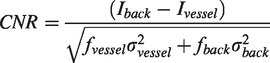Abstract
Cortical visualization is essential to understand the dynamic changes in brain microenvironment under physiopathological conditions. However, the turbid scalp and skull severely limit the imaging depth and resolution. Existing cranial windows require invasive scalp excision and various subsequent skull treatments. Non-invasive in vivo imaging of skull bone marrow, meninges, and cortex through scalp and skull with high resolution yet remains a challenge. In this work, a non-invasive trans-scalp/skull optical clearing imaging window is proposed for cortical and calvarial imaging, which is achieved by applying a novel skin optical clearing reagent. The imaging depth and resolution are greatly enhanced in near infrared imaging and optical coherence tomography imaging. Combining this imaging window with adaptive optics, we achieve the visualization and manipulation of the calvarial and cortical microenvironment through the scalp and skull using two-photon imaging for the first time. Our method provides a well-performed imaging window and paves the way for intravital brain studies with the advantages of easy-operation, convenience and non-invasiveness.
Keywords
Introduction
In vivo non-invasive imaging of cerebral vascular structure and dynamical observation of its functions are critical to understand normal brain physiology as well as further progression of microvascular dysfunction and neurodegeneration in various brain diseases.1,2 However, the turbid scalp and skull severely limits the optical imaging performance. Generally, researchers need to consecutively implement the scalp removal and skull thinning 3 or removal4,5 before imaging. Such processes are invasive and detrimental because the skull has been verified to be a reservoir for supplying immune cells that mediate brain immune surveillance.6,7 And there exist direct vascular channels between the brain and the skull bone marrow through the meninges for cell migration.7 –9 The damages to the skull bone marrow and dura mater thus breaks the channels and disrupts the brain immunity integrity, which may lead to misinterpretations of events related to brain diseases.10 –12
The tissue optical clearing technique provides a promising way for improving the imaging qualities.13,14 Various optical clearing skull windows have been proposed for realizing the visualization of calvarial and cerebral microenvironment with high resolution,15 –17 which are achieved by using specific chemical solutions to treat the skull. These methods have been demonstrated to be safe that they do not cause any side effects to the brain. Thus, optical clearing skull window is a promising approach for intravital cortical imaging in neuroscience research. 18 However, scalp removal and skull exposure for establishing the optical clearing skull window inevitably causes an invasion of the body. Besides, in vivo skull optical clearing techniques can realize imaging of the cortex through the skull, but the resolution and signal intensity severely decrease with depth as a result of cleared tissue scattering and optical aberrations. 19 Thus, there still exists a challenge to fully non-invasively monitor the microenvironment of skull bone marrow, meninges and cortex through the intact scalp and skull with high resolution.
In this work, we developed a novel trans-scalp/skull clearing window (TSSCW) based on the in vivo tissue optical clearing technique, compatible with visible (Vis)- and near-infrared (NIR) imaging. TSSCW significantly improves the NIR imaging performance, and enables optical coherence tomography (OCT) imaging to observe the microvasculature through the scalp and skull with high resolution. We further implement the adaptive optics (AO) devices in the two-photon imaging system to eliminate aberrations from the tissue. With the assistant of TSSCW, the calvarial/cortical vessels and cells are clearly observed, and the cortical microglia cells can be manipulated precisely, whereafter the dynamic changes of microglia cells are recorded. Our technique for the first time provides an imaging window that enables visualization of calvarial and cortical microenvironment through both scalp and skull with high resolution. We consider it a powerful tool in broad intravital animal imaging studies.
Materials and methods
Animal
The experimental procedures were performed with the approval of the Guangdong Medical University Animal Ethics Committee (Approval number: GDY2002085), and experiments were conducted in compliance with the animal experiment guidelines of the Experimental Animal Management Ordinance of Guangdong Province, P. R. China and General Requirements for Laboratory Animal Experiment of P. R. China (GB/T 35823-2018). The researchers are approved by the Institutional Animal Ethics Committee of Central People's Hospital of Zhanjiang. All animal experiments data are analyzed and reported according to ARRIVE 2.0 guidelines. 20 Cx3cr1EGFP/+ transgenic mice (Genetic background: C57BL/6, male, 3.5 weeks old) bought from Jarvis Bio-pharmaceutical Co., LTD (Wuhan, China) are applied for microglia/monocytes imaging. The wide type BALB/c mice (male, 10 weeks old) are used for NIR-II imaging and OCT imaging. The mice are fed under the specified pathogen free (SPF) condition, with 12 h/12 h light and dark cycle. During the experiments, the mice are anesthetized by using 4% isoflurane for induction, and 1.5% for maintenance.
Preparation of optical clearing agents (OCA)
The saturated fructose (78.9%, wt/wt) (Sinopharm, China) and sucrose (67.1%, wt/wt) (Sinopharm, China) solutions are prepared by dissolving them into water (H2O) and deuteroxide (D2O, Sigma-Aldrich, 151882), respectively. The reagent is made up of sucrose and PEG-400 (analytically pure, Sinopharm, China) in proportion of 9:1(v/v).
Evaluation of optical clearing efficacy of reagents
After hair removal, fresh mouse scalp samples are harvested from 7-week-old male BALB/c mice (n = 19), which are treated with saline and different optical clearing agents (fructose, sucrose and sucrose + PEG-400) for 15 min, respectively. Then they are put on the 1951 USAF target and imaged.
To quantitatively evaluate the optical clearing efficacy of different reagents on excised skin, the resolution of 1951 USAF target (United States Air Force) through the skin samples is calculated according to the Rayleigh criterion. 21 For each element, the intensity profile along the bar is analyzed. The bars and the spaces in the resolution target corresponds to the intensity valleys and peaks. If the intensity at the valley region is no more than 8/π2 times of that at the peak region, we consider the bar can be distinguished, and the corresponding optical resolution is subsequently determined.
Measurement of the transmittances of OCA and mouse scalp
The transmittances of the optical clearing agents are measured by using the spectrophotometer (Lambda1050+, PerkinElmer, USA). First, we obtain a transmittance baseline using a blank cuvette. The transmittances of H2O, D2O, fructose (H2O), sucrose (H2O), sucrose (D2O) and sucrose (D2O)+PEG 400 are successively detected within the range of 400 nm–2500 nm (step size: 5 nm).
Similarly, we measured the transmittances of saline and sucrose (H2O) + PEG 400 treated mice scalp that derived from telogen (∼7 weeks old), and anagen (∼6 weeks old) BALB/c (n = 10) and C57BL/6 mice (n = 10), respectively. The blank holding plate is used to measure the transmittance baseline. Following that, the scalp samples are placed on the plate for measurement through the optical range of 400 nm–1400 nm (step size: 10 nm).
Preparation of TSSCW
The skin state is critical for establishment of TSSCW. We choose the mice whose skin are at the telogen phase (∼3.5 weeks or ∼7–10 weeks old), which looks pink and touches thin. After the isoflurane anesthesia, the residual hair on the scalp is removed with depilatory cream (sensitive hair removal cream, Veet), and the residue is cleaned by scrubbing with alcohol. The stratum corneum in the region of interest (ROI) is removed gently by tape stripping 2 to 3 times. The scalp is subsequently flattened as much as possible, and a custom-made circular tinfoil holder is glued to the skin (Ergo 5400, Switzerland).
The optical clearing agent (OCA) is the mixture of saturated sucrose (D2O) and PEG-400 with the volume ratio of 9:1(v/v). The OCA is applied on the ROI. About 15 min later, the scalp becomes transparent, and the TSSCW is fully established for imaging. Here, a stereotaxic apparatus (TwInter-MSA, TimWinter (Beijing) Science and Technology Ltd., China) is used to immobilize the mouse, which can effectively reduce the movement induced by breathing and the heart beats of mice during the imaging procedure. After the experiment, OCA is removed, and the ROI is washed by saline repeatedly. About 10 min later, the ROI almost recovers from the procedure. For repeated imaging, we smear the Vaseline on ROI to keep skin moisturizing. The applied Vaseline has to be removed before OCA treatment in next imaging session.
NIR-II imaging of brain
The NIR-II imaging system (NIR II MS, Sunny Optical Technology (Group) Co., LTD., China) is employed for brain imaging. It is equipped with four objectives: 1 × (NA = 0.02, WD = 148 mm, 900–1700 nm), 5 × (NA =0.15, WD = 19.8 mm, 450–1700 nm), 10 × (NA = 0.3, WD = 1.3 mm, 450–1700 nm) and 20 × (NA = 0.45, WD = 7.3–7.6 mm, 450–1700 nm). It also comes with an 808 nm laser and a 1000 nm long-pass (LP) filter. 0.2 mL bright NIR-II emission nanoparticles TTQiT ((4,4′-(5,5′-([1,2,5] thiadiazolo [3,4-i] dithieno [2,3-a:3′,2′-c] phenazine -8,12-diyl) bis (4-octylthiophene-5,2-diyl)) bis (N, N-diphenylaniline) (1 mg/mL) is administered intravenously for vascular labelling.
22
A 5-minute circulation is required before imaging. The image processing is performed by using ImageJ, and the contrast-noise ratio (CNR) of images is used to evaluate the imaging performance. The CNR is defined as the following:
23
OCT imaging of cerebral microvessels
A spectral domain OCT system (TEL221PSC1, Thorlabs, USA) is employed for cerebral vascular imaging. The light source has a central wavelength of 1300 nm and a full width at half maximum bandwidth of 100 nm. It is equipped with a single objective (OCT-LK3, 5X) for probing light beam on the ROI, which could offer a high axial resolution of 5.5 μm and a lateral resolution of 13 μm in air. The vascular map can be obtained by using the mode of speckle variance angiography, and the OCT angiographic images are processed by using ImageJ for vascular reconstruction. Vessel density is obtained by AngioTool vascular analysis software. And the CNR of images are calculated for assessing the imaging performance.
Wavefront-sensorless adaptive optics
We used an AO device (AOL1821A, Dynamic Optics Srl, Italy) to eliminate aberrations. It is installed in a two-photon microscope through an additional refractive adaptive element behind the microscope objective (Figure S1(a)). A stand-alone control software performs optimization based on the acquired images. The algorithm for sensorless AO optimization is based on the Zernike polynomials 24 (Figure S1(b)). The basic idea is to scan each Zernike mode 25 with the lens deformation, and find the maximum for best correction. For each scanning mode, the best correction is reserved based on image metric information. Once the whole process is finished, we will obtain an optimal imaging configuration that minimizes image aberrations. The acquisition parameters for imaging fluorescent beads are set to: 1024 × 1024 pixels, 0.06 μm/pixel; Excitation: 920 nm, Detection: 575 ±12.5 nm, Objective: 25 × W, 1.1 NA, Zoom factor: 7.752. For cerebral blood vessel and microglia imaging, the acquisition parameters are set as: 1024 × 1024 pixels, 0.44 μm/pixel; Excitation: 920 nm, Detection: 575 ± 12.5 nm and 525 ± 25 nm, Objective: 25 × W, 1.1 NA, Zoom factor: 1 or 2. The acquisition and processing of image datasets are handled by the software of NIS and ImageJ.
Two-photon imaging of the cortical and calvarial microenvironment
The Cx3cr1EGFP/+ mice (male, ∼3.5 weeks) are used to in vivo monitor the microglia/monocytes activation both in cerebral cortex and skull marrow cavity. And the two-photon microscope (A1+, Nikon, Japan) together with a Ti: sapphire laser (Chameleon Vision II, Coherent, USA) is used in cortical and calvarial microenvironment imaging. Image stacks are obtained with a step size of 2 μm using a water-immersion objective (25×, 1.1 NA, WD 2.0 mm, Nikon). The commercial NIS software controls the instrumentation during acquisition, processing and storage of image datasets. ImageJ is used for the image analysis. After TSSCW establishment, 100 μL tetramethyl rhodamine-conjugated dextran (3 mg/mL, 70 kDa, Sigma Aldrich, USA) is injected through the tail vein for blood vessels labelling. For two-photon imaging of cortical and calvarial bone marrow microenvironment through the scalp and skull, imaging parameters are set to: 1024 × 1024 pixels, 0.5 μm/pixel; Z-step: 2 μm, Excitation: 920 nm, Detection: 492 SP, 525 ± 25 nm, 575 ± 12.5 nm, Zoom factor: 1.
Laser lesion model
A highly localized injury is achieved by focusing a two-photon laser beam in the superficial layer of the cortex through scalp and skull. The ND stimulation function in two-photon microscope (A1+, Nikon, Japan) is applied. The wavelength is set to 800 nm and the laser power is 60–80 mW. Besides, the injured area is set to 17.4 μm × 17.4 μm, located at the depth of 200–300 μm below the skull. The scan speed is 1 s/frame, and 6 loops are applied to create a small injury site. Imaging parameters are set to: 1024 × 1024 pixels, 0.3 μm/pixel; Excitation: 920 nm, Detection: 525 ± 25 nm, 575 ± 12.5 nm, Objective: 25 × W, 1.1 NA, Zoom factor: 1.645.
Monitoring the diffusion of fluorescent dye
Here, we used the rhodamine (average mol wt 155,000, Sigma-Aldrich, USA) as the indicator to evaluate the permeability of OCA in 10-week-old male BALB/c mice. The mixture of 2 μL rhodamine (1 mg/ml) and 0.5 mL OCA is topically applied to the ROI. When the ROI becomes transparent, we monitored the distribution of dye molecules in the scalp. The NIS software controls the instrumentation during acquisition, processing, and storage of image datasets. The acquisition parameters for two-photon imaging are set to: 512 × 512 pixels, 0.99 μm/pixel; Z-step: 2 μm, Excitation: 920 nm, Detection: 492 SP, 575 ± 12.5 nm; Objective: 25 × W, 1.1 NA, Zoom factor: 1.
The establishment of open-skull glass window and thinned-skull window
The open-skull glass window 4 and thinned-skull window 3 are established following the standard protocols as previously reported. For the open-skull glass window, the anesthetized mice underwent hair and scalp removal is stabilized in the stereotaxic frame firstly. Then a small piece of skull (∼3 mm) above the brain cortex is removed by using a dental drill, and a small amount of melted 1.2% agarose (Sigma-Aldrich, A9793) is applied to the exposed cortex. Followingly, the window is sealed by a coverslip (∼4 mm) using the cyanoacrylate and dental cement all around it. For establishment of the thinned-skull window, the scalp of depilated mice is dissected along the middle line for exposing the skull. Then, a small area (∼3 mm) of skull is polished to ∼25 μm using the dental drill and microsurgical blade. Finally, the scalp is sutured with 6-0 silk.
Evaluating cranial windows safety
10-week-old male Cx3cr1EGFP/+ and BALB/c mice are respectively used in microglia and astrocyte imaging for evaluating the safety of different cranial windows. The mice are repeatedly treated by OCA once a day for one week, or undergo partly skull thinning or removing and recover for one week. The mice are deeply anesthetized, given bilateral thoracotomy, and transcardially perfused with ice-cold phosphate buffer solution (PBS) followed by 4% formaldehyde (PFA). The brain is taken out and post-fixed in 4% PFA overnight at 4°C. For Cx3cr1EGFP/+ mice, the brains are sliced into 50 μm in the coronal plane for imaging. As for BALB/c mice, 50 μm coronal brain sections are successively immunofluorescent stained by primary antibody (GFAP polyclonal antibody, 1:100, Invitrongen) and secondary antibody (A-21244, 1:500, Invitrongen) for glial fibrillary acidic protein (GFAP) labelling. The confocal microscope (A1-SHR-LFOV, Nikon, Japan) is employed for images acquisition.
Statistical analysis
Statistical comparisons are done by using GraphPad (Prism 8, GraphPad Software, Inc., CA, USA)). The sample sizes and other statistical information are specified in the figure legends. The evaluation of repeated imaging performance based on vessels density and CNR are performed, shown as box plots with 10th and 90th percentiles. The normality of data distribution in each group is checked by Shapiro-Wilk test. For comparison among multiple groups, the P values are calculated by using one-way ANOVA. For comparison between two groups, the P values were calculated using by two-tail t-test. In this work, P value >0.05 is considered to have no significant difference (N.S.: no significance).
Results
Screening tissue optical clearing reagent for Vis-NIR imaging window
Sugar molecules, rich in hydroxyl groups, have excellent optical clearing potentials in skin tissue, in which the fructose and sucrose have been widely used. Here, we compared the clearing capability of saturated fructose and sucrose solutions. The excised scalp samples are immersed in different reagents, including saline, saturated fructose, saturated sucrose, and saturated sucrose mixed with penetration enhancer-PEG-400 (volume ratio: 9:1) for 15 min, and the images of skin-covered resolution target are recorded as shown in Figure 1(a). The images show that the sucrose has a better skin optical clearing capability than the fructose. Additionally, with the blending of PEG-400, the optical clearing effect of sucrose is improved further. To quantitatively characterize the clearing efficacy of different reagents, the optical resolutions of the USAF target through the skin are evaluated according to the intensity profiles as shown in Figure 1(b). The resolution of USAF target through the skin treated by sucrose is significantly smaller than fructose (P < 0.001), while the smallest resolution corresponds to the group of skin treated by sucrose + PEG-400 (Figure 1(c)) (P < 0.001). Therefore, the sucrose +PEG-400 is the optimal optical clearing agent for the scalp among them.
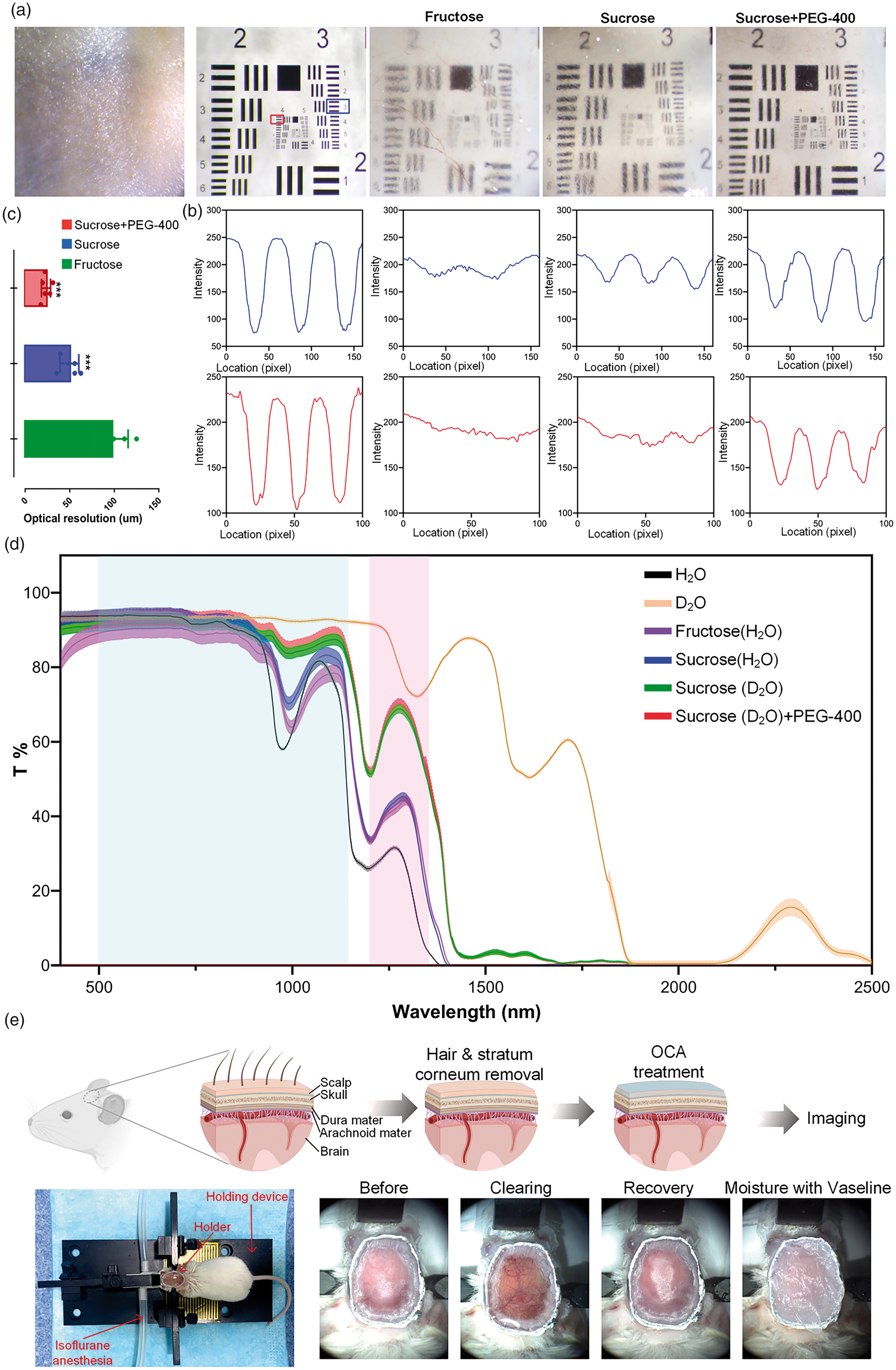
Screening tissue optical clearing reagent for scalp. (a) White-light images of excised skin before and after treatment with different reagents. The background is the central region of the 1951 USAF target. (b) The intensity profiles of elements in the blue and red rectangular boxes in (a). (c) The optical resolutions of the USAF target through the skin treated by different reagents (n = 6, mean ± standard deviation, ***: P < 0.001, One-way ANOVA multiple comparison test). (d) Transmittance of different tissue optical clearing reagents. The curves and the width of shadows are mean and standard deviation, respectively. (n = 6, mean ± standard deviation) (Blue shade: 500–1140 nm, red shade: 1200–1340 nm (Transmittance >50%)) and (e) Schematic of the TSSCW establishment procedure. Representative photograph of mouse immobilization, and the TSSCW at different states.
In general, the solvent for these sugar reagents is purified H2O, however, it has a strong absorption across the infrared band. Thus, the imaging quality may be limited by using the conventional optical clearing method in near infrared imaging. Compared to the H2O, the D2O has similar physicochemical property but higher transmittance than the water in near infrared band (Figure 1(d)). We quantitatively compare the transmittance of different clearing reagents and find that the D2O-dissolved saturated sucrose reagent containing PEG-400 has superior transmittance (Figure 1(d)). Using this clearing reagent, we can establish an imaging window that is suitable for both Vis‐ and NIR‐imaging.
The procedure of TSSCW establishment is shown in Figure 1(e). First, the hair and stratum corneum on the scalp is removed, and a custom-made holder is glued on the head. Then the OCA is applied to the scalp for about 15 min. After the application of OCA, the scalp becomes transparent. The mouse is then immobilized and ready for imaging. After imaging, the scalp has to be recovered and sealed by Vaseline for moisturization. This procedure allows repeated operations, therefore it is a switchable imaging window.
Improvement of NIR-II fluorescent imaging for brain
NIR-II fluorescence imaging has a unique superiority in in vivo deep tissue imaging owing to the low scattering, deep penetration, and high spatiotemporal resolution. Here, we applied the TSSCW to further improve the NIR-II imaging performance of the mouse brain angiography through scalp and skull. The nanoparticles TTQiT is intravenously injected for fluorescence labelling of the vessels. Figure 2(a) shows clear enhancement of the image quality. With the application of TSSCW, both big and small vessels can be clearly observed, and the bloodstream can also be monitored (movie S1). Moreover, with the TSSCW, the intensity of fluorescence signal and the contrast of blood vessels are greatly increased, which directly allow us to easily distinguish adjacent vessels (indicated by green arrows in Figure 2(b)). Further, the CNR of images is calculated to evaluate the NIR-II imaging improvement (Figure 2(c)). The result shows that the CNR of cerebral vascular images with TSSCW is significantly enhanced (P < 0.001). These results clearly indicate that the TSSCW improves the NIR-II imaging capacity.
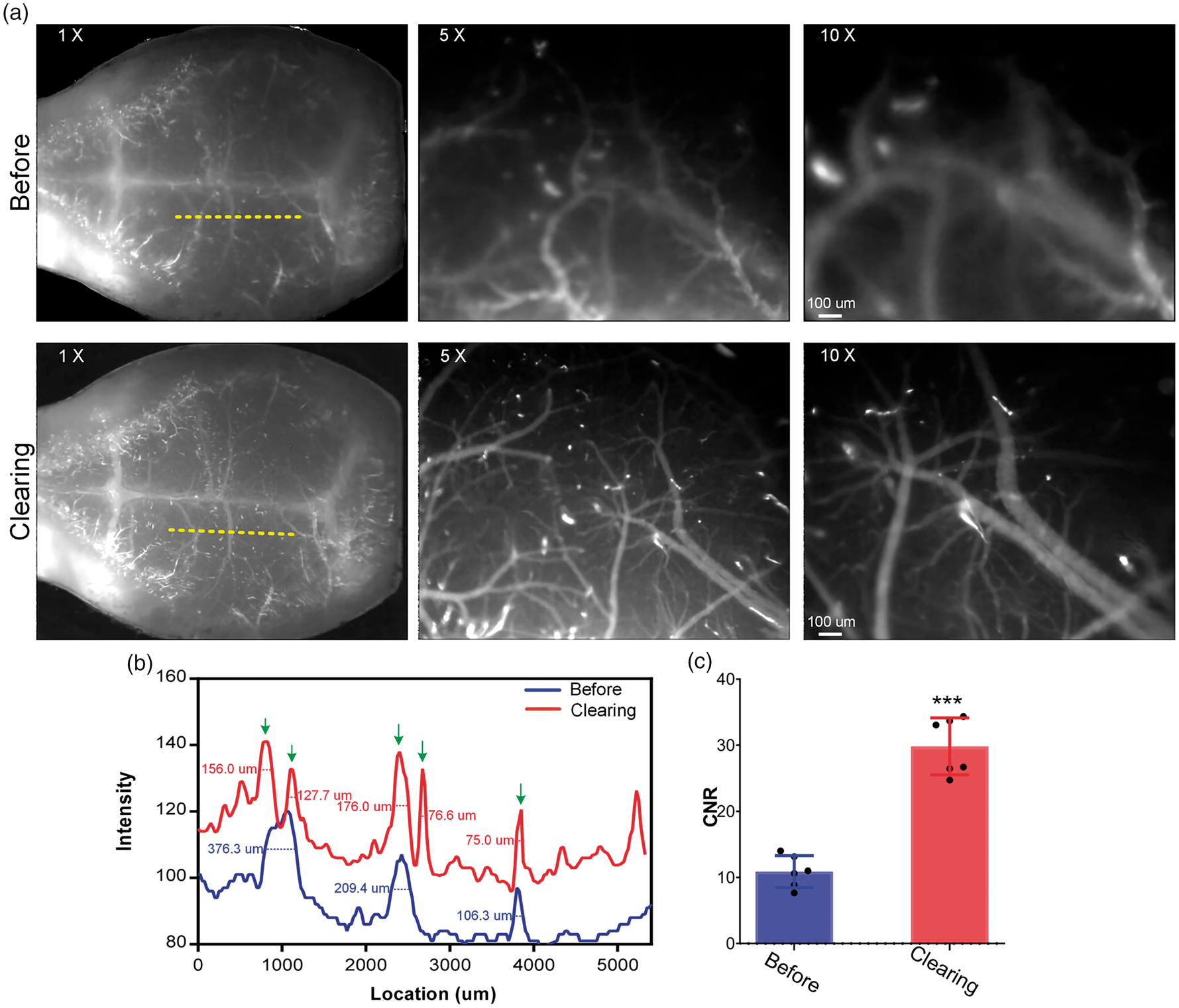
Improvement of NIR-II imaging performance. (a) NIR-II fluorescent angiography of brain before and after optical clearing treatment. (Views under 1 × /5 × /10 × objectives) (b) The intensity profiles along the yellow dotted line in (a). The numbers are full width at half maximum (FWHM) of the vessels. (The green arrows point to the distinguishable vessels after clearing.) and (c) The CNR values of images. (n = 6, mean ± standard deviation, ***: P < 0.001, One-way ANOVA multiple comparison test).
OCT imaging of cerebral microvessels through scalp and skull
In order to further characterize the performance of TSSCW with the OCT imaging, the 1300 nm-OCT is employed to image the cortical microvessels through scalp and skull. As shown in Figure 3(a), due to the high scattering and absorption from scalp and skull, the imaging depth is seriously limited, and the blood vessels in angiographic images are nearly indistinguishable. After the establishment of TSSCW, the cerebral blood vessels could be obtained with high resolution (Figure 3(b)), even a 19.5 μm-diameter vessel can be well distinguished. Also, the imaging depth is strongly enhanced (Figure 3(c) and (d)).
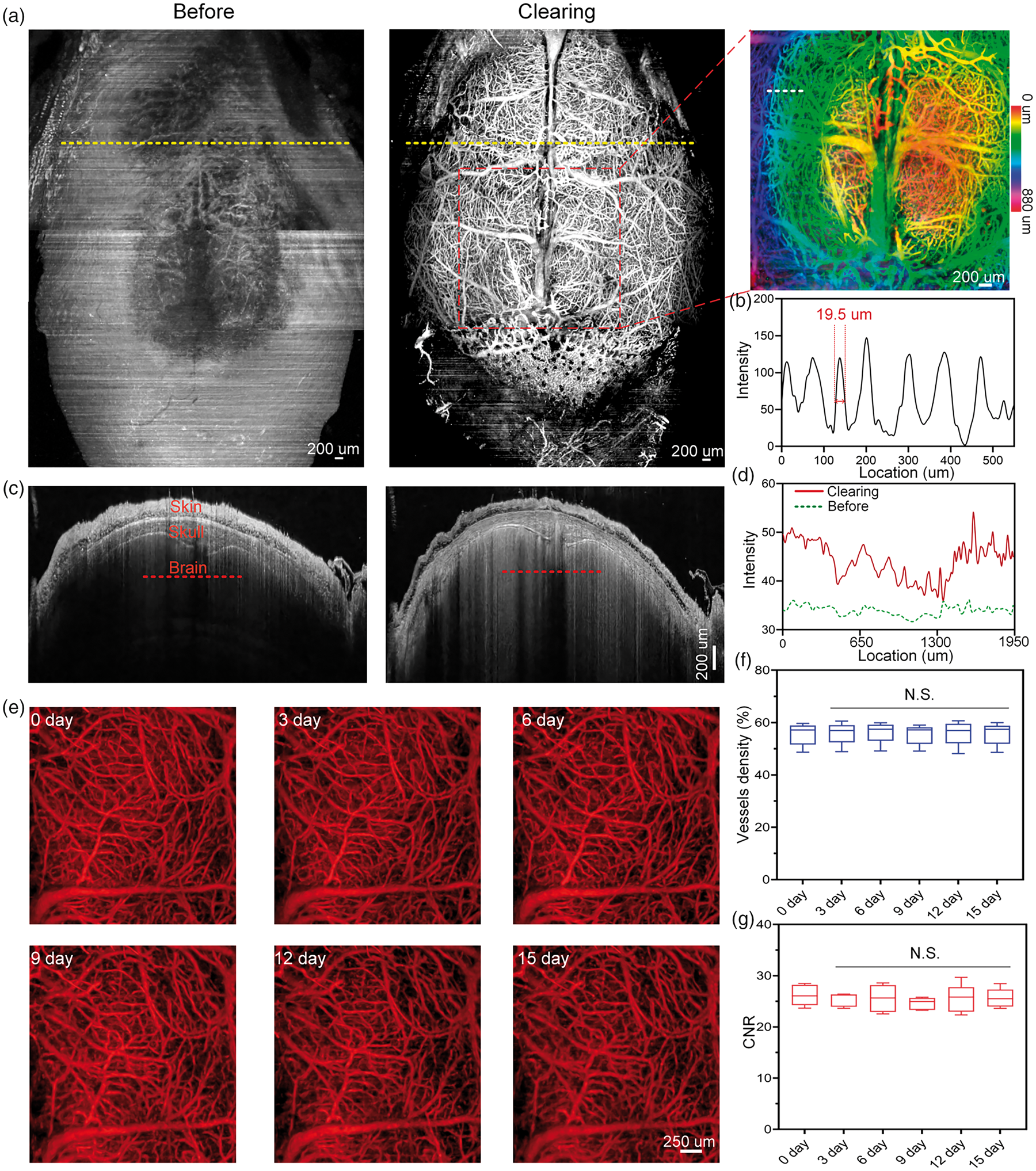
TSSCW enables cerebral vascular imaging through scalp and skull. (a) OCT angiography of cerebral and calvarial vessels before and after the clearing treatment. (The large-scale cortical vascular maps are stitched by five regional images) (b) The intensity profile along the white dotted line in the magnification of boxed region in (a). (c) The cross-sectional OCT intensity of yellow dotted lines in (a). (d) The intensity profiles along the red lines in (c). (e) Representative angiograms of cerebral blood vessels at different time points (vascular maximum intensity projection (MIP) (from surface to depth 530 um)). The changes in vessels density (f) and the CNR values (g). (Data are represented as box plots with 10th and 90th percentiles, n = 5, One-way ANOVA multiple comparison test; N.S.: no significance (P > 0.05)).
To explore the repeatability and imaging stability of TSSCW, we image the cerebral vessels at the same region in adult mice over a period of 15 days using OCT imaging system (Figure 3(e)). The results shows that the cerebral microvascular distribution patterns and morphology have no obvious changes. Further quantitative analysis indicates that the vascular density and CNR values over this period almost shows no changes as well (Figure 3(f) and (g)). These results indicate that the TSSCW not only enables OCT to obtain high-resolution images through the scalp and skull but also permits a repeatable mouse brain imaging over an extensive time period.
Two-photon imaging through scalp and skull
Although in vivo tissue optical clearing technique can improve light penetration in tissues, aberrations arising from optical heterogeneity in the chemically cleared tissue impedes the transcranial brain imaging performance in both resolution and depth. Adaptive optics, a widely-used technique in astronomical telescopes, recently has been introduced to improve two-photon imaging by correcting the aberration. We implement the AO by introducing a deformable lens between the microscope and the objective lens as indicated in Figure S1 (a). As shown in Figure 4(a), the AO can greatly enhance the two-photon imaging performance, both the signal intensity and aberrations of fluorescence beads are improved greatly. Moreover, with the AO, the two-photon imaging performance of cortical microvessels and microglia cells is further enhanced, manifesting as the well-distinguished microglia cells foot (Figure 4(b)).
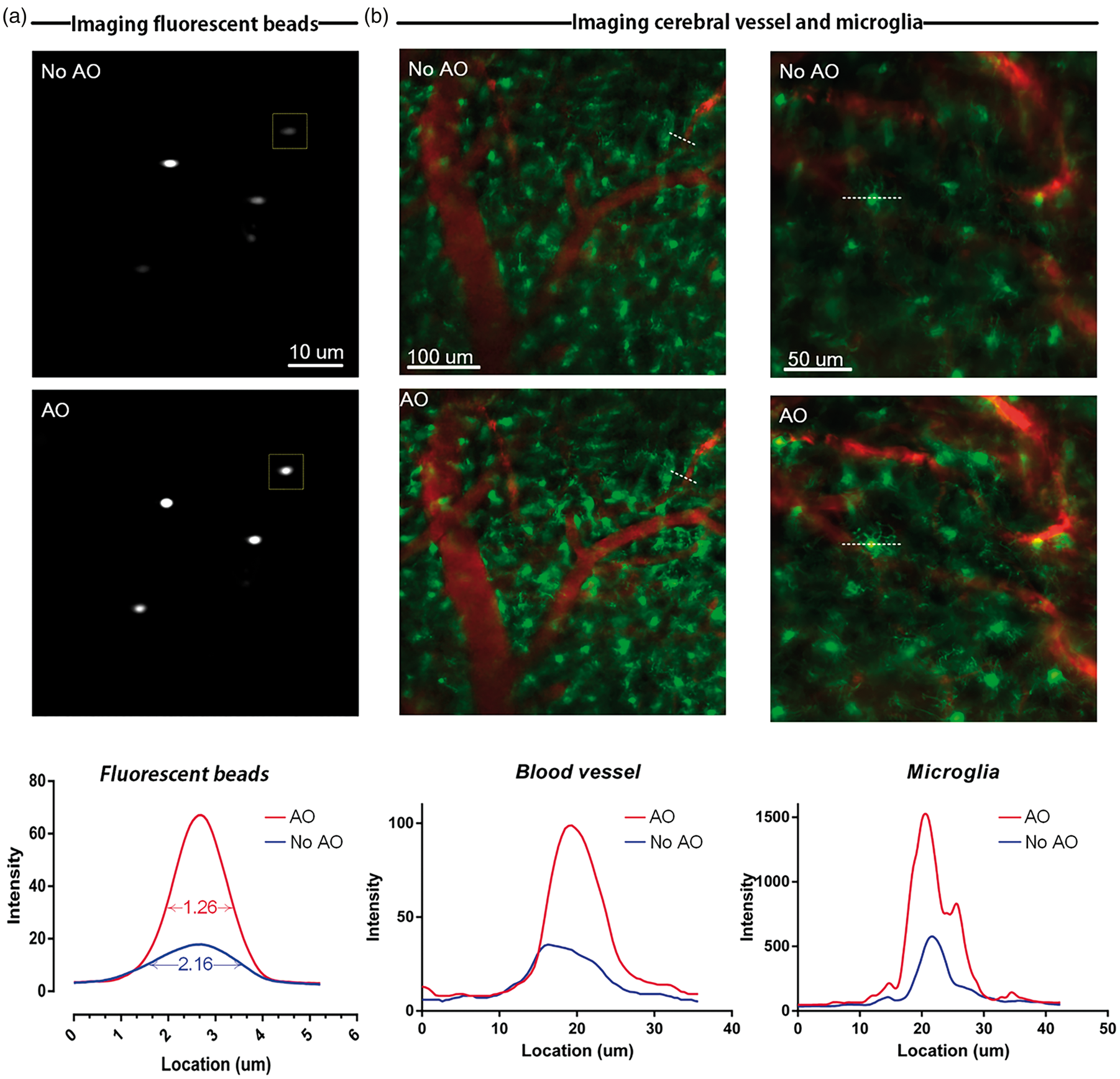
AO improves two-photon imaging performance. (a) Imaging of 1 um fluorescent beads in the hydrogel with and without AO. And signal profiles of the fluorescent bead in rectangular boxes in (a) and (b) Imaging of cortical blood vessels and microglia cells under TSSCW with and without AO. (zoom = 1 and 2) The signal profiles of vessel and microglia along the white dotted lines.
Additionally, we evaluate the two-photon imaging performance with and without TSSCW and monitor the dynamic changes in cerebral microvessels, microglia and calvarial bone marrow monocytes. With the help of AO, TSSCW largely improves the two-photon imaging depth and signal intensity (Figure 5(a) and (b)). We monitor the dynamic changes in cortical microglia within one hour (Figure 5(c)). It shows that the microglia in the cortex barely moves, except for the foot swing over time. This means that TSSCW does not affect the cortical microenvironment. Additionally, we also monitor the skull bone marrow microenvironment as indicated in Figure 5(d). The cutaneous and calvarial blood vessel can be seen in YZ-view (dotted circles). In some specific positions such as the circle indicated by the arrow (Figure 5(e)), the monocyte cells exhibit a tiny movement due to the blood flow (Movie S2). In summary, combining the TSSCW with AO enables two-photon imaging to dynamically monitor the brain and skull bone marrow microenvironment with high resolutions through scalp and skull.
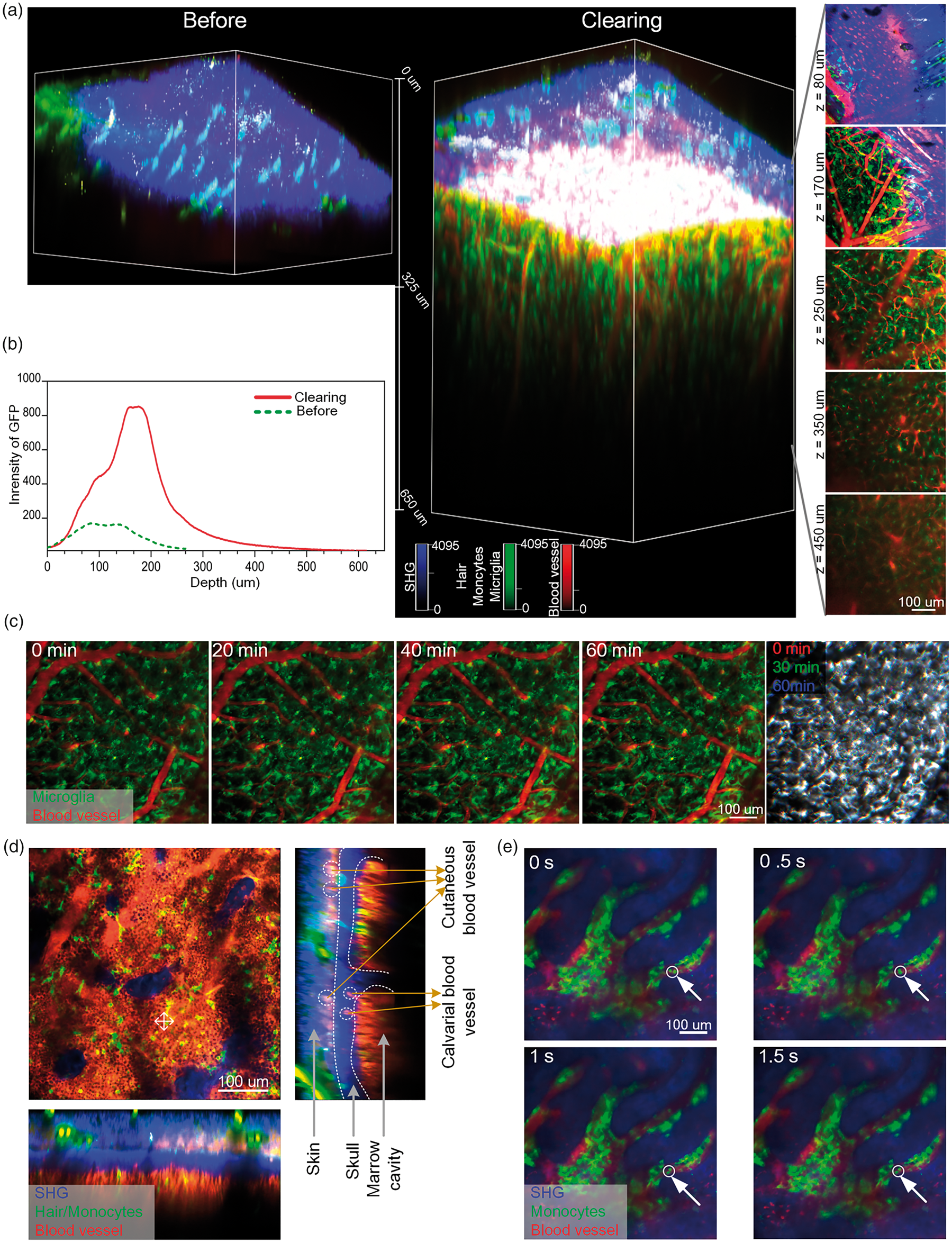
Two-photon imaging of cortical and calvarial bone marrow microenvironment through scalp and skull by combining TSSCW with AO. (a) Typical 3 D views of blood vessels and microglia in 3.5-week-old Cx3cr1EGFP/+ mice before and after the OCA treatment (Raw images: 1024 × 1024 pixels, 0.5 μm/pixel; Excitation: 920 nm, Detection: 525 ± 25 nm (Green: microglia/monocytes/hair); 575 ± 12.5 nm (Red: vessel); 492 SP (Blue: skin/skull SHG), Objective: 25 × W, 1.1 NA). (b) Depth-directional profiles of GFP fluorescence signal intensity of microglia. (c) Monitoring the activation of microglia in cortex within 1 hour (The last panel is obtained by merging the 0 min (red), 30 min (green) and 60 min (blue) images of microglia). (d) and (e) Imaging of calvarial bone marrow microenvironment.
Light manipulation of cortex through TSSCW
We implement light-mediated brain manipulation because of the enhancement of light penetration through the TSSCW. Here, the two-photon-induced laser lesion model is used because it has extremely high spatial accuracy and controllable damage. After establishing TSSCW, high-power two-photon excitation laser is projected to the cortex for focal damage, and the response of microglia is subsequently monitored (Figure 6(a)). As shown in Figure 6(b), at the initial state, the resting cortical microglia cells distribute uniformly and extensively. After the laser stimulation, fluorescence signal at the injured area disappears instantly (0 min), followed by immediate response from the adjacent microglia. The neighboring cells extends their processes toward the damaged site (2–6 min) and gradually tightens the encirclement. These cells later appear to fuse together, forming a spherical containment around the injured area for tissue repair (8-16 min). These results indicate that the highly localized signals released by laser injured site are able to rapidly attract individual microglial processes. Moreover, this clearing window not only permits the visualization of cortical vessels and cells but also has a great potential in realizing the light-mediated brain control.
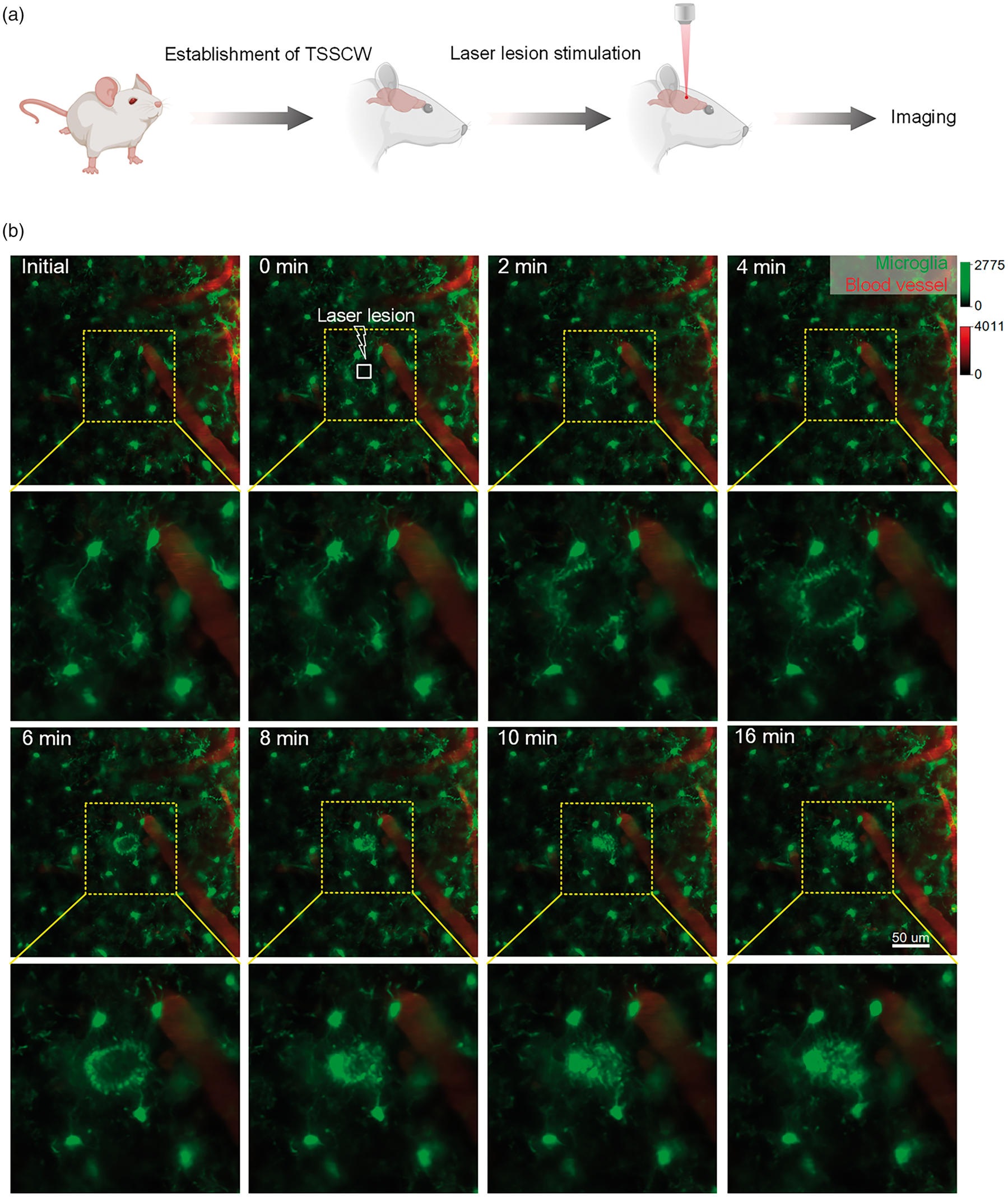
Two-photon imaging of cortical microglia dynamics after laser lesion. (a) The schematic of the laser lesion model establishment and imaging and (b) The dynamic changes of cortical microglia after two-photon-mediated lesion on 3.5-week-old Cx3cr1EGFP/+ mice (Raw images: 1024 × 1024 pixels, 0.3 μm/pixel; Excitation: 920 nm, Detection: 525 ± 25 nm (Green: microglia); 575 ± 12.5 nm (Red: vessel); Objective: 25 × W, 1.1 NA).
Discussion
At present, in vivo optical imaging of cortical microvasculature and cells usually relies on various cranial windows, including open-skull glass window,4,26 thinned-skull window, 3 and their variants.27 –29 The thinned-skull window is widely-used for brain imaging because it satisfies requirements for instant imaging. 3 However, skull has to be re-polished before each imaging trial, which limits the imaging frequency. Meanwhile, polishing the skull generates heat that could increase the chance of cortical vascular proliferation and deformation.12,30 Another widely-used method for brain imaging is open-skull glass window.4,26 But the imaging cannot be performed immediately after imaging window establishment, which needs to take ∼2 weeks for recovery. Sometimes the procedure may damage cortical or dural vessels and induce inflammations.11,12 Recent studies have demonstrated that the skull bone marrow contains the pools of myeloid and B cells, and the rich vascular channels serve as pathway for cell migration.7,31 Thus, the skull plays an important role in brain immunity. The skull thinning or removal will inevitably affect the skull integrity, bringing some brain immunity damages, which may lead to misunderstanding to the real physiopathological processes.
The emergence of in vivo tissue optical clearing technique provides a new way to overcome the high scattering of tissue, 32 which mainly includes the in vivo skin33,34 and skull optical clearing methods.15,16 It makes tissues transparent by matching the refractive index of various components via the introduction of biocompatible reagents.35,36 In vivo skin optical clearing technique has been widely used in improving imaging performance of cutaneous cells and blood vessels on rodent animal. It even has the potential to be applied on human skin. 6 The skull optical clearing techniques allow the imaging of the structural and functional changes in cortical vessels and neurons through skull. Nevertheless, for most skull optical clearing methods, the transparent states of skull cannot be preserved for days or even months. They require repeated clearing operation in each imaging session. Recently, we proposed a novel long-term optical clearing skull window, dedicating to chronic skull transparency maintenance for months with single operation, 17 which allowed monitoring the dynamic changes of cortical and calvarial cells and blood vessels with high resolution.
Up to now, these proposed optical clearing skull window16,18,37 still requires scalp excision and skull exposure, which will damage the periosteum on the surface of skull. Periosteum contributes to the blood supply of bone, and plays an important role in growth and development of bone. 38 Thus, there is an urgent need to develop an optical clearing cranial window that can preserve the integrity of living body. In this work, we develop an innovative imaging window – TSSCW based on an optimized in vivo skin optical clearing technique, which is realized by applying OCA on the scalp. We assess the OCA diffusion by monitoring the permeation of fluorescent dye into the scalp (Figure S2 (a)). The results show that after optical clearing, the fluorescent molecules are distributed only in the derma and do not diffuse into the skull. This indicates that the depth of OCA diffusion is limited in the skin, which is consistent with our previous study. 39 Additionally, we analyze the status of microglia and the expression of GFAP under the different cranial windows (7 days after establishment of cranial windows). For the open-skull glass window, microglia on the contralateral control side of the cortex appear normal, with many ramified branches project from somata (Figure S2 (b)). However, on the operated side, microglia seem abnormal, having either assumed amoeboid shapes or extending most of their processes toward the pial surface. For the thinned-skull window (polish only once) and TSSCW, there are no reactive phenotypes of microglia. Figure S2 (c) shows little GFAP expression in astrocytes on the contralateral control side (right hemisphere) but extensive GFAP expression in the operated hemisphere of the cortex subjected to open-skull glass window (left hemisphere). And little or no GFAP expression in astrocytes is found under the thinned-skull window and TSSCW. Further, we quantitatively analyze the GFAP and microglia density as shown in Figure S2 (d) and (e), and the results indicate that only the open-skull glass window causes significant changes (P < 0.001) to the cortical microenvironment. By contrast, Holtmaat A. et al. demonstrated that the open-skull glass window caused extensive activation of astrocytes until 30 days after this cranial window establishment based on the GFAP immunostaining. 4 In a previous study, Wang J. et al. proposed a switchable skin optical clearing window, and showed it had no potential toxicity of OCA to the body. 33 And our recent work also demonstrated that skin structure and function could recover after in vivo skin optical clearing. 39 Therefore, TSSCW based on biocompatible reagents has a good safety.
In previous study, sugars, rich in hydroxyl groups, were demonstrated to have a good potential in skin optical clearing. 40 By comparing the skin optical clearing ability of monosaccharide, Wang, J. et al. found that fructose had the superior skin optical clearing capability. 41 And Shi, R. et al also proposed a footpad skin optical clearing window based on fructose. 34 More following studies indicated that the disaccharides showed a better skin optical clearing capability than monosaccharide.42,43 Our results again shows that sucrose has a better optical clearing effect for scalp tissue than fructose, and sucrose+PEG-400 can further improve imaging performance of subcutaneous vessels in the dorsal skin of mouse. 44 Additionally, the majority of OCAs use pure water as the solvent. However, water shows strong absorption in the near infrared band. A recent study reported that the skull optical clearing reagent compounded by deuteroxide showed better imaging performance in NIR-II imaging. 45 Thus, this newly-developed in vivo optical clearing technique based on sucrose and deuteroxide can not only makes the scalp transparent but also better fit for Vis and NIR imaging.
To establish the TSSCW, the hair growth cycle of mouse should be paid great attentions. This is because that the hair cycle can affect the thickness and pigmentation of the skin.46 –48 When the skin is in telogen phase, it is very thin and looks pink in color, which will be beneficial for optical clearing. Figure S3 shows the direct viewings and the transmittance of skin at different hair cycle in BALB/c and C57BL/6 mice. The results shows that the skin of BALB/c mice at the telogen phase looks in a deeper pink color than that at the anagen phase. As for C57BL/6, the skin is pink at the telogen phase but shows obvious pigmentation at the anagen phase. Moreover, our results also show that the skin at the anagen phase has less transmittance than that at the telogen phase. After the optical clearing treatment, the transmittance of BALB/c mouse skin at the anagen is greatly increased. However, the transmittance of C57BL/6 mouse skin at the anagen does not exceed 10% even after clearing. For the skin at telogen, the transmittances of scalp for both BALB/c and C57BL/6 are greatly increased after clearing. Overall, the mice at telogen are more suitable for TSSCW establishment.
Two-photon laser scanning microscopy is essential for deep brain imaging with subcellular resolution.49,50 Although this TSSCW makes it possible to realize two-photon imaging of the brain cortex through scalp and skull, the resolution and signal intensity severely decrease with depth as a result of tissue scattering and optical aberrations. 19 Recently, AO has been introduced in the two-photon imaging by using a compensatory wavefront distortion to the excitation laser, which can eliminate the system- or specimen-induced aberrations.51,52 In this work, we combine the TSSCW with the AO device to improve two-photon imaging ability through scalp and skull. Although we can clearly observe the cortical and calvarial microvasculature and cells through scalp and skull, the two-photon imaging depth is still limited to the superficial layers of the cortex. Some studies reported that the thickness and composition of mouse skull had changes with age.53,54 Moreover, the mouse skin will go into the next anagen stage at ∼12 weeks after birth, 48 which has extremely low transmittance. Thus, the mice we use are all younger than 12 weeks old in this study.
Conclusions
In this study, we propose a novel imaging window named TSSCW for in vivo cortical and calvarial imaging and manipulation through scalp and skull. It not only can greatly enhance the NIR-II fluorescent imaging ability, but also allows OCT imaging of cerebral microvessels with high resolution through scalp and skull. By combing TSSCW with AO, we firstly realize the two-photon imaging of the calvarial and cortical microenvironment through scalp and skull. Moreover, the cortex can be precisely manipulated by laser, and the spatial-temporal changes of microglia have also been monitored. This technique possesses the advantages of easy-operation, convenience and noninvasiveness, providing a useful way to realize the high-resolution brain imaging and manipulation in intact mouse.
Supplemental Material
sj-jpg-1-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-jpg-1-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-2-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-jpg-2-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-jpg-3-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-jpg-3-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-mp4-4-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-mp4-4-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-mp4-5-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-mp4-5-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-6-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-pdf-6-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-7-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-pdf-7-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-8-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-pdf-8-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Supplemental Material
sj-pdf-2-jcb-10.1177_0271678X231167729 - Supplemental material for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull
Supplemental material, sj-pdf-2-jcb-10.1177_0271678X231167729 for An optical clearing imaging window: Realization of mouse brain imaging and manipulation through scalp and skull by Wei Feng, Chun-jie Liu, Lisi Wang and Chao Zhang in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by National Natural Science Foundation of China (NSFC) (Grant Nos. 82001266 to CZ), Guangdong Basic and Applied Basic Research Foundation (Grant Nos. 2019A1515110699 and 2021A1515011783 to WF, and 2019A1515110803 and 2021A1515011443 to CZ).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
CZ and WF conceptualized and designed the experiments. Two-photon imaging and image analysis was done by WF. OCT imaging and NIR imaging was performed by CZ. Immunostaining was done by LW. Statistical analysis was done by WF. Manuscript was written by CL and CZ with editing and contributions from all authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.