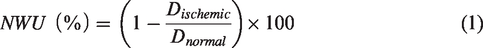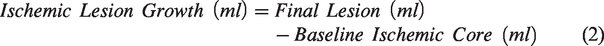Abstract
Ischemic lesion net water uptake (NWU) represents a quantitative imaging biomarker for cerebral edema in acute ischemic stroke. Data on NWU for distinct occlusion locations remain scarce, but might help to improve the prognostic value of NWU. In this retrospective multicenter cohort study, we compared NWU between patients with proximal large vessel occlusion (pLVO; ICA or proximal M1) and distal large vessel occlusion (dLVO; distal M1 or M2). NWU was quantified by densitometric measurements of the early ischemic region. Arterial collateral status was assessed using the Maas scale. Regression analysis was used to investigate the relationship between occlusion location, NWU and ischemic lesion growth. A total of 685 patients met inclusion criteria. Early ischemic lesion NWU was higher in patients with pLVO compared with dLVO (7.7% vs 3.9%, P < .001). The relationship between occlusion location and NWU was partially mediated by arterial collateral status. NWU was associated with absolute ischemic lesion growth between admission and follow-up imaging (β estimate, 5.50, 95% CI, 3.81–7.19, P < .001). This study establishes a framework for the relationship between occlusion location, arterial collateral status, early ischemic lesion NWU and ischemic lesion growth. Future prognostic thresholds for NWU might be optimized by adjusting for the occlusion location.
Introduction
In acute ischemic stroke due to anterior circulation large vessel occlusion (ACLVO), cytotoxic cerebral edema constitutes a leading pathomechanism of clinical deterioration and death. 1 The identification of imaging biomarkers for cerebral edema and its progression is crucial to improve acute treatment, medical aftercare, and, ultimately, functional outcomes of ischemic stroke patients.
Previous studies introduced ischemic lesion net water uptake (NWU) at the time of hospital admission as a prognostic imaging biomarker in ACLVO.2,3 The ischemic lesion NWU per volume is quantified by densitometric measurements on admission non-contrast computed tomography. It has been shown that this widely available imaging parameter is associated with lesion age,4 –6 collateral status,7,8 hemorrhagic transformation, 9 and space-occupying malignant infarction in acute ischemic stroke.2,3 First approaches for automated NWU estimation show potential to implement this parameter into clinical practice.10,11
The question remains how to make effective use of this quantitative imaging biomarker to support treatment decision making and outcome prediction in endovascular therapy for ACLVO. For example, a recent study established an early ischemic lesion NWU of >12.7% as threshold value to predict malignant infarction after endovascular therapy. 2 Whether ischemic lesion NWU differs between proximal and distal occlusion locations in the anterior circulation remains unclear, but might have important implications for the definition of optimal threshold values.
We sought to provide an in-depth analysis of the relationship between the occlusion location and early ischemic lesion NWU in ACLVO patients who received endovascular therapy. We hypothesized that more proximal vessel occlusions are associated with greater voxel-wise cerebral edema, potentially explained by the more likely failure of primary and secondary arterial collaterals.
Materials and methods
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request after approval of both participating centers and ethic committees.
Study design
This retrospective multicenter cohort study included patients with anterior circulation large vessel occlusion who underwent endovascular therapy between October 1, 2013 and January 31, 2021 at one of two participating stroke centers (University Medical Center Hamburg-Eppendorf, Germany and Stanford University School of Medicine, California). Baseline patient, imaging, and treatment characteristics were obtained from electronic medical records and imaging assessment. This study was approved by the institutional review boards at each participating center and was conducted in accordance with the Declaration of Helsinki and the Health Insurance Portability and Accountability Act (HIPAA). Informed consent was waived by the institutional review boards due to the retrospective design of the study. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline was used for reporting this observational study. 12
Study cohort
Inclusion criteria were defined as follows: [1] triage for endovascular therapy with non-contrast head computed tomography (NCCT), single-phase computed tomography angiography (CTA), and computed tomography perfusion (CTP); [2] acute ischemic stroke in the anterior circulation due to proximal large vessel occlusion (pLVO; internal carotid artery (ICA) and proximal M1 segment of the middle cerebral artery) or due to distal large vessel occlusion (dLVO; distal M1 segment and M2 segment of the middle cerebral artery); [3] endovascular therapy; [4] follow-up imaging within 48 hours after endovascular therapy. Supplementary Figure S1 provides a detailed flowchart of the inclusion and exclusion criteria.
Assessment of net water uptake
The NWU of the early ischemic region was quantified on admission NCCT using a standardized procedure as described previously.2,4 Briefly, we performed densitometric measurements of the hypoattenuated ischemic region on NCCT (Dischemic) and of a mirrored region of interest within the contralateral non-ischemic hemisphere (Dnormal). Voxels with Hounsfield units <20 were considered as cerebrospinal fluid and removed from the regions of interest. In patients without identifiable hypoattenuation on NCCT, cerebral blood volume (CBV) maps derived from CTP imaging were used to define the ischemic region (Figure 1). Ischemic lesion NWU was calculated according to the formula (1):
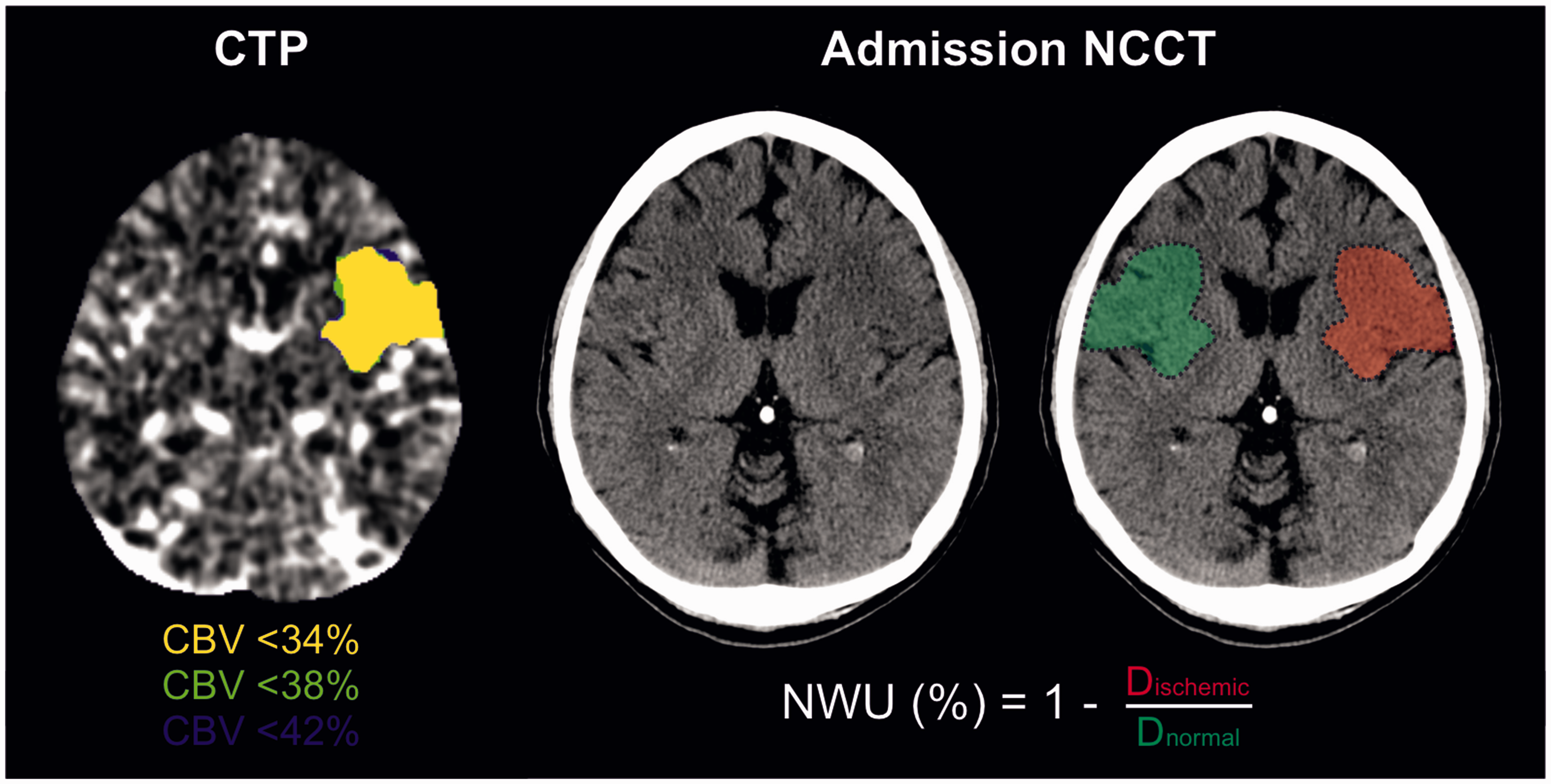
Assessment of net water uptake on admission non-contrast computed tomography.
The early edema progression rate was determined as NWU on admission NCCT divided by time from symptom onset to imaging.
Assessment of arterial collaterals
The extent of pial arterial collaterals was assessed on admission CTA using the Maas scale. 13 Favorable arterial collateral status was defined as a Maas score of ≥3, which indicates an equal or greater filling of the collateral vessels in the Sylvian fissure and leptomeningeal convexity on the affected hemisphere compared to the unaffected hemisphere. Ratings were performed by two experienced neuroradiologists (T.D.F. and J.J.H., with 12 and 17 years of working experience).
Assessment of ischemic lesion growth
The software platform RAPID (iSchemiaView, Menlo Park, California) was used for automated CTP analysis. Baseline ischemic core volume was identified as brain tissue with a relative cerebral blood flow (CBF) of less than 30% compared with the average cerebral blood flow of brain tissue in any region without Tmax delay. Penumbra volume was defined as the volume of tissue with time-to-maximum (Tmax) delay >6 seconds minus the volume of the baseline ischemic core. Final lesion volume was determined by volumetric measurement of the hypoattenuated ischemic region on follow-up NCCT, corresponding to the subacute ischemic lesion according to previous literature.
14
The absolute ischemic lesion growth was defined as the difference between the final lesion volume on follow-up imaging and the baseline ischemic core volume on admission imaging according to formula (2):
Outcome measures
Primary outcome was ischemic lesion NWU on admission NCCT. Secondary outcome was the ischemic lesion growth between admission and follow-up imaging.
Statistical analysis
Descriptive statistics were used to analyze the study cohort subdivided into patients with pLVO and dLVO. Categorical variables were reported as counts and percentages. Continuous variables were reported as median and interquartile range (IQR). Categorical variables were compared between subgroups using chi-squared test. Continuous variables were compared between groups using Mann-Whitney U test after rejection of the normality assumption using Kolmogorov-Smirnov test (Table 1).
Baseline, imaging, treatment characteristics and outcome measures dichotomized by occlusion location.
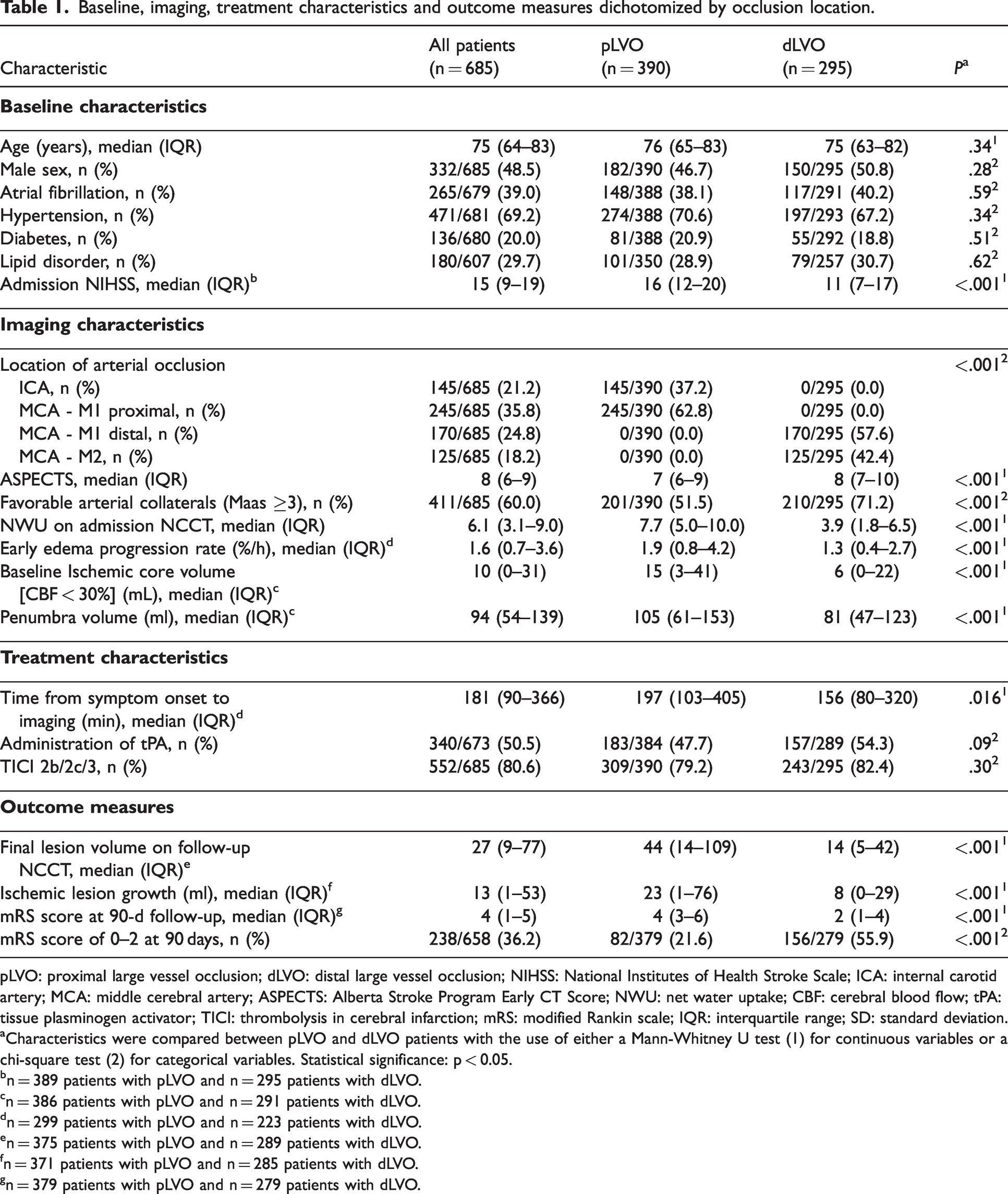
pLVO: proximal large vessel occlusion; dLVO: distal large vessel occlusion; NIHSS: National Institutes of Health Stroke Scale; ICA: internal carotid artery; MCA: middle cerebral artery; ASPECTS: Alberta Stroke Program Early CT Score; NWU: net water uptake; CBF: cerebral blood flow; tPA: tissue plasminogen activator; TICI: thrombolysis in cerebral infarction; mRS: modified Rankin scale; IQR: interquartile range; SD: standard deviation.
Characteristics were compared between pLVO and dLVO patients with the use of either a Mann-Whitney U test (1) for continuous variables or a chi-square test (2) for categorical variables. Statistical significance: p < 0.05.
n = 389 patients with pLVO and n = 295 patients with dLVO.
n = 386 patients with pLVO and n = 291 patients with dLVO.
n = 299 patients with pLVO and n = 223 patients with dLVO.
n = 375 patients with pLVO and n = 289 patients with dLVO.
n = 371 patients with pLVO and n = 285 patients with dLVO.
n = 379 patients with pLVO and n = 279 patients with dLVO.
Univariable and multivariable linear regression was conducted to identify factors associated with ischemic lesion NWU (Model A) and ischemic lesion growth between admission and follow-up imaging (Model B; Table 2). Unadjusted and adjusted β estimates were reported with 95% confidence intervals (CI). For multivariable regression analyses, independent variables were selected a priori. The following independent variables were included in Model A and Model B: Age, sex, admission National Institutes of Health Stroke Scale (NIHSS), time from symptom onset to imaging, site of vessel occlusion (pLVO vs dLVO), baseline ASPECTS, baseline ischemic core volume, and arterial collateral status. The results of Model B were additionally adjusted for: Ischemic lesion NWU, treatment with intravenous thrombolysis, and successful endovascular therapy defined as final Thrombolysis in Cerebral Infarction (TICI) grade 2 b, 2c, or 3. A sensitivity analysis was conducted to support the robustness of the main findings of Model A by excluding patients with small ischemic core volume <10 ml (Supplementary Table 1).
Univariable and multivariable regression analyses to identify factors independently associated with ischemic lesion net water uptake (NWU) on admission (Model A) and ischemic lesion growth between admission and follow-up imaging (model B).
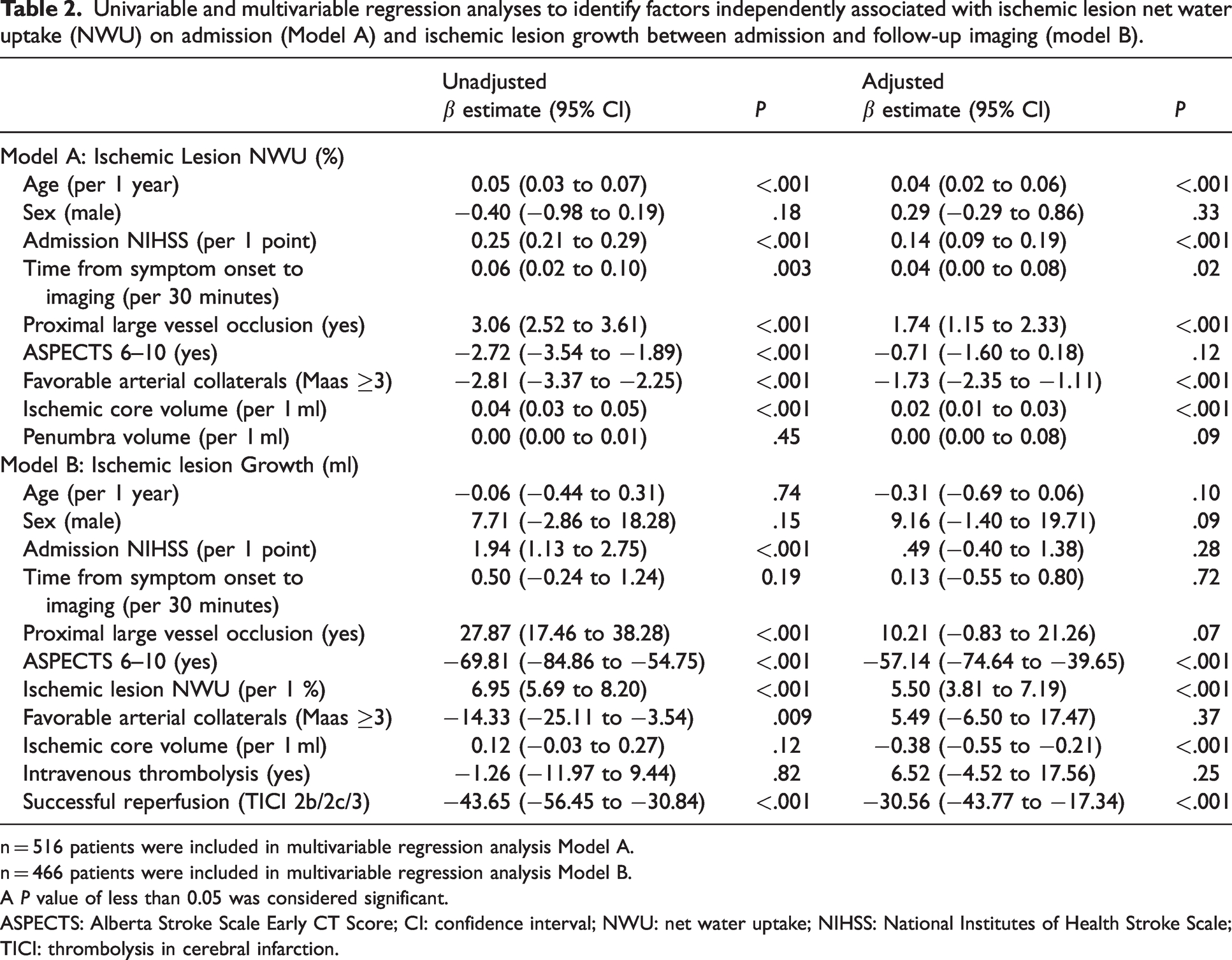
n = 516 patients were included in multivariable regression analysis Model A.
n = 466 patients were included in multivariable regression analysis Model B.
A P value of less than 0.05 was considered significant.
ASPECTS: Alberta Stroke Scale Early CT Score; CI: confidence interval; NWU: net water uptake; NIHSS: National Institutes of Health Stroke Scale; TICI: thrombolysis in cerebral infarction.
Mediation analysis was used to evaluate to what extent arterial collaterals explain the relationship between the site of vessel occlusion and ischemic lesion NWU. 15 The package ‘mediation’ in R statistical software was used to assess the total, direct and indirect effect of the specified mediation model (Figure 3 and Supplementary Table 2). Results of the mediation analysis were adjusted for age, sex, admission National Institutes Health Stroke Scale (NIHSS), time between symptom onset and imaging, baseline Alberta Stroke Program Early CT Score (ASPECTS), baseline ischemic core volume, occlusion location and arterial collateral status.
A two-tailed P < .05 was considered significant for all statistical tests. Complete-case analysis was performed for all regression models. R statistical software (version 4.3.0, R Project for Statistical Computing) and Rstudio statistical software (version 2023.03.0 + 386, Rstudio) were used for all statistical analyses and data visualization.
Results
Patient characteristics
A total of 685 patients met the inclusion criteria and were included for further analysis. Among those, the median age was 75 (IQR, 64–83), 332 (48.5%) were men and 353 (51.5%) were women. The median NIHSS score on admission was 15 (IQR, 9–19) and the median baseline ASPECTS was 8 (IQR, 6–9). Successful mechanical reperfusion (TICI 2b/2c/3) was achieved in 552 (80.6%) patients. At 90 days after stroke, 238 (36.2%) patients were functionally independent, defined as mRS score of 0 to 2. Please refer to Table 1 for more information about baseline, imaging, and treatment characteristics.
Patient characteristics stratified by occlusion location
The study cohort was dichotomized into patients with pLVO (390 [56.9%] patients) and dLVO (295 [43.1%] patients). Patients with pLVO demonstrated longer time from symptom onset to imaging (median, 197 vs 156, P = .016), higher NIHSS on admission (median, 16 vs 11, P < .001) and lower baseline ASPECTS (median, 7 vs 8, P < .001). Favorable arterial collaterals were more likely in patients with dLVO (51.5% vs 71.2%, P < .001). The median ischemic lesion NWU on admission was 7.7% in pLVO patients and thus twice as large as in dLVO patients (median, 7.7% vs 3.9%, P < .001). Ischemic lesion NWU was significantly lower in both pLVO and dLVO in presence of favorable arterial collaterals (Figure 2).
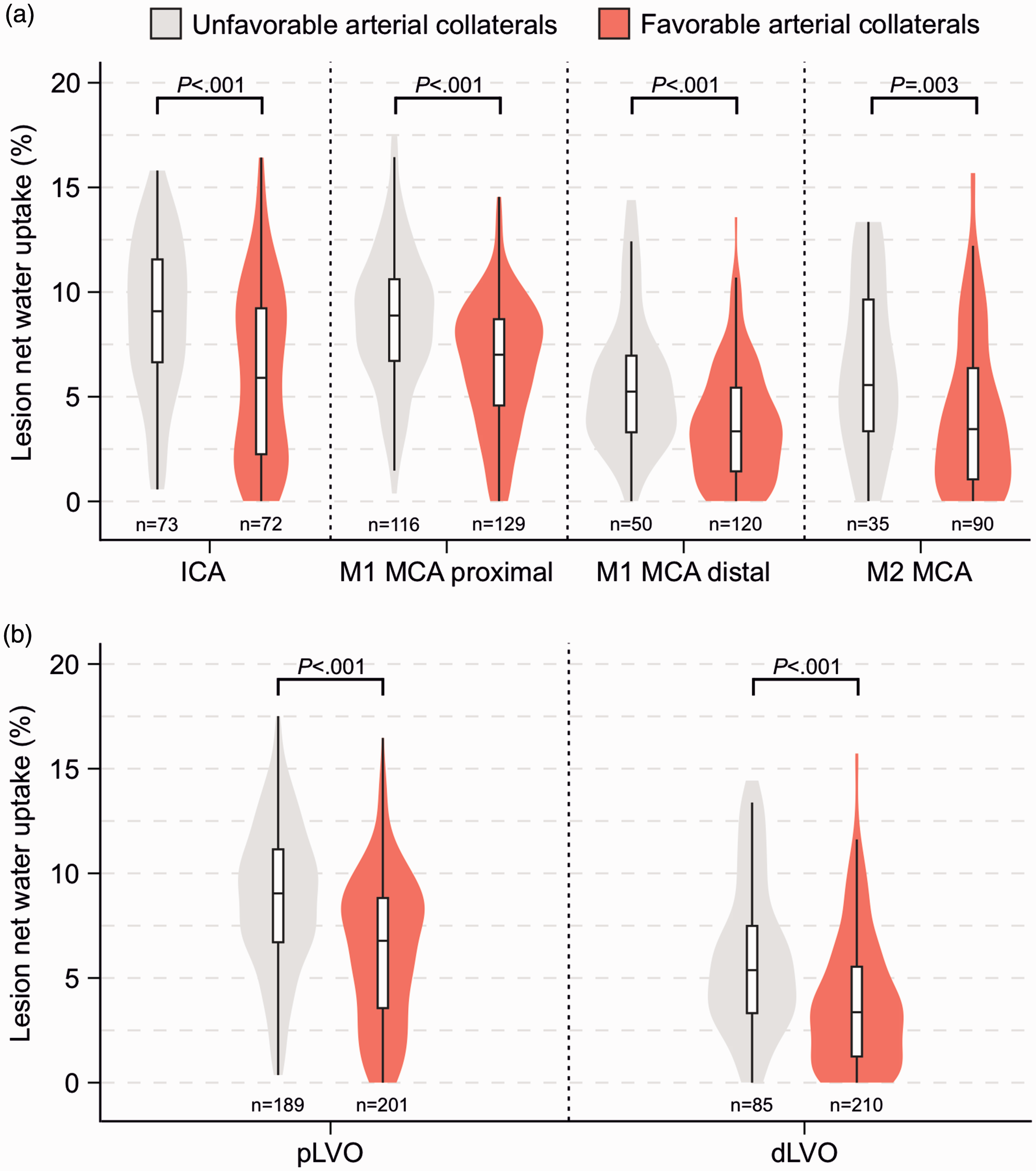
Relationship between occlusion location, arterial collateral status, and ischemic lesion net water uptake. (a) Violin plots comparing the probability distributions of ischemic lesion net water uptake on admission NCCT stratified by detailed occlusion location and arterial collateral status. Please note that ischemic lesion net water uptake was significantly lower in presence of arterial collaterals across all occlusion locations and (b) Same as Figure 2(a) but stratified by proximal large vessel occlusion (pLVO; ICA and M1 MCA proximal) and distal large vessel occlusion (dLVO; M1 MCA distal and M2 MCA). Consistently, ischemic lesion net water uptake was significantly lower in presence of arterial collaterals.
Predictors of ischemic lesion net water uptake
Multivariable linear regression was performed to identify factors independently associated with ischemic lesion NWU on admission NCCT. Older age, higher admission NIHSS, longer time from symptom onset to imaging, and higher baseline ischemic core volume correlated with higher baseline NWU, while favorable arterial collaterals were associated with lower baseline NWU (Table 2). Notably, the presence of pLVO was found to increase baseline NWU even after adjustment for covariates (β estimate, 1.74, 95% CI, 1.15 to 2.33, P < .001). The association between occlusion location and ischemic lesion NWU remained significant after excluding patients with small baseline ischemic core volumes (Supplementary Table 1).
Mediation between occlusion location and net water uptake
We performed a mediation analysis to further investigate the association between the occlusion location and early ischemic lesion NWU. The regression coefficients of the total effect (c), the direct effect (c′) and indirect effect (a1b1) were significant (Figure 3 and Supplementary Table 2). Thus, the relationship between occlusion location and NWU was partially mediated by the degree of arterial collaterals.
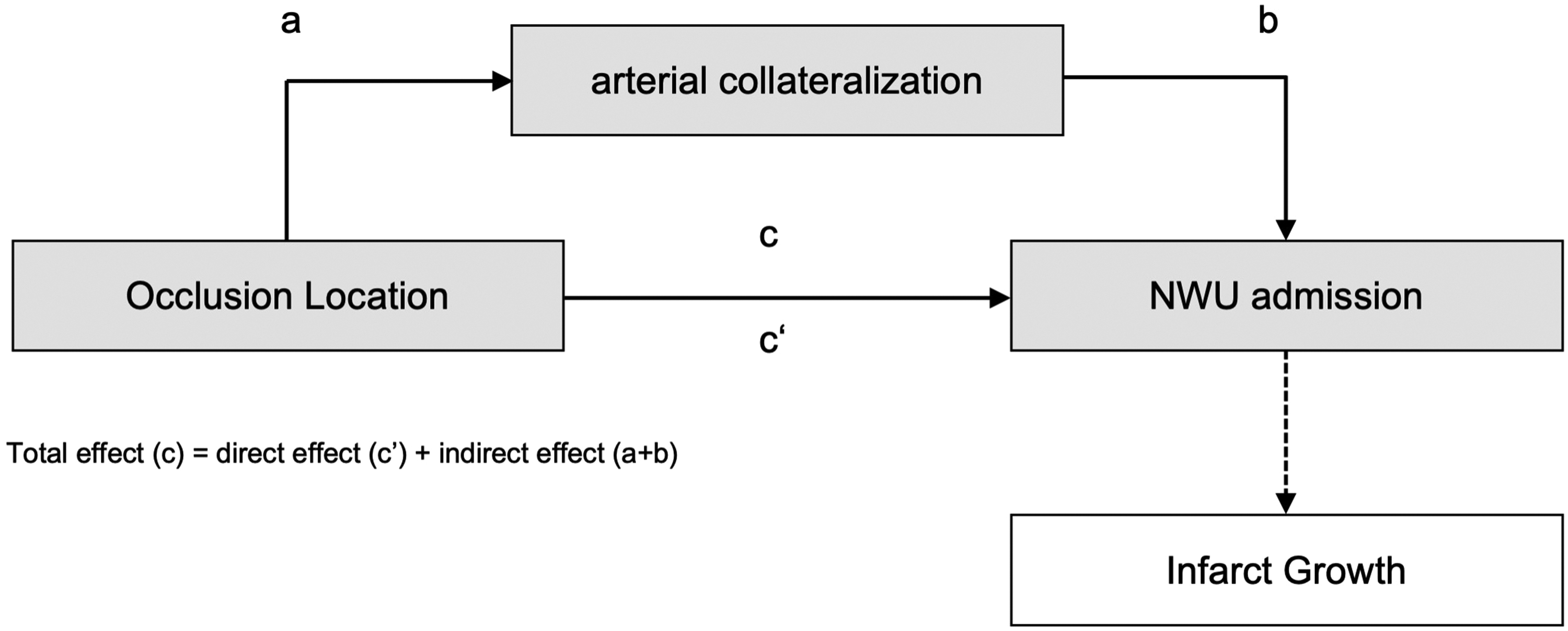
Mediation model of the relationship between occlusion location and ischemic lesion net water uptake in acute ischemic stroke.
Predictors of ischemic lesion growth
The volume of ischemic lesion growth between admission and follow-up imaging differed significantly between pLVO and dLVO (23 ml vs 8 ml, P < .001). In addition to the baseline ASPECTS, ischemic core volume, and successful mechanical reperfusion, higher early NWU was an independent predictor of larger ischemic lesion growth between admission and follow-up imaging (β estimate, 5.50, 95% CI, 3.81 to 7.19, P < .001).
Discussion
This retrospective multicenter study investigates the relationship between occlusion location, ischemic lesion NWU, and ischemic lesion growth in ACLVO. We found that proximal occlusion sites in the anterior circulation were associated with greater ischemic lesion NWU. The association between occlusion location and the extent of NWU was partially mediated by the arterial collaterals status. Voxel-wise NWU within the early ischemic lesion was correlated with the volume-based ischemic lesion growth between admission and follow-up imaging. Our findings provide new insights into early ischemic lesion NWU, which might help to establish more accurate prognostic thresholds for this quantitative imaging biomarker of cerebral edema.
It could be argued that the occlusion location influences, first and foremost, the absolute volume of edematous brain tissue in the acute phase of ACLVO. Our findings suggest that the occlusion location is additionally associated with the voxel-wise extent of cerebral edema approximated by NWU, even after adjustment for covariates such as time between symptom onset and imaging. Higher baseline NWU in patients with more proximal occlusion sites, and its mediation by arterial collateral flow, might be explained on various levels of collateral flow.
First, more proximal vessel occlusions in the anterior circulation reduce the collateral ability of arterial segments of the circle of Willis, such as the anterior communicating artery. These so-called primary collaterals might rapidly compensate for decreased cerebral blood flow of the affected hemisphere.16,17 For instance, a thrombus in the terminal bifurcation of the ICA (Carotis-T occlusion) results in an occlusion of the M1 segment of MCA and of the A1 segment of the anterior cerebral artery (ACA), eliminating antegrade flow via the A1 segment into the ipsilateral ACA territory. In this scenario, collateral flow into the ipsilateral ACA territory depends on the patency of the anterior communicating artery. There is similar dependency on primary collaterals in more proximal ICA occlusions, in which reverse blood flow via the anterior or posterior communicating artery might be crucial to supply the affected MCA territory. The prognostic value of patent primary collaterals is underscored by recent studies, which found superior functional outcomes in patients with complete circle of Willis.18,19
The presence and patency of primary collaterals, in turn, might facilitate the recruitment of cortical or leptomeningeal collaterals, often referred to as secondary collaterals. These anastomoses between distal parts of the major cerebral arteries, most numerous between ACA and MCA, may pave the way for reverse arterial flow into the hypoperfused territory, thereby sustaining tissue viability. Beyond that, previous studies highlighted the role of deep subcortical collaterals in lacunar strokes.20,21 It is conceivable that the more perforating arteries of the M1 segment are occluded, the worse is the degree of subcortical collaterals in ACLVO, and, ultimately, the higher NWU of ischemic lesions in the basal ganglia. Technical progress, including ultra-high field strengths to achieve higher spatial resolution in 4 D flow MRI, might advance research on deep collateral flow in future studies.
Thus, more proximal occlusion locations might cause deterioration of both primary and secondary collaterals, associated with greater voxel-wise cerebral edema as suggested by our study. The failure of arterial collaterals is known to reduce the blood flow in the further downstream capillary and venous system, which has been found to be a significant predictor of cerebral edema formation and ischemic lesion NWU in ACLVO.8,22,23 According to these findings, the failure of arterial collaterals has been linked to worse cerebral perfusion parameters derived from advanced imaging.24,25 In summary, the deterioration of collateral flow via the arterial, capillary and venous system provides a convincing explanation for greater ischemic lesion NWU in patients with more proximal ACLVO.
Our study highlights ischemic lesion NWU on admission NCCT as a significant predictor of ischemic lesion growth within 24 hours after stroke (Figure 4). This finding establishes a connection between (1) voxel-wise NWU, which does not take the volume of the initial brain lesion into account, and (2) the volume-based ischemic lesion progression, which can range from harmless focal edema to malignant infarction and death. Again, the relationship between NWU and ischemic lesion growth might be explained by the fact that NWU is highly associated with the occlusion location and serves as a surrogate for the capacity of arterial collaterals to sustain collateral blood flow (Figure 3). In line with these considerations, both more proximal occlusion location and poorer arterial collaterals have been described as strong predictors of fast ischemic lesion progression.26,27
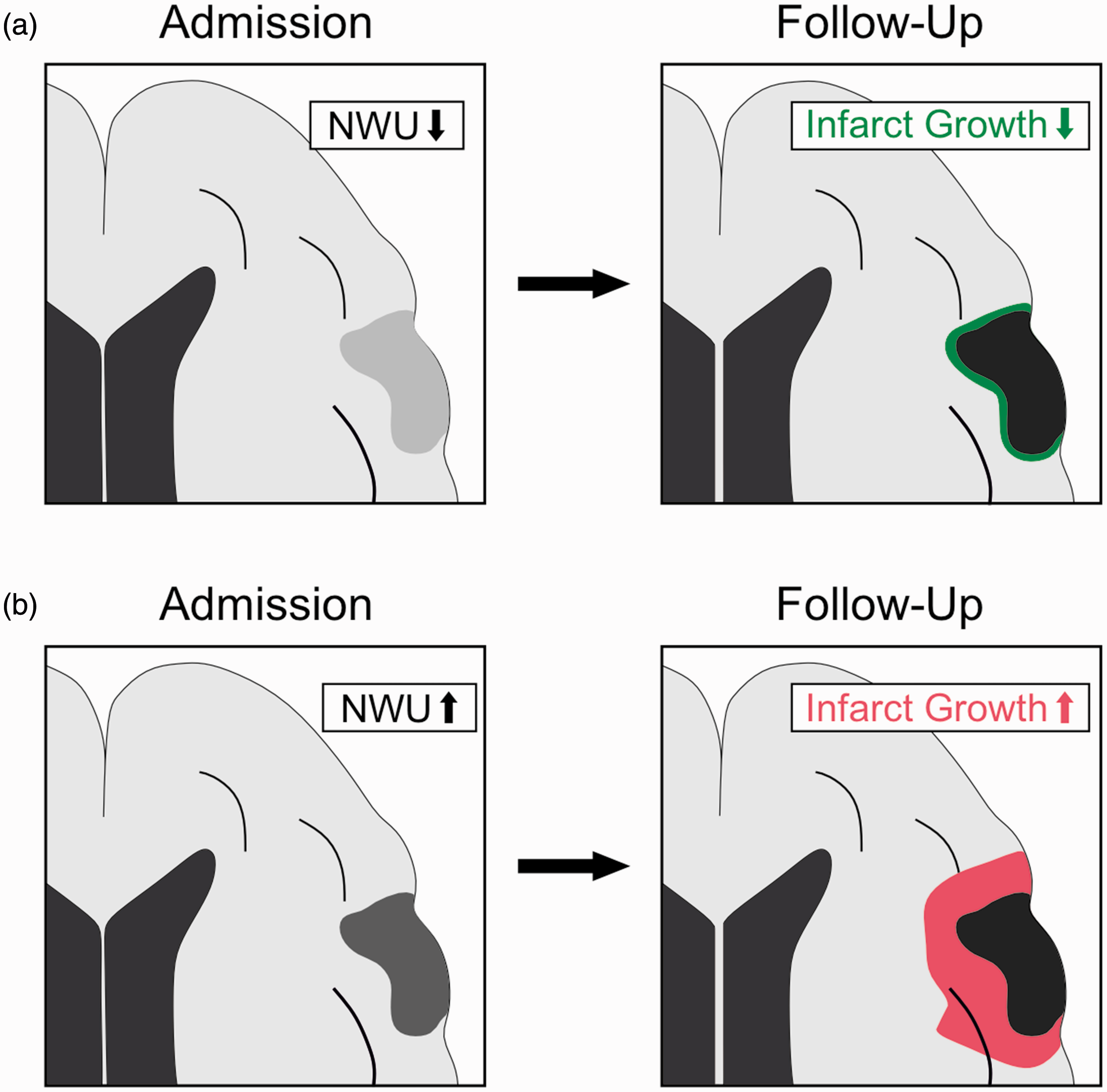
Ischemic lesion net water uptake as predictor of lesion growth. (a) Illustration of an ischemic lesion with low admission net water uptake (NWU; light grey area) in the territory of the middle cerebral artery. Lesion growth between admission and follow-up imaging (green area) was relatively small and (b) Alternative scenario of an ischemic lesion with high admission NWU (dark grey area). Please note that lesion growth was relatively greater.
In summary, diagnostic thresholds adjusted for the occlusion location might further improve the prognostic value of NWU for ischemic lesion growth and adverse events. For instance, a distal M1 occlusion with ischemic lesion NWU of 8% should be less likely to develop malignant infarction compared with a more proximal ICA occlusion exhibiting the same NWU. To date, however, ischemic lesion NWU remains an experimental imaging biomarker of cerebral edema without application in daily clinical practice. The manual and thus work-intensive postprocessing currently precludes its use and validation as prognostic parameter in prospective study designs. Future approaches for automated and fast estimation might leverage the potential of ischemic lesion NWU to improve the treatment of ACLVO.
The present study has certain limitations. First, the retrospective study design could introduce selection bias and reduce the generalizability of the main findings. Second, the manual definition of the region of interest for densitometric measurement of NWU is prone to significant interrater variability and inaccuracies caused by very small lesions and other, primarily hypoattenuating pathologies, such as cerebral small vessel disease. Third, the assessment of arterial collateral status was assessed on single-phase CTA and might therefore be influenced by the timing of the image acquisition. Fourth, the baseline ischemic core volume was required to calculate the ischemic lesion growth and was approximated by CTP imaging, which might have led to overestimation as reported previously. 28
Conclusion
This study suggests that more proximal vessel occlusions are independently associated with higher early ischemic lesion NWU, partially mediated by the extent of arterial collaterals. It highlights the connection between the voxel-wise NWU without volume dimension and the ischemic lesion growth as a volume-based parameter. Future studies on ischemic lesion NWU should consider the occlusion location to define more accurate thresholds, with the final aim to improve treatment decision making and outcome prediction by using this quantitative marker of cerebral edema within an automated diagnostic workflow.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241232193 - Supplemental material for Association between occlusion location, net water uptake and ischemic lesion growth in large vessel anterior circulation strokes
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241232193 for Association between occlusion location, net water uptake and ischemic lesion growth in large vessel anterior circulation strokes by Laurens Winkelmeier, Jeremy J Heit, Gabriel Broocks, Julia Prüter, Christian Heitkamp, Maximilian Schell, Gregory W Albers, Maarten G Lansberg, Max Wintermark, André Kemmling, Christian Paul Stracke, Adrien Guenego, Daniel Paech, Jens Fiehler and Tobias D Faizy in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Tobias Djamsched Faizy was funded by the German Research Foundation (DFG) for his work as a postdoctoral research scholar at Stanford University, Department of Neuroradiology (Project Number: 411621970).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Laurens WINKELMEIER reports no disclosure.
Dr Jeremy J. HEIT reports consulting for Medtronic and MicroVention and Medical and Scientific Advisory Board membership for iSchemaView.
Dr Gabriel BROOCKS reported receiving personal fees from Eppdata GmbH and compensation as a speaker from Balt outside the submitted work.
Julia PRÜTER reports no disclosure.
Dr Christian HEITKAMP reports no disclosure.
Maximilian SCHELL reports no disclosure.
Dr Gregory W. ALBERS reports equity and consulting for iSchemaView and consulting from Medtronic.
Dr Maarten G. LANSBERG reports no disclosure.
Dr Max WINTERMARK reports grants and funding from the NIH under the grant numbers (1U01 NS086872-01, 1U01 NS087748-01 and 1R01 NS104094).
Dr André KEMMLING reports a research collaboration agreement with Siemens Healthcare (company involved in CT/MRI distribution).
Dr Christian Paul STRACKE reports no disclosure.
Dr Adrien GUENEGO reports no disclosure.
Dr Daniel PAECH reports no disclosure.
Dr Jens FIEHLER reports grants and personal fees from Acandis, Cerenovus, MicroVention, Medtronic, Stryker, Phenox and grants from Route 92 outside the submitted work.
Dr Tobias D. FAIZY reports grants from the German Research Foundation (DFG) during the conduct of the study.
Authors’ contributions
Conception/design of the work: LW, JH, JF, TF. Data collection: TF, JH, JP. Interpretation of data: LW, JH, GB, JP, CH, MS, GA, ML, MW, AK, CS, AG, DP, JF, TF. Drafting the work: LW, TF. Revising the work for important intellectual content: JH, GB, JP, CH, MS, GA, ML, MW, AK, CS, AG, DP, JF. Supervision: JF, TF.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.