Abstract
To investigate whether unfavorable cerebral venous outflow (VO) predicts reperfusion hemorrhage after endovascular treatment (EVT), we conducted a retrospective multicenter cohort study of patients with acute ischemic stroke and large vessel occlusion (AIS-LVO). 629 AIS-LVO patients met inclusion criteria. VO profiles were assessed on admission CT angiography using the Cortical Vein Opacification Score (COVES). Unfavorable VO was defined as COVES ≤ 2. Reperfusion hemorrhages on follow-up imaging were subdivided into no hemorrhage (noRH), hemorrhagic infarction (HI) and parenchymal hematoma (PH). Patients with PH and HI less frequently achieved good clinical outcomes defined as 90-day modified Rankin Scale scores of ≤ 2 (PH: 13.6% vs. HI: 24.6% vs. noRH: 44.1%; p < 0.001). The occurrence of HI and PH on follow-up imaging was more likely in patients with unfavorable compared to patients with favorable VO (HI: 25.1% vs. 17.4%, p = 0.023; PH: 18.3% vs. 8.5%; p = <0.001). In multivariable regression analyses, unfavorable VO increased the likelihood of PH (aOR: 1.84; 95% CI: 1.03–3.37, p = 0.044) and HI (aOR: 2.05; 95% CI: 1.25–3.43, p = 0.005), independent of age, sex, admission National Institutes Health Stroke Scale scores and arterial collateral status. We conclude that unfavorable VO was associated with the occurrence of HI and PH, both related to worse clinical outcomes.
Introduction
Reperfusion hemorrhages after endovascular treatment (EVT) are a common complication which may be associated with poor clinical outcomes in patients suffering from acute ischemic stroke due to large vessel occlusion (AIS-LVO).1,2 Recent studies found that age, elevated blood glucose, large baseline ischemic cores, extensive cerebral edema formation and poor thrombolysis in cerebral infarction (TICI) scores predict reperfusion hemorrhage after EVT.2–8 In addition, poor pial arterial collateral status is associated with a higher risk of reperfusion hemorrhages following EVT.2,4,6,9–12
Recent investigations shed more light on venous outflow (VO) profiles as a new surrogate for cerebral collateral status and microvascular blood flow in AIS-LVO patients. Favorable VO profiles assessed before EVT were found to correlate with arterial collateral status and tissue microperfusion and were linked to smaller ischemic core volumes, higher penumbra volumes, and ultimately greater success for vessel reperfusion during thrombectomy.13–20 Whether pre-treatment VO profiles predict the occurrence of reperfusion hemorrhage in AIS-LVO patients who underwent EVT has not been reported. While several studies suggested that especially the occurrence of severe parenchymal hematoma (PH) seems to be linked to poor outcomes, other studies presumed that any form of reperfusion hemorrhage including milder forms such as hemorrhagic infarctions (HI) may be associated with less favorable neurological status 90-days after mechanical recanalization.1–3,21,22 Consequently, the identification of potential predictors and mechanisms that govern reperfusion hemorrhage is of great interest for medical aftercare of AIS-LVO patients.
We aimed to investigate whether VO profiles determined by the Cortical Vein Opacification Score (COVES) 20 on admission CT angiography images correlate with the occurrence of PH and HI in AIS-LVO patients after EVT. We hypothesized that compared to patients with poor VO, reperfusion hemorrhage is less likely to occur in patients with favorable VO profiles.
Materials and methods
Study design
In this multicenter retrospective cohort study, we analyzed the continuously maintained databases of two high-volume stroke centers between October 2013 and January 2021 (University Medical Center Hamburg-Eppendorf and Stanford University School of Medicine). We included patients with acute ischemic stroke meeting the following criteria: [1] EVT triage within 16 hours after symptom onset; [2] multimodal computed tomography comprising pretreatment non-contrast computed tomography of the head (NCCT), single-phase computed tomography angiography (CTA) and computed tomography perfusion (CTP); [3] acute ischemic stroke due to anterior circulation large vessel occlusion of the internal carotid artery (ICA) or of the M1 or M2 segment of the middle cerebral artery (MCA); [4] EVT; [5] follow-up imaging (NCCT or magnetic resonance imaging) for assessment of reperfusion hemorrhages within 48 hours after EVT. Patients who were not treated by EVT including those who showed spontaneous recanalization of the occluded vessel on the first run of the digital subtraction angiography, were not included into our study. We excluded 9 patients due to the detection of intraprocedural bleedings in the angiography suite (all of them subarachnoid hemorrhage; median COVES of 1 [interquartile range 0–2]). A total of 629 patients met the inclusion criteria (Supplementary Figure 1).
Of the 796 patients which were assessed for eligibility, data from 649 patients were reported in a previous study on the correlation between tissue-level collaterals and VO profiles in AIS-LVO patients. 14 Data from 565 patients were reported in another study which assessed the correlation between pre-treatment VO profiles and thrombectomy success. 15 A total of 580 patients were included into a previous study which assessed the correlation between VO profiles and ischemic edema formation in AIS-LVO patients after vessel reperfusion treatment. 16 A recent study which introduced a comprehensive approach of cerebal blood flow in ischemic stroke included 647 patients. 23
Subjects were divided into patients with favorable venous outflow (COVES ≥3; VO+) and unfavorable venous outflow (COVES ≤ 2; VO−) based on the threshold of previous studies. 15 Successful mechanical recanalization was defined as TICI grade of ≥2b on digital subtraction angiography images. The 90-day modified Rankin Scale (mRS) scores were determined by a stroke neurologist or registered study nurse either at a follow-up examination in the respective stroke center or by a standardized phone interview.
The study was approved by the institutional review boards at each of the included centers (University Medical Center Hamburg-Eppendorf and Stanford University School of Medicine). The study was conducted in accordance with the ethical guidelines of the local ethics committees and in accordance with the Declaration of Helsinki and the Health Insurance Portability and Accountability Act (HIPAA). Informed consent was waived by our institutional review boards due to the retrospective design of the study.
Assessment of reperfusion hemorrhages after reperfusion therapy
Follow-up imaging (NCCT or magnetic resonance imaging) was obtained within 48 hours after EVT. Reperfusion hemorrhages were classified on follow-up imaging according to the Heidelberg bleeding classification criteria based on the radiographic distinction between hemorrhagic infarction (HI) and parenchymal hematoma (PH).1,24 Both HI and PH were initially divided into the grades 1 and 2: Hemorrhagic infarction 1 (HI1) was defined as small petechiae along the margins of the infarct; hemorrhagic infarction 2 (HI2) as confluent petechiae within the infarcted area without mass effect; parenchymal hematoma 1 (PH1) as hematoma occupying 30 % or less of the infarcted tissue with no substantial mass effect; and parenchymal hematoma 2 (PH2) as hematoma occupying 30% or more of the infarcted tissue with obvious mass effect. The classification of reperfusion hemorrhages was performed by three neuroradiologists (L.W., V.G. and T.D.F.).
Symptomatic intracranial hemorrhage (sICH) was defined according to ECASS III as a new parenchymal hemorrhage on follow-up imaging after EVT together with a clinically evident neurological worsening presumably caused by the hemorrhage (i.e. a minimum of ≥4 points increase of 24 h NIHSS compared to admission NIHSS). 25
Assessment of cerebral venous outflow
VO profiles were evaluated on admission single-phase CTA using the Cortical Vein Opacification Score (COVES), which reflects the major venous drainage pathways of the MCA territory. 19 COVES is obtained by grading the opacification of three major cortical veins, namely the superficial middle cerebral vein, vein of Labbé and sphenoparietal sinus, on a scale from 0 to 2 points (0: not visible, 1: moderate opacification, 2: full opacification). Consequently, COVES ranges from 0 to 6 points. Unfavorable VO was defined as COVES ≤2 based on previous studies. 14
VO assessment was independently performed by two experienced neuroradiologists (T.D.F. and J.J.H., with 10 and 15 years of working experience). Inter-rater reliability for COVES determination was substantial (kappa = 0.71) and thus in line with previous studies.14,20 Discrepancies were settled by consensus readings. Only patients with sufficient CTA imaging quality to determine cortical VO profiles were included, as described in previous studies (complete opacification of the dural and sigmoid sinuses).14,16
Assessment of arterial collateralization
Pial arterial collaterals were assessed on admission CTA imaging using the Maas system. 26 A favorable Maas Score was defined as an equal or greater arterial filling of the affected hemisphere compared to the unaffected hemisphere (Maas score ≥3). Maas scoring was independently performed by two experienced neuroradiologists (T.D.F. and J.J.H., with 10 and 15 years of working experience).
CT perfusion image analysis
Fully automated software (RAPID, iSchemaView, Menlo Park, CA) was used to analyze CT perfusion (CTP) studies. Baseline ischemic core volume was identified by RAPID as tissue with a relative cerebral blood flow (CBF) of <30% compared to mean CBF of the contralateral unaffected hemisphere. Penumbral tissue at risk was defined as tissue with a time to maximum (Tmax) of >6 s and the target mismatch profiles were determined in each patient.
Outcome measures
The occurrence of PH (PH1 or PH2) and HI (HI1 or HI2) on follow-up imaging were the primary outcome measures. Secondary outcome measure was good clinical outcome defined as 90-day mRS score ≤2.
Statistical analysis
Statistical tests and data visualization were performed using R statistical software (version 4.1.2, R Project for Statistical Computing) and RStudio statistical software (version 2021.09.1 + 372, Rstudio). We compared demographics, medical history, current stroke event characteristics, imaging findings and treatment outcomes between patients with favorable and unfavorable venous outflow (see Tables 1 and 2). For sensitivity analysis, we dichotomized EVT patients according to [1] the time from symptom onset to imaging (see Supplementary Table 1) and [2] the TICI score (see Supplementary Table 2). Categorical variables were reported as counts and percentages. Continuous variables were reported as mean and standard deviation or as median and interquartile range. Categorical variables and continuous variables were compared using chi-squared test (χ2) and Mann-Whitney U test, respectively. Multivariable logistic regression models were built to determine variables significantly associated with the occurrence of PH (PH1 or PH2; model PH) or HI (HI1 or HI2; model HI). A total of 600 patients was included into the model PH (after excluding patients with unknown blood glucose levels, unknown admission NIHSS and unusable CTP studies) and a total of 505 patients was included into the model HI (after excluding patients presenting with PH and unknown anesthesia regime). Candidate variables were selected based on previous studies:6,21,22,27 Admission National Institutes of Health Stroke Scale (NIHSS), hypertension, smoking behavior, usage of antiplatelet or anticoagulant agents, blood glucose on admission, extend of baseline infarction, arterial collateral status, administration of tissue plasminogen activator (tPA), general anesthesia, time from symptom onset to imaging, time to groin puncture, mechanical recanalization status. Candidate variables with a p-value of ≤0.2 in the univariate logistic regression analysis were included as independent variables into the multivariable regression models (see Supplementary Table 3). Both multivariable regression analyses were adjusted for the effects of age, sex and study center. The model PH included the following independent variables: Age, sex, usage of antiplatelet or anticoagulant agents, admission NIHSS, blood glucose on admission, baseline ischemic core volume, arterial collateral status, VO profiles and study center (see Table 3). The model HI included the same independent variables as the model PH and additionally the use of general anesthesia and the time window from symptom onset to imaging (see Table 4). In addition, we performed a multivariable regression analysis to identify independent determinants of 90-day mRS score ≤ 2 (see Supplementary Table 4). We calculated the variance of inflation factor (VIF) for each independent variable to exclude multicollinearity in the regression models. A two-tailed p-value of <0.05 was considered significant for all statistical tests.
Patient and imaging characteristics dichotomized by venous outflow.
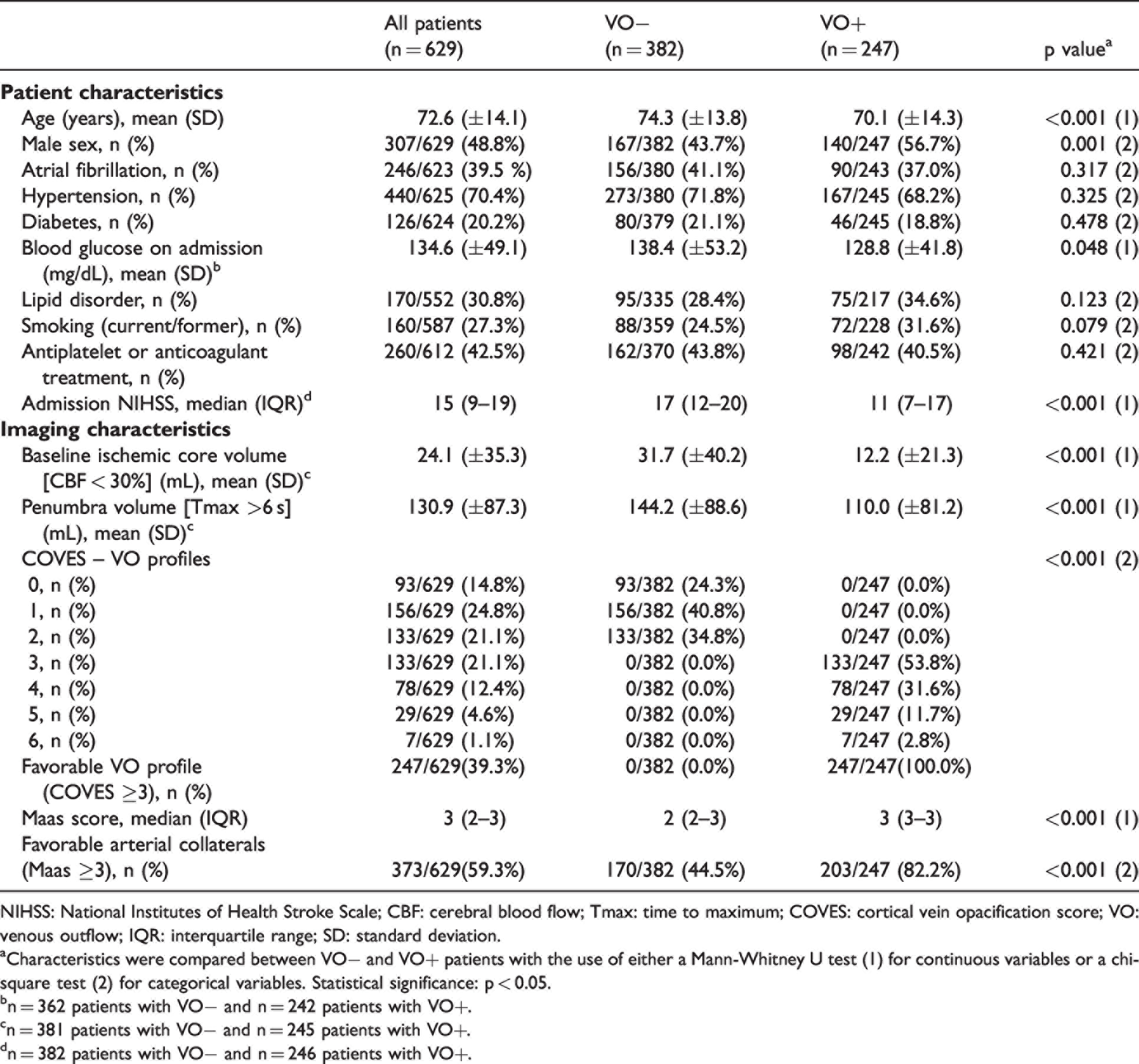
NIHSS: National Institutes of Health Stroke Scale; CBF: cerebral blood flow; Tmax: time to maximum; COVES: cortical vein opacification score; VO: venous outflow; IQR: interquartile range; SD: standard deviation.
aCharacteristics were compared between VO− and VO+ patients with the use of either a Mann-Whitney U test (1) for continuous variables or a chi-square test (2) for categorical variables. Statistical significance: p < 0.05.
bn = 362 patients with VO− and n = 242 patients with VO+.
cn = 381 patients with VO− and n = 245 patients with VO+.
dn = 382 patients with VO− and n = 246 patients with VO+.
Procedural characteristics, follow-up imaging and clinical outcomes.
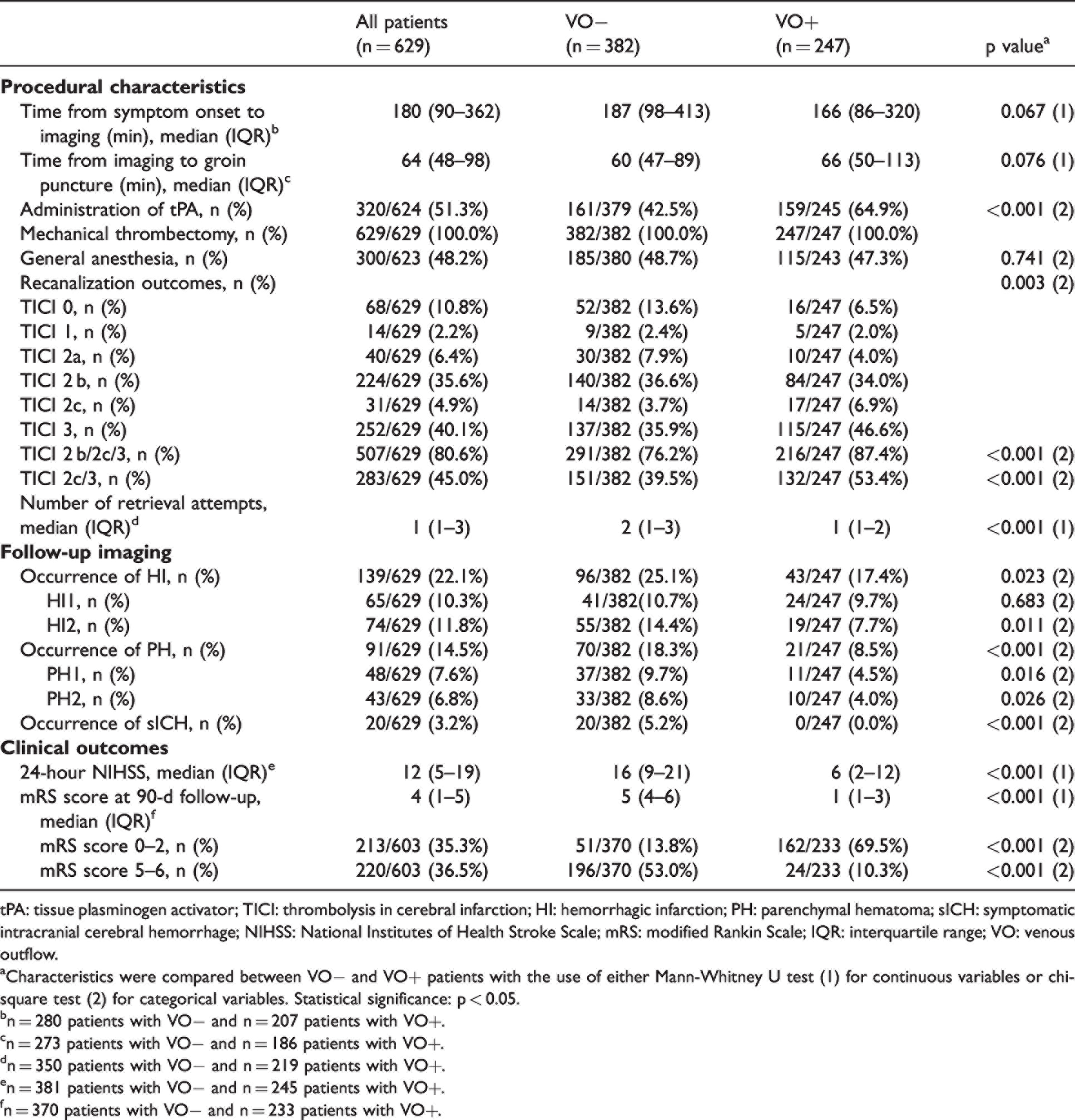
tPA: tissue plasminogen activator; TICI: thrombolysis in cerebral infarction; HI: hemorrhagic infarction; PH: parenchymal hematoma; sICH: symptomatic intracranial cerebral hemorrhage; NIHSS: National Institutes of Health Stroke Scale; mRS: modified Rankin Scale; IQR: interquartile range; VO: venous outflow.
aCharacteristics were compared between VO− and VO+ patients with the use of either Mann-Whitney U test (1) for continuous variables or chi-square test (2) for categorical variables. Statistical significance: p < 0.05.
bn = 280 patients with VO− and n = 207 patients with VO+.
cn = 273 patients with VO− and n = 186 patients with VO+.
dn = 350 patients with VO− and n = 219 patients with VO+.
en = 381 patients with VO− and n = 245 patients with VO+.
fn = 370 patients with VO− and n = 233 patients with VO+.
Multivariable logistic regression to predict parenchymal hematoma (PH) on follow-up imaging in patients with acute ischemic stroke (model PH).
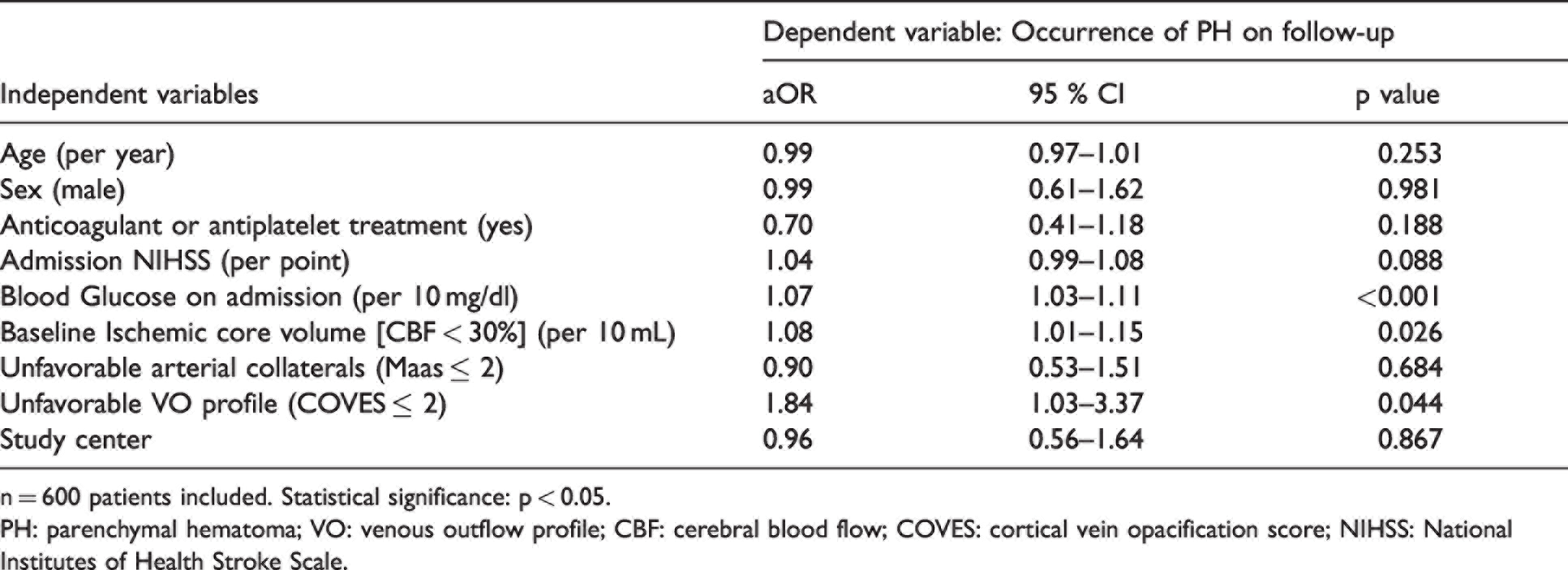
n = 600 patients included. Statistical significance: p < 0.05.
PH: parenchymal hematoma; VO: venous outflow profile; CBF: cerebral blood flow; COVES: cortical vein opacification score; NIHSS: National Institutes of Health Stroke Scale.
Multivariable logistic regression to predict hemorrhagic infarction (HI) on follow-up imaging in patients with acute ischemic stroke (model HI).
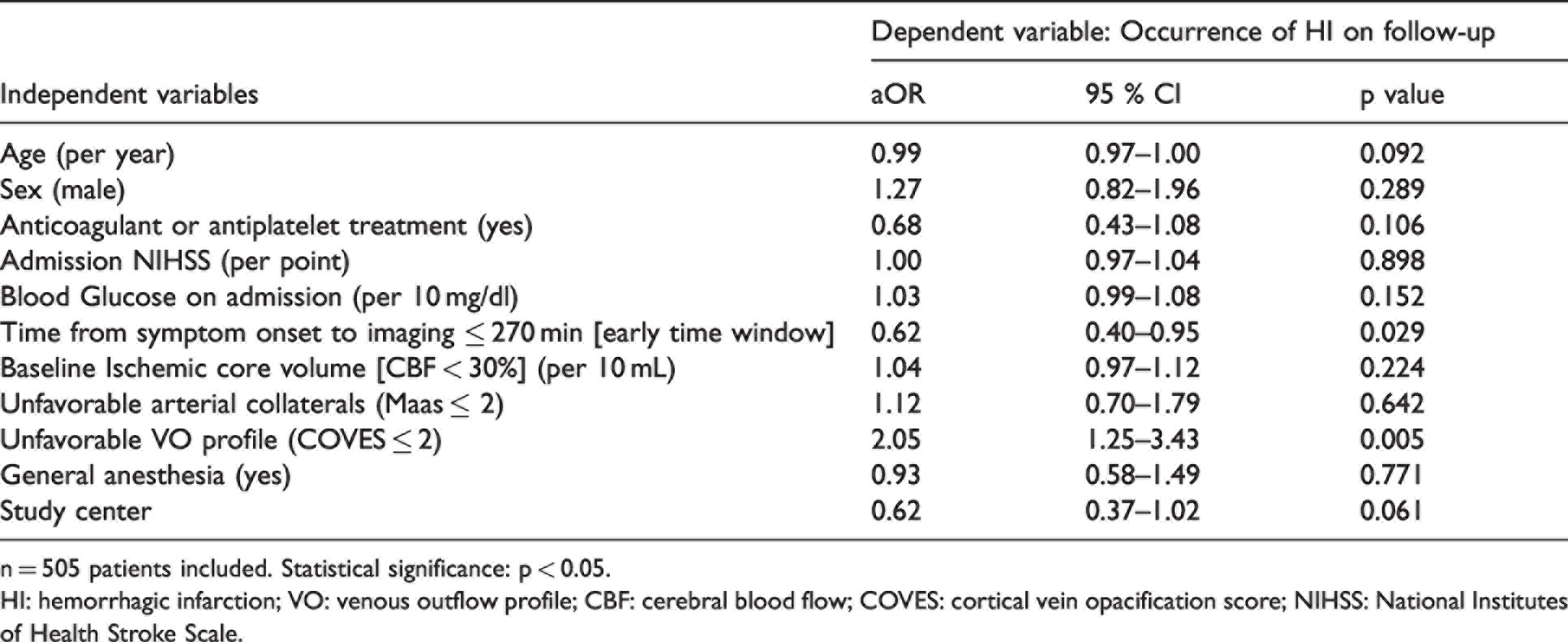
n = 505 patients included. Statistical significance: p < 0.05.
HI: hemorrhagic infarction; VO: venous outflow profile; CBF: cerebral blood flow; COVES: cortical vein opacification score; NIHSS: National Institutes of Health Stroke Scale.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Results
Patient characteristics
A total of 629 patients presenting with AIS-LVO met the inclusion criteria (Supplementary Figure 1). Among these patients, the mean age was 73 years and 307 (48.8%) were male. On pretreatment CTA, favorable arterial collaterals (Maas score ≥ 3) were present in 373 (59.3%) patients and therefore more frequent than favorable VO profiles (COVES ≥ 3) which were observed in 247 (39.3%) patients. At clinical evaluation after 90 days, 213 (35.3%) patients achieved good clinical outcomes defined as mRS score ≤2. Please refer to Table 1 and Table 2 for detailed information about patient, imaging and treatment characteristics.
Association between reperfusion hemorrhages and clinical outcomes
Figure 1(a) displays the frequencies of reperfusion hemorrhages after EVT. No reperfusion hemorrhage (noRH) was observed in 399 (63.4%) patients while any type of reperfusion hemorrhage after EVT was found in 230 (36.6%) patients on follow-up imaging. Of these patients, 139 (22.1%) patients exhibited HI and 91 (14.5%) patients PH. The occurrence of HI and PH did not differ significantly between patients with near complete (TICI 2 b) and complete reperfusion (TICI 2c/3) (see supplementary Table 2). We further analyzed whether the 90-day clinical outcomes differed according to the extent of hemorrhagic transformation after EVT. Patients with HI or PH on follow-up imaging showed a higher rate of poor clinical outcomes defined as mRS scores of 5–6 (PH: 55.7% vs. HI: 44.8% vs. noRH: 29.1%; p = <0.001) and a lower rate of good clinical outcomes defined as mRS scores of 0–2 (PH: 13.6% vs. HI: 24.6% vs. noRH: 44.1%; p = <0.001) compared to noRH patients. The distributions of mRS scores with respect to the type of reperfusion hemorrhage are displayed in Figure 1(b). Both the occurrence of PH (adjusted odds ratio [aOR]: 0.26; 95% confidence interval [CI]: 0.11–0.57, p = <0.001) and HI (aOR: 0.32, 95% CI: 0.17–0.60, p = <0.001) were independently associated with lower odds of achieving good clinical outcomes 90 days after stroke (see Supplementary Table 4).
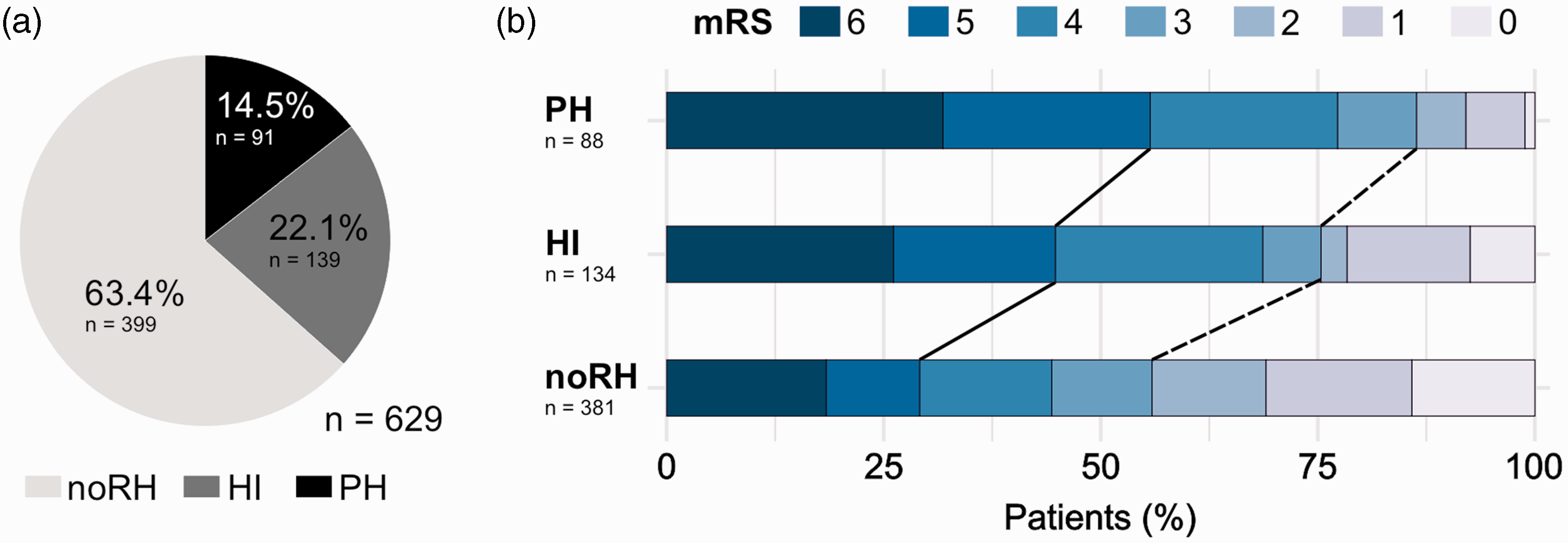
Association between Reperfusion Hemorrhages and Clinical Outcomes. (a) Pie chart illustrating the frequency of HI and PH detected on follow-up imaging in AIS-LVO patients after EVT and (b) 90-day clinical outcomes stratified by reperfusion hemorrhage types. Note that the rate of poor clinical outcomes (90-day mRS 5–6) followed PH > HI > noRH (p = <0.001; black solid line), while the rate of good clinical outcomes (90-day mRS 0–2) inversely followed PH < HI < noRH (p = <0.001; black dashed line).
Association between reperfusion hemorrhages and cerebral venous outflow
The relationship between the extent of reperfusion hemorrhage and functional outcomes highlights the possible benefit of predictive biomarkers in clinical practice. We hypothesized that the status of cerebral venous drainage correlates with the occurrence of HI and PH. Therefore, the study cohort was dichotomized into patients with favorable (VO+) and unfavorable (VO−) venous outflow profiles on admission CTA (see Figure 2(a) and (b)). Please refer to Table 1 and Table 2 for detailed information about patient, imaging and treatment characteristics. As hypothesized, both the occurrence of HI (25.1% vs. 17.4%; p = 0.023) and PH (18.3% vs. 8.5%; p = <0.001) was more frequent in the VO- group on follow-up imaging (see Figure 2(c)).
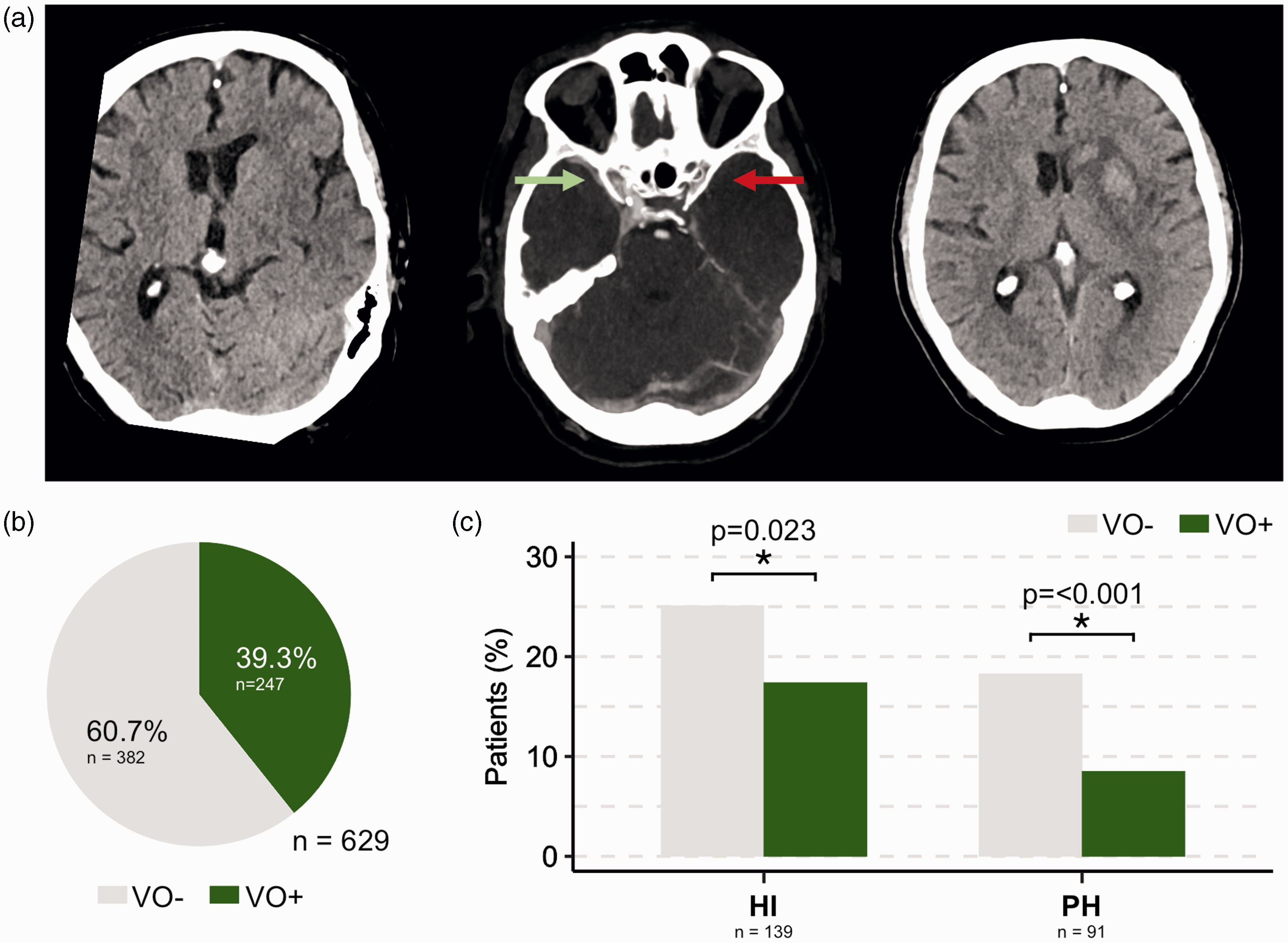
Association between Reperfusion Hemorrhages and Venous Outflow. (a) Patient with acute ischemic stroke due to occlusion of the left MCA in segment M1. Left: No early infarct signs were present on admission NCCT. Middle: Admission CTA revealed a poor cerebral venous outflow of the affected left hemisphere with missing contrast filling of the left sinus sphenoparietalis (red arrow) and full contrast filling of its right counterpart. Right: Parenchymal hematoma within the infarcted tissue was observed on follow-up imaging. (b) Pie chart illustrating the rate of favorable (VO+; green) and unfavorable (VO−; grey) venous outflow on admission CTA and (c) Bar charts indicating the frequency of HI (left) and PH (right) after EVT on follow-up imaging. Note that both the occurrence of HI and PH were significantly less frequent in AIS-LVO patients with favorable venous drainage (see Table 2 for statistics).
Predictors of parenchymal hematoma after mechanical thrombectomy
Our descriptive statistics suggest a relevant association between unfavorable cerebral VO and PH after EVT. For further analysis, a multivariable logistic regression model was designed to determine factors that were independently associated with the occurrence of PH (model PH). A total of 600 AIS-LVO patients was included into the model. Variable selection was based on prior studies and the results from univariate regression analyses (see Methods and Supplementary Table 3). We found that unfavorable VO profiles (COVES ≤2) significantly increased the likelihood of PH by the factor 1.84 (adjusted odds ratio [aOR], 95% confidence interval [CI]: 1.03–3.37, p = 0.044). Higher blood glucose on admission (aOR: 1.07 [per 10 mg/dl], 95% CI: 1.03–1.11, p = <0.001) and larger baseline ischemic core volume on admission CTP (aOR: 1.08 [per 10 ml], 95% CI: 1.01–1.15, p = 0.026) were independently associated with greater odds of PH. Model estimates were adjusted for age, sex, usage of antiplatelet or anticoagulant agents, admission NIHSS, arterial collateral status and study center. Predicted probabilities from the logistic regression model are illustrated in Figure 3, showing the impact of blood glucose levels and baseline ischemic core volumes on the likelihood of PH in dependence of VO status.
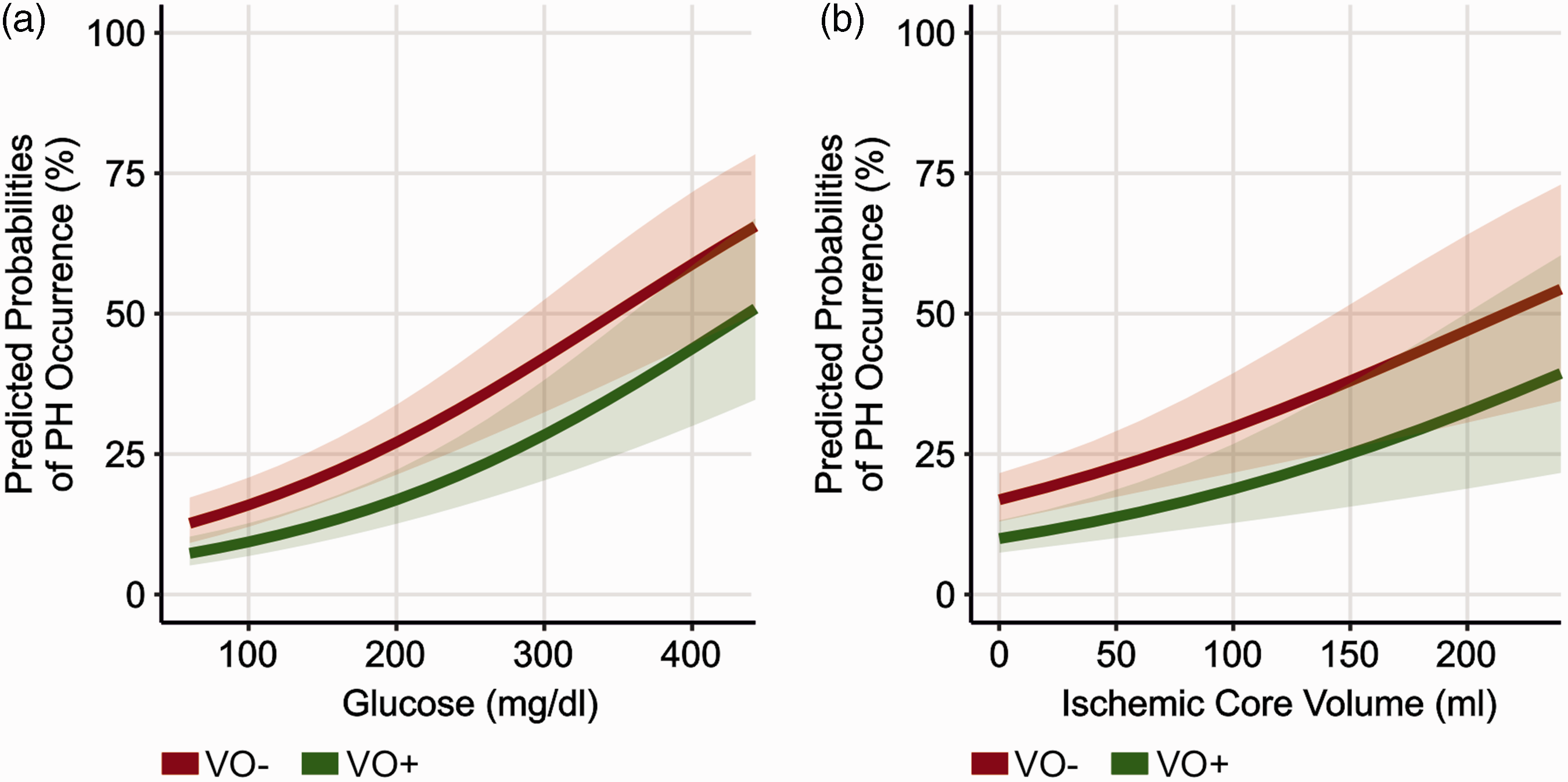
Predictors of Parenchymal Hematoma after Mechanical Thrombectomy. (a) Predicted probabilities from the multivariable logistic regression displayed in Table 3 (model PH). Note that poor cerebral VO profiles increased the predicted probability of PH on follow-up imaging depending on blood glucose levels on admission. Shaded error bars represent one standard deviation and
Predictors of hemorrhagic infarction after mechanical thrombectomy
A second multivariable logistic regression was performed to determine predictive variables for the occurrence of HI on follow-up imaging after EVT. 505 patients were included into a second multivariable logistic regression model (model HI; see Table 4). Noteworthy, unfavorable VO status was independently associated with a higher likelihood of HI on follow-up imaging (aOR: 2.05, 95% CI: 1.25–3.43; p = 0.005). In addition, patients within the early time window (time from symptom onset to imaging ≤ 270 minutes) were less likely to exhibit HI after EVT.
Predictors of sICH after mechanical thrombectomy
We aimed to identify determinants of sICH after EVT by means of univariable logistic regression. Interestingly, we found that unfavorable VO profiles, higher blood glucose on admission and larger baseline ischemic core volume were significantly correlated with an increased risk of sICH (see Supplementary Table 3). Please note that a multivariable logistic regression was not performed given the small number of n = 20 patients presenting with sICH.
Discussion
In this multicenter cohort study of AIS-LVO patients, we investigated whether VO profiles determined on admission CT angiography imaging predict the occurrence of reperfusion hemorrhages after thrombectomy treatment. We found that unfavorable VO profiles, elevated baseline blood glucose and large ischemic core volumes were independent determinants of PH after EVT regardless of arterial CTA collaterals. The independent determinants of PH, namely unfavorable VO profiles, higher blood glucose levels and larger baseline ischemic core volume, were also correlated with sICH occurrence (unadjusted results). Unfavorable VO profiles were also significantly associated with the occurrence of HI after EVT. In addition, unfavorable VO profiles, HI and PH were independently associated with lower odds of achieving good clinical outcomes (90-day mRS 0–2). A significantly higher number of patients with HI or PH after treatment exhibited poor clinical outcomes (90-day mRS 5–6) compared to patients who had no reperfusion hemorrhage. Our study may have important implications for the management of AIS-LVO patients after EVT and reveals new insights into stroke pathophysiology with respect to the role of VO profiles and the emergence of reperfusion bleedings.
The most striking and novel finding of our study was that unfavorable VO profiles (i.e. COVES of 0–2) were associated with the occurrence of any reperfusion hemorrhage after treatment regardless of pial arterial collateral profiles on CTA. Cortical vein opacification is thought to be a more sensitive biomarker of microvascular blood transit compared to conventional collateral biomarkers on CTA, as it reflects blood flow after permeating the brain tissue. 14 In addition, VO was found to correlate with other biomarkers that strongly predict reperfusion hemorrhage after EVT, such as admission NIHSS, quantitative ischemic lesion formation on follow-up imaging, as well as baseline ischemic core and penumbra volumes.13,14,16,17,19,20 Favorable VO profiles have also been reported to correlate with successful vessel reperfusion rates, shorter procedural times and first pass clearance of the occluded vessel, which are other procedural parameters that have been found to influence the occurrence of reperfusion hemorrhages following EVT.15,18 Pathophysiologically, one can speculate that favorable VO profiles correlate with prolonged tissue viability and reduced ischemic damage to the brain, which would be in line with the assumptions of previous studies. 14 Consequently, the reduced likelihood for reperfusion hemorrhages may result from less disruption of the blood-brain barrier and maintenance of microvascular blood flow within the respective ischemic regions.
The administration of tPA was not found to be a significant determinant of reperfusion hemorrhages after EVT, as reported by previous studies.21,27 However, we observed a higher rate of tPA administration in patients with favorable compared to unfavorable VO profiles. This finding is in line with a previous study of our group, suggesting that the thrombolytic activity of tPA may reduce arteriole and venuole microvascular thrombosis in the setting of a proximal artery occlusion, which then allows for more favorable VO. 28
While most studies in the recent literature focused on the identification of factors that predict symptomatic or severe reperfusion hemorrhages, we found that unfavorable VO profiles were a predictor of HI after EVT. Noteworthy, we found that patients who developed any reperfusion hemorrhages (either PH or HI) were likely to have worse clinical outcomes. These findings are in line with a recent investigation of the DEFUSE 3 cohort that found that the occurrence of any reperfusion bleeding after thrombectomy predicted poor long-term functional outcomes.2,4 The importance of mild reperfusion hemorrhages after stroke treatment still remains unclear. While some studies found mild reperfusion hemorrhages after stroke treatment to be an indicator for successful vessel reperfusion and a predictor of good functional outcomes, 29 several studies reported that the occurrence of post-treatment HI was directly related to poor outcomes and to lower rates of excellent clinical outcomes after 90 days.2,7,30–34 The potential impact of mild reperfusion hemorrhage after stroke treatment on functional outcomes requires further study. Nevertheless, it is likely that the development of post-EVT hemorrhage is mediated by a multifactorial genesis, driven by an interplay of baseline clinical and imaging parameters, collateral blood flow and procedural outcomes in AIS-LVO patients. More studies are needed to investigate the underlying mechanisms in more detail.
Our findings are in line with previous studies that found extensive baseline infarct core volumes on perfusion imaging to be correlated with parenchymal hematoma after endovascular reperfusion therapy.2,4 Large infarct core volumes were reported to correlate strongly with impaired tissue microperfusion in numerous studies.6,7,10,14,35
Our findings also indicate that elevated blood glucose measures were associated with the occurrence of PH after treatment. The mechanisms of how blood glucose is related to PH are not fully understood. Yet, several studies found a strong correlation of elevated blood glucose levels with extended baseline edema formation on non-contrast head CT, poor pial arterial collateral profiles on CTA, higher infarct core volumes and a reduced likelihood of successful recanalization during thrombectomy, all of which are associated with more extensive tissue damage and consequently greater likelihoods of reperfusion hemorrhage.2–5,7,9,10,36 Based on these findings, it has been speculated whether immediate and extensive antihyperglycemic treatment of increased blood glucose levels in AIS-LVO patients would improve clinical outcomes, but ambiguous findings have been published in the literature.37–39 Moreover, is remains unclear whether elevated blood glucose profiles affect cerebral VO. Further studies are warranted to assess a potential correlation between these two biomarkers in detail.
This study has certain limitations. Importantly, due to the retrospective study design and the lack of a control group, our data does not justify to preclude stroke patients with a high predicted probability of PH from EVT. VO assessment is known to be affected by the selected CTA imaging protocol, including acquisition timing and rate of contrast injection. 19 Anatomical variants of cortical veins may introduce bias to VO determination. The inclusion of patients from two comprehensive stroke centers which routinely perform perfusion imaging may limit the generalizability of our results.
Conclusion
In this multicenter cohort study of AIS-LVO patients, we found poor VO profiles to be associated with an increased risk of reperfusion hemorrhages after thrombectomy treatment, independent from conventional pial arterial collateral measurements on CT angiography. Patients in which HI or PH bleeds occurred had significantly worse clinical outcomes compared to patients who did not develop any reperfusion hemorrhage after treatment. VO profiles may serve as a biomarker for risk stratification of reperfusion hemorrhages and help to assess the need for intensified monitoring in the ICU after EVT.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221127089 - Supplemental material for Poor venous outflow profiles increase the risk of reperfusion hemorrhage after endovascular treatment
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221127089 for Poor venous outflow profiles increase the risk of reperfusion hemorrhage after endovascular treatment by Laurens Winkelmeier, Jeremy J Heit, Gautam Adusumilli, Vincent Geest, Adrien Guenego, Gabriel Broocks, Julia Prüter, Nils-Ole Gloyer, Lukas Meyer, Helge Kniep, Maarten G Lansberg, Gregory W Albers, Max Wintermark, Jens Fiehler and Tobias D Faizy in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Tobias Djamsched Faizy was funded by the German Research Foundation (DFG) for his work as a postdoctoral research scholar at Stanford University, Department of Neuroradiology (Project Number: 411621970).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Laurens WINKELMEIER reports no disclosure.
Dr Jeremy J. HEIT reports consulting for Medtronic and MicroVention and Medical and Scientific Advisory Board membership for iSchemaView.
Dr Gautam ADUSUMILI reports no disclosure.
Dr Vincent GEEST reports no disclosure.
Dr Adrien GUENEGO reports no disclosure.
Dr Gabriel BROOCKS reports no disclosure.
Julia PRÜTER reports no disclosure.
Nils-Ole GLOYER reports no disclosure.
Dr Lukas MEYER reports no disclosure.
Dr Helge KNIEP reports no disclosure.
Dr Maarten G. LANSBERG reports no disclosure.
Dr Gregory W. ALBERS reports equity and consulting for iSchemaView and consulting from Medtronic.
Dr Max WINTERMARK reports grants and funding from the NIH under the grant numbers (1U01 NS086872-01, 1U01 NS087748-01 and 1R01 NS104094).
Dr Jens FIEHLER reports grants and personal fees from Acandis, Cerenovus, MicroVention, Medtronic, Stryker, Phenox and grants from Route 92 outside the submitted work.
Dr Tobias D. FAIZY reports grants from the German Research Foundation (DFG) during the conduct of the study.
Authors’ contributions
We affirm that all individuals listed as authors agree that they have met the criteria for authorship, agree to the conclusions of the study, and that no individual meeting the criteria for authorship has been omitted. LW, JJH, JF and TDF conceived the project. LW, JJH, GA, VG, JP, NOG and TDF acquired, analyzed and interpreted the data.
LW, JJH and TDF drafted the manuscript. LW, JJH, VG, AG, GB, LM, HK, MGL, GWA, MW, JF and TDF revised the manuscript and contributed important intellectual content. The project was supervised by JJH, JF and TDF
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
