Abstract
Aneurysmal subarachnoid hemorrhage (aSAH) is a devastating disease with high morbidity and mortality rates. Within 24 hours after aSAH, monocytes are recruited and enter the subarachnoid space, where they mature into macrophages, increasing the inflammatory response and contributing, along with other factors, to delayed neurological dysfunction and poor outcomes. High-density lipoproteins (HDL) are lipid-protein complexes that exert anti-inflammatory effects but under pathological conditions undergo structural alterations that have been associated with loss of functionality. Plasma HDL were isolated from patients with aSAH and analyzed for their anti-inflammatory activity and protein composition. HDL isolated from patients lost the ability to prevent VCAM-1 expression in endothelial cells (HUVEC) and subsequent adhesion of THP-1 monocytes to the endothelium. Proteomic analysis showed that HDL particles from patients had an altered composition compared to those of healthy subjects. We confirmed by western blot that low levels of apolipoprotein A4 (APOA4) and high of serum amyloid A1 (SAA1) in HDL were associated with the lack of anti-inflammatory function observed in aSAH. Our results indicate that the study of HDL in the pathophysiology of aSAH is needed, and functional HDL supplementation could be considered a novel therapeutic approach to the treatment of the inflammatory response after aSAH.
Keywords
Introduction
Aneurysmal subarachnoid hemorrhage (aSAH) accounts for 5% of all types of stroke, has a mortality of 35%, and most survivors have long-term disabilities or cognitive dysfuntion.1–3 The mechanisms of secondary brain injury after aSAH are multifactorial and start immediately after bleeding. Recent evidence shows that inflammation mediates many of the vascular and neural changes observed in the acute phase after aSAH (0–24 hours after hemorrhage).4,5 The inflammatory response after aSAH leads to increased permeability of the blood-brain barrier and increased expression of cellular adhesion molecules.6,7 These molecules allow infiltration and activation of innate immune cells that exacerbate intracerebral inflammation and lead to neuronal cell death and motor deficits after aSAH. 8 To date, studies that focus specifically on monocytes after aSAH are limited, but it is likely that these cells play an important role in inflammation after aSAH and can contribute to early and delayed brain damage.9–13
A recent preliminary report shows that HDL-cholesterol (HDL-C) levels are decreased in aSAH and are associated with poor outcomes and mortality. 14 However, there is accumulating evidence that targeting HDL functionality rather than concentration may represent a better therapeutic target. 15 As research in the field of lipoproteins and new powered omics tools progresses, it becomes apparent that HDL particles possess different functions that depend on their structural components. During HDL biogenesis and metabolism, a family of numerous subclasses of particles is generated, which vary in size, shape, and composition. Therefore, the lipid and protein content of the HDL particle is not uniform during its lifecycle, reflecting its continuous remodeling through interactions with other circulating lipoproteins and tissues, and ultimately affecting its functional properties. 16 In addition to their well-characterized cholesterol transport function, HDL possess anti-inflammatory properties, including their ability to limit monocyte adhesion,17,18 thus HDL can play a role in aSAH. Furthermore, HDL anti-inflammatory capacity may provide independent clinical information for the prediction of cardiovascular disease risk. 19 However, in diseases characterized by inflammation, HDL can become dysfunctional or even pro-inflammatory independently of HDL-C levels, all linked to modifications in the HDL-associated proteome and lipidome.20,21
Given that HDL protein composition changes in inflammatory states and has been shown to be related to altered functionality, 22 the aim of this work was to investigate the anti-inflammatory capacity of HDL in patients with aSAH and its association with their proteome. This is the first study to focus on HDL as potential therapeutic targets in the inflammatory response after aSAH.
Material and methods
Patients selection
This was a case-control study that included a cohort of 28 patients with severe aSAH hospitalized in the intensive care unit of the University Hospital Virgen del Rocío (Sevilla, Spain), from March 2019 to February 2020. Glasgow Coma Scale (GCS), Hunt-Hess, the World Federation of Neurological Surgeons (WFNS), and modified Fisher scales were used to evaluate patients during the first 24 hours after hospital admission, after hemodynamic and metabolic resuscitation to rule out pharmacological influence, such as sedation. The functional outcome was assessed using the Glasgow Outcome Scale (GOS). A group of 20 healthy controls of similar age and sex to the patients group was also evaluated. This study was conducted in accordance with the Declaration of Helsinki and the protocol was approved by the Ethics Review Board of the University Hospital Virgen del Rocío (0586-N-16). All relatives and legal representatives signed the informed consent in writing.
Blood samples were obtained from patients within the first 24 hours after the appearance of symptoms, and from healthy control subjects at one time. Plasma was stored at −80°C until analysis. Plasma concentrations of total cholesterol (TC), HDL-C, and LDL/VLDL levels were measured enzymatically using the EnzyChromTM Assay Kit (BioAssay Systems, USA) following the manufacturer's protocol.
HDL isolation from plasma
HDL were isolated from 1 ml of plasma by classical 2-step sequential density ultracentrifugation as previously described. 23 In summary, plasma density was adjusted to 1.063 g/ml using potassium bromide (KBr) and overlayed with KBr saline solution (d = 1.063 g/ml). Ultracentrifugation was performed in a 70.1Ti rotor using a Beckman Optima L-100XP ultracentrifuge at 100,000 g for 18 hours at 10°C to separate LDL from the rest of the plasma. The density of the bottom fraction resulting from the first ultracentrifugation and containing HDL was adjusted to 1.21 g/ml with KBr (d = 1.35 g/ml) and overlaid with KBr saline solution (d = 1.21 g/ml). The second ultracentrifugation was also performed at 100,000 g for 20 hours at 10°C. After this step, the upper fraction containing HDL was recovered and rinsed extensively with saline and concentrated using a centrifugal filter device (Amicon Ultra, 3 KDa cut off). The purity of isolated HDL was confirmed by sodium dodecyl sulfate-12% polyacrylamide gel electrophoresis (SDS-PAGE) and the total protein concentration was determined in triplicate using the PierceTM BCA Protein Assay Kit (Thermo Scientific, USA) with bovine serum albumin as standard.
HDL functionality assay
Monocyte adhesion assay
HDL anti-inflammatory capacity was evaluated using the CytoSelectTM Leukocyte-Endothelium Adhesion Assay (Cell Biolabs Inc, USA) as previously described. 24 Human umbilical vein endothelial cells (5 × 105 cells/well; HUVEC, Lonza Clonetics, USA), were cultured in 200 µl supplemented EBM medium to confluence in 96-well plates coated with 100 µl gelatin. Cells were cultured for 48 hours until a monolayer was formed. For adhesion assays, HUVEC were preincubated with 300 µg/ml of HDL for 16 hours. Subsequently, HUVEC were activated with 20 ng/mL of TNFα for an additional 6 hours and then washed twice with serum free medium. THP-1 monocytes (1 × 105 cells/well; ATCC, USA) were labeled with LeukoTrakerTM, seeded on the HUVEC monolayer and incubated for 45 minutes at 37°C. The unattached cells were removed, and the remaining cells were washed 3 times and lysed with lysing buffer. Cell lysate fluorescence, measured at 480/520 nm, was recorded on a CLARIOstar Plus fluorescence reader (BMG LABTECH, Germany).
Flow cytometry for VCAM-1 measurement
The HUVEC subjected to the different treatments were recovered using 0.5% trypsin/EDTA, washed, and then labeled by incubation for 30 minutes with the anti-VCAM1/CD106-FITC antibody (20 µl per sample, BD Biosciences, USA). Cells were washed, resuspended in 200 µl of PBS, and directly used for flow cytometry. Data were collected on a Cytoflex S flow cytometer (Beckman Coulter, USA), and the expression of the adhesion molecules was quantified by measuring the median fluorescent intensity (MFI). Non-stained samples were used as negative controls. Data were analyzed with Kaluza 2.1 software (Beckman Coulter, USA).
Proteomic analysis of HDL
HDL proteomic profiling was carried out in two different batches several weeks apart at the Research Support Central Service (SCAI) Proteomic Facility of the University of Córdoba as previously described.
25
HDL were digested with trypsin and the resulting peptides were scanned and fragmented with the Orbitrap Fusion mass spectrometer (Thermo Fisher Scientific, USA) equipped with a Dionex Ultimate 3000 UHPLC system (Thermo Fisher Scientific, USA). The analysis of the raw mass spectrometry data was performed using MaxQuant software, and proteins were identified by searching the raw data against the human UniprotKB/Swiss-Prot protein database. The Maxquant output was preprocessed prior to statistical analysis.
26
Potential contaminants resulting from manipulation of the samples and of the proteomic process were manually excluded from the analysis.
21
All protein values (LFQ intensities) were log2 transformed to achieve an approximately normal distribution and missing protein values were imputed independently in cases and controls using the median value of each sub-cohort.
27
The batch effect was removed using the combat algorithm integrated in the sva package.28–31 Principal component analysis (PCA) was performed to observe the clustering data among isolated HDL. Known protein-protein interactions of relevant proteins were explored using STRINGdb.
32
A systems biology approach based on Ingenuity System Pathway Analysis (IPA) software (Qiagen, Germany) was used to determine statistically significant associated network functions and canonical pathways in which the identified proteins were involved.
Statistical analysis
Most statistical analyzes and graphs were performed using GraphPad Prism 5.0 (USA). PCA analysis was performed with the statistical software R. 33 Normality was determined with the Shapiro-Wilk test to guide the choice of parametric or non-parametric statistics. Differences were evaluated by ANOVA or Kruskal–Wallis test followed by a Student’s t test or Mann–Whitney U test when corresponding. Differences between pairs were tested using the Student’s t test or Mann-Whitney test. Statistical analysis of the qualitative variable was performed using Fisher's exact test. The correlation analysis was performed using the Spearman test. Differences were considered statistically significant when p-Value <0.05.
Results
The main characteristics of the patients with aSAH and healthy controls included in the study are detailed in Table 1. Patients with aSAH were comparable in age, sex, prevalence of diabetes mellitus, and alcohol intake to controls. Patients with aSAH have a mean age of 55.6 years and women represented 61% of the population. Among the patients, 46.43% had a documented history of hypertension and 53.57% were smokers. The aSAH lipid profile was characterized by lower HDL-C levels compared to controls. The mean HDL-C of aSAH patients was 36.22 mg/dl (95% CI: 29.06–43.37) and of controls was 61.89 mg/dl (95% CI: 53.14–69.65). The mean TC and LDL in the patients were 162.3 mg/dl (95% CI: 139.2–185.3) and 77.59 mg/dl (95% CI: 64.24–90.93), respectively, without differences from controls. The median GCS (interquartile range) at the time of presentation was 13 (9–15), while the median Hunt-Hess scale was 2 (2–3), the median Fisher grade was 4 (3–4), and the median WFNS was 2 (1–4). The GOS at 6 months was 5 (3–5). During hospitalization, cerebral vasospasm was detected in 50% of patients with aSAH. Interestingly, HDL-C levels were significantly lower in aSAH patients who developed cerebral vasospasm compared to those who did not: 27.27 mg/dl (95% CI: 21.84–33.10) vs. 47.59 mg/dl (95% CI: 35.20–59.98), respectively.
Baseline characteristics of patients with aSAH and controls.
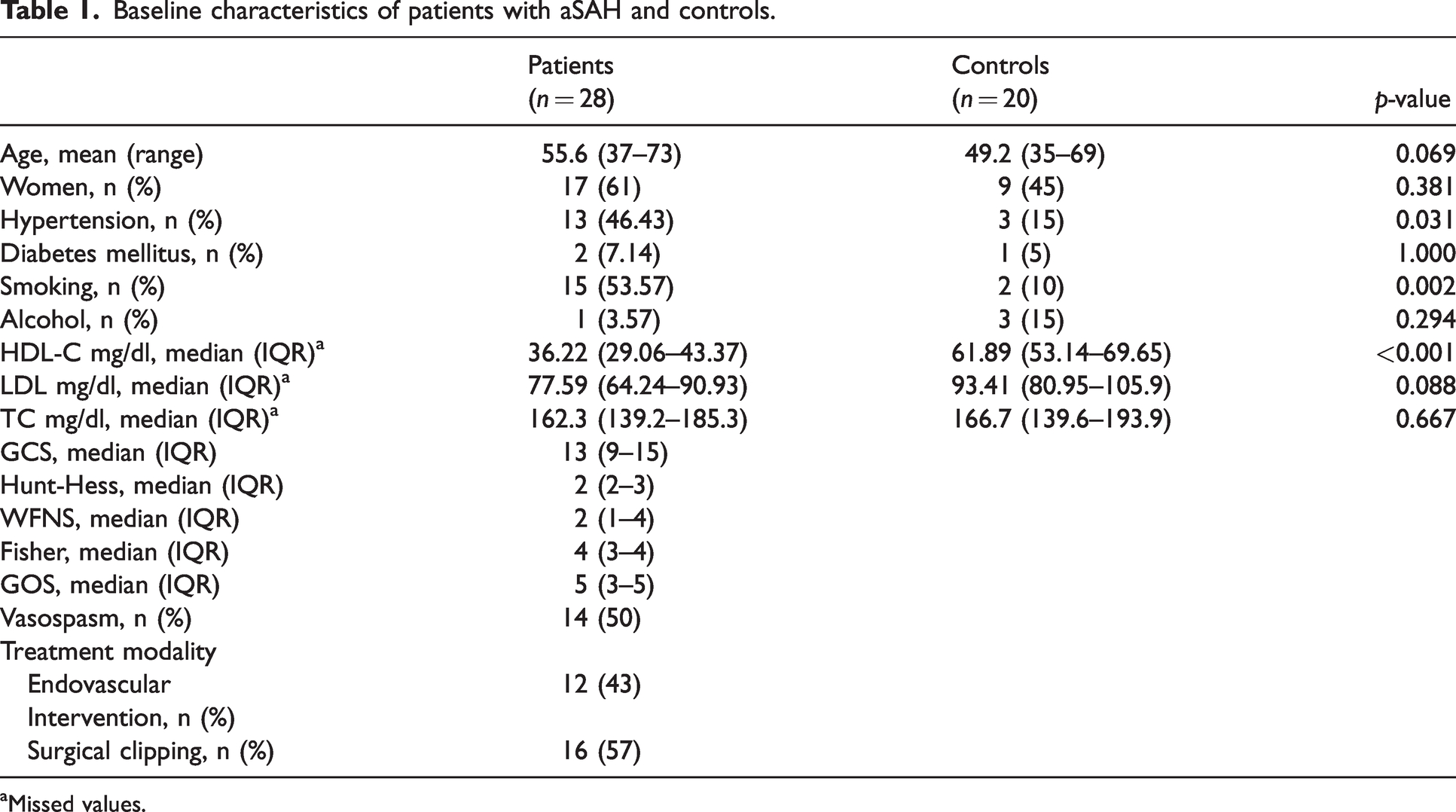
Missed values.
HDL is known to prevent leukocyte adhesion by regulating cellular adhesion molecules in endothelial cells. To address whether HDL function is altered in our cohort of patients, we performed an in vitro adhesion assay. Figures 1(a) and (b) show that HDL isolated from control plasmas decreased THP-1 adhesion to TNFα-activated HUVEC. However, when HDL from patients with aSAH were used, the inhibitory effect on adhesion was abrogated. Interestingly, this effect was related to a higher expression of VCAM-1 in HUVEC (Figure 1(c)). Furthermore, an inverse correlation was found between HDL-C plasma levels and adhesion of THP-1 to HUVEC (Figure 1(d)). These findings suggest that HDL anti-inflammatory capacity is significantly lower in patients with aSAH compared to controls.
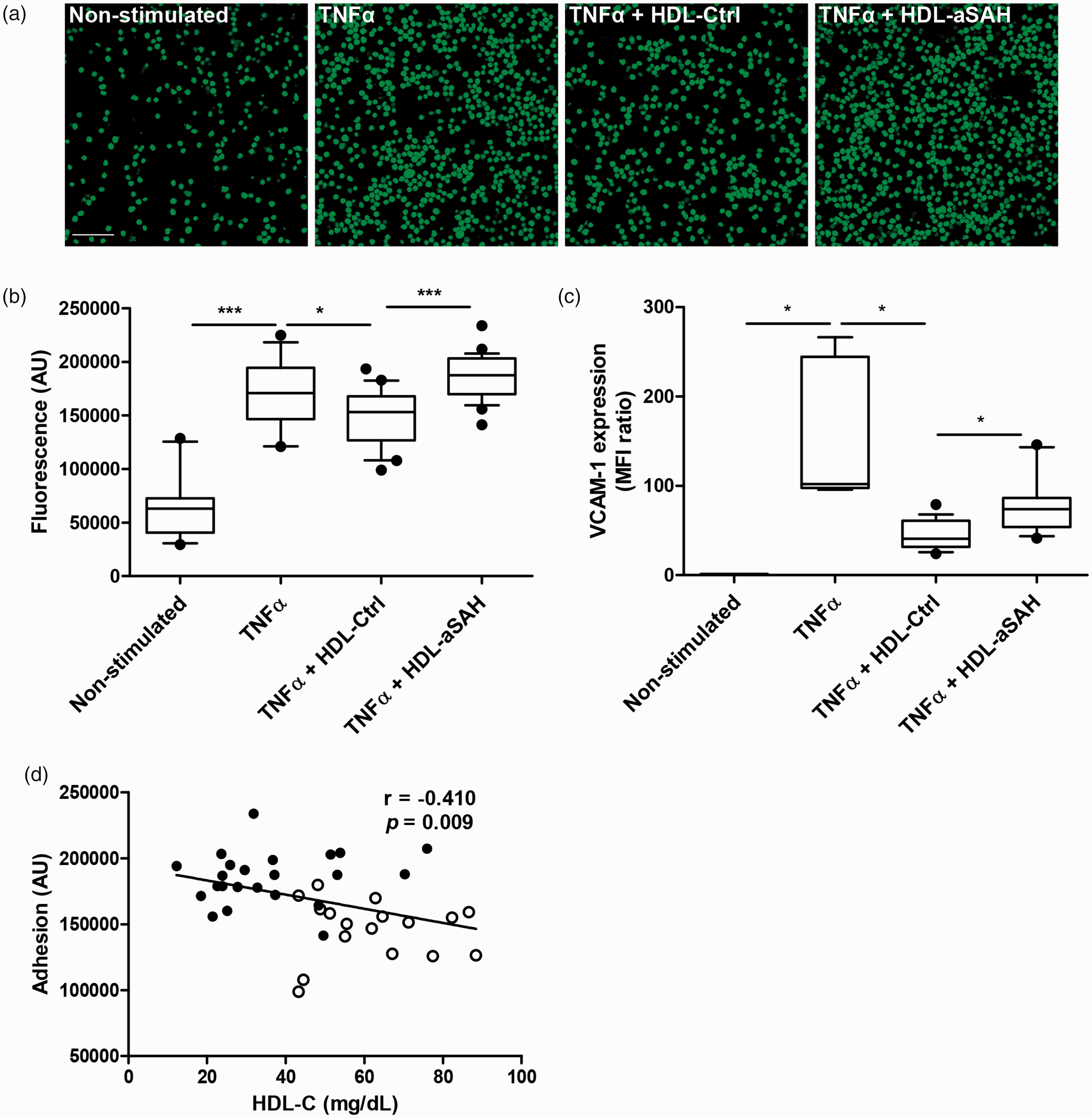
Altered anti-inflammatory function in HDL from patients with aSAH. HUVEC were pretreated with and without HDL (300 µg/ml) for 18 hours and then activated with 20 ng/ml TNFα for 6 hours. Representative images of THP-1 monocytes adhered to HUVEC (magnification ×20, scale bar = 100 µm; (a) and fluorescence of cell lysates (b). HDL-Ctrl (healthy donors), n = 20; HDL-aSAH (patients with aSAH), n = 28. (c) Expression of VCAM-1 in HUVEC, analyzed by flow cytometry. HDL-Ctrl (healthy donors), n = 16; HDL-aSAH (patients with aSAH), n = 18 and (d) Spearman correlation between HDL-C levels and adhesion of THP-1 monocytes to HUVEC. White dots are results from healthy controls and black dots from patients with aSAH. *p < 0.05; ***p < 0.001. For the boxplots in B and C, the centre line indicates the median, the box limits indicate the first and third quartiles, and the whiskers indicate the 10th and 90th percentiles.
Since HDL proteome composition can directly influence the biological functions of these lipoproteins, analysis of HDL protein composition can be useful in identifying new markers of HDL dysfunction among aSAH patients. We used a targeted proteomic analysis to investigate the proteomic composition of HDL isolated from 13 patients with aSAH and 7 healthy controls. Proteomic analysis allowed the identification of 84 proteins in HDL (Table S1). Among them, 17 proteins showed a missing value rate ≥15% in the global cohort: SELL, APOC2, C4A, LPA, APOC1, CRP, F12, IGHG4, CETP, SAA2, A2M, IGFALS, ANTXR2, IGHM, PLG, CP and ITIH1 (Figure S1). PCA was applied to identify groups of subjects based on similarities in their HDL proteome. The dimensional image of PCA showed the distinction between patients with aSAH and controls, thus ensuring a high quality of proteomic data. Figure 2(a) shows the contribution of ten dimensions to the profiling classification in the plot of the percentage of explained variances. The two first dimensions (Dim1 and Dim2), explained 22.7% and 12.3% of the HDL proteome variance, respectively, and clearly allowed the differentiation of patients with aSAH from controls (Figure 2(b)).
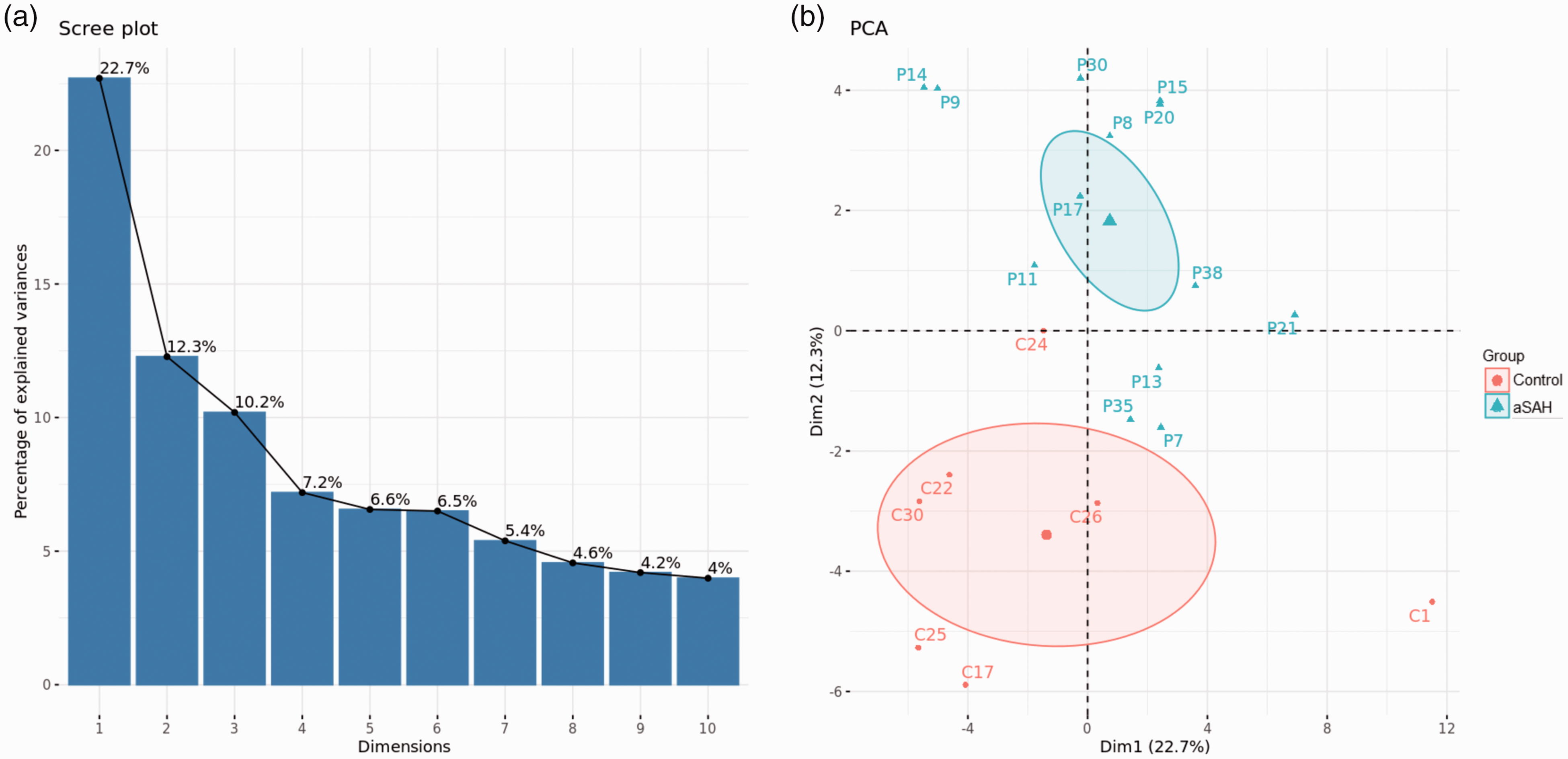
Principal Component Analysis (PCA) of HDL proteomic data. (a) Scree plot of the percentage of explained variances after PCA. This plot shows the proportion of total variance in the data included in the PCA for each principal component (dimensions), in descending order of magnitude and (b) Score plot of the two first principal components of the dataset. The first and second components explain 22.7% and 12.3% of the variation, respectively. Different colors and shapes correspond to patients with aSAH (turquoise triangles) and controls (red circles). Healthy donors, n = 7; Patients with aSAH, n = 13.
To examine possible signaling pathways that can be altered as a consequence of the changes in the HDL proteome observed in patients with aSAH, we used a system biology approach. IPA-based protein network analysis was performed using all identified proteins, and this analysis revealed pathways related to the inflammatory response, infiltration, and activation of immune cells (Figure S2A). The top molecular and cellular functions identified that were related to changes in HDL-associated proteins included lipid metabolism (p = 3.58E-06–1.04E-24), molecular transport (p =4.10E-06–1.04E-24), small molecule biochemistry (p = 3.63E-06–1.04E-24), cell-to-cell signaling and interaction (p = 3.19E-06–8.94E-24), and cell movement (p = 3.08E-06–1.99E-19). Finally, the IPA analysis of the proteomic data analyzed by IPA revealed eight significant networks (Table S2). Figure S2B shows the top three networks with the highest scores (scores ≥3 are considered significant with 99% confidence).
The differential expression analysis of proteomic data revealed that of the 84 proteins identified, 14 protein levels differed significantly in patients with aSAH compared to controls (Figure 3). Among these, 11 proteins were significantly more abundant (C3, SERPINA3, AGT, APOH, ITIH4, LRG1, SAA1, SAA2, SAA4, SELL and CRP) and 3 were less abundant (ITHIH1, APOA4 and APOC2) in HDL from patients with aSAH compared to controls (Figure 3(a)). We subsequently used the STRING database to analyze the known protein-protein interactions between differentially expressed proteins (Figure 3(b)). Finally, correlations between candidate proteins were assessed showing that some of them were highly correlated, either positively or negatively. Specifically, the most positive correlation coefficient between proteins was that for SAA1–SAA4 (0.751, p = 0.00014), and the most negative for SAA1–APOA4 (−0.694, p = 0.0007) (Tables S3–S4).
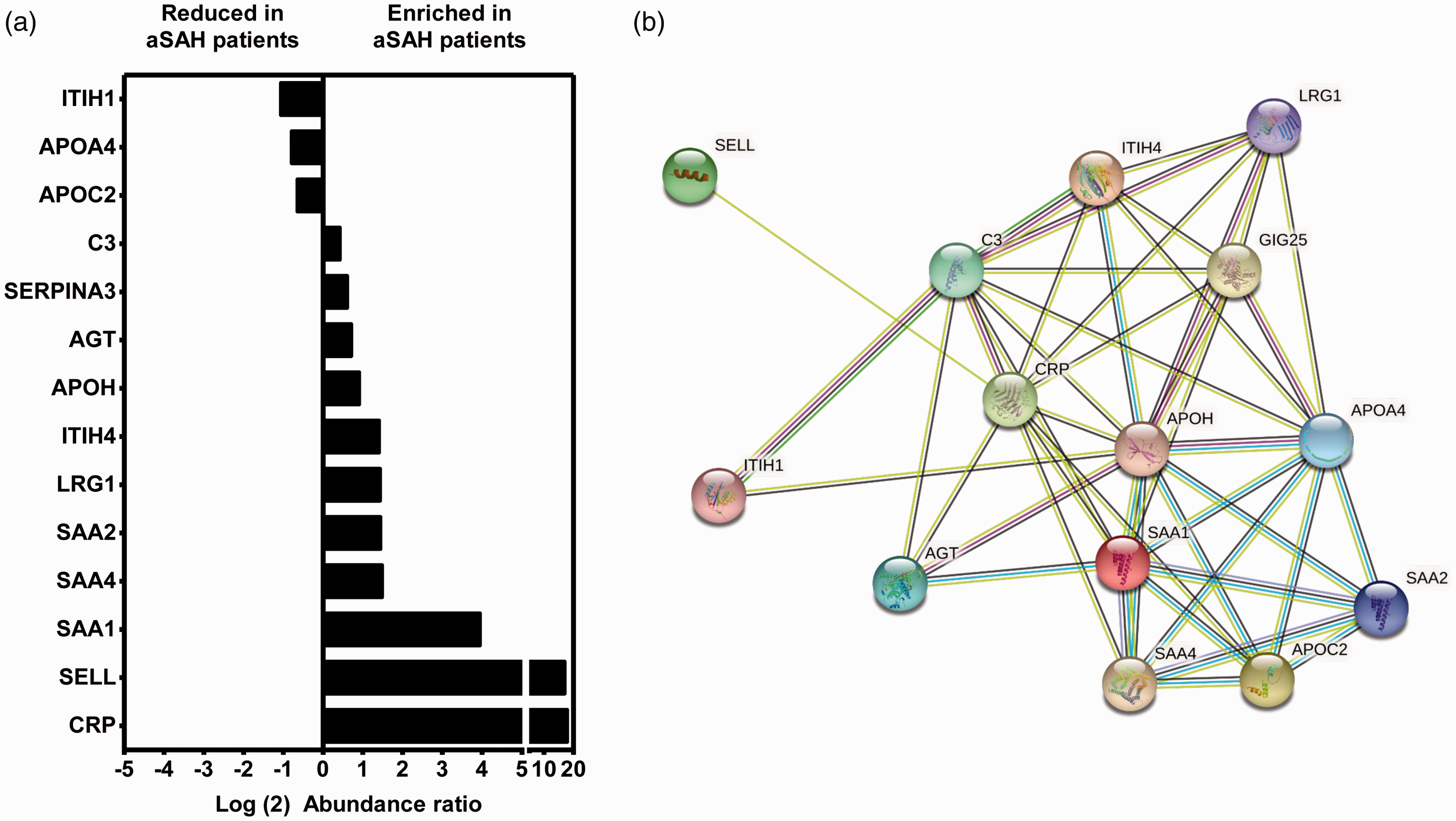
HDL protein levels and protein interactions in patients with aSAH. (a) Relative expression levels of proteins in HDL isolated from controls (n = 7), and patients with aSAH (n = 13) and (b) STRING interactions between proteins differentially expressed in patients with aSAH and controls. The nodes (spheres) represent proteins and the edges (lines) represent functional and physical protein–protein associations with a significant contribution of proteins to a common function, regardless of their physical binding to each other. The color of the lines indicates the type of interaction. Olive green line: neighborhood evidence; blue line: cooccurrence evidence; purple line: experimental evidence; yellowish green line: textmining evidence; black line: coexpression evidence.
To further explore the functional importance of these changes in the HDL proteome, we also analyzed the statistical correlations between the levels of the 14 differentially expressed proteins and the adhesion of THP-1 to HUVEC. Among these proteins, the expression of two of the three previous correlated proteins showed strong correlations. APOA4 showed the most negative (−0.472, p = 0.036) and SAA1 the most positive (0.584, p = 0.007) (Figure 4(a)). Therefore, using western blots, we analyzed the expression of APOA4 and SAA1 and confirmed that APOA4 had a lower expression and SAA1 a much higher level, in HDL from patients with aSAH (Figure 4(b)). Noticeably, we observed that SAA1 expression was significantly higher in patients with aSAH who developed vasospasm compared to those who did not (Figure 5). We did not find any difference in APOA4 levels between patients in relation to the development of cerebral vasospasm.
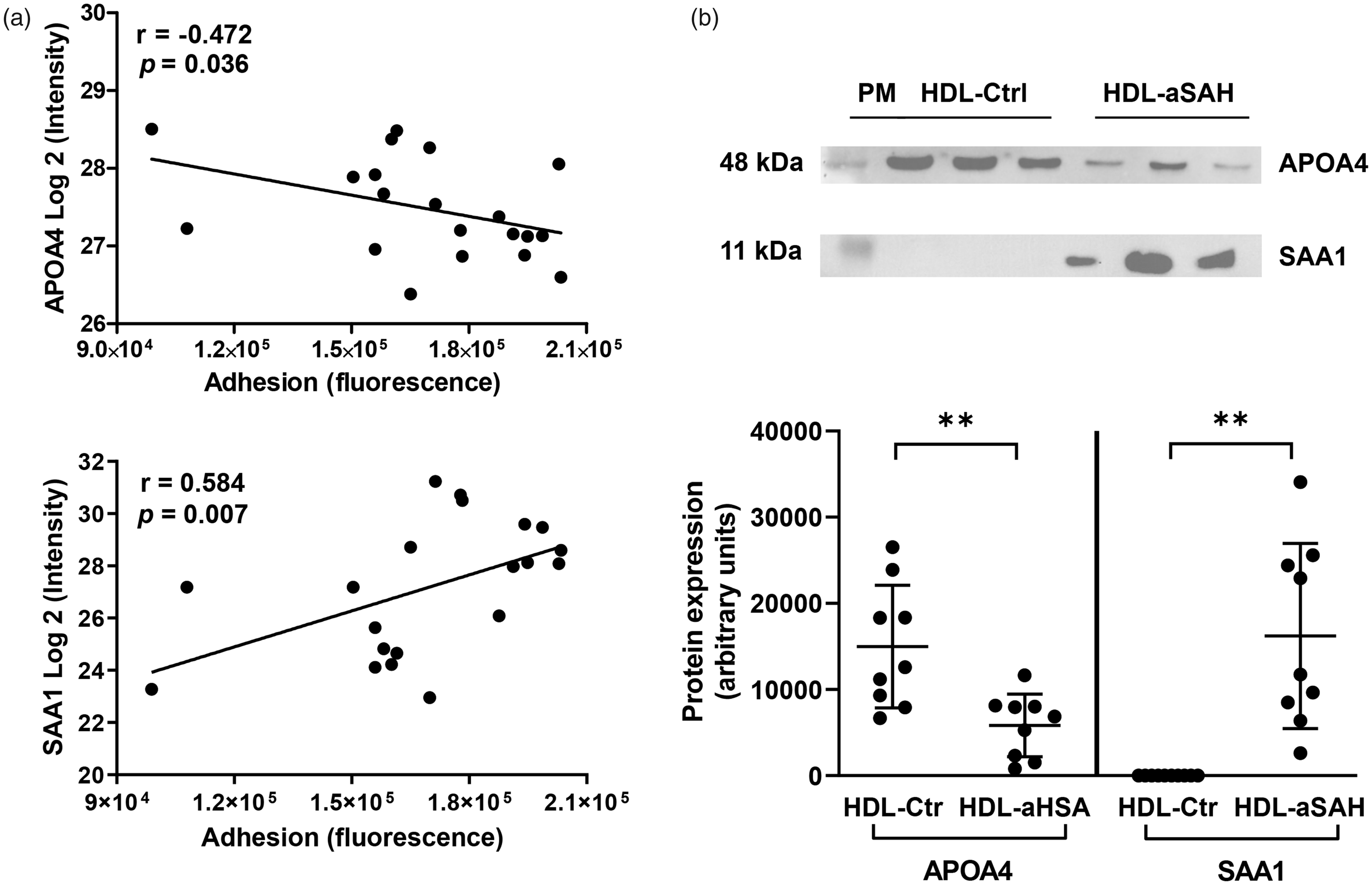
Levels of APOA4 and SAA1 proteins and their correlation with the adhesion of THP-1 to HUVEC. (a) Spearman correlation analysis of APOA4 and SAA levels (Log2 ratio) and adhesion of THP-1 to HUVEC and (b) Western blot analysis for the detection of APOA4 and SAA1 in HDL from healthy controls (HDL-Ctrl) and patients with aSAH (HDL-aSAH). Values are shown as mean ± standard deviation, n = 9; **p < 0.01. PM: protein marker. Uncropped blots are presented in Supplemental files.
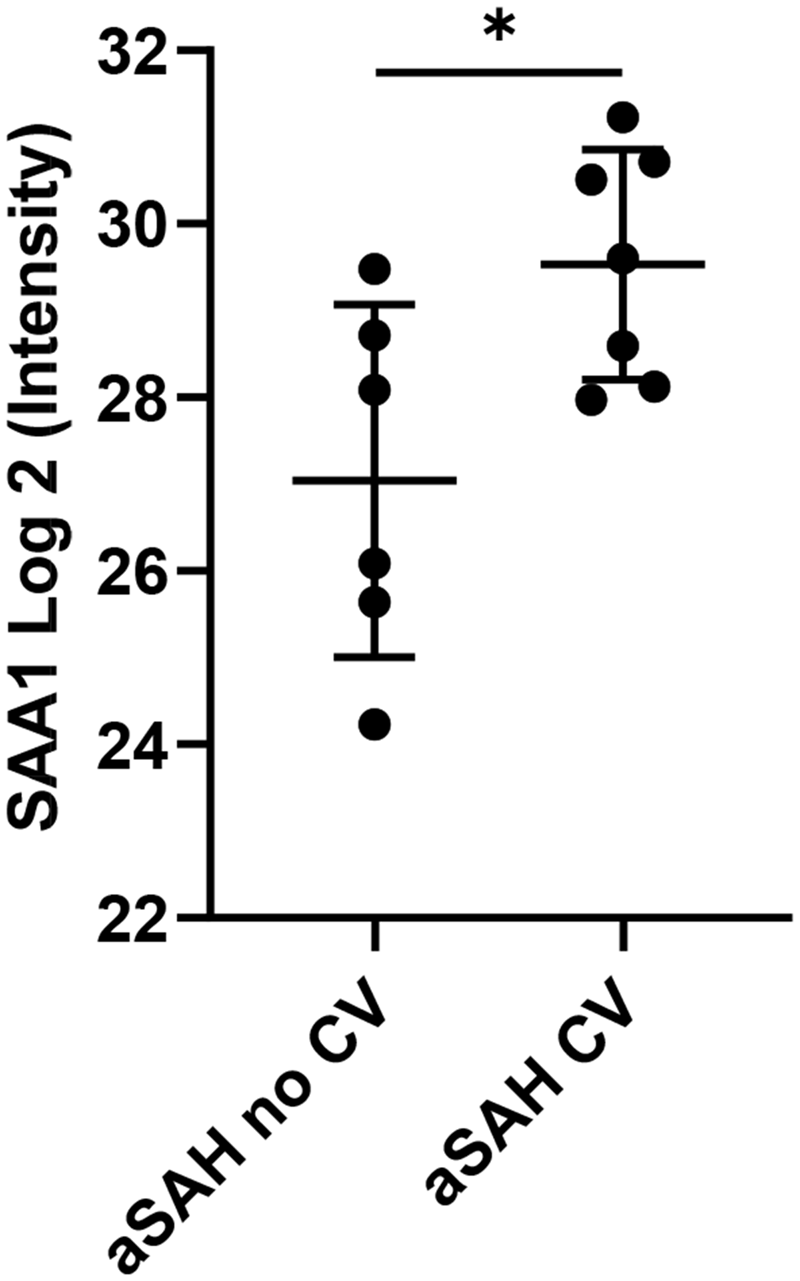
SAA1 levels in HDL from patients with aSAH. Relative expression levels of SAA1 in HDL isolated from patients with aSAH with (n = 7) or without cerebral vasospasm (n = 6). Values are shown as mean ± standard deviation, *p < 0.05.
Discussion
In this study, we found that patients with aSAH have low levels of HDL-C and their HDL particles are dysfunctional. HDL from patients with aSAH lost the ability to prevent VCAM-1 expression in HUVEC, with a subsequent increased adhesion of THP-1 monocytes to the endothelium. This effect was associated with changes in the protein composition of HDL particles, among which we observed higher levels of SAA1 and lower levels of APOA4 in HDL isolated from patients with aSAH. Finally, we observed that SAA1 in HDL could be a potential biomarker for cerebral vasospasm in aSAH.
The association between cholesterol (TC, HDL-C, or LDL) and the risk of aSAH is not completely defined. Most studies focused on the relationship between lipid profile and stroke have established two types of stroke: ischemic and hemorrhagic.34,35 However, hemorrhagic stroke is a mixed term, since it can include aSAH, intracerebral hemorrhage, subdural or epidural hemorrhages, and hemorrhagic transformation after ischemic stroke. Only a few studies distinguish between hemorrhage subtypes. All of this generates inconsistent results when establishing associations. Recently, a study aimed at clarifying the association between the lipid profile and the risk of aSAH reported that an adverse lipid profile was associated with an elevated risk of aSAH: low HDL-C elevated the risk of aSAH in both sexes, while high TC and high LDL elevated the risk only in men. 36 Another study, which included medical records from patients diagnosed with intracranial aneurysms, concluded that HDL-C and the use of lipid-lowering agents were associated with a significantly lower risk of aSAH. 37 In our study, we have observed that patients with aSAH have lower HDL-C levels at admission, with no differences in TC or LDL in both sexes, which is consistent with the above-mentioned studies. Furthermore, a recent preliminary report highlighted a direct relationship between HDL-C at admission and poor outcomes after aSAH, indicating that clinicians may consider an admission HDL-C < 40 mg/dL when evaluating delayed cerebral ischemia and mortality risk after aSAH. 14 Our results support this indication, as we observed lower HDL-C levels in aSAH patients who developed vasospasm during hospitalization, which is a prognostic factor for the poor outcome itself.
In addition to changes in HDL-C levels, under some pathological conditions alterations in structure and, as a consequence, in HDL functions have also been observed. 38 There are several mechanisms by which HDL protect against cardiovascular diseases, including their anti-inflammatory capacity, determined as their ability to suppress TNFα-induced VCAM-1 expression in endothelial cells.39,40 Furthermore, HDL’s anti-inflammatory capacity has been considered a new pharmacological intervention target to reduce the risk of cardiovascular disease. 18 However, studies addressing the functionality of HDL in aSAH have not been addressed before, which confers novelty on the present work. It is known that immediately after aSAH, blood in the subarachnoid space stimulates upregulation of adhesion molecules in endothelial cells, such as VCAM-1, intercellular adhesion molecule-1 (ICAM-1) and endothelial (E)-selectin.41,42 These molecules play a key role in the interaction between endothelial cells and leukocytes and the transendothelial migration of the latter to the site of injury, resulting in the development of inflammatory reactions after aSAH. 19 An important finding of our study is that HDL from patients with aSAH exhibit reduced anti-inflammatory function, due to the altered composition of the associated proteins. We observed that HDL from patients with aSAH were ineffective in inhibiting TNFα-induced monocyte adhesion and decreasing the abundance of the VCAM-1 protein in the plasma membrane of HUVEC. Although dysfunctional HDL have also been described in a variety of diseases, including coronary artery disease, ischemic stroke, diabetes mellitus, lupus erythematosus, and rheumatoid artritis,23,43–45 to our knowledge, this is the first report to describe dysfunctional HDL in aSAH.
To identify possible features that could distinguish healthy controls from patients with aSAH, we analyzed the protein composition of HDL by shotgun proteomics. Most studies of HDL-associated proteins have focused on the analysis of single proteins. However, proteomics is a revolutionary technique that provides a better understanding of protein expression, as it allows the quantification of many proteins in a single analysis and transcends the analysis of individual components. Our proteomic analysis of HDL, isolated by ultracentrifugation, unequivocally identified and quantified 84 proteins that had been previously associated with these particles. 22 Our pathway analysis confirmed that many of these HDL-associated proteins are related not only to the role of HDL in lipid metabolism, but also to the immune response and inflammation. In recent years, HDL have been shown to play a role in the immune inflammatory response, not only modulating the anti-inflammatory potential of endothelial cells, but also removing cholesterol from the cell membrane, regulating the abundance of lipid rafts and their cholesterol content, and thus controlling immune cell activation. 46 Interestingly, HDL have been shown to influence CD11b expression in human monocytes, reducing adhesion to endothelial cells. 47 Our quantitative proteomics results show that HDL in patients with aSAH are particularly enriched in C3, SERPINA3, AGT, APOH, ITIH4, LRG1, SAA1, SAA2, SAA3, SAA4, SELL and CRP, and are depleted in ITHIH1, APOA4 and APOC2. Protein-protein interactions ensure the integrity and stability of HDL particles, as such interactions can induce conformational changes, which in turn may affect functionality. 45 Interactions between differentially expressed proteins were identified by applying the STRING tool, and the established network showed that all of these proteins have known relationships. Of particular importance, we found a positive significant correlation between the SAA1 and SAA4 proteins in HDL. Previous studies have shown that a high concentration of HDL-bound SAA modifies the functionality of HDL from anti-inflammatory to pro-inflammatory properties. The accumulation of SAA in HDL from patients with kidney disease has been associated with a reduced cholesterol efflux capacity and anti-inflammatory activity of HDL.48,49 In fact, HDL in patients with chronic kidney disease has been shown to inhibit endothelial nitric oxide production and concomitantly increase the endothelial production of reactive oxygen species. 50 This is in line with our current finding that SAA accumulates in dysfunctional HDL particles in patients with aSAH.
Proteins associated with HDL allow, in part, to understand their functionality. When we analyzed the functional connection of HDL proteome changes, we found significant correlations between HDL-associated proteins and THP-1 adhesion to HUVEC. Both SAA1 and APOA4 play an important role in inflammation and the acute phase response.51,52 As mentioned above, SAA1 has been consistently found in HDL from individuals with high cardiovascular risk and has been associated with loss of anti-inflammatory function. 53 The presence of SAA in HDL reduces their affinity for hepatocytes but increases their binding affinity to macrophages.54,55 In this way, HDL clearing is redirected from the liver to macrophages, which has been postulated to induce a functional change from anti-inflammatory to pro-atherogenic HDL. 56 On the other hand, the results of APOA4 in HDL are controversial, as its expression in cardiovascular disease decreases in some studies but increases in others. 22 However, there is evidence for an inhibitory role of APOA4 in leukocyte adhesion to endothelial cells. 57 In a mouse model of acute colitis, APOA4 knockout mice exhibited a significantly greater inflammatory response to dextran sulfate sodium than their wild-type littermates, mainly by downregulating P-selectin and therefore the adhesion of leukocytes to the site of infection. 58
HDL proteome changes have been associated with various functional outcomes in human cohorts. A recent study showed that the altered HDL proteome was able to classify patients with COVID-19 according to the severity of the disease and that SAA1 and SAA2 levels in HDL particles were directly associated with an increased probability of death due to COVID-19 complications. 59 Interestingly, HDL-related biomarkers (HDL-C and APOA1) were robust predictors of disease progression and survival in patients with chronic liver failure. 60 Currently, there are no established biomarkers for diagnosing cerebral vasospasm or monitoring its progression. Some studies have reported that inflammatory markers can be used as a predictor of disease outcome. Luo et al. 61 reported that IL-6, IL-10, IL-8, IL-2 and TNF-α, might predict the poor prognosis of aSAH. In terms of inflammatory indices, the neutrophil-lymphocyte ratio (NLR) has been found to be capable of predicting poor functional outcomes in aSAH patients, 62 and more recently, it has been reported that the NLR could even distinguish between aSAH and non-aneurysmal SAH. 63 In our study, SAA1 levels were elevated in HDL particles from patients with aSAH who suffered cerebral vasospasm. However, its value as a biomarker for predicting the clinical outcome of patients with aSAH needs to be further investigated in larger clinical cohorts.
Smoking and elevated blood pressure are strong risk factors for aSAH.64,65 Furthermore, smoking is also associated with reduced HDL-C levels and has a negative impact on HDL function. 66 Although our cohort of patients presented an increased number of smokers and hypertensive subjects, we did not observe differences between HDL-C, HDL anti-inflammatory function, and SAA1 and APOA4 associated with HDL particles when these subjects were excluded from the analyzes, suggesting that the changes are a consequence of aSAH.
We are aware of the limitations of the current work. The primary limitations of our study were its small patient population and its one center rather than multicenter participation which can limit the generalization of conclusions. Another potential limitation is the use of ultracentrifugation for HDL isolation, which could strip off proteins during the two steps of ultracentrifugation, possibly resulting in an underestimation of the HDL composition as a result of the small amount of plasma initially available. However, we would like to note that ultracentrifugation is the gold standard for lipoprotein isolation, and under these conditions, we identified 84 proteins associated with HDL.
In summary, this study shows that patients with aSAH have low levels of HDL-C and particles that lose their anti-inflammatory capacity and are characterized by an altered proteome compared to healthy subjects. We identified two specific candidate proteins, SAA1 and APOA4, which are associated with loss of HDL functionality. Between them, SAA1 can be useful as a reliable biomarker to guide the stratification of patients into risk categories for developing cerebral vasospasm after aSAH. Despite the limitations of the study in terms of the number of samples and detected proteins, these findings suggest that supplementation with functional HDL particles may represent a novel therapy against the inflammatory response after aSAH which should be validated in independent studies.
Supplemental Material
sj-pptx-1-jcb-10.1177_0271678X231184806 - Supplemental material for HDL anti-inflammatory function is impaired and associated with high SAA1 and low APOA4 levels in aneurysmal subarachnoid hemorrhage
Supplemental material, sj-pptx-1-jcb-10.1177_0271678X231184806 for HDL anti-inflammatory function is impaired and associated with high SAA1 and low APOA4 levels in aneurysmal subarachnoid hemorrhage by Zaida Ruiz de Azúa-López, M Rosa Pezzotti, Ángela González-Díaz, Olivier Meilhac, Juan Ureña, Rosario Amaya-Villar, Antonio Castellano and Lourdes M Varela in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pptx-2-jcb-10.1177_0271678X231184806 - Supplemental material for HDL anti-inflammatory function is impaired and associated with high SAA1 and low APOA4 levels in aneurysmal subarachnoid hemorrhage
Supplemental material, sj-pptx-2-jcb-10.1177_0271678X231184806 for HDL anti-inflammatory function is impaired and associated with high SAA1 and low APOA4 levels in aneurysmal subarachnoid hemorrhage by Zaida Ruiz de Azúa-López, M Rosa Pezzotti, Ángela González-Díaz, Olivier Meilhac, Juan Ureña, Rosario Amaya-Villar, Antonio Castellano and Lourdes M Varela in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Grant US-1381231 (Proyectos I + D+i FEDER Andalucía 2014–2020) and the Embassy of France (2021). LM Varela acknowledges financial support from VI-PPIT-US (University of Seville).
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Acknowledgements
The authors thank Joaquín Navarro-Rodríguez, María Luisa Gascón-Castillo, and Raquel García-Ríos for their help in blood collection, and Ma Eugenia Saéz-Goñi (CAEBi Bioinformática) for her help with proteomic analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Conceptualization, L.M.V., A.C., J.U., R.A.V.; data collection and analysis: Z.R-d.A.L., M.R.P., A.G.D., and L.M.V.; data interpretation: Z.R-d.A.L., M.R.P., A.G.D, O.M., J.U., R.A.V., A.C., and L.M.V.; writing-review and editing, Z.R-d.A.L., M.R.P., A.G.D, O.M., J.U., R.A.V., A.C., and L.M.V.; funding acquisition, L.M.V. and A.C. All authors read and approved the final manuscript.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
