Abstract
Cerebral hypoxic vasodilation is poorly understood in humans, which undermines the development of therapeutics to optimize cerebral oxygen delivery. Across four investigations (total n = 195) we investigated the role of nitric oxide (NO) and hemoglobin-based S-nitrosothiol (RSNO) and nitrite (
Introduction
Hypoxic vasodilation is a fundamental physiologic function conserved across mammalian species. 1 Integral to the maintenance of systemic oxygen (O2) delivery, hypoxic vasodilation is requisite to maintain cerebral homeostasis. A high oxygen demand and limited ability for storage of glycolytic substrate underlies the brain’s vulnerability to reductions in oxygen supply. Indeed, complete interruption of cerebral blood flow (CBF), and thus cerebral delivery of oxygen (CDO2) precipitates unconsciousness in as little as 4-seconds, with adverse consequences ensuing shortly thereafter. 2 Clinically, optimizing CDO2 remains an elusive target in the setting of acute ischemic brain diseases (e.g. focal ischemic stroke or global hypoxic-ischemic brain injury), while chronic impairments in cerebral perfusion have been associated with increased risk of neurodegenerative disease. 3 Delineation of the mechanisms regulating cerebral hypoxic vasodilation is necessary to determine future therapies for the optimization of cerebral perfusion.
Nitric oxide (NO) has been demonstrated as a key regulator of cerebral hypoxic vasodilation in animal models.4,5 S-nitrosohemoglobin, an NO moiety covalently bound to the cysteine 93 residue of the hemoglobin (Hb) β-chain, is now well described to subserve NO-mediated vascular signaling and is a fundamental regulator of hypoxic vasodilation.6,7 Allostery dependent, this Hb-based signaling mechanism functions to ‘protect’ NO during circulation
8
and facilitate export of vasodilatory S-nitrosothiols (RSNO) from the erythrocyte during gas exchange at the tissue level.7–9 Indeed, pre-clinical studies have established that RSNO release from hemoglobin during circulatory transit across a vascular bed occurs commensurate to oxygen demand.8,9 Hypoxic vasodilation may also be transduced through a nitrite (
Given the apparent multi-dimensional regulation of cerebral hypoxic vasodilation in humans, we conducted a series of four inter-related studies with the central aim of determining the regulation of cerebral hypoxic vasodilation by Hb and NO. We first aimed to determine the roles that RSNO and
Methods
We conducted four separate investigations wherein we examined: 1) the association between RSNOs and cerebral hypoxic vasodilation, 2) the NO-dependence of cerebral hypoxic vasodilation, 3) the association between [Hb] and cerebral hypoxic vasodilation, and 4) cerebral hypoxic vasodilation in high-altitude native Andeans with and without excessive erythrocytosis. This study adhered to the standards outlined in the Declaration of Helsinki (except registry in a database) and the Canadian Tri-council Policy Statement for Integrity in Research. All protocols and retrospective analyses were approved by the University of British Columbia’s Clinical Research Ethics Board (H16-01028; H18-01755; H18-01404; H18-01764), Duke University Institutional Review Board (Protocol: 00091879), and the Universidad Peruana Cayetano Heredia Comité de Ética (no. 101686). 14 All participants read an in-depth study information form, spoke with an investigator, and provided written informed consent prior to participation (in their native language). Some aspects of investigations 1 and 2 have been reported previously (e.g. blood gas data),15,16 however, the main outcomes reported herein and the hypotheses addressed hold no overlap.
Investigation 1 – Isovolumic hemodilution
Ten young, healthy males were recruited to participate (Age: 29 ± 7 years; body mass index, BMI: 23 ± 2 kg · m−2). Following instrumentation with radial arterial, jugular venous (see Supplemental Figure 1), and ante-cubital venous catheters, participants had ≥20 minutes to rest while the remaining experimental set-up for monitoring minute ventilation (V̇E), the partial pressures of end-tidal carbon dioxide (PETCO2) and oxygen (PETO2), heart rate (HR), radial artery mean arterial pressure (MAP), jugular venous blood pressure (JVBP), and cerebral perfusion pressure (CPP, i.e. MAP – JVBP) (see Supplemental Methods). Moreover, during each measurement period, vertebral (VA) and internal carotid artery (ICA) blood flow was quantified to determine CBF in conjunction with blood gas analysis (see Supplemental Methods). For each experimental manipulation, end-tidal forcing was utilized to control arterial blood gases. After 5-minutes of eucapnic and normoxic baseline measurements, each participant completed an isocapnic hypoxemia test with targeted arterial oxygen saturation (SaO2) values of 90, 85, and 75%. Isovolumic hemodilution was then performed, where 10% of whole blood volume was removed and replaced with an equal volume of 5% human serum albumin (Alburex® 5%, Canadian Blood Services) in two successive stages, resulting in a total removal and replacement of 20% of whole blood volume. Baseline (eucapnic normoxia) measures were acquired following the first stage of 10% blood removal/replacement, while a second isocapnic hypoxemia trial was conducted following the second stage of hemodilution (see Supplemental Methods). At each baseline and during each hypoxemia test, V̇E, PETCO2, PETO2, HR, MAP, JVBP, VA, ICA, and blood sampling measures were performed, with data averaged over the last minute of each stage.
Investigation 2 – Nitric oxide synthase inhibition
Eleven young, healthy males were recruited to participate (Age: 25 ± 5 years; BMI: 24 ± 3 kg/m2). Following instrumentation (consistent with Investigation 1), either a control (volume matched saline, 0.9% NaCl), NO synthase inhibitor (NG-monomethyl-L-arginine, L-NMMA) or α1-adrenoceptor agonist (phenylephrine, n = 5 subset) infusion was initiated (singled-blinded, counter-balanced design). The L-NMMA infusion was initiated with a 5 mg/kg bolus delivered over 5 min followed by a 50 μg/kg/min maintenance dose throughout the protocol until completion. Phenylephrine was infused at 0.1 to 0.6 μg/kg/min to serve as an equipotent vasoconstrictor control for the L-NMMA trial. 12 During each drug infusion we utilized end-tidal forcing to conduct an isocapnic hypoxemia test with a baseline period (eucapnic normoxia) followed by two hypoxic stages lasting 5-minutes each where SaO2 was targeted to 85% and 75%. At baseline and each stage of hypoxemia, V̇E, PETCO2, PETO2, HR, MAP, JVBP, MCA, PCA, VA, ICA, and blood sampling measures were performed.
Investigation 3 – Baseline hemoglobin concentration and cerebral hypoxic vasodilation
We conducted a retrospective analysis of nine experiments wherein participants were instrumented with a radial artery and jugular venous catheter and completed an isocapnic hypoxemia test. From these nine separate experiments conducted between 2004 and 2016, we assessed all data files (n = 199) with 134 participants included in our final analysis (79 male, 55 female; Age: 26 ± 6 years; BMI: 24 ± 3 kg/m2) (see Supplemental Methods for exclusion criteria). Concurrent arterial and jugular venous blood samples were acquired serially at each stage of hypoxemia, wherein hypoxia was induced utilizing a sequential gas delivery system (RespirAct®, Thornhill Medical, Toronto, Canada).
17
Percent changes in CBF were calculated based upon the Fick principle under the assumption that cerebral metabolism of oxygen remains constant during the level of hypoxemia employed in this study
18
(i.e., CBF ∝
Investigation 4 – cerebral hypoxic vasodilation in the setting of lifelong hypoxia
In order to isolate the role of elevations in [Hb] on cerebral hypoxic vasodilation, high-altitude native Andeans with (n = 15; all male; Age: 39 ± 15 years; BMI: 25 ± 3 kg/m2) and without (n = 25; 2 female; Age: 29 ± 12 years; BMI: 24 ± 3 kg/m2) excessive erythrocytosis (EE+/EE−) were recruited to participate in the Cerro de Pasco region of Peru (4,300 m above sea-level). Venous blood was collected for the measurement of Hct and [Hb], and CBF was measured with duplex ultrasound. Utilizing an end-tidal forcing system, an isocapnic hypoxemia test was performed in 15 participants, wherein PETO2 was targeted to 100 mmHg to restore SaO2 to normal (i.e. 98%), and then reduced to a PETO2 of 50 and 40 mmHg, with each stage lasting 5-minutes following the attainment of steady-state. At each stage, V̇E, PETCO2, PETO2, HR, MAP, VA, and ICA measures were made (see Supplemental Methods). Further, a subset of six EE+ males were instrumented with a radial artery catheter and completed an isovolumic hemodilution protocol (Age: 44 ± 19 years; BMI: 25 ± 3 kg/m2). Therein, 25% of blood volume was removed and replaced with human serum albumin (5%) as described in Investigation 1. Prior to and following hemodilution these participants underwent an isocapnic hypoxemia test as described above.
Statistical analyses
For all statistical tests, significance was set at an alpha level of α = 0.05. Data were assessed for normality using a Shapiro-Wilk test. Normal data are reported as mean ± standard deviation. When data significantly differed from a normal distribution, non-parametric analyses were employed and data are presented as median [interquartile range]. Across all investigations, pre vs. post intervention or between group comparisons were performed using two-tailed t-tests or non-parametric equivalents (Mann Whitney U test or Wilcoxon Signed Ranks test). Responses during hypoxemia tests prior to and following an intervention were made using linear mixed models or two-way ANOVAs. Finally, correlational analyses were conducted with Pearson r correlations or repeated measures correlations. 19 See Supplemental Methods.
Results
Investigation 1 – Cerebral hypoxic vasodilation is associated with trans-cerebral S-nitrosothiol release
We utilized our isovolumic hemodilution protocol to match changes in CaO2 between hypoxemic and anemic stages of hypoxia. Neither PaO2 (93.3 ± 4.0 vs. 92.4 ± 4.4 mmHg; P = 0.27) nor SaO2 (97.6 ± 0.4 vs. 97.6 ± 0.6%; P = 0.75) were altered pre-to post hemodilution (Figure 1(a) and (b)). However, the reduction in [Hb] from 14.2 ± 0.9 to 11.3 ± 0.5 mL/dL (P < 0.001) led to a 20 ± 2% reduction in CaO2 (19.3 ± 1.1 vs. 15.4 ± 0.7 mL/dL; P < 0.001) (Figure 1(c) and (d)). There was a 19 ± 7% increase in CBF following hemodilution (765 ± 105 vs. 912 ± 120 mL/min; P < 0.001).
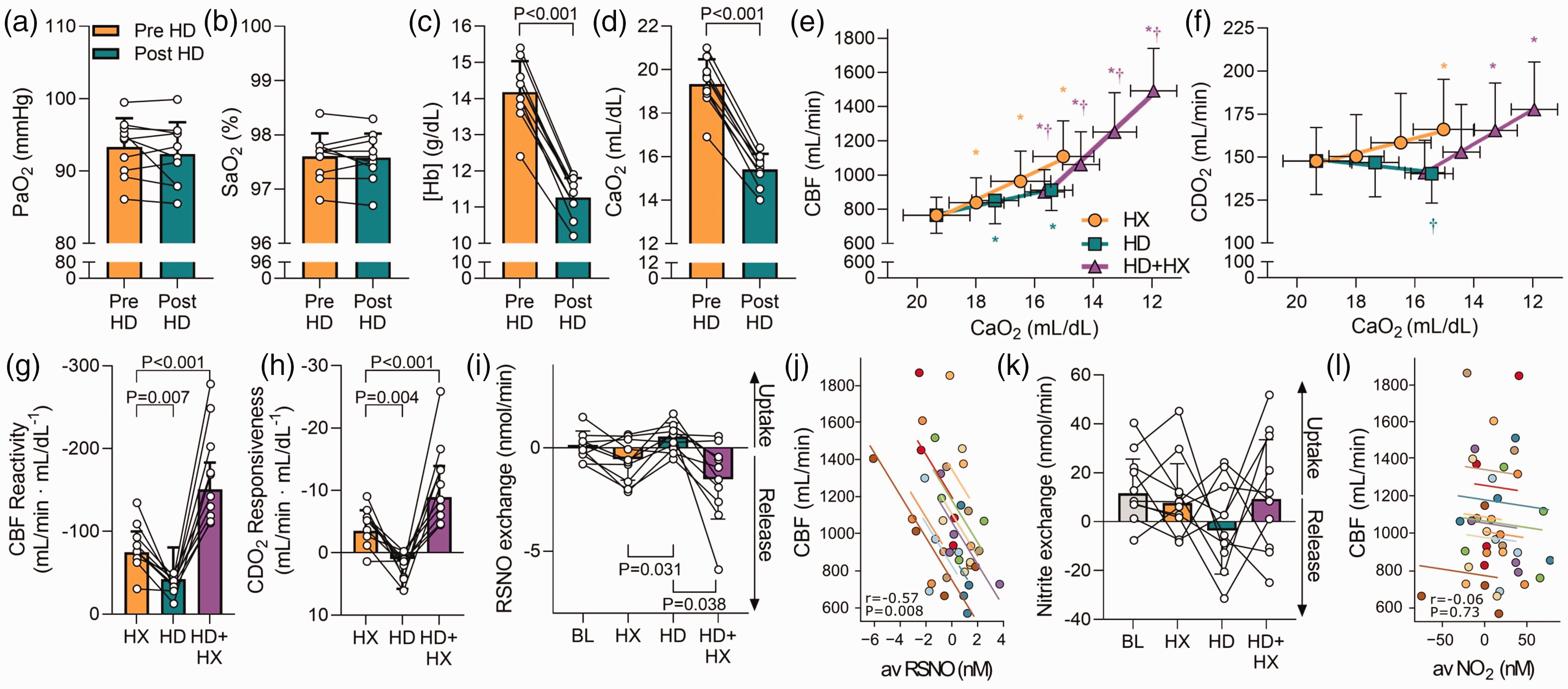
Cerebral hypoxic vasodilation is associated with trans-cerebral release of S-nitrosothiols. Panels (a–d) depict PaO2, SaO2, [Hb], and CaO2 at baseline pre- and post-hemodilution, respectively. Panel e depicts CBF during each stage across all trials. CBF increased at each stage of hypoxemia (HX), hemodilution (HD), and combined hemodilution and hypoxemia (HD + HX) compared to their respective eucapnic and normoxic baselines (P < 0.05 for all). Further, CBF was higher at each stage of combined HD + HX compared to each SaO2 matched stage of HX (P < 0.05 for all). Panel f depicts CDO2 during each stage across all trials. CDO2 was increased from baseline (148 ± 19 mL/min) at the third HX stage (166 ± 29 mL/min; P = 0.002), while CDO2 was lower at the third HD stage compared to the CaO2 matched HX stage (140 ± 17 mL/min; P = 0.001). Similarly, following hemodilution, hypoxemia increased CDO2 from the post-hemodilution baseline (141 ± 19 mL/min) at the second (166 ± 28 mL/min; P < 0.001) and third stage of hypoxemia (178 ± 28 mL/min; P < 0.001). Panels g and h depict the individual CBF and CDO2 slope responses to reductions in CaO2 across each trial, respectively. Panels i and k depict the trans-cerebral exchange of S-nitrosothiols (RSNO) and
The CBF and CDO2 responses during the three hypoxic tests are depicted in Figure 1(e) and (f). The CBF reactivity to hypoxemia prior to hemodilution was −74.8 ± 25.0 mL/min · mL/dL−1. Reactivity to anemic hypoxia (the hemodilution protocol) was −42.1 ± 38.4 mL/min−1 · mL/dL−1, ∼45% lower than that during hypoxemia (P = 0.007). Reactivity to hypoxemia following hemodilution was ∼100% greater than hypoxemia prior to isovolumic hemodilution (−150.6 ± 32.4 mL/min · mL/dL−1; P < 0.001; Figure 1(g)). Inclusion of CPP as a covariate did not alter these differences. The slope response of CDO2 (i.e. CDO2 reactivity) to hypoxemia prior to hemodilution was −3.5 ± 3.3 mL/min · mL/dL−1. Conversely, hemodilution caused a progressive decrease in CDO2 in concert with CaO2 (CDO2 reactivity, 1.0 ± 4.8 mL/min · mL/dL−1; P = 0.004). Indeed, CDO2 was lower during anemic hypoxia (140.3 ± 17.0 mL/dL) compared to hypoxemia (166.1 ± 29.2 mL/dL; P = 0.001; Figure 1(e)) despite matched CaO2. Finally, CDO2 reactivity to hypoxemia following hemodilution was ∼150% greater than pre hemodilution (−8.9 ± 5.0 mL/min · mL/dL−1; P < 0.001; Figure 1(h)).
Cerebral release of RSNO was greater during both the final stage of hypoxemia pre-hemodilution (−0.54 ± 1.03 nmol/min; P = 0.031) and final stage of hypoxemia post-hemodilution (−1.51 ± 1.92 nmol/min; P = 0.038) compared to the final stage of hemodilution (0.54 ± 0.66 nmol/min) (Figure 1(i)). This was reflected in a correlation
19
between the magnitude of the cerebral a-v RSNO difference and CBF (r = −0.57; P = 0.0080; Figure 1(j)). No differences in the cerebral net exchange of
Investigation 2 – Nitric oxide synthase inhibition reduces cerebral hypoxic vasodilation
Arterial plasma total NO (
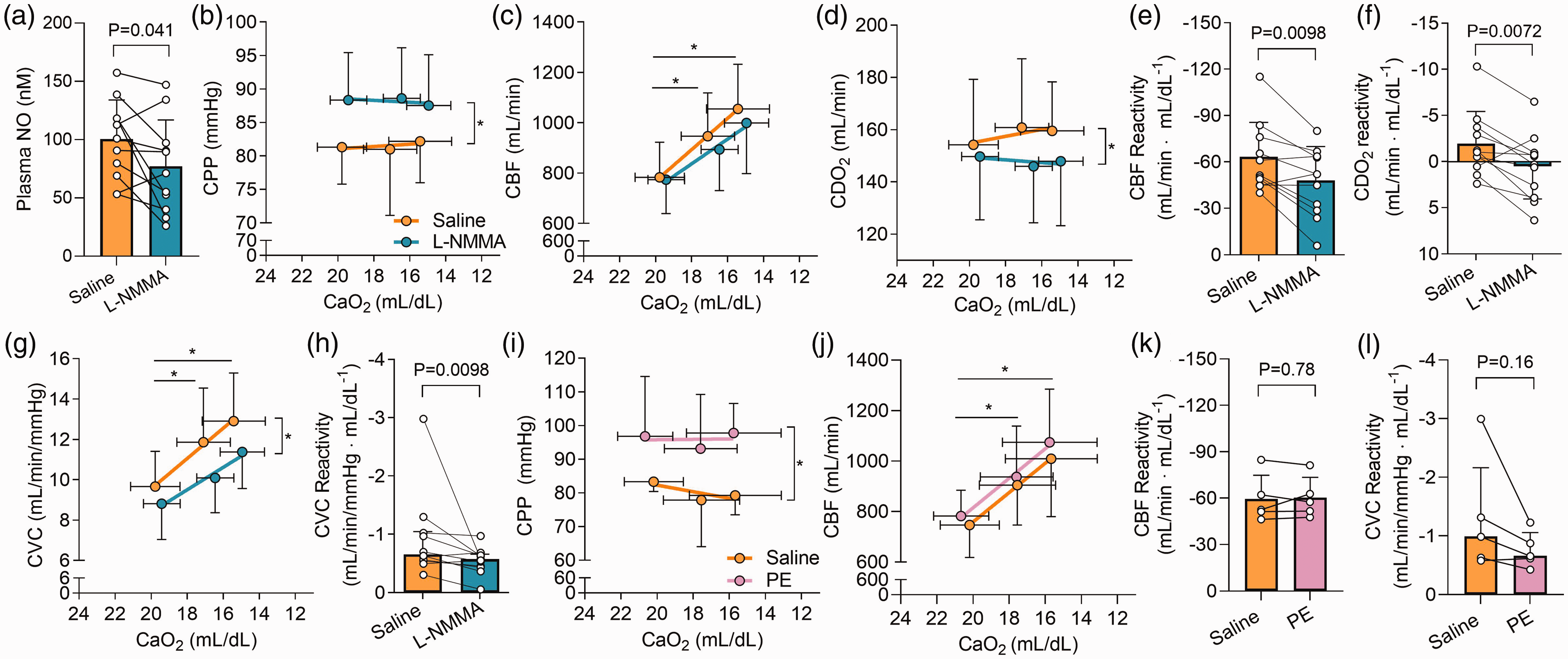
Nitric oxide synthase inhibition reduces cerebral hypoxic vasodilation in response to progressive hypoxemia. Panel a depicts plasma NO (
We assessed the influence of phenylephrine infusion, a vasoconstrictor control, on cerebral hypoxic vasodilation compared to saline. Phenylephrine led to a higher CPP compared to saline (P < 0.001; Figure 2(i)). However, while CBF was increased with hypoxemia (P < 0.001), there was no difference in CBF between saline and phenylephrine trials (P = 0.147; Figure 2(j)). Ultimately, neither CBF reactivity (−59.6 ± 15.3 vs. −60.4 ± 13.1 mL/min · mL/dL−1; P = 0.78; Figure 2(k)) or CVC reactivity (−0.99 [−2.16–−0.61] vs. −0.66 [−1.06–−0.53] mL/min/mmHg · mL/dL−1; P = 0.125; Figure 2(l)) were different between saline and phenylephrine trials.
Investigation 3 – Lower hemoglobin concentration is associated with increased cerebral hypoxic vasodilation
In Investigation 1, the CBF response to hypoxemia following hemodilution (i.e. during concurrent anemia) was greater than pre-hemodilution, indicating a modulatory role of [Hb] in the regulation of cerebral hypoxic vasodilation. To confirm this, we assessed the potential relationship between [Hb] and cerebral hypoxic vasodilation across a larger cohort of individuals. As we have previously demonstrated the stability of CMRO2 during equivalent hypoxemia, 18 and herein confirm in 23 participants (from studies 1 & 2; Figure 3(a)) that CMRO2 is unaltered with hypoxemia (P = 0.892), we are able to infer percent changes in CBF from changes in the a-vDO2 (see Supplemental Methods). Therefore, in 134 individuals (79 male, 55 female; see Supplemental Table 2 for details) we retrospectively analyzed arterial and jugular venous blood gas data to calculate a-vDO2 (Figure 3(b)) and percent changes in CBF during hypoxemia (Figure 3(c)). Males had a higher [Hb] (14.8 ± 1.0 g/dL) compared to females (13.1 ± 1.2 g/dL; P = 4.3e−14; Figure 3(d)), but lower cerebral hypoxic vasodilation (−6.9 ± 2.1 vs. −8.3 ± 2.7%ΔCBF · mL/dL−1; P = 0.0014; Figure 3(e)). Across all individuals we observed an inverse relationship between cerebral hypoxic vasodilation and [Hb] (r2 = 0.12; P < 0.01; Figure 3(f)). This relationship remained evident when males were assessed independently (r2 = 0.25; P < 0.01; Figure 3(f)); however, it was absent in the female participants (r2 = 0.01; P = 0.50; Figure 3(f)).
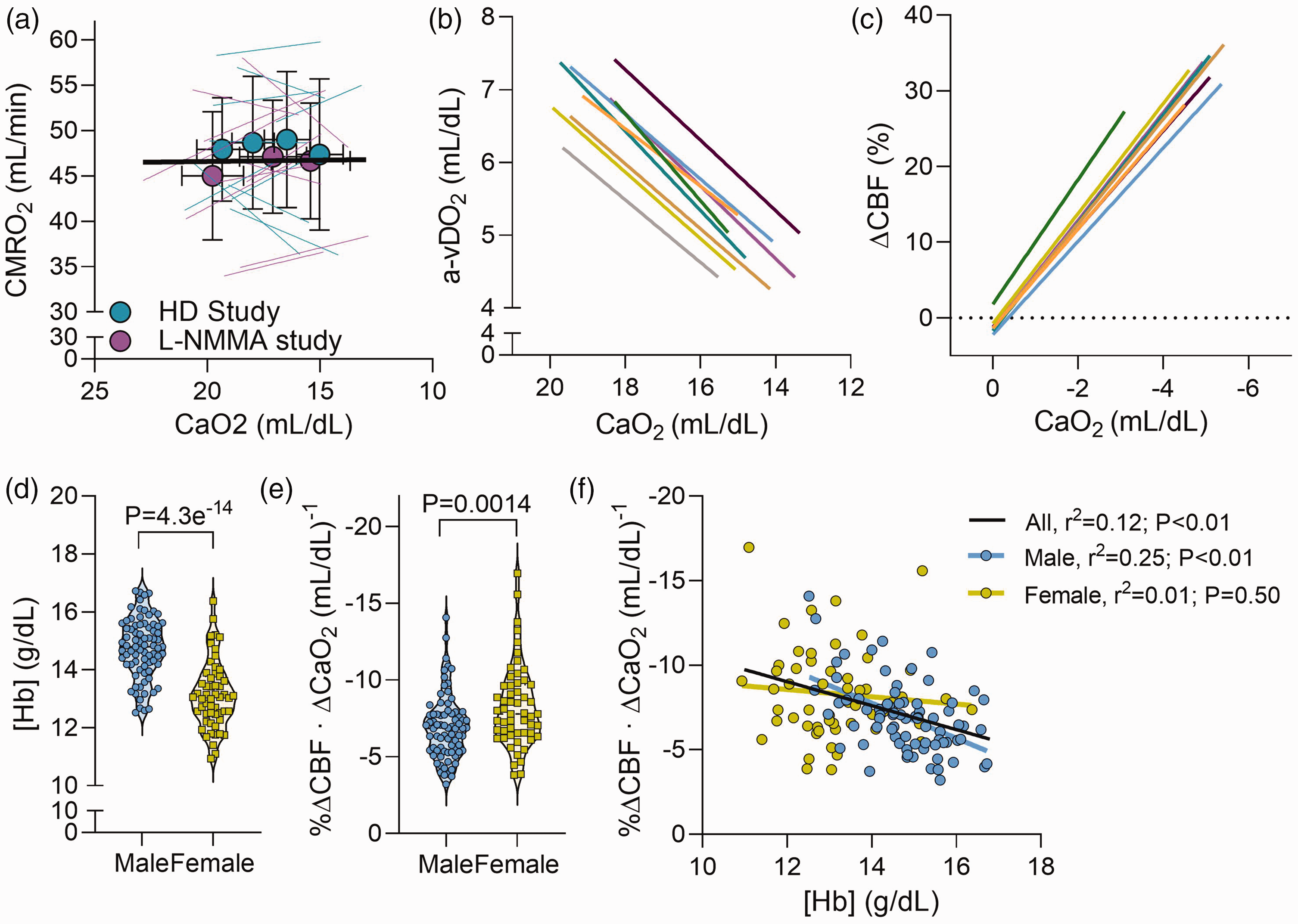
Association between hemoglobin concentration and cerebral hypoxic vasodilation. Panel a depicts the cerebral metabolic rate of oxygen (CMRO2) in the 21 participants combined from the hemodilution study (HD Study; cyan) and the nitric oxide synthase inhibition study (L-NMMA Study; magenta). Mean ± standard deviation is presented for each study, overlaid on individual participant responses. The black line represents the overall linear regression for the entire n = 21 sample (linear mixed model analysis). Panel b depicts the relationship between CaO2 and the a-vDO2 during progressive hypoxemia for each study included in this analysis (a different colour indicates a different study). These data are collated from nine separate studies conducted by our group over the last two decades (see Supplemental Methods). As per the Fick Principle (CMRO2 = a-vDO2 × CBF), in the setting of a constant metabolism, CBF is inversely proportional to a-vDO2 (a-vDO2 ∝ 1/CBF). Thus, the percent change in CBF can be determined, as depicted in Panel c. Panel d and e depict [Hb] and the magnitude change in CBF change per unit reduction in CaO2 (i.e. magnitude of cerebral hypoxic vasodilation), respectively for both males (blue symbols; n = 79) and females (yellow symbols; n = 55). The relationship between [Hb] and the magnitude of cerebral hypoxic vasodilation is depicted in Panel f. The black line represents the regression for all individuals, while the blue and yellow regressions are specific to the male and female participants, respectively. N=134. Comparisons without an associated p-value or symbol were not significant.
Investigation 4 – Reductions in hemoglobin concentration increase cerebral hypoxic vasodilation in patients with excessive erythrocytosis
To translate our findings from Investigations 1–3 to a clinical population we assessed the potential relationship between [Hb] and cerebral hypoxic vasodilation in high-altitude native Andeans with and without excessive erythrocytosis. 20 The EE+ participants had a [Hb] of 21.8 [21.4–23.9] g/dL while the EE- participants had a [Hb] of 18.7 [17.3–19.5] g/dL (P = 2.4e−9; Figure 4(a)), which corresponded to a higher CaO2 (P = 5.7e−6; Figure 4(b)). Neither CBF (516 [445–661] vs. 512 [465–682] mL/min; P = 0.93; Figure 4(c)) nor CDO2 (115 [99–138] vs. 123 [116–157] mL/min; P = 0.11; Figure 4(d)) differed between the EE− and EE+ groups. There was no correlation between CaO2 and CBF at rest (r = 0.133; P = 0.45; Figure 4(e)). However, in agreement with our observations in Investigation 3 (Figure 3(f)), we observed an inverse relationship between arterial [Hb] and CBF reactivity (r = −0.617; P = 0.014; Figure 4(f)).
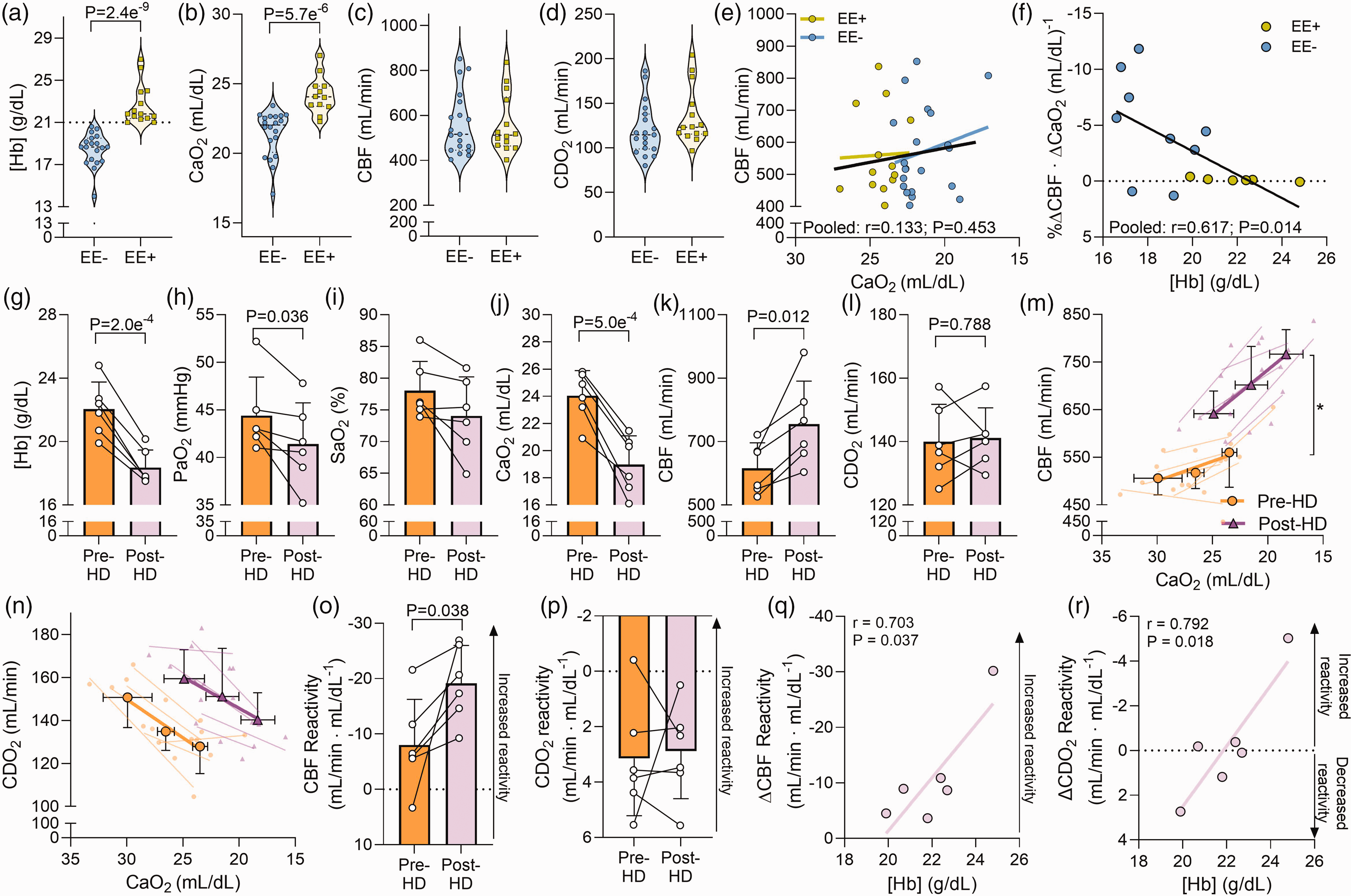
Isovolumic hemodilution increases cerebral hypoxic vasodilation in high-altitude Andeans with excessive erythrocytosis. Panels a–d depict violin plots of hemoglobin concentration [Hb], arterial oxygen content (CaO2), cerebral blood flow (CBF), and cerebral delivery of oxygen (CDO2) for high-altitude native Andeans without (EE−, n = 19) and with (EE+, n = 14) excessive erythrocytosis. Panel e depicts the relationship between CaO2 and CBF under resting conditions (r = 0.133; P = 0.45), while Panel f depicts the relationship between [Hb] and CBF reactivity in a subset of the Andean participants with (n = 6) and without EE (n = 9). Panels g to m depict bar graphs (mean ± standard deviation) with individual data overlaid for [Hb], the partial pressure of arterial oxygen (PaO2), arterial oxygen saturation (SaO2), CaO2, CBF, and CDO2, respectively, prior to and following isovolumic hemodilution (HD) in the EE+ group (n = 6). Panels m and n display CBF and CDO2 during graded hypoxemia (reduced CaO2) prior to and following isovolumic hemodilution, respectively and Panels o and p depicts the CBF and CDO2 reactivity, respectively, while Panels q and r depict the relationship between the change in CBF and CDO2 reactivity following hemodilution with the pre-hemodilution [Hb]. An asterisk (*) indicates a significant main effect of isovolumic hemodilution (P < 0.05). Comparisons without an associated p-value or symbol were not significant.
To assess the influence of reducing [Hb] on CBF reactivity, 6 of the EE+ patients underwent isovolumic hemodilution. This led to a reduction in [Hb] (Figure 4(g)) and CaO2 (Figure 4(j)), while SaO2 was unaltered (Figure 4(i)) despite a nominal reduction in PaO2 (Figure 4(h)). Isovolumic hemodilution was associated with a 22 ± 13% increase in CBF (616.1 ± 79.9 vs. 755.3 ± 135.2 mL/min; P = 0.012; Figure 4(k)) whereas CDO2 did not change (Figure 4(l)). In response to hypoxemia, CBF was increased prior to and following isovolumic hemodilution (main effect, P = 0.005; Figure 4(m)) while CDO2 decreased (main effect, P = 0.001; Figure 4(n)) both before and after hemodilution. However, comparison of reactivity slopes demonstrated that isovolumic hemodilution increased CBF reactivity by ∼140% (−8.0 ± 8.2 vs. −19.2 ± 6.8 mL/min · mL/dL−1; P = 0.038; Figure 4(o)), despite an unaltered CDO2 reactivity (P = 0.812; Figure 4(p)). Individual changes in CBF and CDO2 reactivity were correlated to pre-hemodilution [Hb], whereby a higher [Hb] prior to hemodilution was associated with a greater improvement in CBF and CDO2 reactivity following isovolumic hemodilution (Figure 4(q) and (r)).
Discussion
Our investigations demonstrate that cerebral hypoxic vasodilation is nitric oxide-dependent, associated with trans-cerebral release of S-nitrosothiols, and inversely related in magnitude to arterial hemoglobin concentration. Specifically, we demonstrated that: 1) greater trans-cerebral release of S-nitrosothiols during hypoxia is associated greater cerebral blood flow; 2) cerebral hypoxic vasodilation is reduced by 25% following nitric oxide synthase inhibition; 3) the magnitude of cerebral hypoxic vasodilation is inversely correlated to arterial hemoglobin concentration; 4) reducing arterial hemoglobin concentration in the setting of excessive erythrocytosis increases cerebrovascular reactivity to hypoxia. Collectively, our integrated approach implicates nitric oxide as a primary regulator of cerebral hypoxic vasodilation in humans.
To what extent NO contributes to cerebral hypoxic vasodilation in humans has remained equivocal. Albeit limited evidence, prior studies employing an NO synthase blockade have demonstrated reductions
13
or no impact
12
on cerebral hypoxic vasodilation, potentially due in part to technical limitations of measuring intra-cranial blood velocity and not flow. We provide clear evidence that NO synthase inhibition reduced cerebral hypoxic vasodilation (Figure 2). As the 23% reduction in plasma total NO with the L-NMMA infusion is not indicative of complete NOS inhibition, the observed 24% reduction in cerebral hypoxic vasodilation with L-NMMA likely underestimates the true magnitude by which NO contributes to cerebral hypoxic vasodilation in humans. This observation is consistent with our hemodilution study where we used differing hypoxic interventions of hypoxemia and hemodilution to elicit CaO2 matched levels of hypoxia with differing trans-cerebral RSNO release. We conducted an additional hypoxic test in the setting of acute anemia (i.e. combined hemodilution and hypoxemia) to investigate the influence of reductions in [Hb] on cerebral hypoxic vasodilation and trans-cerebral RSNO release. During these studies we simultaneously collected arterial and jugular bulb blood samples to quantify the arterial-to-venous differences of NO species (RSNO &
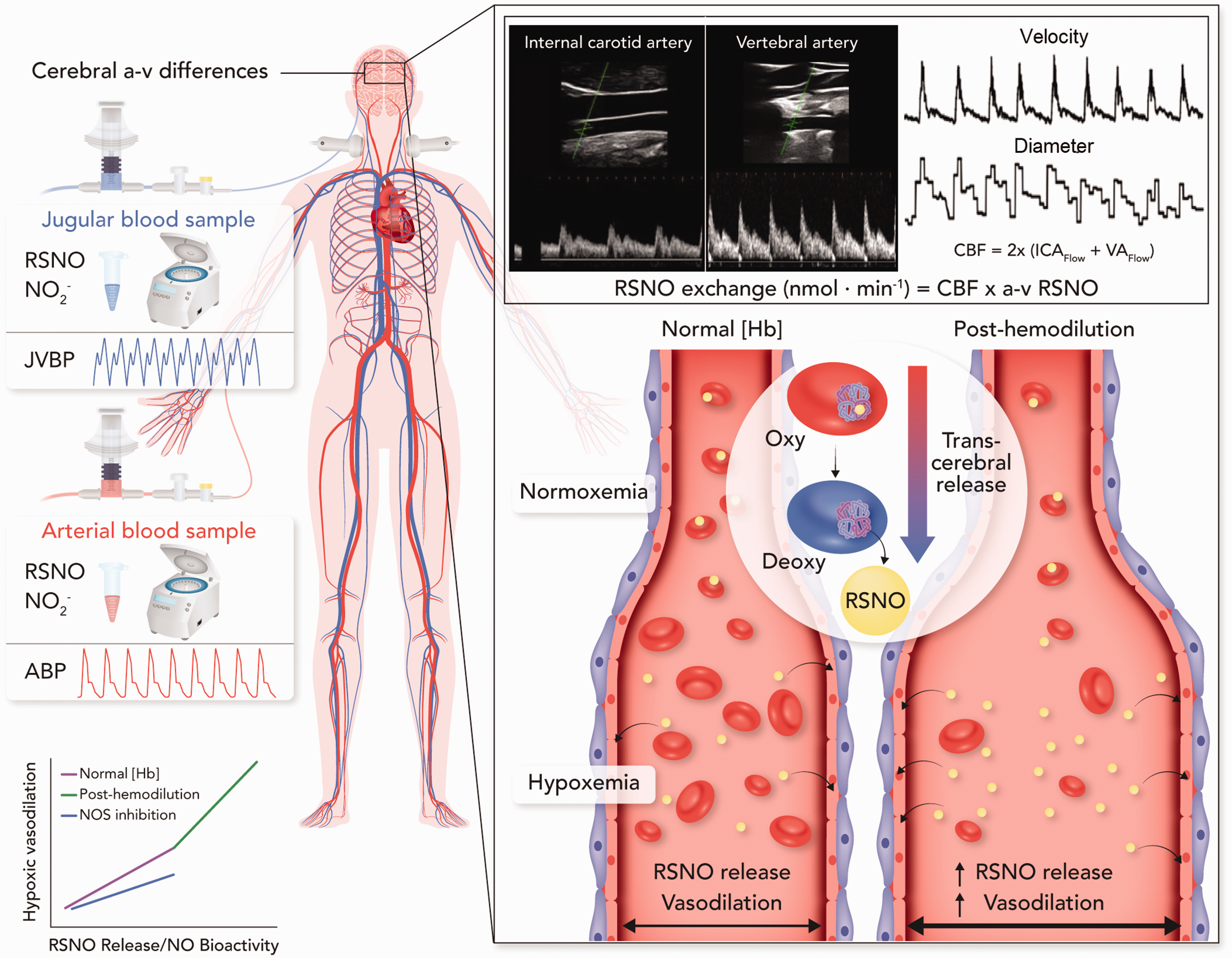
Assessing trans-cerebral exchange kinetics in humans. This schematic depicts our experimental model for assessing the trans-cerebral exchange of signaling molecules (e.g. nitric oxide) in humans. Cerebral arterial-to-venous (a-v) differences were quantified using concurrent arterial and jugular bulb blood sampling. Coupling of the a-v difference of S-nitrosothiols (RSNOs) and cerebral blood flow (CBF) measures acquired with duplex ultrasound allows for the quantification of the molar exchange of RSNOs during cerebral circulatory transit. This model of simultaneous arterial and jugular bulb blood sampling allows for experimental isolation of the cerebrovascular bed during a systemic stressor such as arterial hypoxemia or hemodilution and a more specific assessment of the signaling pathways involved in cerebrovascular control. In the present study we investigated the trans-cerebral exchange kinetics of RSNOs and
The observation that the magnitude of cerebral hypoxic vasodilation was associated with the magnitude of trans-cerebral RSNO release is in agreement with work by Stamler’s group.
7
SNO-Hb-mediated hypoxic vasodilation has been demonstrated in pre-clinical ex vivo
22
and in vivo models,8,9 and verified with genetic knockouts.1,23 In humans, however, we saw no such relationship between trans-cerebral exchange of
While the focus of our investigations was to assess the role of NO in regulating cerebral hypoxic vasodilation, the role of other prototypical vasodilators should not be disregarded. Previous studies conducted in humans have largely demonstrated that neither adenosine, 31 prostaglandins,32–34 or reactive oxygen species,35,36 are obligatory for cerebral hypoxic vasodilation. However, Harrell and colleagues recently demonstrated that combined prostaglandin synthesis inhibition and ascorbic acid infusion results in a marked blunting of cerebral hypoxic vasodilation in humans (albeit with transcranial doppler blood velocity estimates of CBF), highlighting that ‘redundancy’ of vasodilatory pathways may underscore the lack of effect from vasodilator pathway blockades typically observed in human studies. 35 Moreover, ATP sensitive potassium channels, which are influenced downstream of NO signaling by cyclic guanosine monophosphate, have been demonstrated to contribute to cerebral hypoxic vasodilation in humans. 37 That ATP sensitive potassium channel blockade reduces cerebral hypoxic vasodilation further supports the role of NO observed in our study, although its effect may have manifested through the multiple other pathways that converge on ATP sensitive potassium channels. 38 Therefore, while it is difficult to disentangle vasodilatory mechanisms in humans given the multiple levels of cross-talk that occur downstream of the ‘surface level’ pharmacological blockades that are safe to employ (e.g. adenosine receptor antagonism or prostaglandin synthesis inhibition), 38 our results link physiologic changes in cerebral hypoxic vasodilation with alterations in NO bioavailability and trans-cerebral RSNO release in humans.
Following our observation that acute anemia (i.e. isovolumic hemodilution) augments the magnitude of cerebral hypoxic vasodilation for a given reduction in CaO2 (Figure 1(g)), and that this augmented cerebral hypoxic vasodilation was linked to greater trans-cerebral RSNO release (Figure 1(i) and (j)), we investigated this relationship further. Consistent across two separate investigations, we determined that a lower Hb concentration is associated with increased cerebral hypoxic vasodilation in both lowlanders (Figure 3(f)) and Andean highlanders (Figure 4(f)). Further, and consistent with investigation 1, an acute reduction in [Hb] (i.e. isovolumic hemodilution) led to an increase in cerebral hypoxic vasodilation in Andean highlanders with excessive erythrocytosis (Figure 4(o)). In both isovolumic hemodilution studies (investigation 1 & 4), the augmented cerebral hypoxic vasodilation may be explained by the greater level of hypoxia, which coincides with potentiated RSNO-mediated vasodilation8,22 as well as greater trans-cerebral RSNO release as observed herein (Figure 1(i)). Moreover, a lower [Hb] may be related to an improvement in NO-mediated signal transduction due to reduced NO scavenging by hemoglobin.39–41 However, we contend that it is unlikely that differences in blood viscosity associated with [Hb] underscore the differences in cerebral hypoxic vasodilation we observed (as reviewed in 42 ).43–46
It is important to note that the relationship between Hb concentration and cerebral hypoxic vasodilation was only observed in males (Figure 3(f)). As investigations 1 and 2 were only conducted in males we are unable to discern if this is due to differences in the NO-dependence of cerebral hypoxic vasodilation between males or females or differing impacts of Hb levels on trans-cerebral RSNO release. Moreover, as menstrual phase was not standardized (or recorded) for investigation 3, it is unknown if the lack of relationship between cerebral hypoxic vasodilation is due to variability in female sex hormones (with potential impacts on NO signaling47–49). Nonetheless, this provides a more representative cross-section of female sex hormone profiles and thus a more generalizable sample of females than if inclusion was restricted to one menstrual phase (e.g. low circulating hormone phase as is common). Despite the lack of control over the menstrual phase, we observed a ∼20% greater cerebral hypoxic vasodilation in females when compared with males. As CaO2 will be lower for a given SaO2 in the females compared to males (due to lower [Hb]), the greater cerebral hypoxic vasodilation in females may be related to potentiated RSNO-mediated vasodilation.8,22
Given the low rate for which pre-clinical studies translate to the development of efficacious clinical therapies (<5%),50,51 demonstrating that an integral physiologic function is modifiable in humans, in this case by NO, is a key step in translation. Multiple cerebral hypoxic vasodilation signaling pathways have been elucidated in ex vivo and pre-clinical experiments (e.g. adenosine, prostaglandins) but when these studies are replicated (as much as one can) in humans, no effect is observed. If the manipulation of a signaling pathway is unable to modify an associated physiologic function in the context of human biology, then a treatment effect in clinical patients is unlikely. Our data provides a bridge between pre-clinical and clinical studies, whereby, in humans we demonstrate that cerebral hypoxic vasodilation is modifiable through changes in NO signaling. This sets a precedent for exploring NO-based therapies in disease populations where resolution of impairments in cerebral perfusion, acute or chronic, is desired (e.g. focal ischemic stroke or global hypoxic-ischemic brain injury).
Conclusion
We determined that the magnitude of cerebral hypoxic vasodilation in humans is linked to trans-cerebral release of S-nitrosothiols. This implicates the erythrocyte as an integral regulator of cerebral blood flow. We further determined this vasodilation to be nitric oxide-dependent, observing a reduction in cerebral hypoxic vasodilation following nitric oxide synthase inhibition. Finally, we demonstrate during both acute experimental anemia, and when comparing across a cohort, that a lower hemoglobin concentration is related to increased cerebral hypoxic vasodilation. Collectively, these results indicate that hemoglobin and nitric oxide hold key roles in the regulation of cerebral hypoxic vasodilation in humans. That changes in nitric oxide-dependent signaling elicit changes in cerebral hypoxic vasodilation in humans indicates the potential feasibility of clinically modifying cerebral perfusion by manipulation of these pathways and thereby provides a promising therapeutic target for the treatment of brain diseases associated with acute or chronic impairments in cerebral perfusion.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231169579 - Supplemental material for Hemoglobin and cerebral hypoxic vasodilation in humans: Evidence for nitric oxide-dependent and S-nitrosothiol mediated signal transduction
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231169579 for Hemoglobin and cerebral hypoxic vasodilation in humans: Evidence for nitric oxide-dependent and S-nitrosothiol mediated signal transduction by Ryan L Hoiland, David B MacLeod, Benjamin S Stacey, Hannah G Caldwell, Connor A Howe, Daniela Nowak-Flück, Jay MJR Carr, Michael M Tymko, Geoff B Coombs, Alexander Patrician, Joshua C Tremblay, Michelle Van Mierlo, Chris Gasho, Mike Stembridge, Mypinder S Sekhon, Damian M Bailey and Philip N Ainslie in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Heart and Stroke Foundation of Canada Grant in Aid (G-18-0022304) and Canada Research Chair in Cerebrovascular Physiology (PNA).
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgements
We would like to thank our research participants for volunteering their time and energy to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
Study Conception: RLH, DBM, DMB, PNA; Data collection: RLH, DBM, BSS, HGC, CAH, DN-F, JMJRC, MMT, GBC, AP, JCT, MvM, CG, MS, MSS, DMB, PNA; Data analysis: RLH, DBM, BSS, MvM; Data interpretation: RLH, DBM, DMB, PNA; Drafted the manuscript: RLH; Revised the manuscript for critical intellectual content: RLH, DBM, BSS, HGC, CAH, DN-F, JMJRC, MMT, GBC, AP, JCT, MvM, CG, MS, MSS, DMB, PNA; Approved the final version: RLH, DBM, BSS, HGC, CAH, DN-F, JMJRC, MMT, GBC, AP, JCT, MvM, CG, MS, MSS, DMB, PNA.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
