Abstract
Astrocytes undergo disease-specific transcriptomic changes upon brain injury. However, phenotypic changes of astrocytes and their functions remain unclear after hemorrhagic stroke. Here we reported hemorrhagic stroke induced a group of inflammatory reactive astrocytes with high expression of Gfap and Vimentin, as well as inflammation-related genes lipocalin-2 (Lcn2), Complement component 3 (C3), and Serpina3n. In addition, we demonstrated that depletion of microglia but not macrophages inhibited the expression of inflammation-related genes in inflammatory reactive astrocytes. RNA sequencing showed that blood-brain barrier (BBB) disruption-related gene matrix metalloproteinase-3 (MMP3) was highly upregulated in inflammatory reactive astrocytes. Pharmacological inhibition of MMP3 in astrocytes or specific deletion of astrocytic MMP3 reduced BBB disruption and improved neurological outcomes of hemorrhagic stroke mice. Our study demonstrated that hemorrhagic stroke induced a group of inflammatory reactive astrocytes that were actively involved in disrupting BBB through MMP3, highlighting a specific group of inflammatory reactive astrocytes as a critical driver for BBB disruption in neurological diseases.
Keywords
Introduction
The blood-brain barrier (BBB) is a biological and functional barrier in the central nervous system (CNS), which plays a fundamental role in maintaining normal brain function. 1 Aging and neurological diseases such as stroke, brain trauma, Alzheimer’s disease, etc. cause BBB breakdown, subsequently leading to the influx of cytotoxic substances, neuroinflammation, and long-term permanent neurological impairment.2–4 Accumulating evidence suggests that preserving the integrity of BBB after CNS injury is beneficial for improving neurological function.5,6 Thus, understanding the mechanism involved in BBB disruption is critical for developing novel therapies to treat different neurological disorders.
As a key component of BBB, astrocytes have been demonstrated to play essential roles in the formation and maintenance of BBB. 7 With their endfeet wrapping around cerebral blood vessels, astrocytes regulate BBB integrity and function by crosstalk with endothelial cells and pericytes. 1 Astrocytes are activated after CNS insults, referred to as reactive astrocytes. 8 However, the function of reactive astrocytes on BBB integrity after CNS injury is still in debate. Growing evidence has suggested a critical role of reactive astrocytes in neurological diseases, such as influencing BBB integrity. Astrocytes have been shown to play either a detrimental or protective role after brain injury. 9 For example, reactive astrocyte-derived vascular endothelial growth factor-A (VEGF-A) enhanced BBB permeability by decreasing tight junction-related proteins following brain injury, and selective inhibition of VEGF-A signaling protects against BBB disruption.10,11 By contrast, Alvarez et al. showed that astrocyte-secreted sonic hedgehog promoted BBB integrity by acting on hedgehog receptors in endothelial cells in the animal model of multiple sclerosis. 12 One of the main reasons for such debate was the heterogeneity of astrocytes in the brain and different subsets of astrocytes could have distinct effects on the function of BBB.
With the development of single-cell RNA sequencing (scRNA-seq), scientists now realize that astrocytes underwent profound transcriptomic changes in response to CNS injury. Recent studies have shown that systemic inflammation induced multiple heterogeneous astrocyte subsets in the brain, and different subsets of astrocytes have distinct functions.13,14 Besides, single-nucleus RNA sequencing (snRNA-seq) reveals a population of disease-associated astrocytes (DAAs) in an Alzheimer’s disease mouse model. 13 The DAA-like population appears in the early stage and increases with disease progression. Similar astrocytes were also detected in normal aging, yet their impact on pathology and lifespan remains unclear. In malignant brain tumors, astrocytes undergo transcriptomic re-programming, and a distinct astrocytic phenotype caused by the coexistence of microglia and astrocytes in the tumor environment was observed. These tumor-associated astrocytes contribute to anti-inflammatory responses and induce an immunosuppressive environment. 14 However, the heterogeneity of astrocytes and their function in regulating BBB integrity in response to hemorrhagic stroke have not been investigated so far.
In the current study, we first performed scRNA-seq to fully characterize the transcriptomic changes of astrocytes in the brain after hemorrhagic stroke. We identified a group of inflammatory reactive astrocytes marked by Gfap and Vimentin (Vim) as well as inflammation-related genes lipocalin-2 (Lcn2), Serpina3n, and Complement component 3 (C3). The expressions of Lcn2, Serpina3n, and C3 in astrocytes were induced by microglia rather than infiltrated macrophages. We further determined the inflammatory reactive astrocytes mediated BBB breakdown via MMP3 in hemorrhagic stroke mice.
Materials and methods
Animals
Animal protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of Shanghai Jiao Tong University, Shanghai, China. Animal studies were performed in accordance with the Animal Research: Reporting of in vivo Experiments (ARRIVE) guidelines. All animal experiments were conducted in accordance with Bioethics/2012022, Shanghai Jiao Tong University. A total of 48 (20–25 g, 8–10 weeks) male C57BL/6 mice were purchased from Vital River Laboratories (Vital River Laboratories, Beijing, China) and 112 astrocyte-specific MMP3 knockout mice (Aldh1l1Cre-ERT 2 ; MMP3flox/flox, 20–25 g, 8–10 weeks) were used in this study. Age- and sex-matched astrocyte-specific MMP3 knockout mice were used in this study. If the sex of the transgenic mice was not specifically elucidated, groups were sex-balanced. All mice were housed under standard 12 h light/dark cycle conditions with free access to food and water. Ambient humidity was 50∼60% and room temperature (RT) was maintained at 18∼22°C.
Generation of loxp floxed MMP3 mice
Briefly, two guide RNA molecules (gRNAs) targeting sites upstream and downstream of the exon(s) were co-injected with appropriate Cas9 into the cytoplasm of fertilized C57BL/6 mouse eggs, followed by implantation of the eggs into surrogate mothers to obtain offspring, knockout pups with MMP3 region deletion could be generated (Cyagen Biosciences, Suzhou, China). The following primers were used for genotyping:
forward: 5′-CCTTGCTTTGGTCCAGTGTAATTT-3′, reverse: 5′-GTGTCTCTGTAGTTTTAGCTTGCC-3′.
To specifically knockout MMP3 in astrocytes, loxp floxed MMP3 mice were crossed with Aldh1l1Cre-ERT2 mice (gift from Dr. Won-Suk Chung). Tamoxifen (Sigma-Aldrich, MO, USA) was prepared in corn oil and allowed to be dissolved at 37°C while shaking. Mice were injected intraperitoneally (0.075 mg/g body weight) once a day for five consecutive days at 2 months of age, and used for experiments at least 1 week after the last dose of tamoxifen.
Collagenase-induced intracerebral hemorrhage
Mice were anesthetized with 1.5% isoflurane in a 30% O2/70% NO mixture by anesthesia machine (RWD Life Science Co., Shenzhen, China) and secured in a stereotaxic Instruments (RWD Life Science Co.). The depth of anesthesia was monitored continuously and adjusted when necessary. The scalp of mice was sterilized with 75% alcohol and then cut by scissors to expose the skull. A 29-gauge needle loaded with collagenase IV dissolved in PBS was inserted into the right striatum (mediolateral, 2.0 mm from bregma; anteroposterior, 0.2 mm; dorsoventral, 3.0 mm). Each mouse was injected with 0.04 U collagenase IV dissolved in 2 μl PBS for 5 min using a micro-infusion pump (WPI, Sarasota, FL, USA). The needle was left in place for 5 min to avoid backflow. After the withdrawal of the needle, the scalp was sutured and sterilized with iodophor. The animals were allowed to regain consciousness on a 37°C heating pad (RWD Life Science Co.) after the operation.
Microglia and macrophage depletion
To pharmacologically ablate brain microglia, mice were given the colony stimulating factor 1 receptor (CSF1R) inhibitor PLX5622 formulated in AIN-76A standard chow (1.2 g PLX5622 per kg, Plexxikon). Respective control groups received a normal AIN-76A diet (Plexxikon). Mice were fed for at least 14 days prior to hemorrhagic stroke to ensure maximum microglia depletion, which was maintained until the mice were sacrificed.
To pharmacologically deplete peripheral macrophages, clodronate liposomes (F70101C-A, FormuMax Scientific, Inc., Palo Alto, CA) were intraperitoneally injected (7 mg/ml, 0.2 ml each mouse) 1 day before hemorrhagic stroke to ensure the depletion of 90% of macrophages within 24 to 36 h.
IgG staining
Immunoglobulin G (IgG) leakage was examined using the immunohistochemistry secondary antibody kit (Absin, Shanghai, China) according to protocols from suppliers. The IgG images were collected using bright-field microscopy (Leica, Solms, Germany). Integrated optical density (IOD) of IgG was quantified using Image J Software (NIH, Bethesda, MD, RRID: SCR_003070) immunohistochemistry image analysis toolbox. For each mouse, four sections with 200 μm interludes were evaluated and four fields at 20× per section were imaged in the hemorrhagic perifocal area. Data were analyzed using Prism Graphpad 9 (GraphPad Software, San Diego).
Gelatin zymography
Equal amounts of protein from each sample were loaded with zymogram sample buffer (Bio-Rad, Heracles, CA, USA) and electrophoresed in SDS-PAGE for 2.5 h. Then the gels were washed in renaturing buffer (2.5% Triton X-100, 50 mmol/L Tris-HCl, 5 mmol/L CaCl2, pH 7.5) for 15 min each time 4 times with agitation. Next, the gels were moved to the developing buffer (1% Triton X-100, 50 mmol/L Tris-HCl, 5 mmol/L CaCl2, pH 7.5) for 30 min with gentle agitation at RT. After that, the fresh developing buffer was replaced and incubated for 2 days at 37°C. The gels were stained with Coomassie blue (0.05% Coomassie Brilliant Blue, 30% methanol, 10% acetic acid) for 3 h with agitation at room temperature, and then incubated with destaining solution (40% methanol, 10% acetic acid) until bands could be seen. 15 The final bands were quantified using Image J software (NIH).
Real-Time PCR analysis
For mice, total RNA was extracted from the brain tissues in the hemorrhagic perifocal area of the striatum following hemorrhagic stroke using TRIzol (Invitrogen, Carlsbad, CA). The in vitro cultured cells were washed with PBS, then resuspend in TRIzol to extract total RNA. RNA quantity and concentration were assessed by spectrophotometer (NanoDrop 1000, Thermo Fisher) and cDNA was generated from 1 μg total RNA per sample using the Hifair II 1st Strand cDNA synthesis kit (Yeasen Biotech, China). Real-time quantitative PCR was conducted using a Hieff qPCR SYBR Green Master Mix (Yeasen Biotech, China) according to the supplier’s protocols and cycling program of 5 min at 95°C followed by 40 cycles of 95°C for 10 s and 60°C for 30 s on a fast real-time PCR system (7900 HT, ABI, Foster City, CA). Primer sequences for mice were listed in Table S1. Quantification was performed by using the comparative CT method (2−ΔΔCT) with all data normalized to GAPDH expression. 16
Evans blue assay
2% Evans blue (Sigma-Aldrich, 4 ml/kg) dissolved in saline was administered via the left jugular vein 3 days after hemorrhagic stroke. After 2 h of circulation, mice were anesthetized for cardiac perfusion. Both ipsilateral and contralateral hemispheres were weighed, and the samples were homogenized in 1 ml of 50% trichloroacetic acid solution followed by centrifuging at 12,000 g for 20 min to extract Evans blue. The supernatant was diluted with 100% ethanol at a ratio of 1:3. The concentration of Evans blue was quantified at 610 nm using a spectrophotometer (Bio-Tek, Winooski, VT).
Transmission electron microscopy
Mice were anesthetized and perfused with ice-cold PBS followed by ice-cold fixation solution (2% PFA and 1% glutaraldehyde in 0.1 M PB). Brain tissues in the perifocal area of the hematoma were cut into 1 cubic millimeter and immediately immersed in 0.1 M PB containing 2.5% glutaraldehyde at 4°C for 12 h. Tissues were washed with PB, then treated with 1% OsO4 for 2 h, dehydrated with a graded series of ethanol (50%, 70%, 90%), and incubated with 100% acetone, a 1:1 mixture of epoxy resin and acetone, and 100% resin. Tissues were then embedded in epoxy resin and kept in a 60°C dryer over 3 days. Semithin sections (1 μm) were cut and stained with 0.1% toluidine blue for accurate localization under a light microscope. Ultrathin sections (100 nm) were cut and collected on 230 hex copper mesh grids, poststained for 6 min with 0.5% lead citrate, and examined under transmission electron microscopy (TEM).
Quantitative assessment of cerebral basement membranes
Capillaries were identified in the transverse section with clear and well-defined basement membrane (BM) structure and a representative image was taken at a magnification of ×9.3 K under TEM. The diameter of all selected capillaries was shorter than 8 micrometers. To quantify BM thickness, electron micrographs of capillaries were analyzed using Image J software (NIH). Ten equidistant perpendicular measurement bars were used to measure the distance between the inside and outside edges of BM and the average values were identified as BM thickness.
MMP3 activity assay
MMP3 activity in brain tissue was assessed using the MMP3 Activity Fluorimetric Assay Kit (ab118972, Abcam), following the manufacturer’s instructions. Data were defined as the amount of enzyme generating 1 μmol of fluorescent substrate per minute per milliliter of the sample at RT (nmol/min/ml).
The culture of primary microglia and astrocytes
Primary microglia and astrocytes were isolated from P0 C57BL/6 mice. Briefly, the cerebral cortices were gently dissected from the pup mice brains and washed with Dulbecco’s modified eagle medium (DMEM, HyClone, Logan, UT) 3 times. The cerebral cortices were enzymatically digested (using 0.25% trypsin) and then mechanically pipetted to generate a single-cell suspension. After being filtered with a 70 μm filter (Millipore), dissociated cortical cells were plated on T75 flasks, 6-well plates, and 24-well plates coated with poly-D-lysine (PDL, Sigma-Aldrich) in DMEM with 10% FBS (Gibco, Carlsbad, NM, USA) and 1% penicillin streptomycin antibiotics (HyClone) at a density of 1 × 107 cells/flask on T75 flasks, 7 × 105 cells/well on 6-well plates, and 2 × 105 cells/well on 24-well plates. The mixed glial cells were cultured in a humidified incubator at 37°C with 5% CO2 for 7–10 days and the culture medium was replaced every 3 days. The primary astrocytes were obtained by patting plates (to remove microglia and oligodendrocyte lineage cells) before changing the culture medium. When high confluency of the mixed glial cultures was observed, the primary microglia were isolated from the cultures by shaking at 180 rpm at 37°C for 30–40 min. The obtained primary microglia were seeded on 6-well plates at a density of 7 × 105 cells/well.
The medium from resting microglia was defined as resting astrocyte-conditioned medium (Resting-MCM). The conditioned medium from the primary microglia treated with LPS (LPS-MCM), hemin (Hemin-MCM), hemin and MMP3 inhibitor (100 nM, Tocris Bioscience, UK 356618) (Hemin-MCM+MMP3 inhibitor) were collected and applied to primary astrocytes for 24 h. We define the conditioned medium from these astrocytes as Resting-MCM-ACM, LPS-MCM-ACM, Hemin-MCM-ACM, and Hemin-MCM + MMP3 inhibitor-ACM.
bEnd.3 cells culture with different types of astrocytes conditioned medium
bEnd.3 cells were purchased from the Shanghai Institute of Biochemistry and Cell Biology (Shanghai, China) and grown in DMEM with 10% FBS (Gibco) and 1% penicillin streptomycin antibiotics (HyClone). After seeding cells at a density of 7 × 105 cells/well on 6-well plates, and 2 × 105 cells/well on 24-well plates for 2 days, bEnd.3 cells were cultured with different types of ACMs for 24 h to prepare for the subsequent experiments. The sources of ACM included Resting-MCM-ACM, LPS-MCM-ACM, Hemin-MCM-ACM, and Hemin-MCM+MMP3 inhibitor-ACM.
Data analysis and statistics
We determined the sample size for animal experiments by two-sided power analysis of the mean difference and standard deviation of the primary parameters (power =80%, a = 0.05). For in vivo experiments, the number of brain sections and animals used (n) is indicated in the Figure legends. At least 3 biologically independent cell cultures were performed in the in vitro study. All statistical analyses were done using GraphPad Prism 9 software. Data were all presented as mean ± standard deviation (SD). All data were subjected to Kolmogorov-Smirnov normality test before statistical analyses. For data exhibiting a normal distribution, we use two-tailed Student's test (equal variances) or Welch's t test (unequal variances) for comparison between two groups, and use one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparisons test (all groups compared with a control group, equal variances) or by Tukey’s multiple comparisons test (comparisons between all groups, equal variances) for more than two groups with one comparison of an independent variable. We used two-way ANOVA with Bonferroni's multiple comparison test for comparisons of more than two groups with two independent variables. We used the two-tailed Mann-Whitney test for non-normally distributed data comparisons. Two-tailed p-value < 0.05 was considered statistically significant.
Availability of data and materials
The scRNA-seq and RNA-seq data generated in this study have been uploaded to the Gene Expression Omunibus under the accession number PRJNA954751 and GSE228927, respectively. Raw images of Western blot are provided in the supplementary materials (Fig. S5).
Results
scRNA-seq revealed a group of reactive astrocytes in hemorrhagic stroke mice
To investigate transcriptomic changes of astrocytes in mice after hemorrhagic stroke, we profiled the transcriptome of 33,048 striatal cells in control and 3 days post hemorrhagic mice using 10× Genomics Chromium technology. Cells were partitioned into 18 clusters, and the identity of these cell clusters was determined by the expression of cell type-specific genes (Figure 1(a)). Single-cell populations with astrocyte-specific gene expression were segregated into three different clusters: cluster 0, cluster 1, and cluster 2 (Figure 1(b)). By comparing the dataset from control mice and hemorrhagic stroke mice, the number of cluster 2 astrocyte was highly increased in hemorrhagic stroke mice brain, and dot plot data demonstrated that cluster 2 astrocytes were enriched in reactive and inflammation-related genes including Gfap, Vim, Lcn2, Serpina3n, H2-D1, S100a10, and C1q, while cluster 0 and 1 showed minimal reactive and inflammation-related genes (Figure 1(c)).
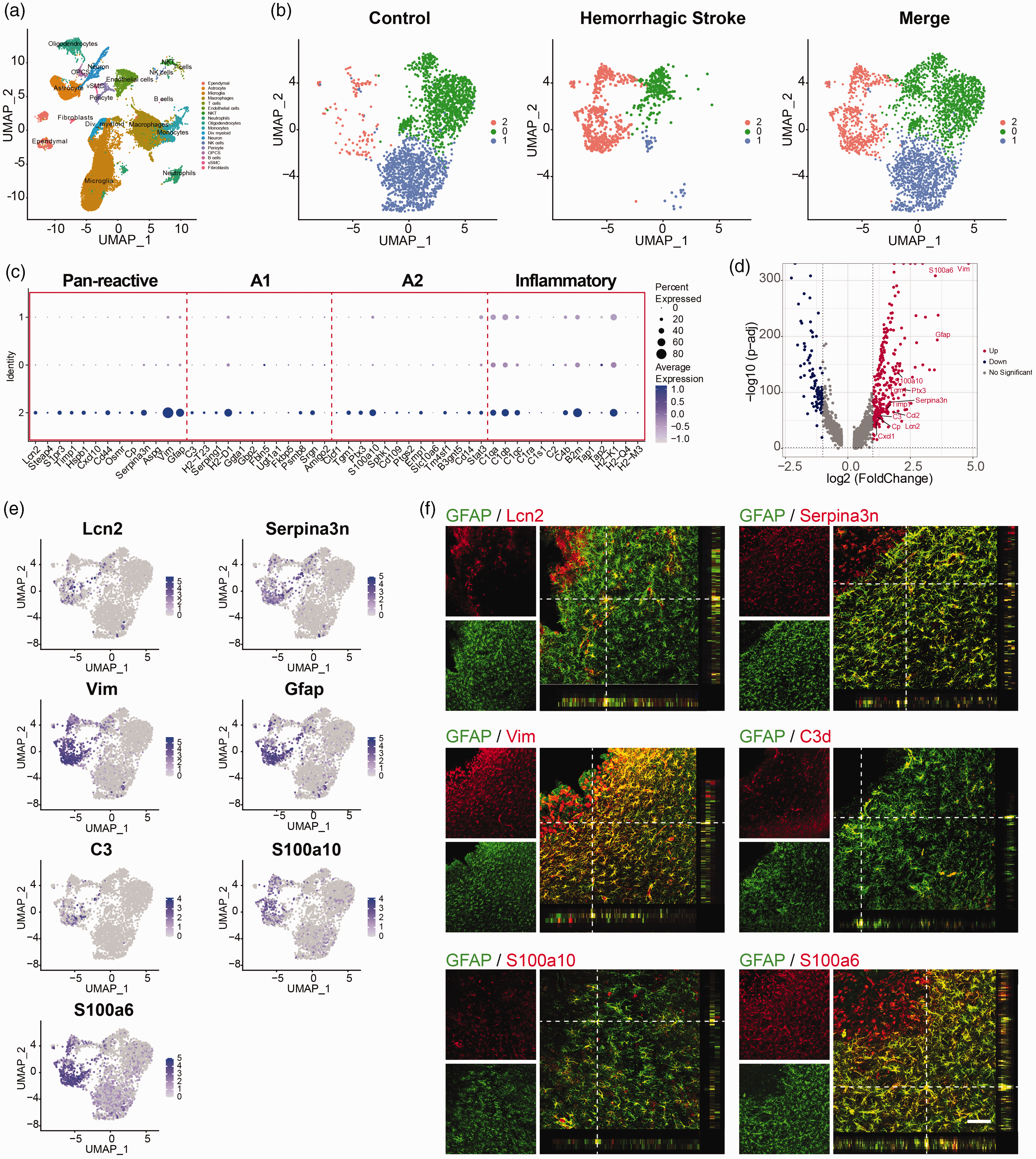
scRNA-seq reveals heterogeneity of astrocytes in the striatum of hemorrhagic stroke mice. (a) A UMAP plot of 33,048 single cells from the striatum in control (11,308) and 3 days post hemorrhagic (21,740) mice. Colors denote different cell clusters. n = 8 mice per group. (b) A UMAP plot of 3,262 astrocytes from the striatum in control and 3 days post hemorrhagic mice. Colors denote different astrocyte phenotypes. (c) Expression of reactive and inflammatory genes in astrocytes. (d) Volcano plots comparing DEGs among cluster 2 versus cluster 0 and 1. (e) Feature plot of Lcn2, Serpina3n, Vim, Gfap, C3, S100a10, S100a6 genes expressed in astrocytes and (f) Representative immunostaining images of astrocytes (GFAP, green) colocalized with Lcn2, Serpina3n, Vim, C3, S100a10, and S100a6 (red) in the peri-lesion area at 3 days after hemorrhagic stroke. Scale bars, 100 μm.
As shown in the volcano plot and feature graphs, classic Pan-reactive astrocyte markers (Lcn2, Serpina3n, Vim, and Gfap), A1 reactive astrocyte markers (C3), A2 reactive astrocyte markers (S100a10) and S100a6 were specifically highly expressed in cluster 2 astrocytes (Figure 1(d) and (e)). Next, we performed immunohistochemistry to validate the presence and location of these reactive astrocytes in mice 3 days after hemorrhagic stroke. We found that hemorrhagic stroke activated numerous astrocytes that expressed reactive astrocyte marker GFAP in the lesion area of the brain (Figure 1(f)). In addition, a part of GFAP+ reactive astrocytes also expressed Lcn2, Serpina3n, Vim, C3, S100a10, and S100a6 (Figure 1(f)), suggesting hemorrhagic stroke induced a group of reactive astrocytes expressed with inflammation-related genes.
Inflammation-related genes in reactive astrocytes were induced by residential microglia but not infiltrated macrophages
Previous studies have suggested that microglia is a key driver for mediating the phenotypic change of astrocytes in mice subjected to neuroinflammation.14,17,18 To explore the effects of residential microglia and infiltrated macrophages on phenotypic change of reactive astrocytes after hemorrhagic stroke, we established microglia and macrophage depletion mice models. PLX5622, a selective inhibitor of colony stimulating factor 1 receptor (CSF1R), was formulated into a standard AIN-76A rodent diet. To deplete microglia, mice were fed with a PLX5622-formulated diet for 14 days before hemorrhagic stroke and continued until the end of the experiments. Clodronate liposomes offer highly efficient in vivo macrophage depletion. To deplete peripheral macrophages during hemorrhagic stroke, the mice were subjected to clodronate liposome F70101C-A injection 1 day before hemorrhagic stroke.
Successful depletion of microglia and macrophages was verified at 3 days after hemorrhagic stroke based on P2RY12 (microglia biomarker) and F4/80 (macrophage biomarker) immunofluorescence staining (Fig. S1A, B). Less than 5% of microglia were found in mice fed with the PLX5622-formulated diet (Fig. S1C). About 85% of F4/80+ macrophages were depleted in the F70101C-A treated mice compared to the control mice (Fig. S1D). F70101C-A slightly reduced the number of microglia while PLX5622 did not affect the number of macrophages (Fig. S1C, D). Interestingly, the number of GFAP+ reactive astrocytes was not affected after F70101C-A or PLX5622 treatment (Fig. S1E).
We then examined the expression of inflammation-related genes on reactive astrocytes in the hemorrhagic perifocal area in microglia-depletion or macrophage-depletion models at 3 days after hemorrhagic stroke (Figure 2(a) to (f)). We found that GFAP+/C3d+ reactive astrocytes almost disappeared in the PLX5622 treated mice, while the number in the F70101C-A treated mice was similar compared to the control (Figure 2(d)). Similarly, the percentage of GFAP+/Lcn2+ and GFAP+/Serpina3n+ astrocytes was decreased after PLX5622 treatment, while no changes were found in the F70101C-A group compared to the vehicle group (Figure 2(a) and (b)). However, the number of GFAP+/Vim+, GFAP+/S100a10+, and GFAP+/S100a6+ reactive astrocytes was not affected by the deletion of microglia or macrophages (Figure 2(c), (e) and (f)). These results indicated that residential microglia rather than infiltrated macrophages contributed to inducing inflammatory reactive astrocytes (GFAP+/Vim+/Lcn2+/Serpina3n+/C3d+) in hemorrhagic stroke mice.

Inflammation-related genes in reactive astrocytes were induced by residential microglia but not infiltrated macrophages. (a–f) Representative immunostaining images of astrocytes (GFAP, green) colocalized with Lcn2 (a), Serpina3n (b), Vim (c), C3d (d), S100a10 (e), and S100a6 (f) (red) in the hemorrhagic perifocal area of mice treated with vehicle, PLX5622, F70101C-A, and PLX5622+F70101C-A at day 3 of hemorrhagic stroke. Scale bars, 100 μm. Percentage of Lcn2+/GFAP+ (a), Serpina3n+/GFAP+ (b), Vim+/GFAP+ (c), C3d+/GFAP+ (d), S100a10+/GFAP+ (e), and S100a6+/GFAP+ (f) cells in the hemorrhagic perifocal area at day 3 of hemorrhagic stroke. n = 6 mice per group. One-way ANOVA followed by Dunnett’s test. *p < 0.05, ***p < 0.001, compared with the Vehicle group. Data are mean ± SD.
Conditioned medium from LPS- or hemin-stimulated microglial induced inflammatory reactive astrocytes in vitro
To investigate if in vitro hemorrhagic stroke microenvironment could induce inflammatory reactive astrocytes, resting astrocytes were treated with conditioned medium from LPS-stimulated microglial (LPS-MCM) or hemin (the degradation product of hemoglobin)-stimulated microglial (Hemin-MCM), respectively. RNA sequencing (RNA-seq) of resting-MCM treated astrocytes, LPS-MCM treated astrocytes, and Hemin-MCM treated astrocytes showed that 3576 differentially expressed genes (fold change >2) in LPS-MCM treated astrocytes compared with the control group, of which 1421 genes were upregulated, and 2155 genes were downregulated (Figure 3(a)). We also found that 2631 genes exhibited differential expression in Hemin-MCM treated astrocytes compared with the control group, among which 1200 genes were upregulated, and 1431 genes were downregulated (Figure 3(d)).
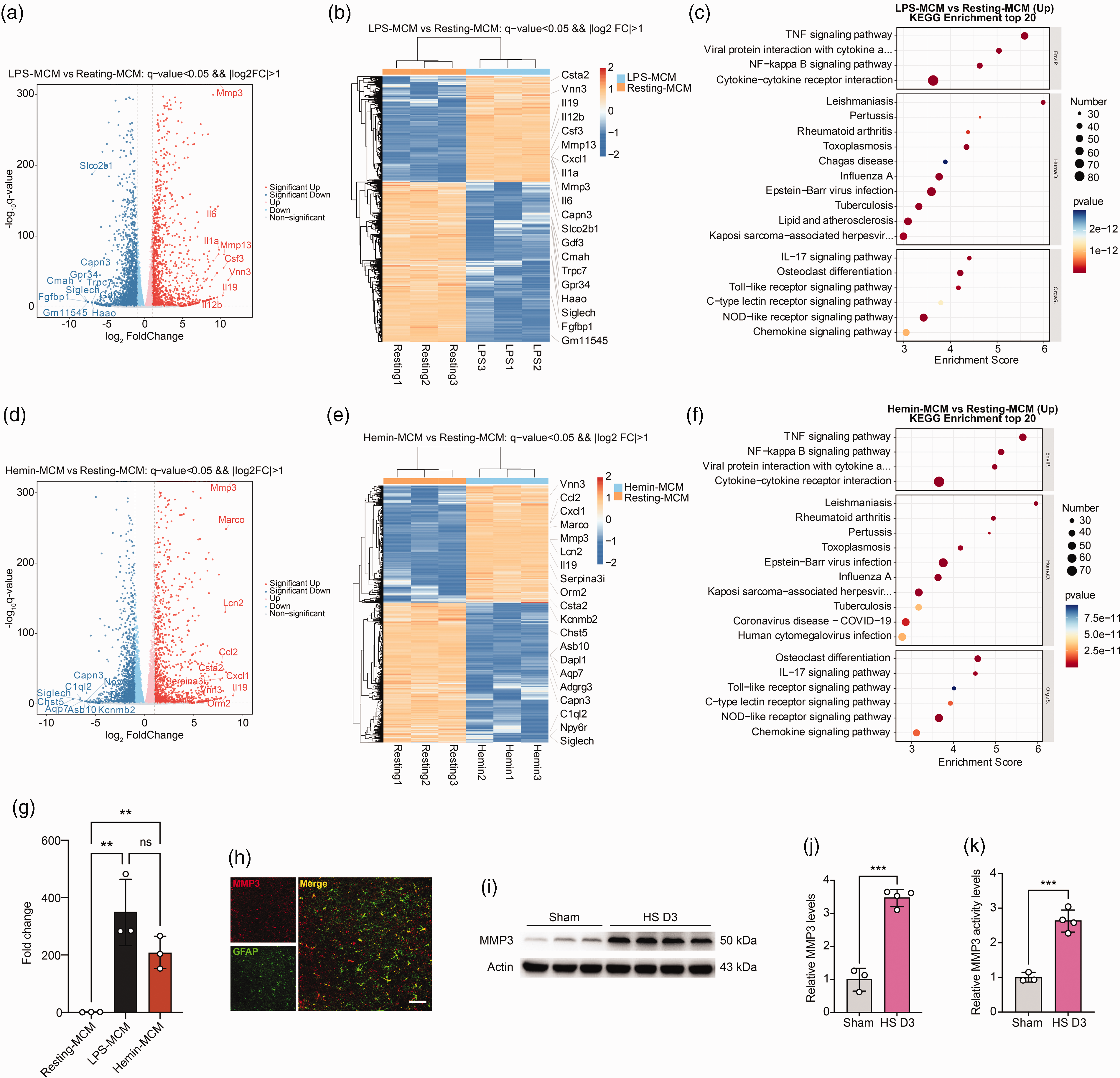
RNA-seq analysis of astrocytes treated with conditioned medium from LPS-stimulated microglia (LPS-MCM) or hemin-stimulated microglia (Hemin-MCM). (a) Volcano plot showed the upregulated (right) and downregulated (left) genes between astrocytes treated with LPS-MCM and astrocytes treated with medium derived from resting microglia (Resting-MCM). The horizontal axis is log2 fold change, and the vertical axis is -log10 p value, p < 0.05. (b) Heatmap showed the overall distribution of differentially expressed genes between LPS-MCM treated astrocytes and Resting-MCM treated astrocytes. Gene expression data was colored in orange for high expression and blue for low expression. (c) KEGG pathway enrichment analysis of differentially expressed genes in LPS-MCM treated astrocytes versus Resting-MCM treated astrocytes. (d) Volcano plot showed the upregulated (right) and downregulated (left) genes between Hemin-stimulated microglia-conditioned medium (Hemin-MCM) treated astrocytes and Resting-MCM treated astrocytes. (e) Heatmap showed the overall distribution of differentially expressed genes between Hemin-MCM treated astrocytes and Resting-MCM treated astrocytes. (f) KEGG pathway enrichment analysis of differentially expressed genes in Hemin-MCM treated astrocytes versus Resting-MCM treated astrocytes. (g) Real-time PCR analysis of MMP3 in Resting-MCM treated, LPS-MCM treated, and Hemin-MCM treated astrocytes. n = 3 biologically independent primary astrocytes cultures. GAPDH was used as an internal control. One-way ANOVA followed by Tukey’s test. **p < 0.01 compared with the Resting-MCM group. (h) Representative immunostaining images of MMP3 (red) and GFAP (green) in the hemorrhagic perifocal area at day 3 of hemorrhagic stroke. Scale bars, 50 μm. (i) Representative immunoblots of MMP3 in the hemorrhagic perifocal area at day 3 of hemorrhagic stroke. (j) Quantification of MMP3 protein levels. n = 3–4 mice per group. β-actin was used as an internal control. Two-sided, unpaired Student’s test. ***p < 0.001 and (k) Quantification of MMP3 activity in the hemorrhagic perifocal area at day 3 of hemorrhagic stroke. n = 3–4 mice per group. Two-sided, unpaired Student’s test. ***p < 0.001. All data are mean ± SD.
We identified increased expression of genes in LPS-MCM and Hemin-MCM treated astrocytes that were previously found in the reactive astrocytes in hemorrhagic stroke brain, such as Cxcl1, Lcn2, Ccl2, Cxcl10, Ptx3, Timp1, Tgm1, Cp, C3 and Serpina3n (Figure 1(e) and S2A). Real-time PCR also verified the results (Fig. S2B). KEGG analyses of the upregulated signaling pathways in LPS- or Hemin-MCM treated astrocytes identified the tumor necrosis factor (TNF) signaling pathway as the top enriched signaling pathway. Other signaling pathways, such as NF-kappa B signaling pathway and chemokine signaling pathway were also enriched in LPS- or Hemin-MCM treated astrocytes (Figure 3(c) and (f)). These data suggested LPS- or Hemin-MCM treated astrocytes and hemorrhagic stroke induced inflammatory reactive astrocytes shared similar reactive and inflammation gene signatures.
Notably, among all the upregulated genes in either LPS- or Hemin-MCM treated astrocytes, the matrix metalloproteinase-3 (MMP3) gene was remarkably upregulated and highly associated with BBB disruption (Figure 3(b) and (e)). Real-time PCR results also verified this upregulation (Figure 3(g)). Matrix metalloproteinases (MMPs) are a gene family of neutral proteases that play pivotal roles in normal development, wound healing, and various pathological processes of brain. 19 Many studies have shown that the mRNA level and the activity of MMPs are upregulated in the hemorrhagic brain.20–22 During hemorrhagic stroke, the activation of MMPs, mainly MMP3 and MMP9 was responsible for BBB breakdown, edema formation, hematoma expansion, and neuron loss through digesting the principal components of the basal lamina (collagen type IV, laminin, and fibronectin) and degrading tight junction proteins such as ZO-1 and Claudin-5.19,20,23–25 Thus, we hypothesize that hemorrhagic stroke-induced inflammatory reactive astrocytes disrupt BBB via MMP3. Our Western blot results showed the increased MMP3 protein level after hemorrhagic stroke in mice brains (Figure 3(i) and (j)). In consistency with the Western blot data, the activity of MMP3 also increased after hemorrhagic stroke (Figure 3(k)). To determine the cellular source of MMP3 after hemorrhagic stroke, MMP3/GFAP, MMP3/Iba-1, MMP3/CD31, and MMP3/NeuN staining were conducted. Immunofluorescence staining results showed that expression of MMP3 was mainly colocalized with reactive astrocytes and microglia in the hemorrhagic perifocal area, but not in endothelial cells and neurons (Figure 3(h) and S3A).
Inflammatory reactive astrocytes reduced endothelial tight junction protein expression in vitro
To investigate the effect of inflammatory reactive astrocytes on BBB integrity, mouse brain capillary endothelial cells (bEnd.3) were treated with medium from astrocytes that were treated with resting microglia medium (Resting-MCM-ACM), LPS-MCM (LPS-MCM-ACM) or Hemin-MCM (Hemin-MCM-ACM) for 24 h (Figure 4(a)), immunofluorescence staining and Western blot results showed that LPS-MCM-ACM treatment and Hemin-MCM-ACM treatment reduced the levels of endothelial cell tight junction-related proteins ZO-1 and Claudin-5 (Figure 4(b) to (d)).
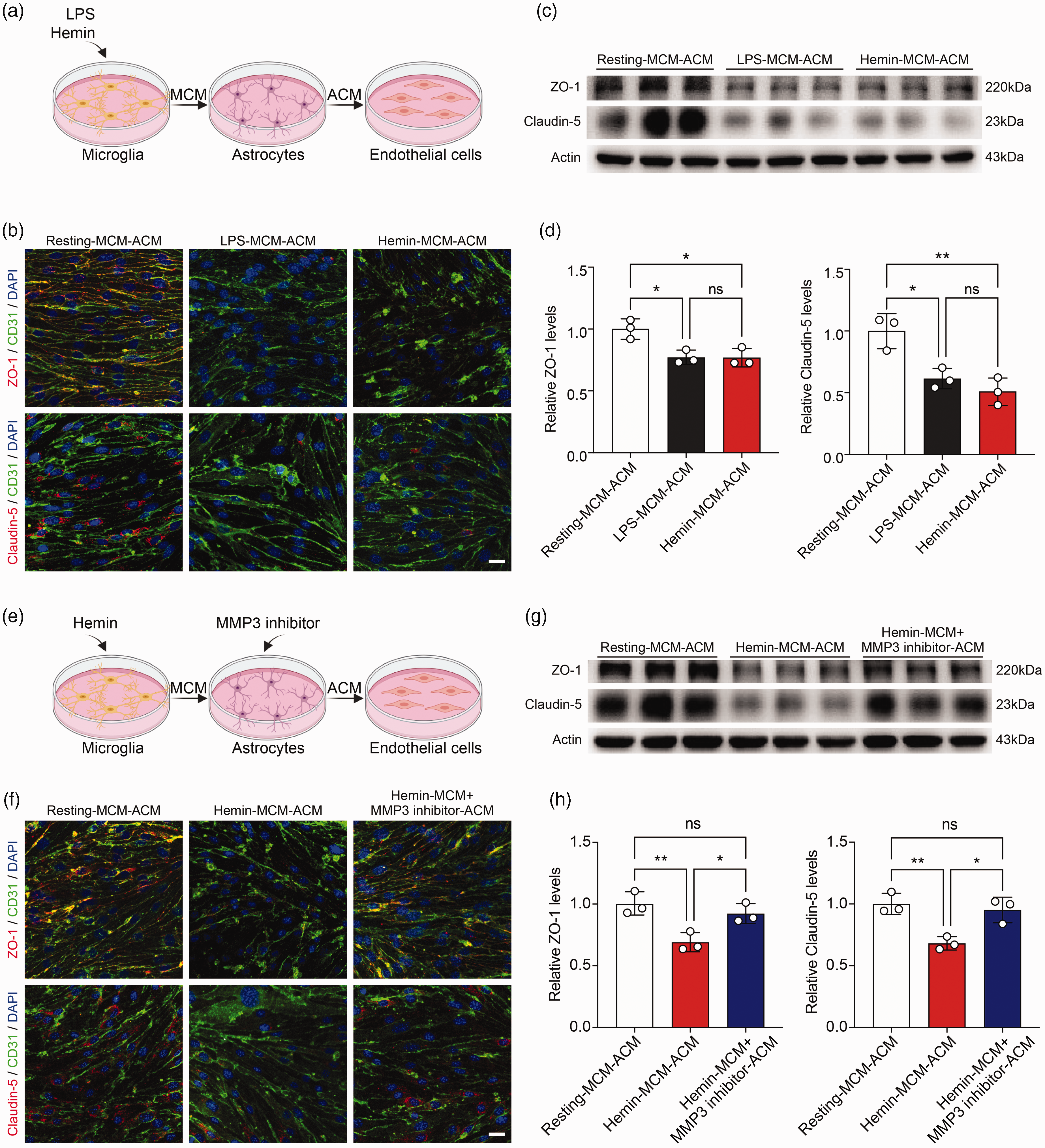
Conditioned medium derived from LPS-MCM or Hemin-MCM treated astrocytes reduced endothelial tight junctions, which reversed by inhibition of MMP3 in vitro. (a) Schematic diagram showed endothelial cells were treated with medium derived from astrocytes that were treated with LPS-MCM (LPS-MCM-ACM) or Hemin-MCM (Hemin-MCM-ACM) for 24 h. (b) Representative immunostaining images showed tight junction proteins ZO-1 (red) and Claudin-5 (red) expressed in CD31+ endothelial cells (green) that were treated by Resting-MCM-ACM, LPS-MCM-ACM, and Hemin-MCM-ACM. Scale bars, 25 μm. (c) Representative immunoblots of ZO-1 and Claudin-5 in endothelial cells that were treated by Resting-MCM-ACM, LPS-MCM-ACM, and Hemin-MCM-ACM. (d) Quantification of ZO-1 and Claudin-5 protein levels in endothelial cells. n = 3 biologically independent endothelial cell cultures. β-actin was used as an internal control. One-way ANOVA followed by Tukey’s test. *p < 0.05, **p < 0.01. (e) Schematic diagram showed treatment of endothelial cells with Hemin-MCM-ACM or medium derived from astrocytes that were treated with Hemin-MCM and MMP3 inhibitor (Hemin-MCM+MMP3 inhibitor-ACM) for 24 h. (f) Representative immunostaining images showed tight junction proteins ZO-1 (red) and Claudin-5 (red) expression in CD31+ endothelial cells (green) that were treated by Resting-MCM-ACM, Hemin-MCM-ACM, Hemin+MCM-MMP3 inhibitor-ACM. Scale bars, 25 μm. (g) Representative immunoblots of ZO-1 and Claudin-5 in endothelial cells that were treated by Resting-MCM-ACM, Hemin-MCM-ACM and Hemin-MCM+MMP3 inhibitor-ACM and (h) Quantification of ZO-1 and Claudin-5 protein levels in endothelial cells. n = 3 biologically independent endothelial cell cultures. β-actin was used as an internal control. One-way ANOVA followed by Tukey’s test. *p < 0.05, **p < 0.01. All data are mean ± SD.
Inhibition of MMP3 secreted from reactive astrocytes attenuated endothelial tight junction protein loss in vitro
To demonstrate whether inflammatory reactive astrocytes disrupt BBB integrity after hemorrhagic stroke through MMP3, MMP3 inhibitor (100 nM) and medium derived from hemin-treated microglia were added to astrocytes for 24 h, and then the medium (Hemin-MCM+MMP3 inhibitor-ACM) was collected and applied to bEnd.3 cells (Figure 4(e)). In endothelial cell cultures, Hemin-MCM+MMP3 inhibitor-ACM treatment ameliorated the loss of tight junction-related proteins in endothelial cells (Figure 4(f) to (h)), indicating that reactive astrocytes reduced endothelial tight junctions via MMP3.
Conditional knockout of MMP3 in astrocytes attenuated BBB disruption and neurobehavioral deficits in hemorrhagic stroke mice
We then generated inducible astrocyte-specific MMP3 knockout mice (Aldh1l1Cre-ERT 2 ; MMP3flox/flox) to further explore its effect on BBB disruption after hemorrhagic stroke. Tamoxifen was injected into Aldh1l1Cre-ERT 2 ; MMP3flox/flox mice once a day for 5 consecutive days, and 1 week after the last dosage of tamoxifen, hemorrhagic stroke surgery was performed. We found that MMP3 was specifically knockout in astrocytes but not in microglia at 3 days after hemorrhagic stroke (Fig. S4A, B). And real-time PCR results exhibited a significant reduction in MMP3 expression 3 days after hemorrhagic stroke in the conditional knockout group, compared with the control groups (Fig. S4C).
We then explored whether conditional knockout of MMP3 in astrocytes affects BBB permeability after hemorrhagic stroke. First, the extravasation of Evans blue was examined to quantify BBB damage. Representative whole mice brain images and quantification data demonstrated that conditional knockout of MMP3 in astrocytes reduced the Evans blue leakage compared to the control groups 3 days after hemorrhagic stroke, while conditional knockout of MMP3 in sham mice did not affect BBB integrity (Figure 5(a) and (b)). Furthermore, the intracranial extravasated plasma-derived IgG was significantly increased in the hemorrhagic perifocal area 3 days after hemorrhagic stroke while it was reduced in the MMP3 conditional knockout mice (Figure 5(c) and (d)). Our Western blot result showed that conditional knockout of MMP3 in astrocytes reduced the degradation of tight junction-related proteins ZO-1 and Claudin-5 in 3 days post hemorrhagic stroke mice compared to the control groups (Figure 5(e) and (f)).
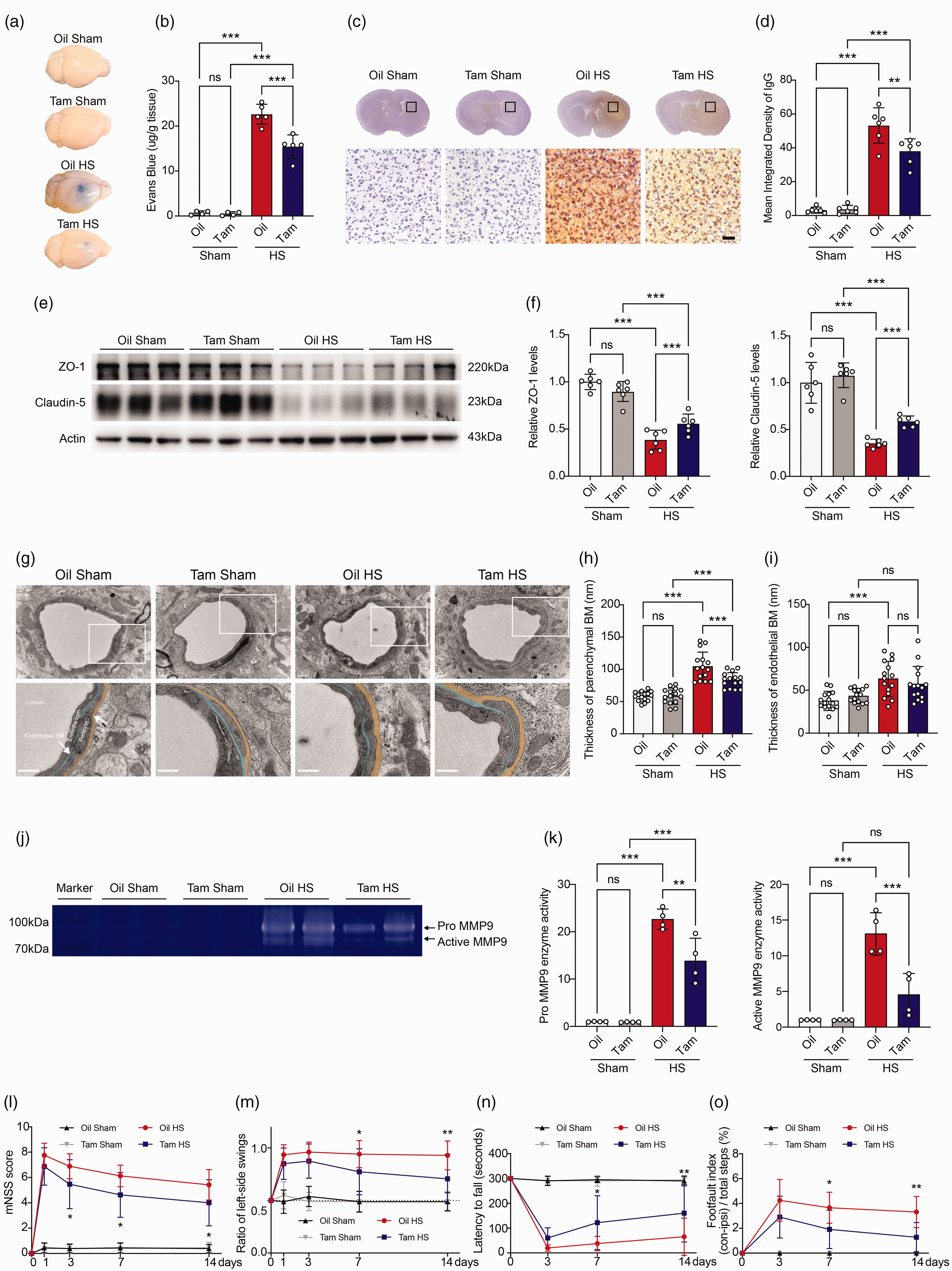
Conditional knockout of MMP3 in astrocytes attenuated BBB disruption and neurobehavioral deficits after hemorrhagic stroke. (a) Representative perfused whole brains of Oil treated sham mice (Oil Sham), tamoxifen treated Aldh1l1Cre-ERT2; MMP3flox/flox sham mice (Tam Sham), Oil treated hemorrhagic stroke mice (Oil HS) and tamoxifen treated Aldh1l1Cre-ERT2; MMP3flox/flox Continued.hemorrhagic stroke mice (Tam HS) after Evans blue injection at day 3 after hemorrhagic stroke. (b) Quantification of extravasated Evans blue in brains at day 3 after hemorrhagic stroke. n = 4–6 mice per group. One-way ANOVA followed by Dunnett’s test. ***p < 0.001. (c) Representative brain sections and immunostaining images of IgG leakage at day 3 after hemorrhagic stroke. Scale bars, 50 μm. (d) Semi-quantification of IgG intensity in the hemorrhagic perifocal area at day 3 after hemorrhagic stroke. Statistics are derived from 16 slices, n = 6 mice per group. One-way ANOVA followed by Tukey’s test. ***p < 0.001. (e) Representative immunoblots of ZO-1 and Claudin-5 in the hemorrhagic perifocal area at day 3 after hemorrhagic stroke. (f) Quantification of ZO-1 and Claudin-5 protein levels. n = 6 mice per group. β-actin was used as an internal control. One-way ANOVA followed by Tukey’s test. ***p < 0.001. (g) TEM images showed representative capillaries in the hemorrhagic perifocal area at day 3 after hemorrhagic stroke. Scale bars, 0.5 μm. BM, basement membrane. Endothelial BM and parenchymal BM is lightly shaded blue and yellow, respectively. (h, i) Quantification of endothelial BM and parenchymal BM thickness in the hemorrhagic perifocal area at day 3 after hemorrhagic stroke. Statistics are derived from 15 capillaries, n = 3 mice per group. One-way ANOVA followed by Tukey’s test. ***p < 0.001. (j) Representative gelatin zymogram showing MMP9 activity in the hemorrhagic perifocal area at day 3 after hemorrhagic stroke. (k) Quantification of pro-MMP9 and active MMP9 activities in the hemorrhagic perifocal area at day 3 of hemorrhagic stroke. n = 4 mice per group. One-way ANOVA followed by Tukey’s test. ***p < 0.001. (l–o) Neurobehavioral outcomes were assessed by four neurobehavioral tests including the mNSS (l), EBST (m), rotarod test (n), and grid walking test (o). n = 10–17 mice per group. Two-way ANOVA followed by Bonferroni’s test. *p < 0.05, **p < 0.01, compared with the Oil HS group. All data are mean ± SD.
Once the endothelial barrier is broken, the basement membrane (BM) acts as another protective layer between blood and nervous tissue. Numerous studies have reported BM dissociation during ischemia, and the BM becomes diffused and thickened after ischemia at ultrastructural levels,26,27 suggesting the thickness could be used as an indicator for assessing BBB integrity. Therefore, we used TEM to observe whether specifically knocking out of MMP3 in astrocytes can alleviate the expansion of BM to assess its effect on BBB integrity. TEM images showed that hemorrhagic stroke led to the increased thickness of both the endothelial BM and the parenchymal BM (Figure 5(g) to (i)). To our surprise, conditional knockout of MMP3 in astrocytes significantly alleviated parenchymal BM expansion at 3 days after hemorrhagic stroke, while having no effect on the thickness of the endothelial BM (Figure 5(h) and (i)). Numerous studies have reported that MMP3 could activate pro-MMP9, which is involved in BBB disruption after brain injury.19,28,29 Thus, the activity of pro-MMP9 and active MMP9 were further evaluated by gelatin zymography at 3 days after hemorrhagic stroke. We found that the activity of pro-MMP9 and active MMP9 decreased after hemorrhagic stroke in the astrocytic MMP3 knockout mice compared with the control groups (Figure 5(j) and (k)).
To evaluate whether conditional knockout of MMP3 in astrocytes affects the neurobehavior in hemorrhagic stroke mice, we examined the neurobehavioral function between astrocyte-specific MMP3 knockout mice and control group mice. We found that MMP3 conditional knockout mice showed better performance in mNSS at 3-, 7-, and 14-day following hemorrhagic stroke (Figure 5(l)). In addition, EBST, rotarod test, and grid walking test showed that conditional knockout of astrocytic MMP3 attenuated neurobehavioral deficits at 7 and 14 days after hemorrhagic stroke compared with the control groups (Figure 5(m) to (o)).
We also examined neuronal apoptosis at 3 days after hemorrhagic stroke by TUNEL staining. We found that the number of apoptotic neurons decreased in the astrocyte-specific MMP3 knockout mice compared with the control group mice (Fig. S5A, B), suggesting that specific knockout of astrocytic MMP3 had neuroprotective effects.
Discussion
In response to pathological alterations, astrocytes undergo specific transformations and adapt to a “reactive state”, which has different functions with high heterogeneity. Recent studies revealed that astrocytes undergo different reactive transformations upon aging, spatial adaptation, and origins of diseases.13,14,30,31 In our study, we identified a group of inflammatory reactive astrocytes (GFAP+/Vim+/Lcn2+/Serpina3n+/C3d+) in hemorrhagic stroke mice brain, and they shared multiple inflammation signatures and neurotoxic related genes with DAA (Vim, Serpina3n) and A1 reactive astrocytes (C3), suggesting their potential overlapping functions. 13 Recent findings revealed that TNF-induced inflammatory reactive astrocytes cause BBB dysfunction through activation of STAT3 and increased expression of Serpina3n. 32 One study performed by our lab demonstrated that stroke-induced C3d+/GFAP+ A1 reactive astrocytes disrupt BBB integrity. Blocking C3d+/GFAP+ A1 reactive astrocyte conversion with semaglutide attenuates BBB disruption in mice after ischemic stroke. 33
Astrocyte activation may involve a cross-talk between various cells in the brain, including microglia. 34 Previous studies demonstrated that inflammatory microglia contribute to the phenotypic change of reactive astrocytes in mice subjected to neuroinflammation. 17 Based on the fact that hemorrhagic stroke activates brain-residential microglia and monocyte-derived macrophages,35,36 we hypothesized that activated microglia and macrophages could interact with astrocytes and induce the phenotypic change of astrocytes in the hemorrhagic brain. Although microglia and macrophages share many gene signatures and similar functions in the diseased brain, to our surprise, they were differentially involved in inducing phenotypic changes of reactive astrocytes. We found that depletion of microglia, but not macrophages abolished the shift of resting astrocytes to reactive astrocytes with inflammation-related gene signatures. One potential reason is that brain-infiltrating macrophages after hemorrhagic stroke are mainly polarized to the M2 phenotype, 37 which had negligible effects on the induction of inflammatory reactive astrocytes.
BBB disruption is involved in the pathogenesis of numerous CNS diseases, including traumatic brain injury, cerebral ischemia, cerebral hemorrhage, etc. Elucidating the role of astrocytes in BBB is critical for developing strategies for protecting BBB integrity subject to brain injury. Whether astrocytes play a beneficial or a detrimental role in regulating BBB after brain injury is still under debate. One of the main reasons is that astrocytes comprise multiple subsets with diverse and potentially opposing roles in diseases based on gene expression. 38
To find out the role of inflammatory reactive astrocytes on BBB disruption in hemorrhagic stroke, RNA-seq of resting and reactive astrocytes was performed, and the number of molecules including MMPs and chemokines was highly increased in inflammatory reactive astrocytes. Among these molecules, MMP3 was ranked as the top distinct gene in both LPS- or Hemin-MCM treated astrocytes, and it has been well documented that MMP3 was closely associated with BBB disruption. 39 Our in vitro study confirmed that factors secreted from reactive astrocytes reduced tight junction protein expression between endothelial cells. By inhibition of MMP3 in reactive astrocytes in vitro and specifically knockout of MMP3 in astrocytes in vivo, we confirmed that MMP3 is the key player that mediates reactive astrocytes-induced BBB disruption in hemorrhagic brain. A recent study showed that the increased MMP9 was mainly derived from reactive astrocytes after subarachnoid hemorrhage. Ndrg2 knockout in astrocytes inhibited MMP9 expression after SAH and attenuated BBB damage. 40 Numerous studies have reported that MMP3 was involved in promoting the maturation of MMP9 during the acute phase of neuroinflammatory diseases.19,28,29 Our gelatin zymography results showed that the activity of pro-MMP9 and active MMP9 were decreased in MMP3 conditional knockout mice after hemorrhagic stroke. These phenomena suggest that astrocyte-derived MMP3 plays a vital role in the maturation of MMP9, which is highly correlated with BBB disruption.
Intriguingly, our TEM data found that astrocyte-derived MMP3 has an inappreciable effect on the thickness of the endothelial BM while swelling the astrocyte-derived parenchymal BM. This phenomenon may be due to the direct contact of reactive astrocytic endfeet with the parenchymal BM, while the endothelial BM was separated by pericytes. 41 Our TEM data implicate reactive astrocytes as critical cellular mediators of vascular BM expansion after brain injury.
In addition, we found that inhibition of MMP3 in reactive astrocytes only partially rescued BBB damage, suggesting other highly expressed molecules such as chemokines cascade genes, including Ccl2, Ccl5, Ccl7, Cxcl1, Cxcl3, and Cxcl5, could also participate in disrupting BBB. Ccl5 promoted the migration of leukocytes into the damaged brain and has been shown to act as a powerful molecule to mediate neuroinflammation, BBB dysfunction, and tissue infarction in ischemic stroke mice. 42 Astrocyte-derived Cxcl1 has been reported to drive neutrophil transmigration and BBB permeability in viral encephalitis. 43 Given these chemokines-related genes were upregulated in reactive astrocytes, future studies should address how these chemokines affect BBB integrity during brain injury.
Overall, our current study highlighted the crucial role of a phenotype of reactive astrocytes in disrupting BBB at the acute stage of CNS injury. Being different from the acute stage, at the chronic stage, neurovascular remodeling was initiated and this process was critical for neurobehavioral recovery. 44 Considering that MMP3, an important mediator for modulating angiogenesis, 45 neurogenesis,46,47 and synaptogenesis, 48 is highly expressed in reactive astrocytes which are expected to be involved in the neurovascular remodeling and contribute to the neurobehavioral recovery. Altogether, our study suggests that reactive astrocytes could be a key driver for disrupting BBB, and serve as a novel therapeutic target for protecting BBB integrity after CNS injury.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241235008 - Supplemental material for Hemorrhagic stroke-induced subtype of inflammatory reactive astrocytes disrupts blood-brain barrier
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241235008 for Hemorrhagic stroke-induced subtype of inflammatory reactive astrocytes disrupts blood-brain barrier by Chang Liu, Yiyan Guo, Shiyu Deng, Shiyi Zhou, Shengju Wu, Tingting Chen, Xiaojing Shi, Muyassar Mamtilahun, Tongtong Xu, Ze Liu, Hanlai Li, Zhijun Zhang, Hengli Tian, Won-Suk Chung, Jixian Wang, Guo-Yuan Yang and Yaohui Tang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from National Key R&D Program of China #2019YFA0112000 (YT), #2022YFA1603600 (YT), the National Natural Science Foundation of China (NSFC) projects 82071284 (YT), 82371307 (YT), 82172529 (JW), 81974179 (ZZ), 82271320 (ZZ), Shanghai Rising-Star Program (21QA1405200, YT), the Scientific Research and Innovation Program of Shanghai Education Commission 2019-01-07-00-02-E00064 (GYY), Science and Technological Innovation Act Program of Shanghai Science and Technology Commission, 20JC1411900 (GYY), Young Leading Scientists Cultivation Plan supported by Shanghai Municipal Education Commission (ZXWH1082101, YT), the Fundamental Research Funds for the Central Universities (YG2023ZD02, YT) and SUITM-202306 (YT).
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
J.W., G.Y.Y. and Y.T. designed the experiments and helped to edit the manuscript finally. C.L. performed the experiments, analyzed data, and wrote the manuscript. Y.G. contributed to bioinformatics analysis. S.D. contributed to animal identification and neurobehavioral tests. S.Z. and M.M. helped with Evans blue assay. S.W. helped with TEM sample collection. T.C. and T.X. helped with primary cell isolation. X.S. and Z.L. participated in data analysis. H.L., Z.Z., H.T., and W.S.C. performed manuscript review and editing.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
