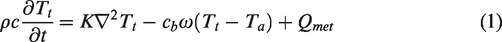Abstract
Brain temperature, regulated by the balance between blood circulation and metabolic heat generation, is an important parameter related to neural activity, cerebral hemodynamics, and neuroinflammation. A key challenge for integrating brain temperature into clinical practice is the lack of reliable and non-invasive brain thermometry. The recognized importance of brain temperature and thermoregulation in both health and disease, combined with limited availability of experimental methods, has motivated the development of computational thermal models using bioheat equations to predict brain temperature. In this mini-review, we describe progress and the current state-of-the-art in brain thermal modeling in humans and discuss potential avenues for clinical applications.
Keywords
Introduction
Brain temperature is an important physiological parameter in humans, particularly after injury.1–5 As brain temperature is regulated by the balance between incoming arterial blood flow, outgoing venous flow, and heat generation from oxygenated metabolism, disruption in hemodynamics or metabolic function after injury can alter brain thermoregulation.6–12 Non-invasive methods using magnetic resonance (MR) thermometry are available but not widely implemented particularly for measurements in the brain. As a result, the use of brain temperature in clinical practice is limited due to a lack of widespread and well-integrated methods for whole-brain thermometry.
To address this challenge, computational models of bioheat transfer in vascularized tissue have been developed. Most rely on the Pennes’ bioheat equation (PBE), 13 with subsequent iterations aiming to develop a more realistic picture of thermal regulation and response to injury.14–16 Challenges in thermal modeling of the human brain, compared to simpler organ systems, include unique vascular features such as the Circle of Willis, higher rates of heat generation from oxygenated metabolism, and a nearly adiabatic environment due to insulation provided by the skull and scalp. In this mini-review, we describe progress in bioheat modeling of the brain and propose avenues for future clinical application.
Heat transfer modeling in vascularized tissue
Basic concepts used in bioheat transfer models
In mathematical or computational models, an energy balance for a differential control volume is usually employed to derive governing equations. Within this control volume, energy is conserved and can be transferred from one volume to another via heat transfer. Temperature is determined by how much heat is stored within a given control volume. The primary modes of heat transfer include conduction, advection, convection, and radiation (Figure 1). Due to relatively low physiological temperatures, radiative heat transfer is insignificant relative to conduction, advection, and convection. Conduction, often referred to as heat diffusion, is the transfer of heat in stationary matter due to a temperature gradient
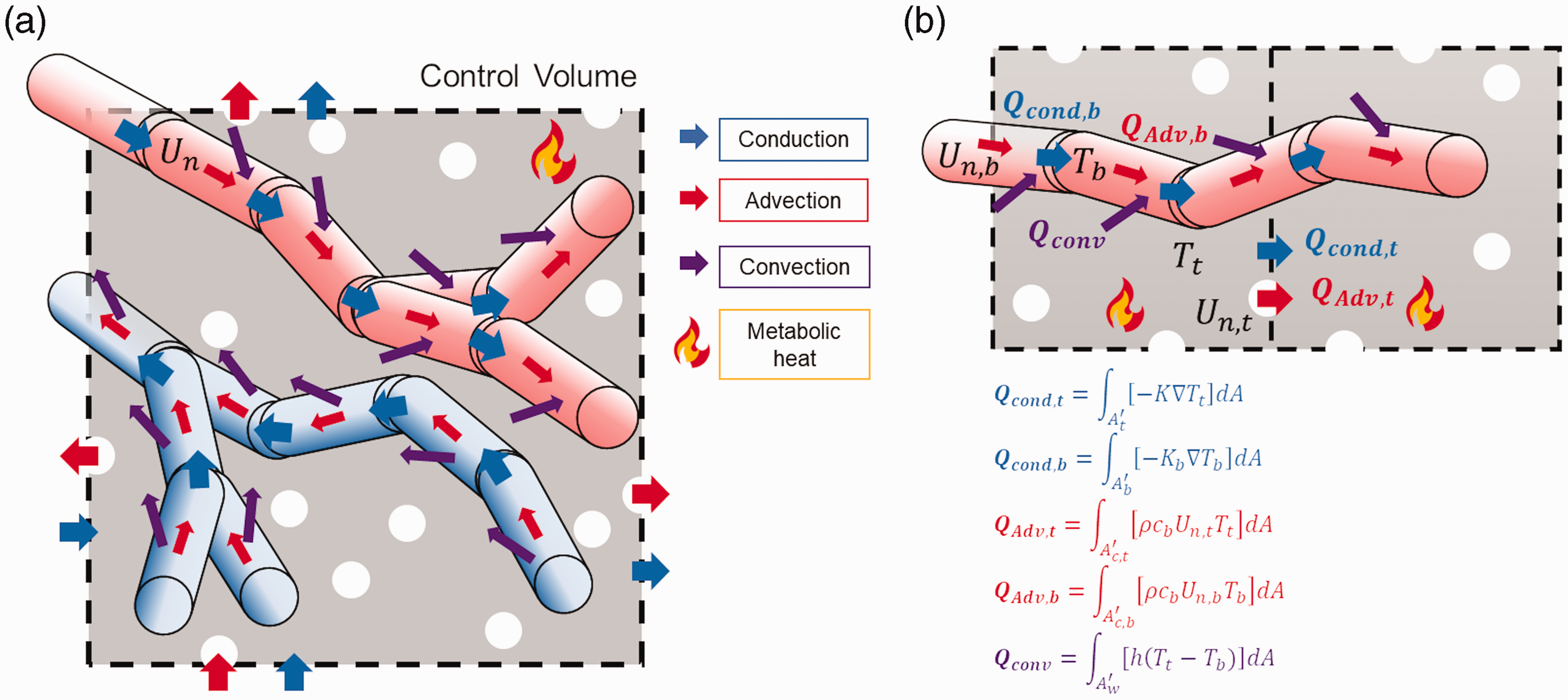
(a) Schematic of conductive, advective, and convective heat transfer in vascularized tissue within a control volume. Arterial vessel segments are depicted with red cylinders as they include oxygenated hemoglobin, and venous vessel segments are depicted with blue cylinders to represent deoxygenated blood. Blue, red, and purple arrows represent conductive, advective, and convective heat flow, respectively. (b) Detailed representation of conductive, advective, and convective heat flow in tissue and blood vessels.
Homogeneous medium approximation
Early bioheat transfer models rely on a homogeneous medium approximation which simplifies the influence of blood perfusion within the control volume by averaging blood flow contributions from arteries, capillaries, and veins. The most fundamental and simplest is the PBE
13
in which tissue temperature,
Vascular structural modeling
Representation of local blood velocity or flow (
One of the limitations in the DIVA model is the excessive computational demand of tracking blood flow and temperature across the complete vascular network including small blood vessels. To mitigate this issue, the concept of thermally significant blood vessels was introduced by Chen and Holmes. 15 Thermal significance was determined by the length15,22,24 or diameters of the vessels in thermal equilibrium with the surrounding tissue.25–27 Shrivastava and Roemer refined the definition of thermal significance and emphasized the need for detailed and realistic vascular networks that include both large (major arteries and veins) and small (arterioles, venules, and capillary beds) vessel structures to accurately predict temperature in vascularized tissue. 28
Porous media integration
Despite the widespread availability of computational resources and improved medical image segmentation, it is challenging to reconstruct entire vascular networks. The use of porous tissue as a heat transfer domain directly addressed this challenge, eliminating the need to account for microvasculature, i.e., structures with diameters less than the spatial resolution of conventional medical imaging such as computed tomography (CT) or MR angiography. The concept of porous media was introduced by Roeztel and Xuan using a two-equation non-equilibrium bioheat model separating blood and tissue phases in thermal interactions. 29 Nakayama and Kuwahara further generalized the porous media approach using volume averaging theory which can be used to average features across vascularized tissue. 30 They incorporated not only a blood perfusion term (commonly used in early models relying on the homogeneous medium approximation) but also interfacial convective heat transfer between blood and tissue phases. To integrate the porous media and DIVA approaches, He and Liu introduced realistic 1 D discrete vasculature within 3 D porous media to capture thermal interactions between blood and tissue. 31 Collectively, these methods laid the groundwork for modeling heat transfer in the brain.
Bioheat transfer models of the human brain
Parallel to the development of bioheat transfer models, representation of accurate physiology and complex anatomical structures used in brain modeling has been refined (Figure 2). In this section, we review progress in brain thermal models towards incorporation of realistic physiology and structure of the human brain, and discuss current challenges and opportunities in human brain modeling.
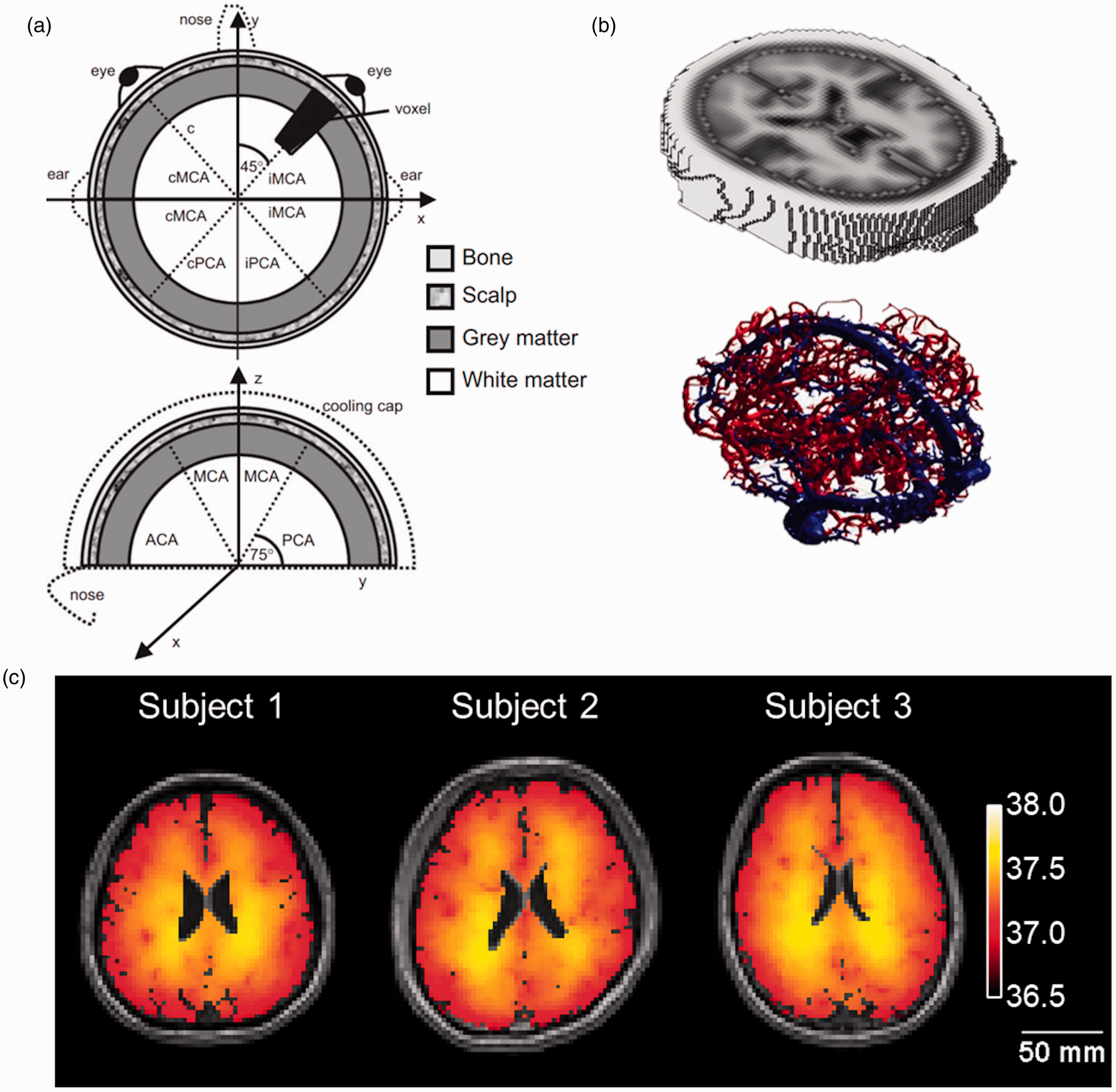
Representations of the human brain used in biophysical modeling. (a) Early representations included simple hemispheric models, 47 and (b) more recently have advanced to realistic human brain models with both tissue and vasculature. 39 (c) Model-predicted brain temperatures in degrees Celsius from three healthy human subjects. 40 Figure 2(a) was adapted and reprinted from literature, 47 with permission from Elsevier. Figure 2(b) was adapted from literature 39 and used under the Creative Commons Attribution 4.0 International (CC BY 4.0) license. Figure 2(c) was adapted from literature 40 and used under the Creative Commons Attribution 4.0 International (CC BY 4.0) license.
Cerebral metabolism and hemodynamics in human brain model
As metabolic heat generation or hemodynamic heat dissipation are important aspects in brain thermal modeling, the effect of various physiological parameters on both of these aspects has been investigated. Sukstanskii and Yablonskiy evaluated several internal and external parameters that affect brain physiology 32 and found inlet arterial temperature is significantly related to deep brain temperature consistent with in vivo findings. 33 Ley and Bayazitoglu demonstrated any regulatory process related to changes in cerebral blood volume can alter brain temperature. 34 Yet, applying transiently evolving metabolism or hemodynamics in computational brain models is challenging. The use of constant values for static physiological parameters (e.g., tissue density, specific heat, thermal conductivity, perfusion rate, metabolic rate) derived from previous in vivo or ex vivo experiments are often averaged within each domain (i.e., skull, scalp, brain) and used to simulate the quasi-steady state physiology.35–38 Recent brain thermal models rely on tissue probability maps representing each tissue type (e.g., gray matter, white matter, cerebrospinal fluid, fat, and skull) at the voxel level to discriminate physiological properties, such as metabolism, between tissue voxels.39–41
Consideration of anatomical structures in human brain models
One of the key structural features of the human head (or brain) is the spherical shape. Initial work used the simplest two-dimensional hemisphere,42,43 and has been further developed into a three-dimensional dome shape consisting of several layers representing skull, scalp, and brain tissue.44,45 Neimark et al. improved the hemispheric (or 3 D dome) model by segmenting brain tissue into gray and white matter and integrating a simplified Circle of Willis structure.46,47 Individual blood flow rates within vessel segments in the Circle of Willis were determined by Kirchoff’s circuit law, enabling calculation of individual temperature and hematocrit for each blood vessel. Nevertheless, the hemispheric model and consideration of the Circle of Willis cannot fully capture the variation in individual anatomy, and more refined anatomical features are necessary.
As medical imaging and post-processing techniques have been improved, more realistic 3 D anatomy has been incorporated into thermal models.48–50 Lutz et al. developed a realistic brain model to investigate the effectiveness of endovascular cooling in ischemic stroke patients with middle cerebral artery occlusion. 51 Using the Colin 27 brain atlas 52 and arterial structure from the Brain Vasculature (BraVa) database, 53 brain tissue was segmented into gray and white matter and incorporated with anterior, middle, and posterior cerebral arteries. Blowers et al. expanded upon these methods, utilizing tissue probability maps from the International Consortium for Brain Mapping 152 brain atlas, 54 arterial structures from the BraVa database (vascular morphology from MR angiography of 61 healthy humans), 53 and venous structures from an MR venography scan 39 to calculate temperature in arteries, tissues, capillary beds, and veins. Similar to He and Liu, 31 Blowers et al. combined the 1 D DIVA model with 3 D porous media and also included counter-current blood flow effects (reverse flow within porous tissue). The use of augmented arterial and venous structure using human MR data resulted in more similar spatial temperature gradients than the Pennes’ model when compared to brain temperature maps acquired from in vivo MR thermometry. 55 The work by Lutz et al. and Blowers et al. represented a major advance in simulating whole brain human temperatures using realistic anatomy at the millimeter scale.39,51 Recently, Sung et al. expanded upon these models by incorporating subject-specific tissue and blood vessel structures acquired from prospective MR imaging. 40 Building on work by Shrivastava and Vaughan, 56 both mass and energy were fully conserved within their model. This advance over previous vascular models using realistic anatomy was achieved by employing the integral (rather than differential) formulation of the governing equations and properly resolving the directionality of thermal energy transport due to blood advection. Model-predicted temperatures were compared with measurements using MR thermometry, with close voxel-wise agreement across three human subjects (±0.8°C). In a follow-up study with a larger number of healthy human participants (N = 30), thermal gradients from model-predictions and in vivo measurements using MR thermometry displayed similar qualitative patterns. Quantitative regional analysis (10 cortical and 12 subcortical regions) showed good agreement (regional mean absolute difference <0.8°C) between model-predicted and MR-measured temperatures when subject-specific anatomy was used. 41 While the model developed by Sung et al. was the first to facilitate individual brain temperature predictions, brain temperature predictions encompassed a narrower range of temperatures (36.95–37.28°C) compared to MR measurements (35.85–39.18°C) as well as other reports in healthy humans,5,57–59 and further refinement of the model may be necessary.
Current challenges and opportunities for brain thermal modeling
Despite the tremendous advancements in brain thermal modeling compared to initial hemispheric models using the PBE, there are several outstanding challenges. Several previous models conserved mass and energy both locally and globally; however, momentum was not fully conserved as this requires detailed vascular information, such as diameter or length of individual vessel segments, and appropriate formulation of the governing momentum conservation equations. To calculate more accurate blood vessel conductance and mass flow rates, more realistic sub-millimeter scale vasculature maps and quantitative metrics of local cerebral blood flow acquired from CT or MR perfusion images will support the development of models that conserve momentum as well as energy and mass.
Heat transfer analysis of the brain requires the use of boundary conditions at all external surfaces. While heat loss via radiation, evaporation, or natural convection are important phenomena in thermoregulation of the entire body, several studies quantified this effect and found that brain temperature is rarely affected by external environments due to insulation by the scalp and skull.32,33,60–62 In recent brain thermal modeling using realistic human anatomy, a specified fixed temperature was used as a self-consistent boundary condition that accounts for the interactions between brain and the external environment through different modes of heat transfer including evaporation, thermal radiation, and natural convection.39–41,51 These studies used empirically determined temperature values as a realistic proxy for the surface boundary condition, which can be improved by using real-time measured scalp temperature (e.g., using IR thermal imaging data). In the cases when brain thermal interactions with a dynamically changing external environment (e.g., ambient temperature) are of interest, this effect can be readily incorporated by changing the boundary conditions from a fixed temperature to Newtonian boundary condition, balancing the heat flux from the brain interior to the heat fluxes to the external environments by all relevant modes (convection, radiation, and evaporation/condensation) of heat transfer.
For clinical applications of the brain thermal models, models must also account for varying physiological conditions. Pharmacological approaches (e.g., anesthesia with benzodiazepine, hemodynamic augmentation using acetazolamide administration, etc.) can be used to temporarily vary physiology, such as metabolism or hemodynamics, and may facilitate direct model evaluation. Slupe et al. showed anesthetic agents can substantially affect physiology, particularly the cerebral metabolic rate of oxygen and cerebral blood flow. 63 While a recent study incorporated effects of anesthesia in a computational model for targeted temperature management after cardiac arrest, 64 validation with experimental measurements is still outstanding. Experimentally, Fleischer et al. found significant relationships between the brain thermal response and hemodynamic augmentation using acetazolamide administration, 12 and brain temperature decreases of ∼ 0.3°C have been observed after increased cerebral blood flow in response to hypercapnia. 65 Future studies comparing model-predicted and experimentally-measured temperatures under dynamic conditions will provide opportunities to develop brain thermal models for direct clinical applications. Furthermore, as models that mimic hemodynamics in the brain for cerebrovascular disease or stroke patients have been developed,49,66,67 these injury models can be embedded into brain thermal models for more realistic implementation. Recent advancements in medical imaging (e.g., quantitative blood oxygenation level dependent imaging or arterial spin labeling) may also aid in quantification of transient metabolic and hemodynamic responses, and future modeling would benefit from incorporating dynamic physiological parameters into simulations of brain temperature evolution.
Clinical applications of brain thermal modeling
Brain thermal modeling has the potential to impact clinical care, particularly in cases where brain temperature may be a key prognostic or treatment stratification parameter such as in thermal ablation, 68 acute ischemic stroke, 69 and therapeutic hypothermia. 70 As brain temperature has been shown to be intimately related to outcomes after injury,12,69,71 there is a clear opportunity for thermal modeling in prognosis and treatment stratification. Two applications are described below.
Thermal ablation
A multitude of thermal ablation techniques have been developed as treatment strategies for a range of diseases including epilepsy and cancer.72–78 Fahrenholtz et al. developed a PBE-based theoretical model for planning laser interstitial thermal therapy (LITT) paired with a damage model to predict the extent of maximum ablation, showing good agreement (Dice similarity coefficient of 0.82) with patient-derived MR thermometry. 79 Similar models have been developed by Jermakowicz et al. 80 which showed features of preoperative MR images can be predictive of LITT ablation dynamics and would be useful in improving the efficacy of LITT when delivered to brain tumors. Thermal ablation modeling utilizing the Pennes’ model may be highly efficacious in the clinical setting to aid neurosurgeons with a priori treatment strategies and patient stratification.
Therapeutic hypothermia
Thermal modeling may also be beneficial for improved implementation of therapeutic hypothermia or prescribed cooling as a neuroprotective measure. While therapeutic hypothermia has been proposed as a treatment strategy following traumatic brain injury (TBI) and acute ischemic stroke, its efficacy is inconclusive potentially due to the use of body temperature to monitor treatment and the known decoupling of brain and body temperature after injury.6,81–84 Li et al. developed a finite element model based on the geometry of brain and carotid artery in the neck acquired from CT images to investigate the effect of cooling methods for hypothermia. 85 Temperature predictions from the finite element model were validated using a 3 D-printed phantom with a skull layer and neck carotid artery bifurification. The finite element model simulations showed good agreement with phantom-based experimental measurements (maximum discrepancy of ∼0.2°C in both tissue and major arteries) acquired with thermocouples from four tissue locations and two major arteries (external and internal carotid artery). As a follow-up study, Yin et al. compared two cooling methods for therapeutic hypothermia using finite element analysis based on a 3D brain model from CT images. 86 Modeling results indicated simultaneous brain and neck cooling is more effective than direct head surface cooling for controlling deep brain temperature. Alves and Mady demonstrated the utility of thermal modeling in improving the methodology of therapeutic hypothermia in post-cardiac arrest patients by testing the capacity of different methods of induction to reduce temperature of hypothalamus, distribution, and stability. 87 Catheter-induced cooling provided faster and more concentrated cooling while a cooling blanket maintains high heat removal capacity and allows for better temperature stability during maintenance and rewarming. While routine clinical applications have yet to be realized, thermal modeling may be beneficial for a number of emerging therapies.
While these previous studies were not validated with in vivo measurements, Baker et al., presented a clinically-validated computational model to predict temperature changes after focal brain cooling. 88 MR-derived individual head models for two clinical cases were used to calculate the transient thermal response using the PBE after cooling. Computational model predictions of local brain temperature as a function of distance from the probe center were compared with in vivo MR thermometry and accurately reproduced the intracranial thermodynamic response after hypothermia in terms of average standard error at each given distance. Although routine clinical applications have yet to be realized, brain thermal modeling may be beneficial for many emerging therapies.
Conclusion
Bioheat transfer models have been developed for over 70 years, yet detailed thermal gradients across the whole brain are still challenging to model and predict. Recent progress has focused on more realistic representations of vasculature and rigorous derivation of governing heat transfer equations, and validation using in vivo experimental measurements has been demonstrated. While clinical integration is currently sparse, opportunities exist to incorporate model-predicted brain temperatures into clinical decision making.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported, in part, by an American Heart Association predoctoral fellowship (Award ID: 909342).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.