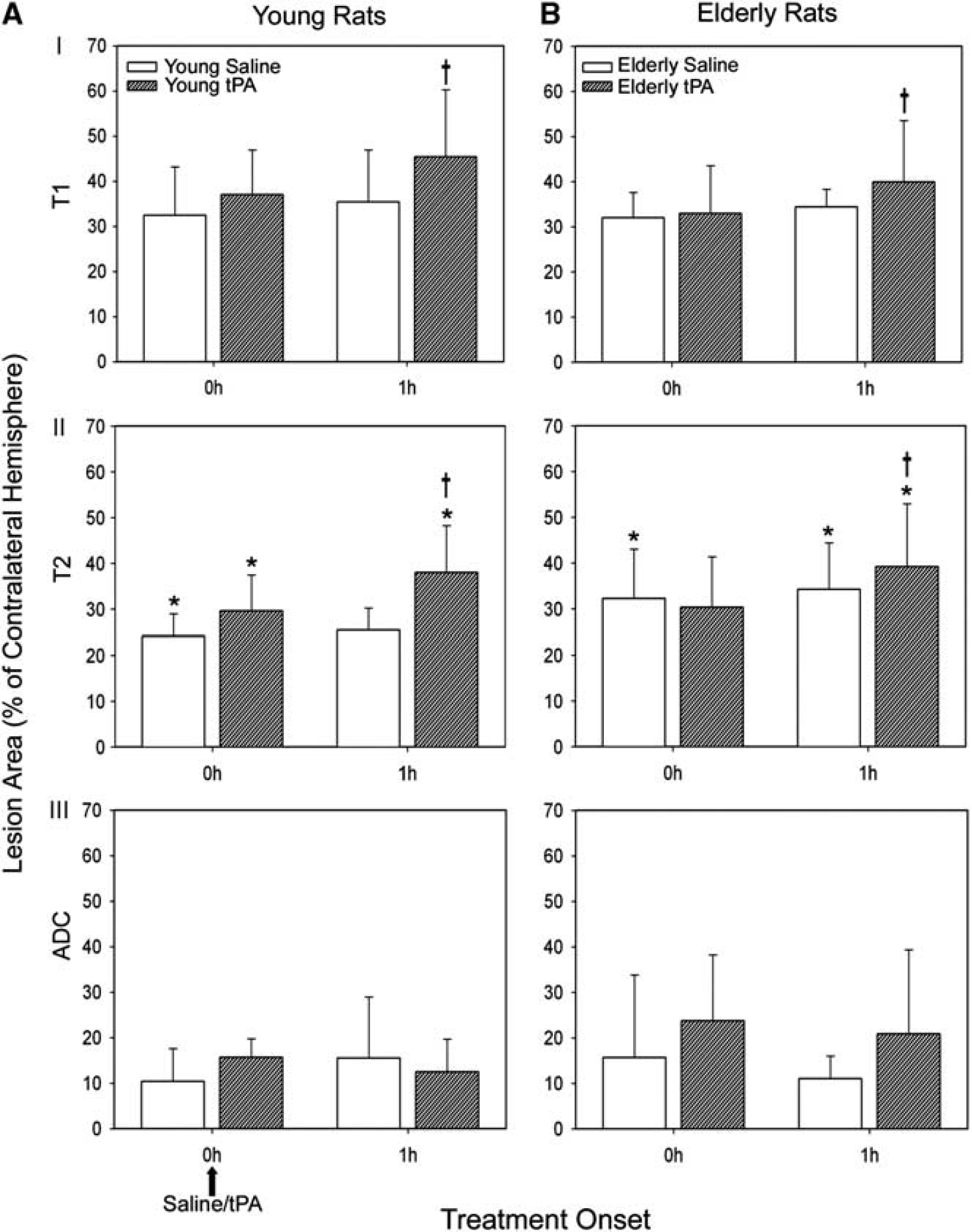
Tissue plasminogen activator (tPA) induces an early increase in T1 and T2 lesion area 1 hour after treatment in young (
Magnetic resonance imaging blood-brain barrier permeability measures (KGd): tissue plasminogen activator treatment effect: Blood-brain barrier permeability detected using Gd-DTPA extravasation (Figure 3A) and measured as KGd was observed to be increased immediately following ischemia reperfusion in the ipsilateral ischemic hemisphere compared with the contralateral hemisphere in all animal groups (P < 0.001) and at 24 hours (P < 0.001) (Figure 3B). KGd was independently enhanced by treatment with tPA in elderly animals (P < 0.001). In the young animals treated with tPA, a trend toward such an increase was observed (P < 0.069). There was also a trend toward an independent effect of increased age (P = 0.098), and an interaction between the elderly animals and treatment (P = 0.069), consistent with and supported by changes in other measures of BBB permeability such as IgG and paracellular tight junction proteins (see below). There was a high correlation between BBB permeability and T2 values (Figure 3C).
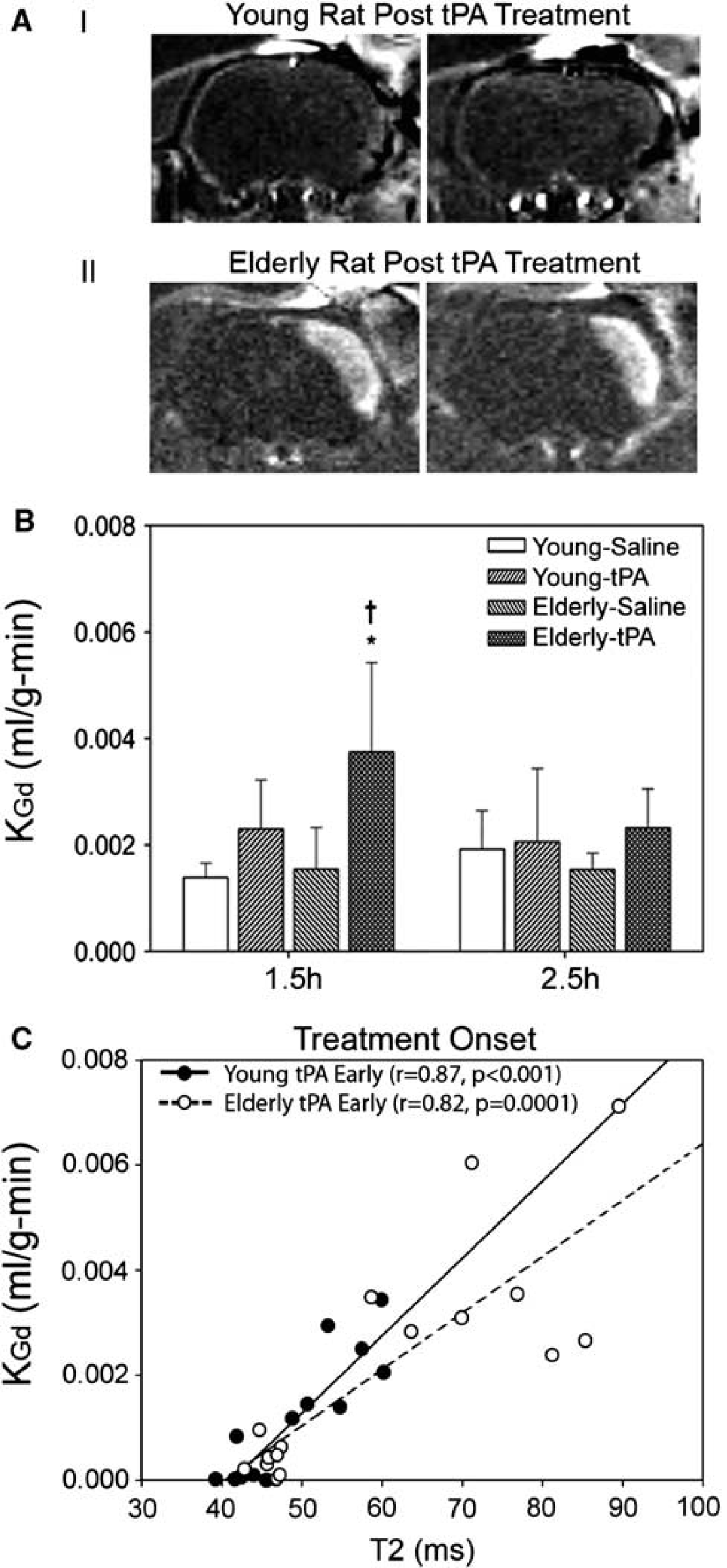
Tissue plasminogen activator (tPA) induces an early increase in blood to brain permeability (KGd). (
Histological assessment of vascular function: Histological measures of BBB breakdown for IgG revealed reduced gray levels of early IgG protein extravasation in the ischemic hemisphere irrespective of animal age or treatment group (P < 0.001) (Figure 4). However, greater early IgG protein extravasation was observed in both elderly saline and tPA-treated rats as compared with young animals (P < 0.05). At 24 hours, tPA treatment led to an exacerbation in IgG extravasation in both young and elderly rats (P < 0.01).
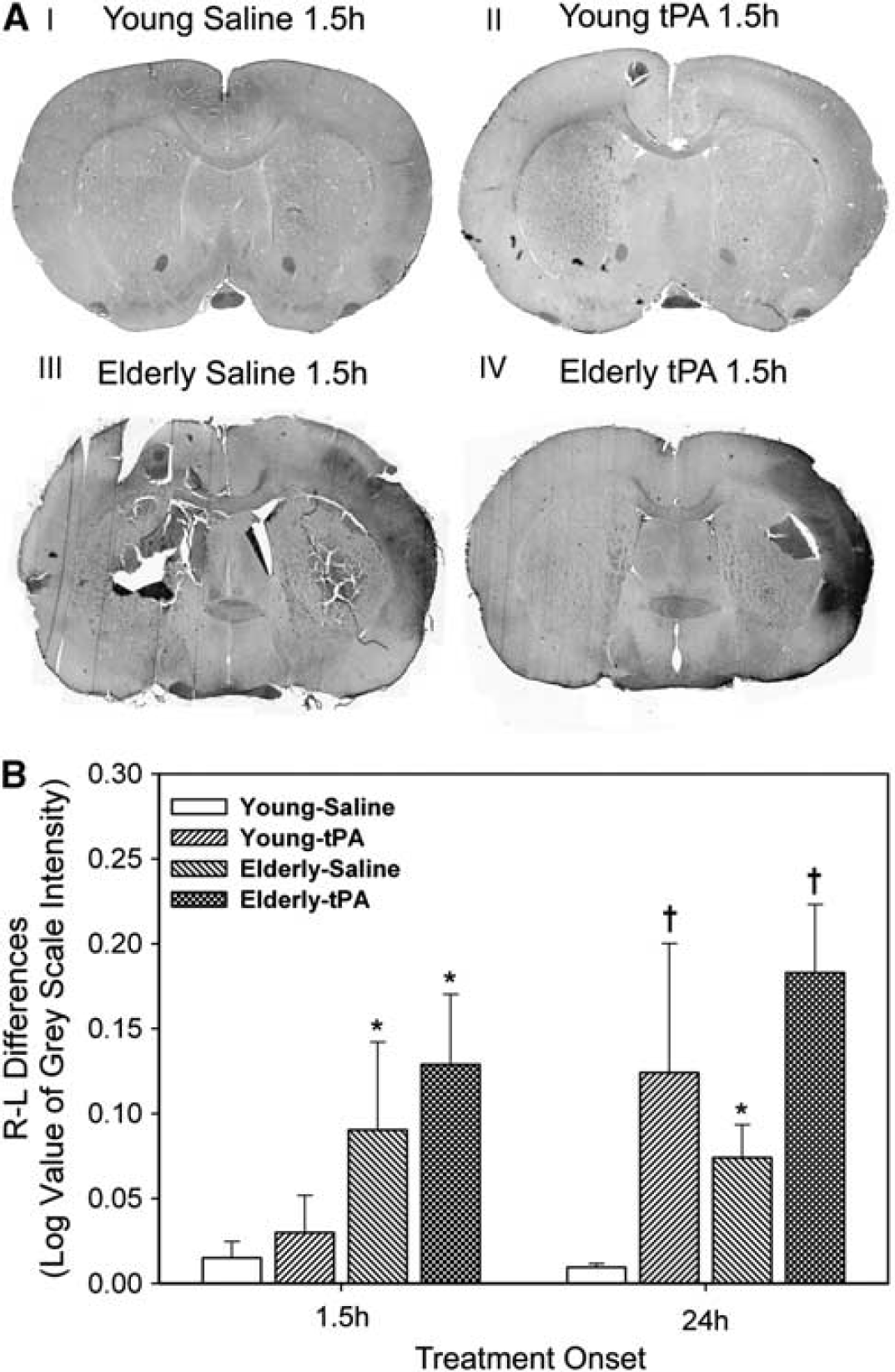
The ischemic elderly brain is associated with early IgG blood-brain extravasation. (
Tissue plasminogen activator administration resulted in greater incidence of hematoma formation in the elderly ischemic brain than the young following MCAO (P < 0.05, Fisher's exact test) (Figures 5A and 5C). Petechial hemorrhage and hematoma frequency was observed in hematoxylin and eosinstained sections following tPA treatment (Figure 5A). Overall, 83% of the tPA-treated elderly animals had evidence of hemorrhagic transformation as opposed to 36% in the saline group (Fisher's exact test, P < 0.05). The incidence of hemorrhagic transformation in young animals treated with tPA was 57% and in controls 50% (Fisher's exact test, P = 0.063). Although, elderly rats treated with tPA had proportionally a greater incidence of hemorrhagic transformation than young rats receiving the same treatment, no statistical difference was observed (P = 0.23). Increased T2 was observed before the formation of tPA hemorrhage (Figure 5B).
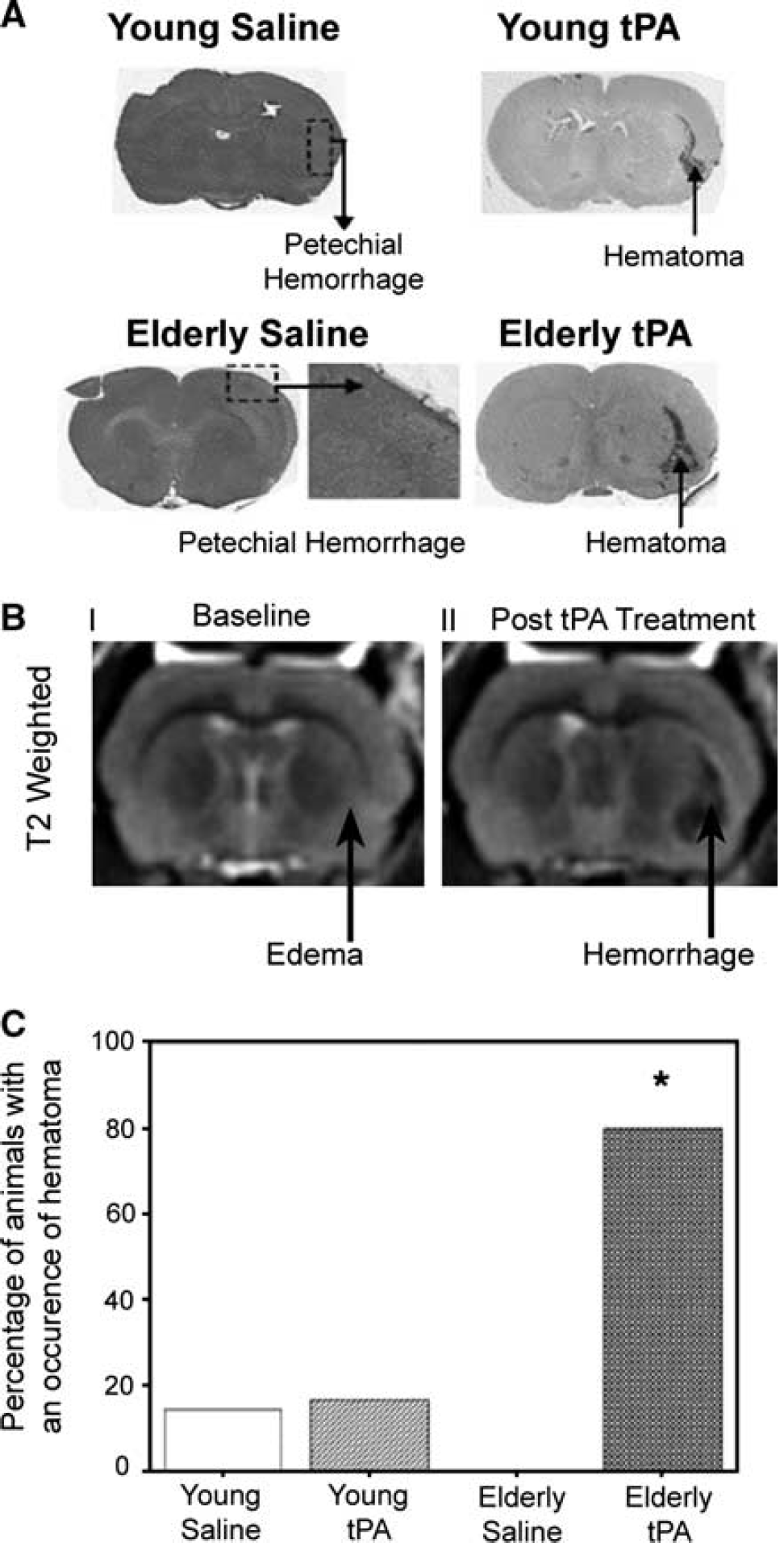
(
Measurement of tight junction proteins occludin and claudin 5: Western blots for claudin 5 protein estimation revealed monomer 19kDa and phosphorylated 25 kDa isoforms in young and elderly rats treated with saline or tPA. Ischemia and tPA induced phosporylation in elderly rats as indicated by an increase in the 25-kDa isoform (Figures 6A and 6B) (McCaffrey et al, 2008). No statistically significant induction of claudin 5 phosporylation was observed in the young rats in either treatment group. Protein quantification for occludin 4 revealed the monomeric isoform (66 kDa) is degraded into lower molecular weight isoforms in elderly rats. There were significant increases in phosphorylated isoforms (24 kDa) of occludin 4 independently related to ischemia, age, and tPA (Figures 6C and 6D).
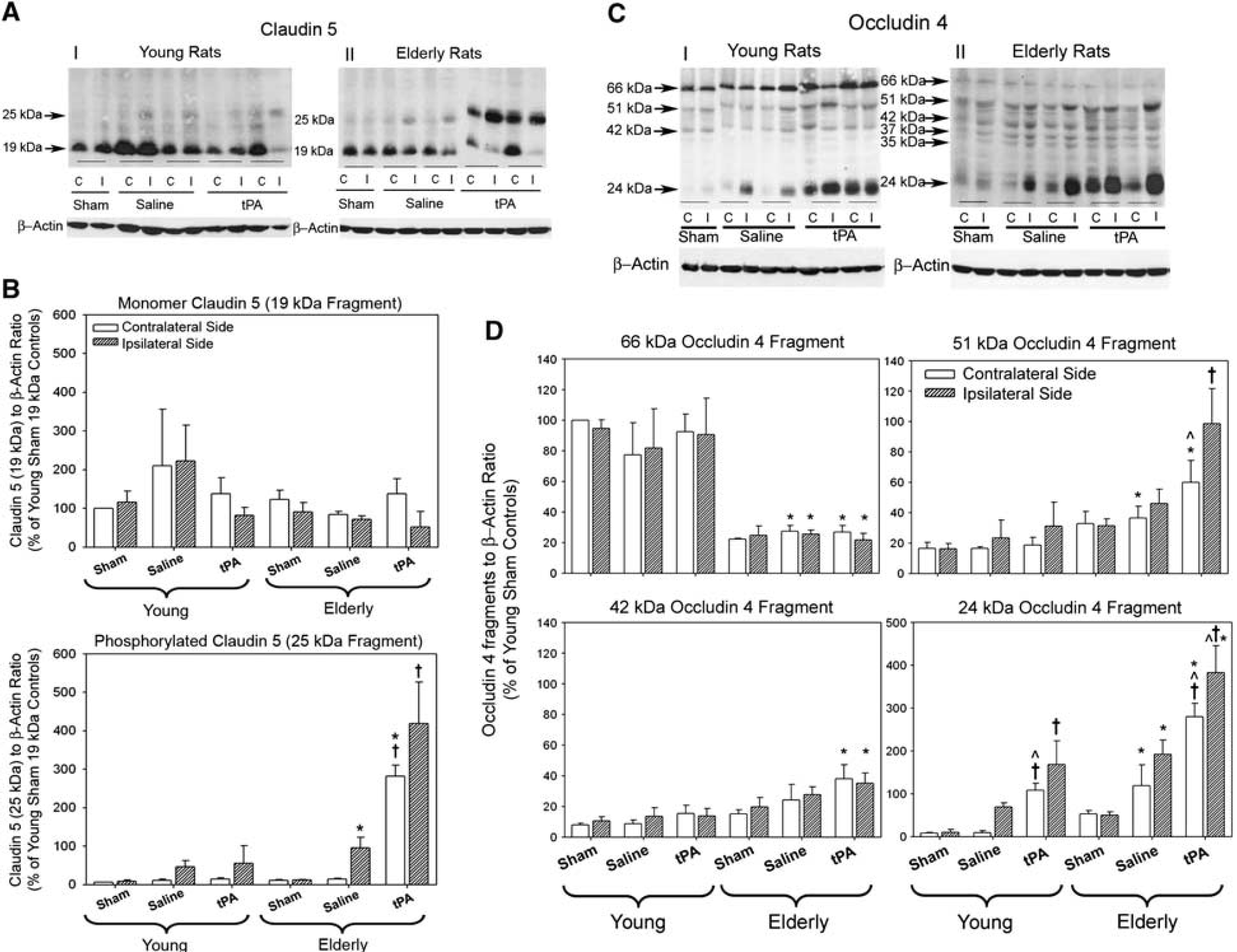
(
Discussion
The current observations have several novel implications. Our results extend our knowledge regarding the effects of ischemia reperfusion in the elderly brain and demonstrate the additive adverse effects of tPA on BBB of the ischemic elderly brain during reperfusion that, to our knowledge, have not been previously documented. Generally, the factor of aging is neglected in animal models of ischemia, despite the substantial clinical problem in an increasing aging society. The importance of performing MRI relaxometry measurements in cerebral ischemia can provide novel insights into the effects of ischemia reperfusion and have the potential to provide practical and clinically relevant surrogates that could be applied to investigate reperfusion-related injury and potential therapies. The permeability of the BBB appears both dynamic and responsive to tPA administration and several lines of evidence implicate enhanced paracellular diffusion of water and protein. Finally, the data support further investigation of related mechanisms and antagonism of occludin and claudin 5 phosphorylation to ameliorate the early BBB permeability and the effects on hemorrhagic transformation following tPA.
Enhanced paracellular permeability reflecting the early increase in BBB permeability is supported by changes in the tight junction phosphorylation status of both claudin 5 and occludin, which were both independently modified by age, tPA, and ischemia. Both claudin 5 and occludin are critical for tight junction BBB function and paracellular diffusion (Furuse et al, 1998; Hawkins and Davis, 2005), and their intricate arrangement of oligomeric transmembrane and accessory proteins rapidly alters in response to external stressors to produce changes in BBB permeability. The observed increase in protein phosphorylation of both claudin 5 and occludin in the ischemic elderly brain corresponds with the marked increase in BBB permeability to IgG observed in the elderly stroke animals and following tPA. There are many factors contributing to BBB disruption in ischemia, including generation of oxygen radicals, nitric oxide, production of vascular endothelial growth factor, and changes in intracellular calcium (del Zoppo, 1994; Brown and Davis, 2005).
The early changes in BBB permeability we have observed and the modification of this increased response in permeability by age during cerebral ischemia and tPA administration implies a synergistic biological response between age and tPA on early paracellular permeability and tight junction structure. Exactly which mechanisms are associated with altered occludin and claudin 5 phosphorylation seen in our analysis related to tPA and the elderly ischemic brain require future clarification.
The other important feature of the BBB damage following ischemia is its regional variability during the acute phase (Figure 3). This observation has been confirmed by several studies that report regional variability of BBB permeability (Preston and Webster, 2002; Nagaraja et al, 2008). A more formal and explorative assessment of claudin and occludin with respect to tight junction disassembly and blood-brain permeability to the temporal relationship of their disassembly is perhaps limited by the severity of stroke in our model as all animal groups had regions of IgG extravasation. It has been shown that in claudin 5 deficiency, the BBB is leaky to substrates of relatively small molecular size (< 800 Da) (Nitta et al, 2003). In our study, we demonstrate permeability to both Gd-DTPA (600 Da) as well as IgG, which has a much greater molecular weight (180 kDa). Therefore, it would be challenging in the context of this study to decipher the course of events with respect to claudin 5 and occludin phosphorylation. Previous studies linking decreased occludin expression to albumin leakage (68 kDa) would provide credence to the hypothesis (Antonetti et al, 1998) that the severity of BBB permeability follows a sequential course initially involving claudin 5 disassembly when leakiness is mild, but during severe breakdown occludin disassembly predominates.
The MRI contrast characteristics of normal diseased tissue are dependent on their relaxation time constants, although T2 and T1 changes obtained commonly from weighted sequences clinically, are largely qualitative in nature. A previous study observed subjective T1W and T2W intensity increases, Gd-DTPA enhancement and regions of IgG extravasation in a transient transorbital model of 4 hours of MCAO in the cat (Lo et al, 1994). Animal studies have indicated that in comparison to T2-weighted images, more subtle T2 increases may be observed much earlier when using quantitative T2 imaging (Hoehn Berlage et al, 1995; Kaur et al, 2009). Our data would support that the quantified T2 changes are a cumulative change in water content and protein in the ischemic tissue associated with significant BBB breakdown. The relation between age of rodent and infarct size has been inconclusive; some studies in rats found that cerebral infarction in aged rats was no different from young animals (Sutherland et al, 1996; Rosen et al, 2005), while others have documented increased infarct size in aged animals (Macri et al, 2006). We observed only increased infarct size in elderly rodents that were administered tPA when compared with their age-matched saline controls, but the survival period of 24 hours was relatively short. We also acknowledge that 90 minutes of MCAO may have resulted in a severe injury that made discrimination between treatment groups difficult. In this study, ADC values after reperfusion were not significant between groups, principally because the ADC values normalized to a variable degree between animals. This can be explained either by a metabolic recovery of the tissue or by an increase in extracellular fluid.
Contrast-enhanced MRI can be used to localize and quantitate the increase in microvascular permeability at the level of the BBB. Previous work by Knight et al (2009) has confirmed the utility and validity of quantitative permeability-related parameters consisting of the transfer constant (KGd) of Gd-DTPA and may be of value for detecting ischemic brain tissue destined for hemorrhagic transformation. Our observations with quantitative contrast-enhanced MRI confirms the increased BBB breakdown in the reperfusion phase with this technique but extends beyond this by revealing enhanced permeability of the BBB with tPA in the elderly animals. Our results also reveal a strong correlation between the transfer constant (KGd) and quantitated T2.
Although KGd provides measures of BBB permeability, the use of quantitative T2 relaxometry measurements may be preferred over quantitative contrast-enhanced MRI for determining BBB dysfunction. First, the image acquisition time required for contrast-enhanced MRI determination of KGd is rather lengthy, as MRI scans are required before and for minutes following the administration of the contrast agent. Second, contrast-enhanced MRI is invasive and therefore carries the risk of contrast reaction. Quantitative T2 relaxometry appears to provide a cumulative measure of the permeability of the BBB to water and proteins over time and is a direct correlate of IgG extravasation that we demonstrated histologically, but is also dynamic to the effects of tPA administration exacerbating vasogenic edema.
Some of the limitations of the experimental design used to investigate the role of tPA and age on BBB disruption following ischemia should be mentioned. The dose of tPA is considered appropriate for the rat but relatively high to that used clinically because Korninger and Collen (1981) showed in vitro that the rat's fibrinolytic system is 10-fold less sensitive to tPA than the human system. Thus, the majority of stroke studies in rodents have been performed with 10 mg/kg tPA instead of 0.9 mg/kg tPA and it is possible that the higher dose could have relatively greater toxic effects on the endothelium than doses used clinically. However, one recent in vivo study did compare these two doses in rats, with the higher dose producing more rapid reperfusion without increasing stroke volume or brain edema (Haelewyn et al, 2010). In addition, the intention of our study design was to observe the effects of treatment on the successfully reperfused brain but to determine the effects of tPA on the parenchyma it was necessary to obtain imaging data before its administration. This required a time difference of ∼30 minutes between reperfusion and the administration of the drug or saline during which time pretreatment MR images were acquired. This replicates fairly closely rapid reperfusion with tPA treatment, although the response of the endothelium to tPA may be very different when the artery is occluded. Note that our experimental design is also highly relevant to the situation where there is spontaneous reperfusion before delivery of the treatment. Another limitation is the severity of stroke induced by the MCAO model, which produced severe cortical injury. Two other studies determined regional variability of BBB damage, but again the degree of ischemia was likely severe in both studies (Jiang et al, 2005; Knight et al, 2005). The question remains as whether quantitative MRI can determine BBB leakage in less severe stroke.
To conclude, thrombolytic therapy for acute ischemic stroke remains underutilized despite convincing evidence as to its benefit (Barber et al, 2001). The use of quantitative MRI might provide a measure linking BBB permeability and stroke severity (by measuring qT2), and therefore, reliably predicting both the response to tPA and risk of hemorrhagic transformation, to allow treatment decisions to be stratified according to the ‘tissue properties’ of cerebral ischemia rather than by arbitrary time windows (Baron et al, 1995). The concept of selecting patients most likely to benefit from thrombolysis independent of the time of stroke and at the same time determining the risk of adverse events using imaging is not new (Baron et al, 1995); but, advances in quantitated MRI may allow such a hypothesis to be tested clinically. Our current results indicate that ischemia reperfusion in the elderly rat brain, treated with tPA administration, are associated with dramatic increases in quantitated T2 and BBB permeability and that the early increase in BBB permeability involves enhanced endothelial paracellular transport evident in the corresponding changes in occludin and claudin 5 proteins. However, the mechanism of the early disassembly of tight junction proteins is poorly understood with respect to stroke, age, and tPA, and requires further investigation to mitigate the process in an attempt to improve safety and efficacy of the current treatment.
Footnotes
Acknowledgements
The authors acknowledge the expert technical contributions of Tadeuz Foniok, David Kirk, Jodie Petersen, and Limei Wang.
Dr Philip A Barber receives salary support from the Alberta Heritage Foundations of Medical Research.
