Abstract
Hypertension is associated with both ageing and dementia. Despite this, optimal blood pressure targets in dementia remain unclear. Both high and low blood pressure are associated with poorer cognition. Changes in vascular physiology in dementia may increase the vulnerability of the brain to hypoperfusion associated with antihypertensives. We discuss the potential risks of antihypertensives in the context of altered cerebral haemodynamics, and evidence from antihypertensive trials in dementia. We suggest that individualised blood pressure targets should be the focus for antihypertensive therapy in dementia, rather than strict control to uniform targets extrapolated from trials in cognitively healthy individuals.
Hypertension affects ∼50% of people aged over 50 across low, middle, and high income countries, and around half of people living with dementia in a systematic review of observational studies 1 (supplementary references [SR] #1,2). Despite this, people with dementia have been under-represented in clinical trials of antihypertensives (2) and the role of antihypertensives in dementia remains unclear. Both high and low blood pressure (BP) are associated with increased dementia risk and cognitive decline. 2 This has led to uncertainty and concern amongst clinicians around the use of antihypertensives, particularly in frailer, older people with established dementia. This commentary explores these concerns and the prevailing arguments against tight BP control for people living with established dementia or cognitive decline.
The majority of studies suggest that dynamic cerebral autoregulation) is maintained in dementia 3 (SR #3-5), but abnormalities are seen in other key aspects of cerebrovascular physiology. Specifically, dementia is associated with reduced cerebral blood flow (CBF), increased cerebrovascular resistance, and reduced CO2 reactivity and neurovascular coupling. 4 Older people with hypertension experience a rightward shift in the static cerebral autoregulation curve, 5 leaving the brain chronically adapted to higher resting perfusion states. This, in combination with reduced CBF and vasoreactivity, may increase the vulnerability of the brain to periods of hypoperfusion associated with the BP lowering effects of antihypertensives. 5 Importantly, studies have largely focussed on dynamic as opposed to static autoregulation. Given the changes demonstrated in static autoregulation amongst hypertensives, understanding this relationship in dementia is key. The brain is also vulnerable during periods of hypotension induced by orthostasis. On a meta-analysis of published trials, including The Hypertension in the Very Elderly Trial (HYVET), orthostatic hypotension was associated with a 21% increased risk of dementia. 2 Even subclinical symptoms (unsteadiness, faintness, or light-headedness) associated with everyday postural changes, such as sitting and standing were predictive of dementia risk. 2 Importantly, cerebral autoregulation is altered in orthostatic hypotension, manifesting as autoregulatory failure or intact autoregulation with an expanded range (compensatory picture) (SR #10). This may further increase the susceptibility of the brain to BP lowering effects of antihypertensives. The combination of orthostatic hypotension and supine hypertension is common in older people and is associated with large reductions in cognitive function. 5 The role for antihypertensives in this setting remains complex to manage both high and low BP simultaneously. 5 Thus, it may be beneficial to focus on identifying and managing individual BP variability, rather than focussing on tight controls or arbitrary cut-offs.
Studies suggest that CBF remains stable or increases with antihypertensives.6,7 However, CBF alone is unlikely to portray the full picture. For example, in a mouse model of hypertension-related small vessel disease, a calcium channel blocker but not angiotensin receptor blocker improved neurovascular coupling, independently of BP lowering through reduced potassium channel activity. 8 This finding demonstrates differential effects of antihypertensives on cerebrovascular physiology between drug classes, with the implication that their therapeutic benefits in dementia may be independent of their antihypertensive properties. Thus, novel therapeutic targets which improve cerebral haemodynamics and cognitive function, without BP lowering effects, may benefit people with dementia. However, to date, few studies have explored this. Studies investigating antihypertensive effects on neurovascular coupling, and other markers of vascular physiology in dementia, are also lacking.
Crucially, the role for antihypertensives in dementia risk reduction remains unclear. Meta-analyses have concluded either no or small (0.5%) reductions in dementia risk, 9 and evidence is particularly limited in established dementia. 1 The only trial with dementia as a primary end-point (SPRINT-MIND) found no benefit to aggressive BP reduction for dementia risk reduction. 10 However, intensive control did reduce the risk of mild cognitive impairment, and mild cognitive impairment combined with probable dementia, suggesting a benefit for the prevention of earlier cognitive decline. 10 Figure 1 summarises the relationship between dementia and cerebral haemodynamics, and how this may be modulated by antihypertensives.
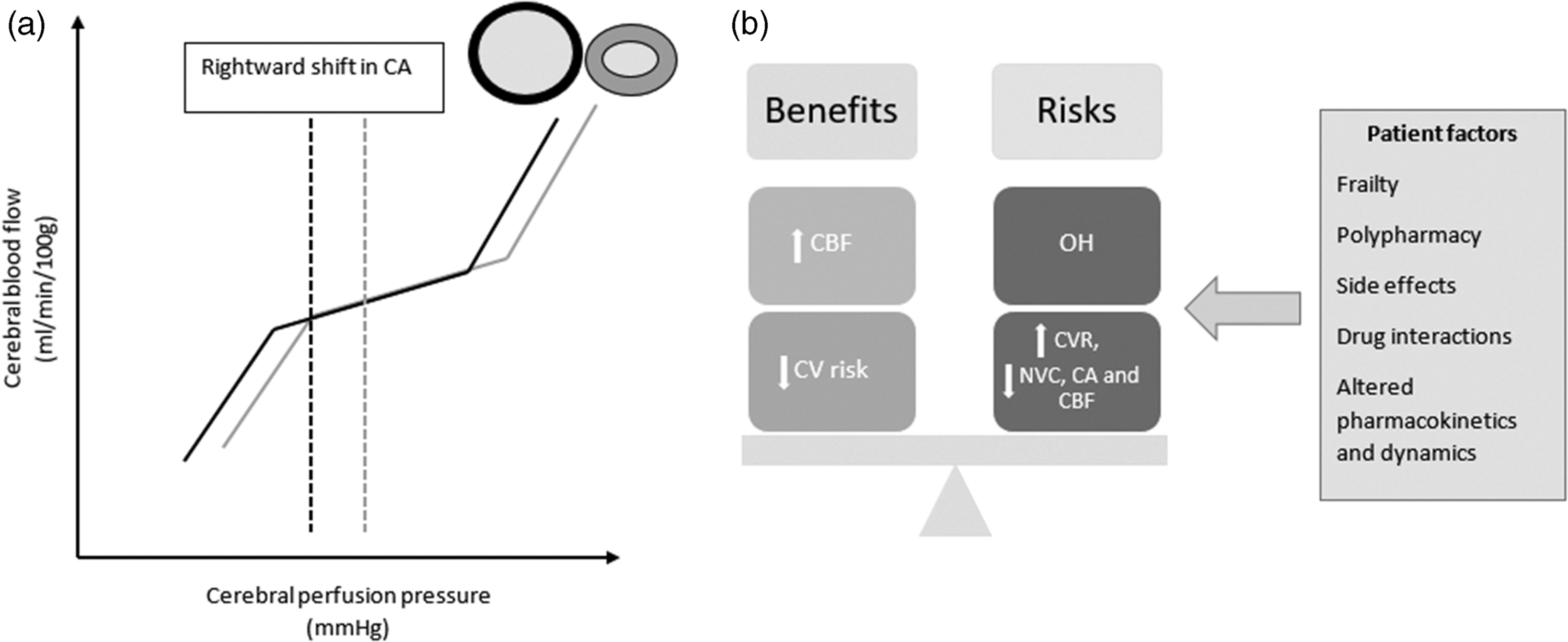
(a) Cerebral autoregulation curve demonstrating the relationship between cerebral blood flow and blood pressure. Black curve = normotensive, grey curve = hypertensive. Vessels above the curve represent lower resistance (black) and higher resistance (grey) corresponding to normotensive and hypertensive-related structural and physiological changes. The dashed line demonstrates the rightward shift in the autoregulation curve with hypertension and (b) The theoretical balance of risks and benefits, and patient factors for cerebral haemodynamics in dementia with antihypertensive treatment.
People with dementia face a number of challenges relating to altered pharmacokinetics and dynamics, multimorbidity, frailty, and polypharmacy. Previous reviews have found antihypertensives are associated with adverse events including: electrolyte disturbances, acute kidney injury, syncope, falls, fractures, and drug interactions, 1 and these effects may be amplified in dementia. 1 A scoping review examined the evidence of benefits and harms associated with antihypertensives in dementia, concluding a lack of firm evidence in this area, attributed to publication bias and the exclusion of people with dementia from these trials. 1 Most clinical trials focus on mild to moderate dementia, and treatment targets are not necessarily applicable to all dementia stages. Thus, the risk benefit ratio of antihypertensives needs to be carefully considered in the individual context.
In conclusion, although autoregulation appears to be intact in dementia, significant changes in other cerebral haemodynamic parameters are apparent. Research to date suggests that CBF remains stable or increases with antihypertensives. However, evidence from clinical trials does not support aggressive treatment of hypertension in established dementia, and significant concerns remain amongst treating clinicians. Future studies should specifically investigate different BP targets in established dementia, and the effects these have on key vascular physiological processes. These questions should be considered in the context of the additional challenges faced by people with dementia, and vascular physiology could play a key role in personalising treatment plans and targets to the individual.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221133473 - Supplemental material for Antihypertensives in dementia: Good or bad for the brain?
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221133473 for Antihypertensives in dementia: Good or bad for the brain? by Lucy Beishon, Victoria J Haunton, Ronney B Panerai in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LB is an Academic Clinical Lecturer funded by the National Institute for Health Research. The views expressed in this article are those of the author(s) and not necessarily of NIHR or the Department of Health and Social Care.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
