Abstract
Selective brain cooling is a promising technique for improving outcomes in ischemic stroke in the area of reperfusion. A recent study described the efficacy of a new method of selective brain cooling via active conductive head cooling. This is a major step forward in the administration of hypothermic treatment during pre-hospital transfer. However, to enhance the benefits of selective therapeutic cooling, a more comprehensive strategy preventing delay in hypothermic induction and increasing the accuracy of selectivity in the brain should be considered to mitigate the side effects related to therapeutic hypothermia.
Therapeutic Hypothermia (TH) has long been demonstrated to be an effective neuroprotective method in acute ischemic stroke (AIS) as shown in a plethora of pre-clinical studies. 1 However, clinical trials have seldom replicated the success of TH in stroke patients. Potential reasons may include undefined parameters of TH, such as onset time, cooling velocity, depth, duration, and rewarming speed, all of which might increase side effects related to hypothermia and reduce its benefits. An earlier onset time is widely considered to be associated with increased ‘freezing’ penumbra tissues and better clinical outcomes. 2 The typical patient loses 1.9 million neurons each minute when stroke is untreated. 3 A potential way to reduce the time from stroke onset to TH treatment is to initiate safe hypothermic therapy during pre-hospital transfer, such as in the ambulance. In addition, many multicenter randomized clinical trials designed to assess the efficacy of a TH device in stroke patients were stopped prematurely, mainly because of increased systemic complications such as pneumonia. 4 Thus, selective brain cooling, or cooling the brain while maintaining the core temperature, is proposed to maximize the benefits of TH therapy and minimize the side effects related to TH treatments. 5
The current challenge for TH in AIS is to optimize hypothermic parameters and establish a systematic program to manage brain temperature from first aid to inter-hospital treatment. A recent study by Diprose et al provides a potential selective cooling method called the WElkins Temperature regulation system, 2nd Gen (TRS-2). 6 In brief, this system consists of a cooling cap that circulates coolant. After wearing the cooling cap for 80 minutes, the mean brain temperature was reduced by 0.9 °C, while the core body temperature only decreased by 0.3 °C. More importantly, the conductive head cooling was well-tolerated in all participants, and no hypothermia-related complications were detected. There are a couple advantages to this active conductive cooling. Firstly, it is convenient and can be induced at an early stage of stroke, before patients have been transferred to the hospital. Secondly, it is non-invasive and is relatively safe. However, the cooling velocity was still slow (>80 min), and a recommended mild hypothermic status (<36°C) was not achieved. Conductive head cooling can thus be considered an important component of selective brain cooling as a viable adjunctive treatment approach, though it will need to be used in conjunction with other limited TH strategies.
Surface hypothermia is the most practical and feasible method to induce TH. The conductive head cooling by TRS-2 is generally just as effective in lowering brain temperature as the traditional brain surface hypothermic methods in patients. 7 Importantly, TRS-2 system extends patient temperature management to the pre-hospital setting, enabling the early induction of therapeutic hypothermia and saving impaired neuron in the penumbra. However, ischemic stroke is a dynamic pathological process, including not only oxygen, glucose, and ATP deprivation during the acute stage, but also reperfusion injuries during the sub-acute stage. 8 Thus, a combined selective brain hypothermia strategy, including pre-hospital and in-hospital approaches, is needed. Combining different hypothermia therapies can promote their advantages while compensating for their disadvantages. For example, the cooling velocity of conductive head cooling is not swift at present as the human skull is too thick compared to rodents. Previous studies have indicated physical cooling with pharmacological hypothermia has been shown to reduce brain temperature more swiftly and smoothly in animal models. 9 Thus, in pre-hospital situations where extra operation cannot be performed, pharmacological hypothermia may be a complementary method to accelerate the cooling of the brain induced by conductive head cooling.
The interhospital treatment of acute ischemic stroke (AIS) has been revolutionized by the development of recanalization therapy. Thus, the most promising selective cooling strategy at present may be intra-arterial selective cooling infusion (IA-SCI). This strategy has been shown to cool the brain to mild hypothermic status in minutes in rodents by directly flushing the ischemic territory with cold solutions. 10 This approach mitigates both ischemia-related damages and reperfusion-associated impairments. More importantly, IA-SCI can be administrated shortly after endovascular thrombectomy through the same micro-catheter without additional procedures. However, image evaluations, such as CT/CTP, are necessary before mechanical thrombectomy in situations when patients arrive at the hospital beyond the time window. Therefore, active conductive cooling may be used to cover the time delay from the ambulance to admission to hospital to re-open the occluded artery. Thus, the combination of conductive head cooling and IA-SCI may maintain selective brain hypothermia, extend its duration, and maximize its translational potential.
Typical side effects of TH include shivering, infection, arrhythmia and so on. Evidences have shown that these systemic side effects are generally correlated to long duration, deep and systematic hypothermia. 1 Focalizing the hypothermic territory and modifying the optimal duration is the greatest challenge at present. Various local hypothermic methods have been introduced to selectively cool the brain while minimizing systemic TH-related complications. Temperature management from first aid to interhospital treatments is necessary to cool the brain as soon as possible.
Selective therapeutic hypothermia, as a promising multi-target neuroprotective strategy, has been demonstrated to be effective in maximizing hypothermic benefits by lowering the brain metabolic rate, inhibiting inflammation and apoptosis, and minimizing the side effects of hypothermia. 8 However, a single TH strategy targeting pathological changes at a specific stage of AIS cannot confer comprehensive neuroprotective effects. Thus, a combined selective brain cooling strategy, including selective head cooling and IA-SCI, may be a viable additional treatment approach for reperfusion therapy after stroke (Figure 1).
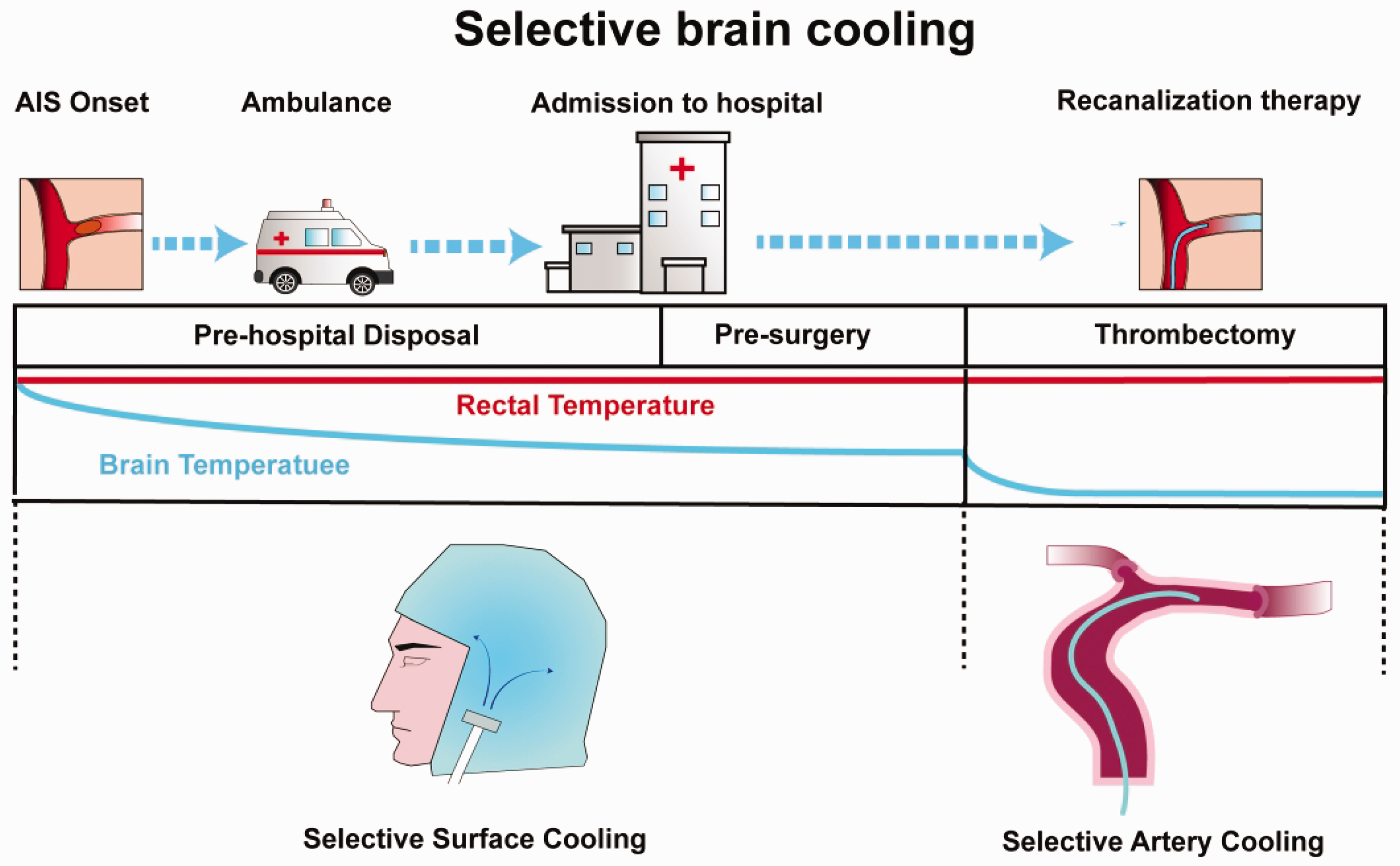
The schematic exhibits a persistent temperature management utilizing selective surface cooling during pre-hospital transit and IA-SCI during thrombectomy.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (82071466,82027802, and 82102220).
Acknowledgements
The authors thank Kelly Chung for language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
